Key Words
external volume expander (EVE), fat grafting, autologous fat transfer, breast reconstruction, tissue expansion, post mastectomy, graft survival, flaps, breast implants, breast sensation
Introduction
To reconstruct total mastectomy defects, implants and flaps were traditionally the only available options. Fat grafting, for total breast reconstruction, is now an evolving third alternative. Over the past 15 years we have refined this procedure and we are now able to, in most cases, restore aesthetically pleasing breasts that feel and look like the original, with the added benefit of normal sensation. Total breast reconstruction with external vacuum expansion (EVE) and autologous fat transfer (AFT) is minimally invasive, and can over multiple stages recreate an autologous breast mound .
Total breast reconstruction with EVE + AFT generates a three-dimensional vascularized and innervated graft recipient scaffold where large amounts of carefully seeded lipoaspirate can survive to regenerate the breast mound.
Recreating a breast mound takes time; although minimally invasive, fat grafting for breast reconstruction is still plastic surgery. We build the mound in a stepwise fashion with 3–6 procedures performed in succession over the course of 9–18 months. Interestingly, many cases of implant and flap breast reconstruction include some fat grafting as an adjunct. There is even a trend towards smaller flaps to enlarge the recipient site along with more fat grafting sessions. The fat grafting procedures are outpatient, and therefore the number of procedures is less of an issue. It is the cost:benefit ratio and the quality of the final result that ultimately matter.
In this chapter we will elaborate on the principles behind large volume AFT and describe our technique that adheres to these principles.
Indications and Contraindications
The technique of total breast reconstruction with fat grafting is indicated for women who wish to avoid invasive procedures and alloplastic implants. The goal is similar to any breast reconstruction: restore her body with natural-looking and sensate breasts. After mastectomy surgery, flap transfer surgery has been the best autologous tissue option. However, the recovery is long and there are donor-site scars distant to the breast. Implant-based reconstruction is considered less surgery, but many women are reluctant to use foreign materials and are not fully satisfied with the results.
The only absolute contraindication to total breast reconstruction with fat grafts is active smoking. Fat grafting has very poor outcomes in smokers. Additionally, blood thinners, anticoagulants, or any vitamins/herbs that may prolong bleeding time must be discontinued well before the surgery. This is primarily due to the fact that extravascular blood from excess bleeding during cannula excursion will compete with the fat for graft-recipient interstitial space. When tissues become taut with extravasated blood, there is less room for the fat grafts.
If a patient has a mastectomy that is irradiated and/or has suffered scarring from prior reconstruction failures, then the patient has to understand that total breast reconstruction with fat graft will involve a minimum of five procedures over 12–24 months.
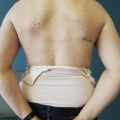
Since EVE is crucial to the success of this technique, inability or unwillingness to comply with the EVE preparation protocol is a major drawback. In order to eliminate non-compliance, patients test-wear EVE in the office for 20–30 minutes. When they become comfortable with wearing the device, we educate them on proper use and consistency with its wear. From our clinical experience, we have found that most intelligent educated women can bear the inconvenience of EVE for a few hours a day over a few weeks. Therefore, EVE is more ideal for educated women who are willing to work with the surgeon and personally contribute to the goal of achieving a breast reconstruction through a sequence of less invasive procedures.
Preoperative Evaluation
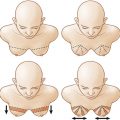
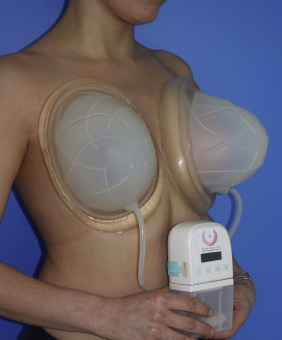
Preoperatively, patient selection and education are paramount and cannot be understated. The patient must understand that successful results depend upon her active contribution, and that reconstruction does not occur overnight. Additionally, unless they have had a total mastectomy, all patients are required to have preoperative MRI in order to exclude occult pathology and obtain a baseline of residual breast tissue quality and volume ( Fig. 19.1 ).
Surgical Techniques
Breast mound reconstruction with EVE + AFT involves minimal incisions; therefore, there is no conventional exposure and no tissue dissection. However, there is much more to successful AFT than syringes, cannulas, and strong arms. The final outcome depends upon a series of steps that need to be individually optimized. The result is the product of the weakest link among these individual steps. Each step in this reconstructive process has the potential for failure and can doom the reconstruction.
The outcome is limited by the rate-limiting step and investing in non-rate-limiting steps is futile. It is important to consider the process in its entirety and to avoid focusing on only one factor while disregarding the others.
Fundamental Concepts of Fat Graft Survival
Avascular free fat grafts cannot survive without revascularization. To ensure that avascular grafts re-vascularize, it is important to understand the fundamentals of 3-D revascularization. Just like the familiar 2-D skin grafts, close graft-to-recipient contact is essential for revascularization. Arbitrary bolus fat injections to plump up a breast mound are doomed for failure, because without restoration of blood supply the result is widespread necrosis.
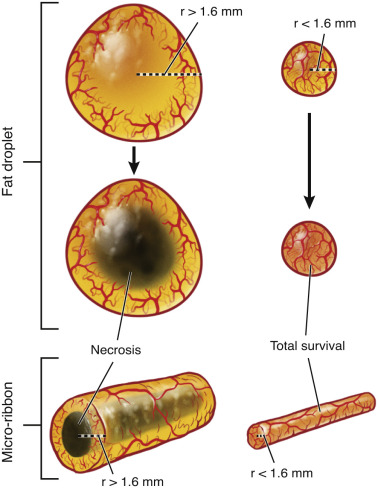
It has been well established that an inserted fat graft droplet has three layers or zones. The most peripheral zone is in direct contact with the recipient bed and will readily re-vascularize through neo-angiogenesis. Unfortunately, neo-angiogenesis is a slow process, and less than 1 mm of the outermost shell can re-establish blood supply before the deeper adipocytes succumb to hypoxia. Just below this layer lies the regenerative zone, where only the more hypoxia-resistant adipose-derived stem cells (ASC) or stromal vascular fraction can re-vascularize, survive, and regenerate a new adipocyte population. Deep to this regenerative zone lies the necrotic zone, where no cells can survive. Studies have demonstrated that even under ideal circumstances, the maximum depth of the graft survival zones is 1.6 mm, and that the rate-limiting step in this process is oxygen diffusion. Revascularization depends upon this critical graft-to-recipient interface, and in large volume fat grafting, only “micro-droplets” or “micro-ribbons” in the 3-mm (1.6 mm × 2) range re-vascularize and survive. In order to optimize this graft-to-recipient interface, large graft volumes must be skillfully and precisely delivered in a 3-D distribution as a fine mist of 17-µL (4/3πr 3 ) micro-droplets, or since we deliver through cannula passes, 3-mm wide micro-ribbons that do not coalesce. Droplets larger than 17 mL or ribbons wider than 3.2 mm will invariably suffer central necrosis ( Fig. 19.2 ).
Principles of Fat Graft Harvesting and Preparation
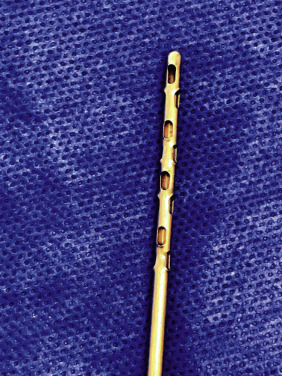
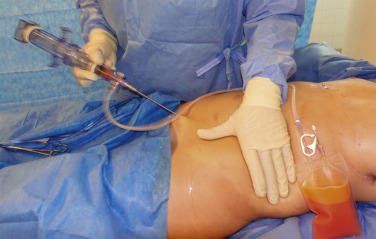
We obtain the fat cells by simple liposuction. In addition to fat cells, the lipoaspirate contains fibroblasts, endothelial cells, pericytes, white blood cells, platelets, and adipose-derived stem cells. In our experience we find it important to graft back the entire group of cells not just the pure adipocytes. In order to get the best graft, we favor extensive tumescence of the donor sites (more than 1 L per donor area) to obtain non-bloody aspirates that can rapidly sediment. We prefer harvesting in a closed system with a constant low-pressure (300-mmHg) spring-activated syringe suction to minimize trauma to the adipocytes. While vacuum pumps can also deliver controlled low pressures they often expose the fat to high airflow in the necessary long tubing before they reach the rigid collection containers. We harvest with a thin, 2.7-mm cannula with 12 holes 1 × 2 mm each. This allows us to harvest smaller fat droplets through tiny puncture wounds that leave minimal scars. Our preferred cannula also has winglets at the opening that invite more fat, while the rough barbs help harvest more stromal vascular tissue. Instead of a scalpel we insert the cannula through 14G needle punctures all around the donor site and we liposuction with criss-crossing passes through these multiple entry sites. This technique provides for the most even harvest with minimal scarring ( Fig. 19.3 ).
While some advocate removing inflammatory cells and debris through filtration or washing, others champion the opposite and add platelets that are rich in inflammatory agents. Efforts to remove from the graft the very same tissue components present at the recipient site are futile. Based upon theoretical and laboratory advantage, some advocate adding stem cells. However, the few controlled clinical studies showed no benefit for this cumbersome and regulatory obstacle-ridden technique. Therefore, since there is no conclusive evidence for the clinical advantage of adding to or removing substances from the harvested adipose tissue, we prefer to take a purist approach, and the leave the graft with its surrounding microenvironment as minimally manipulated as possible. As a matter of fact, there is mounting evidence that the entire harvested liposuction matrix is required for optimal clinical result.
We allow the lipoaspirate to gravity sediment in the collection bag for a few minutes and simply drain the infranatant fluid and collect the supernatant fat into a lipografting bag. Along with most surgeons with extensive experience with large volume fat transfer, we moved away from centrifugation. This is supported by a recent prospective controlled randomized study. We use the Lipografter (MTF, Edison, NJ) to perform the graft harvesting, preparation, and reinjection in a closed system ( Fig. 19.4 ).
Principles of Recipient Site Preparation
Just as in 2-D grafting where the size of the graft must match the surface of the wound, in 3-D grafting, we cannot graft more than what the recipient can expand to accommodate. In post-mastectomy breast reconstruction, the recipient site capacity is quite limited. We use EVE to prepare the recipient bed by increasing its compliance. This increases the amount of graft the mastectomy defect can accept without crowding and without increasing the interstitial pressure to levels that jeopardize perfusion. EVE stretches blood vessels and creates new ones, and this angiogenesis primes the tissues for better revascularization. Along with the blood vessels, sensory nerves are also stretched and it is this neo-neurogenesis that restores sensation to the regenerated breast mound. EVE also creates the skin envelope required for forming a breast mound. There is also evidence that it primes the recipient tissue to accept more grafts by activating pathways independent from the enhanced vascularity.
EVE is worn like a bra for a few hours a day, every day for about 2–3 weeks prior to surgery. From our extensive empiric experience, we determined that the optimal therapeutic dose that prepares the breast for autologous fat transfer is a minimum of 200 hours over the 15 days immediately preceding the grafting with a vacuum pressure that alternates 60–0–60 mmHg for 3 minutes on and one minute off.
Principles of Fat Graft Delivery
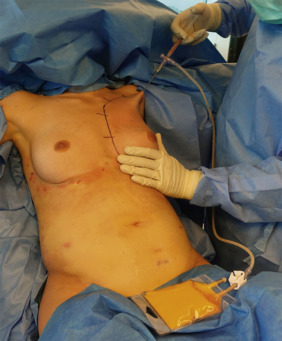
Fat grafting for breast reconstruction is a blind procedure that requires craftsmanship, artistry, and adherence to fundamental principles. Since the tip of the cannula is not exposed, the surgeon does not see the graft as it is delivered under the skin, and is therefore never certain that distribution is even or optimal. However, just like the liposuction technique, where the tip of the cannula is not visualized but even and diffuse harvesting can be achieved through multiple entry sites and criss-crossing passes; we graft the fat in a similar fashion. The goal is to deliver micro-ribbons of fat into separate planes, to avoid coalescence, and to maintain an organized pattern of grafting so that there is no under- or over-grafted site. Like a sprinkler system, the way to evenly and diffusely deliver specks over a broad expanse is to randomly spray out a fine mist through many sprinkler heads, rather than massively pouring a stream out of a few large-bore hoses. With this multiple fine sprinkler system arrangement, we achieve “evenness through randomness” ( Fig. 19.5 ).
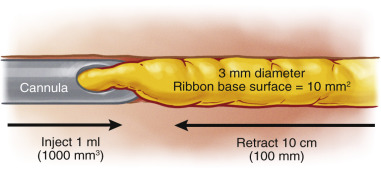
Graft delivery is of paramount importance and is the least studied component of AFT. If the surgeon injects the fat without moving the cannula, the result is a blob of fat. If, on the other hand the surgeon retracts the cannula over a large distance as he injects the same amount, the result is a thin ribbon. The size of the ribbon delivered is therefore a function of volume injected per distance of cannula travelled. Recognizing that 3 mm is the widest ribbon that can re-vascularize, we calculate its surface area to be approximately 10 mm 2 . Therefore, for each 1000 mm 3 (1 mL) delivered, the cannula has to retract in its channel for 100 mm or 10 cm. This is how we derive the cardinal rule of 10 cm of cannula excursion per 1 mL of fat graft delivery. Note that this rule applies regardless of the cannula diameter, the size of its hole, the pressure of injection, or the rate of injection in mL/sec ( Fig. 19.6 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree