Trigeminal anesthesia may yield blindness and facial disfigurement, secondary to neurotrophic keratopathy and trigeminal trophic syndrome. This article summarizes contemporary medical and emerging surgical approaches for the therapeutic management of this rare and devastating disease state.
Key points
- •
Trigeminal anesthesia may yield blindness and facial disfigurement.
- •
The pathophysiology of the tissue insult in trigeminal anesthesia comprises a loss of protective sensory feedback and loss of afferent neural trophic support.
- •
Mounting evidence supports a role for sensory nerve transfers in the management of trigeminal anesthesia.
Introduction
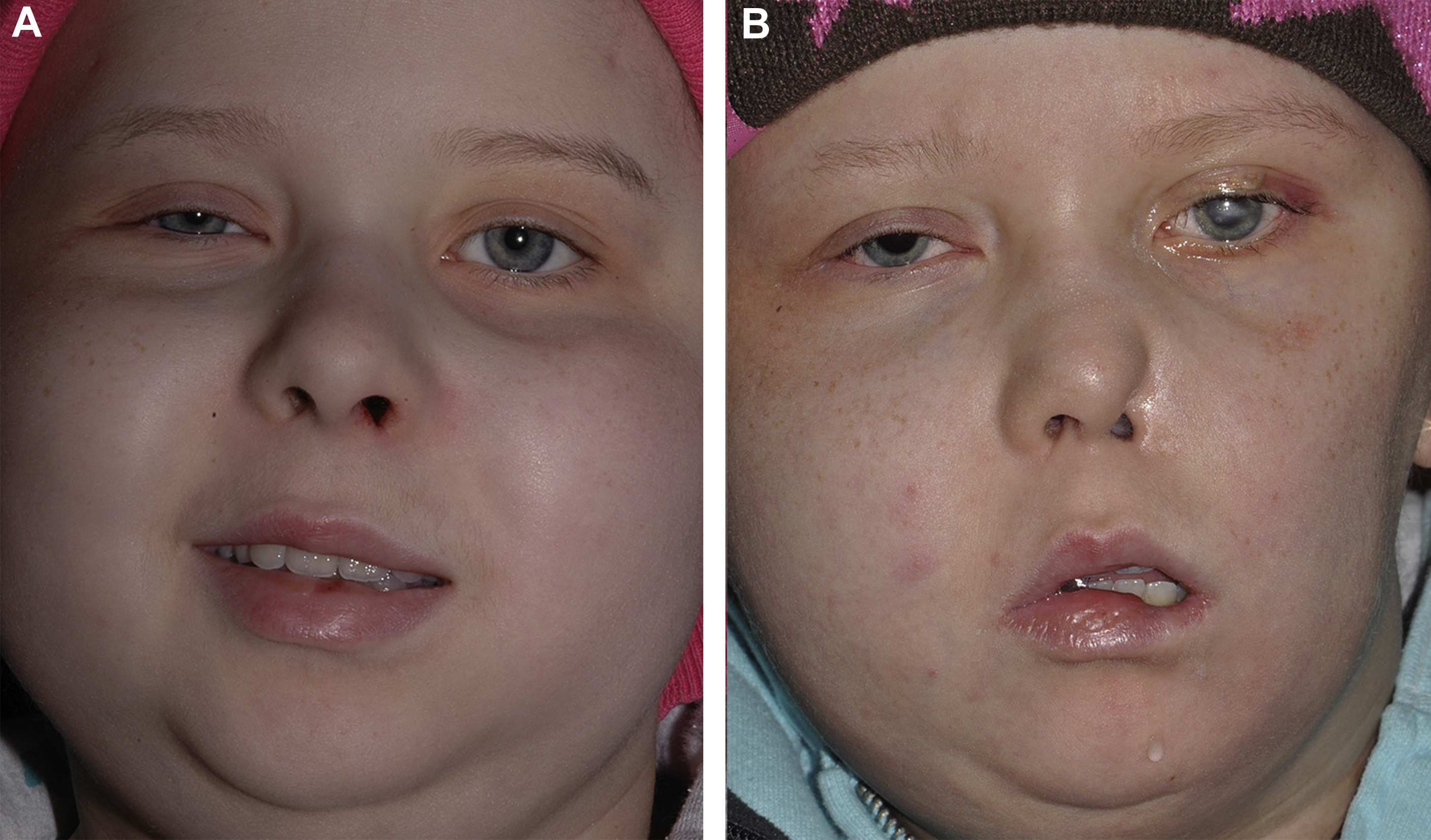
The somatosensory system is highly conserved in evolution. Touch is the first sensation to develop in humans and plays a pivotal role in normal growth and development. A congenital absence of or insult to the somatosensory fibers of the trigeminal nerve yields facial anesthesia. Trigeminal neuropathy may arise from viral or bacterial infection (eg, herpes zoster, leprosy, syphilis), neuroinflammatory disorders (eg, multiple sclerosis, sarcoidosis), rheumatologic disorders (eg, systemic lupus erythematosus, vasculitis), cerebrovascular arteriovenous malformations and infarcts, skull base tumors and their extirpation (eg, meningiomas, schwannomas), and trigeminal ganglion ablation for therapeutic management of trigeminal neuralgia. Beyond its potential role in the growth and development and sense of self, facial sensation is necessary for the protection and normal function of the ocular surface and facial soft tissues. Trigeminal anesthesia may cause corneal blindness, functional impairment with the oral phase of deglutition, and facial disfigurement ( Fig. 1 ).

Trigeminal afferent loss yields neurotrophic keratopathy, a degenerative disease of the ocular surface. The cornea and overlying tear film are key components of the ocular surface, and comprise the principal refractive optics of the visual system. Given its exposure, the ocular surface is prone to injury. Afferent neural input to the ocular surface provides critical regulatory and protective functions. The pathophysiology of neurotrophic keratopathy comprises a loss of sensory feedback and trophic support to the ocular surface. Sensory feedback regulates the protective blink and tearing reflexes in response to ocular foreign bodies and desiccation. Sensory branches of the trigeminal nerve also produce trophic neuropeptides that stimulate wound healing in response to injury and maintain the integrity of the ocular surface in the uninjured state. Neuromediators released from corneal nerves are manifold, and include substance P, calcitonin gene-related peptide, neuropeptide Y, brain natriuretic peptide, vasointestinal peptide, and acetylcholine. In turn, corneal epithelial cells and keratocytes regulate the survival and maturation of corneal nerve fibers by release of several neurotrophins and neuropeptides, including nerve growth factor, neutrohin-3 and neutrohin-4/5, brain-derived neurotrophic factor, and glial cell–derived neurotrophic factor. Inadequate healing and progressive ocular surface degeneration in neurotrophic keratopathy may occur in the absence of mechanical insult, owing to morphologic and metabolic disturbances resulting from loss of trigeminal afferent neural trophic support. ,
The clinical presentation of neurotrophic keratopathy ranges from subtle corneal surface irregularities to corneal melting or perforation ( Fig. 2 ). Vision loss in neurotrophic keratopathy is insidious; erosion of the ocular surface occurs is painless owing to absence of sensory feedback. The disease is typically graded in 3 stages according to Mackie’s classification. Stage I is characterized by hyperplasia or irregularity of the corneal epithelium, evolving to punctate keratopathy, corneal edema, neovascularization, and stromal scarring. Stage II is defined by recurrent or persistent epithelial defects. In stage III, stromal involvement leads to corneal ulceration, melting, and perforation. Concomitant facial paralysis and trigeminal anesthesia places patients at high risk of vision loss; paralytic lagophthalmos accelerates the progression of neurotrophic keratopathy.
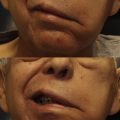
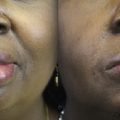
Beyond corneal blindness, trigeminal anesthesia places the facial soft tissues at risk of injury. Biting of the lip, cheek, and tongue on the affected side occurs without nociceptive feedback. Absent vermillion sensation increases the propensity for oral incompetence and social embarrassment as food debris on the lips goes unnoticed. A subset of patients with congenital or acquired trigeminal dysfunction will develop trigeminal trophic syndrome ( Fig. 3 ). , This devastating condition is believed to be the result of self-traumatization of insensate regions of the involved hemiface, knowingly or unknowingly, in response to paresthesiae. The onset of trigeminal trophic syndrome may occur weeks to years after trigeminal nerve injury, although typically within 1 to 2 years. The nasal base and nasal ala are commonly involved, and demonstrate crescenteric ulcerations. , Left untreated, the disease often results in complete destruction of the nasal ala and may also involve the lips, cheek, forehead, and orbit on the affected side. Concurrent neurotrophic keratopathy is typical.
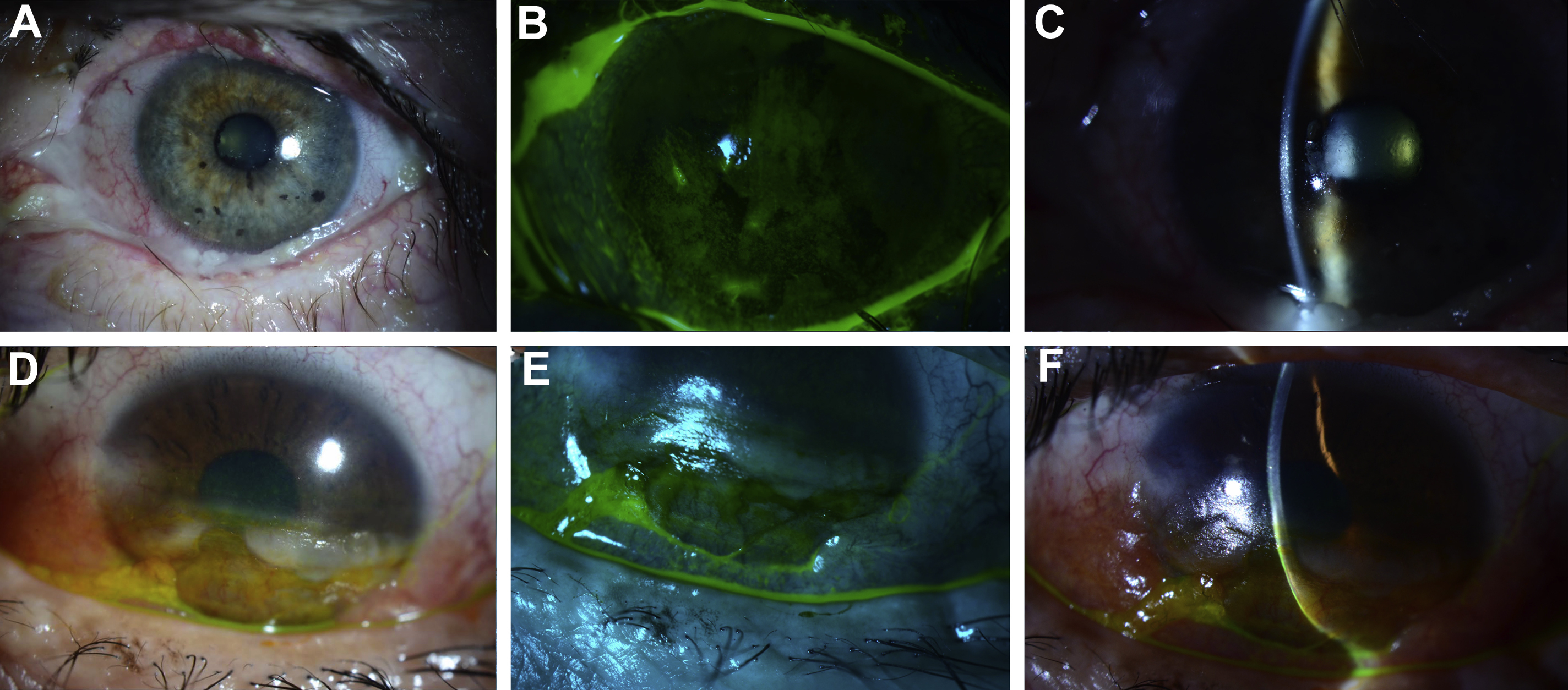
Therapeutic management of neurotrophic keratopathy
Medical Management
Therapeutic management of neurotrophic keratopathy aims to promote corneal healing while avoiding further insult to the ocular surface. Therapy of neurotrophic keratopathy depends on disease stage. In stage I disease, therapy aims to improve the integrity and transparency of the corneal epithelium and avoid further epithelial breakdown. Therapy involves the application of artificial tears during waking hours and gels or ointments overnight. Topical lubricants should be free of preservatives, because they may yield toxic or atopic keratoconjunctivitis. Topical autologous serum and therapeutic soft contact lenses may be used for persistent keratopathy.
In stage II disease, therapy aims to promote the healing of persistent epithelial defects and prevent ulceration. In addition to stage I treatments, medical therapies may include topical recombinant human nerve growth factor administration (eg, cenegermin), levator muscle chemodenervation, the judicious use of topical antibiotics and corticosteroids, and scleral lenses. Scleral lenses are gas-permeable rigid devices that lie on the sclera and vault over the cornea; prosthetic replacement of the ocular surface ecosystem devices are custom-tailored scleral lenses that may yield improved performance. The disadvantages of rigid lenses include a heightened risk of microbial keratopathy and corneal hypoxia, yielding neovascularization.
Therapy in stage III disease aims to promote ulcer healing and avoid perforation. In stage III, additional medical therapies may include topical N -acetylcysteine and systemic medroxyprogesterone or tetracycline in cases of corneal melt. , Surgical interventions are often indicated in stages II and III neurotrophic keratopathy owing to the limitations of medical therapy.
Surgical Management
Adjunctive surgical treatments
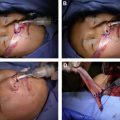
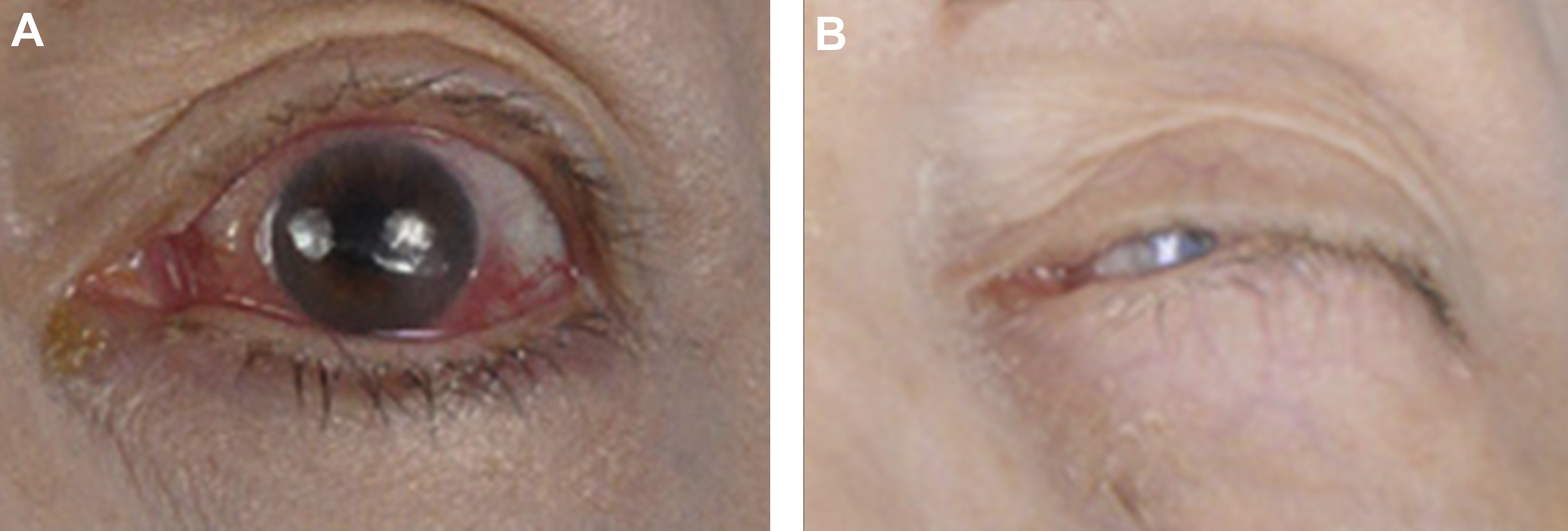
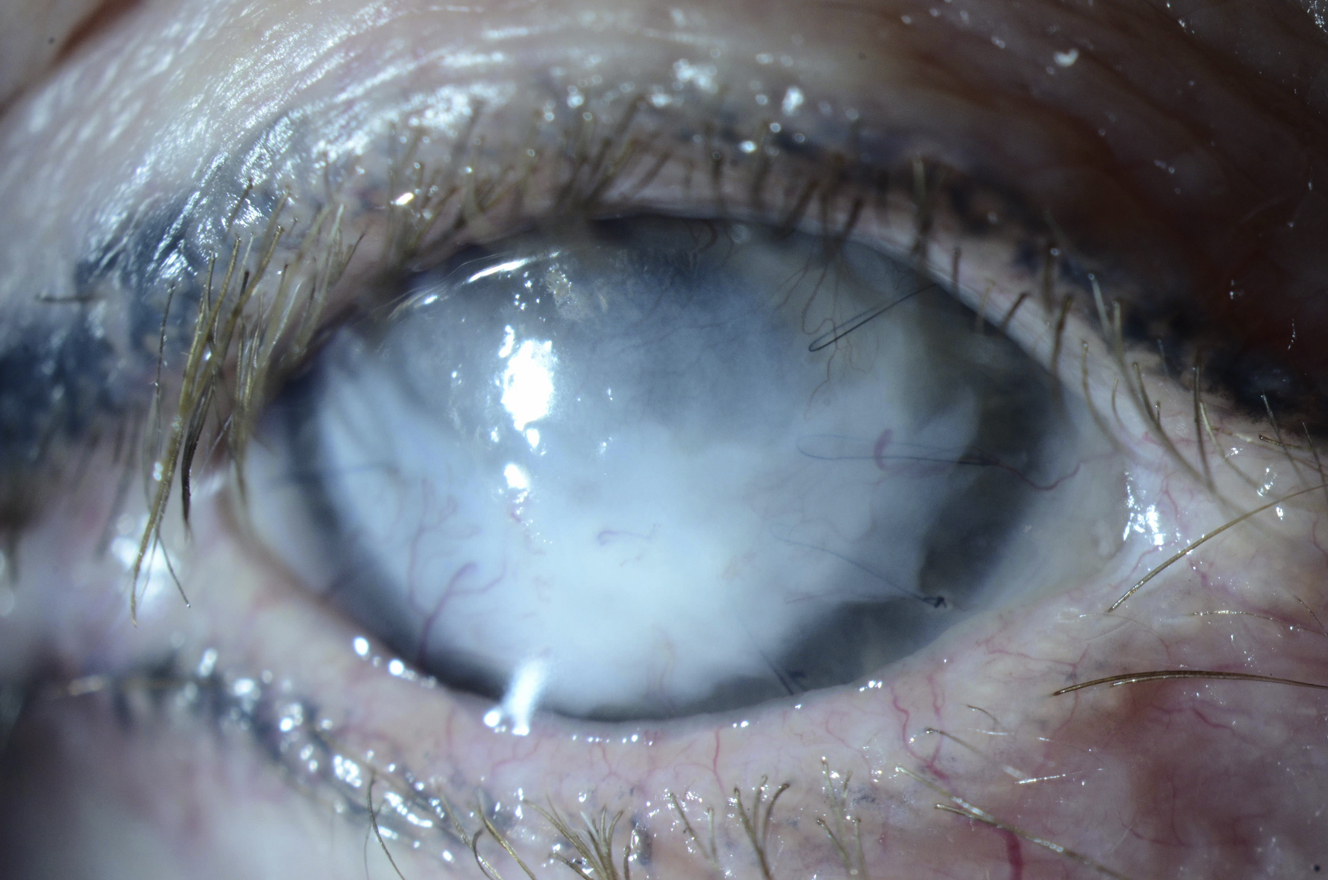
Surgical therapy of advanced neurotrophic keratopathy (stages II and III) is targeted at promoting healing of the ocular surface or replacing damaged or missing tissues. Decreasing ocular surface exposure through levator lengthening or tarsorrhaphy (lateral, medial, or central) establishes an optimal healing environment by minimizing desiccation and foreign body insult. The disadvantages of such procedures include poor aesthetics and decreased visual fields ( Fig. 4 ). Where conservative measures have failed, adhesive therapy (cyanoacrylate or fibrin glue), conjunctival flap surgery, amniotic membrane transplantation, and lamellar or penetrating keratoplasty may be indicated. Corneal transplantation carries a high risk of failure in neurotrophic keratopathy where sensory neural input has not been reestablished ( Fig. 5 ). When present, paralytic lagophthalmos should be addressed immediately using static periocular reanimation techniques and motor neurotization of the orbicularis oculi muscle where indicated.
Corneal neurotization
The management of neurotrophic keratopathy is challenging and costly. Although conventional therapies may halt disease progression and stimulate corneal healing in neurotrophic keratopathy, they do not address its underlying cause. In 2009, Terzis and colleagues demonstrated that the progression of neurotrophic keratopathy could be reversed via by surgical transfer of contralateral supraorbital and supratrochlear nerve branches to the affected side perilimbal region. Widespread adaptation of this remarkable technique was initially hindered by its donor site morbidity and technical challenge, requiring a bicoronal incision and meticulous dissection of the terminal sensory nerve branches yielding anesthesia of the contralateral forehead. International interest in the approach was sparked when Elbaz and colleagues and Bains and associates described the use of an interposition nerve autograft between the cornea and donor supratrochlear nerve to decrease procedural morbidity.
Indications
Indications for corneal neurotization have not been established definitively. In cases of acquired trigeminal anesthesia, there exists the potential for the spontaneous recovery of sensory function where the continuity of corneal nerve branches with the brainstem and cortex exists. Examples include recent-onset neurotrophic keratopathy secondary to herpes keratopathy and skull base surgery where anatomic continuity of the trigeminal nerve was preserved. In such cases, watchful waiting with conservative medical and surgical measures to protect the ocular surface should be considered for at least 1 year. Indications for corneal neurotization are clear in cases of advanced neurotrophic keratopathy (stages II and III) refractory to conservative measures where corneal anesthesia is complete and long standing. Because patients with corneal anesthesia are at constant risk of rapid progression of neurotrophic keratopathy yielding corneal blindness, consideration of corneal neurotization is appropriate in all cases of corneal anesthesia without the potential for sensory recovery. In cases of long-standing corneal hypoesthesia (ie, some preserved sensation to the ocular surface), neurotization may be considered for neurotrophic keratopathy refractive to conservative measures. Corneal neurotization may yield particular benefit in patients with advanced cornea opacification, wherein the goal is to render them candidates for second-stage corneal transplant for vision restoration.
It is unknown whether corneal neurotization procedures impact the recovery of native corneal nerve function after a transient insult, although emerging data suggest alternate neurotization of the ocular surface may provide trophic support that enhances remnant corneal nerve function in cases of corneal hypoesthesia. Anecdotal evidence suggests that nerve transfers may unmask remnant corneal nerve function in select cases. In our own experience, we have noted at least 1 patient with long-standing corneal anesthesia who recovered sensation localizing to the ocular surface in addition to the native dermatome of the donor nerve after corneal neurotization.
Approaches
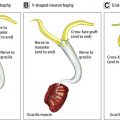
Corneal neurotization approaches vary according to donor nerve source, use and type of interposition graft, and the methods to secure the transferred nerve fascicles to the ocular surface. Donor nerve options in corneal neurotization procedures are dictated by the pattern of sensory loss noted on meticulous physical examination. Potential donor nerves include the contralateral supratrochlear, supraorbital branches, and ipsilateral great auricular nerve branches. In select cases, the ipsilateral supratrochlear, supraorbital, or infraorbital branches of the trigeminal nerve may be suitable for use. Corneal neurotization may be achieved via direct transposition of the distal segments of donor nerves or via the use of an interposition graft to bridge the gap between the donor neurotization source and the ocular surface. Owing to their length, ease of harvest, and low morbidity, sural nerve autografts are used commonly. The use of acellular nerve allografts in place of nerve autografts carries an increased risk of suboptimal neurotization in graft lengths exceeding 3 to 4 cm, yet several centers have used them for corneal neurotization. Transferred nerve fascicles are routed from the facial soft tissues to the subconjunctival space; they may be secured to the perilimbal sclera using microsutures or alternatively placed directly within the corneal stroma via scleral–corneal tunnels and secured using fibrin glue.
Corneal neurotization procedures carry a favorable safety profile. In their pioneering work using a bicoronal approach, Terzis and colleagues reported 1 subgaleal hematoma requiring drainage, and 1 asymptomatic subconjunctival neuroma requiring no treatment. Using an interposition graft to bridge the fibers of the contralateral supratrochlear nerve to the perilimbal region, Catapano and colleagues reported conjunctival suture exposure in several patients where nylon sutures were used. Postoperative persistence of epithelial defects was also noted in several patients, all resolving with bandage contact lens therapy. No autograft donor site morbidity was encountered. In our center’s experience (8 cases to date), we noted 1 case of persistent focal conjunctival chemosis that resolved with topical steroids and simple excision.
Outcome measures
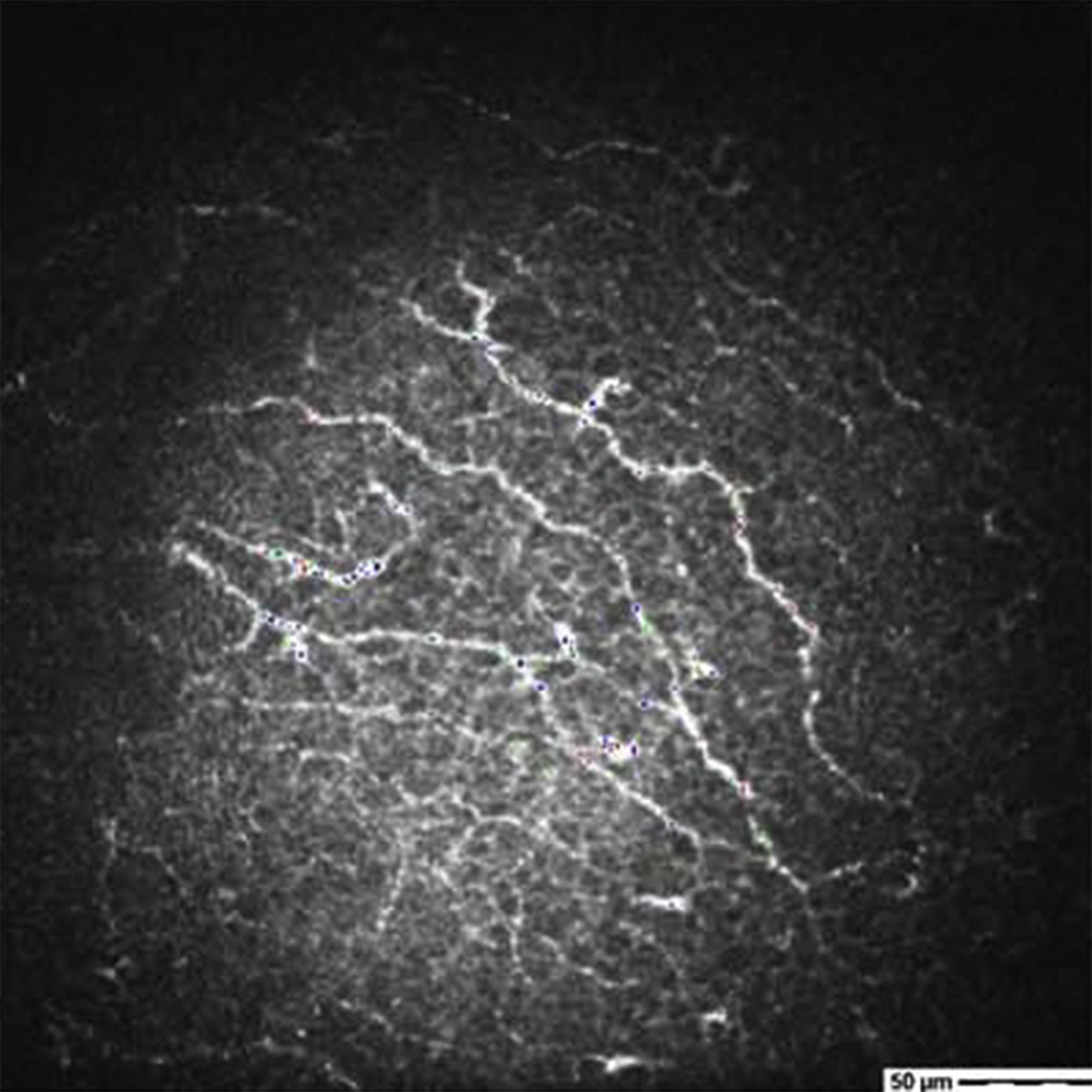
Clinical and research outcome measures in corneal neurotization are manifold. Clinical photography with slit-lamp examination with and without fluorescein together with assessment of visual acuity is regularly performed. Corneal sensation (native and referred) may be quantified and longitudinally tracked using Cochet–Bonnet esthesiometry. In vivo corneal confocal microscopy may be used to visualize regenerating nerve fibers within the cornea ( Fig. 6 ). Pachymetry may be used to assess corneal thickness. The histopathologic examination of explanted tissue obtained during subsequent corneal transplant procedures may confirm the presence of nerve fibers. Noninvasive functional imaging of the brain during tactile stimulation of the ocular surface has been used in research settings to confirm transferred nerves yield sensation.