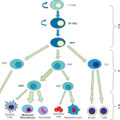
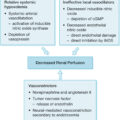
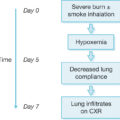
Introduction
Burn injury is the fourth most common type of traumatic injury worldwide, following traffic collisions, falls, and interpersonal violence. , Pediatric burn injury is among the top 10 causes of unintentional injury and death in children. Massive burn injuries in children require a very high number of surgical procedures and hospital resources. The pattern of burn injuries affecting children differs from the pattern observed in adult burns.
In the pediatric population, scald burn is the most frequent mechanism of burn (42%) followed by flame (29%) and contact burn (10%). Thermal burns (scald and contact burns) make up most pediatric burn injuries, especially in children 6 years old or younger, with the majority measuring less than 10% total body surface area (TBSA), whereas flame burns are seen more often in adolescents. , Scald burns are attributable to hot liquids related to cooking, including spilling hot coffee or water, or to children reaching up to countertops, pulling pot handles or cords attached to cooking appliances, and spilling the contents onto themselves. Other causes include unknowingly putting body parts under a hot water faucet or climbing into a bathtub with hot water and intentionally or unintentionally being placed into or brought in contact with a hot substance by another individual.
Children with severe burns have higher mortality than nonelderly adults with similar burns, and the mortality increases with higher TBSA ( Figs. 29.1 and 29.2 ). Drastic changes in burn care over the past decades, which include aggressive and early fluid resuscitation, timely excision and grafting of burn wounds, improved infection control, modulation of the hypermetabolic response, and management of inhalation injury, have promoted a substantial decline in mortality after burn injury. ,
Mortality in burns of more than 80% total body surface area for various ages.
Mortality for increasing burn size.
%TBSA , Percentage of total body surface area.
In addition to biologic derangements, a severe burn produces tremendous psychological challenges to a child. Long-term quality of life is considerably affected. , Patients with large burns are at risk for developing extensive scarring, contractures with restricted movement, itch, pain, and decreased satisfaction with their appearance in the long term.
Anatomic and psychological differences specific to the pediatric population make procedures and interventions more challenging, and there is less room for error compared with the adult patients. The burn care process can be divided into four phases: initial evaluation and resuscitation, initial wound excision and temporary closure, definitive wound closure, and rehabilitation and reconstruction.
Infants
Infants can be considered children anywhere from birth to 1 year old. Several studies have focused on burn injuries in infants. Children younger than 1 year are a specific age group in which they begin to develop mobility, starting from rolling, crawling, and finally walking around the age of 1 year. First-born 9- to 12-month-old males of young mothers of low socioeconomic status are at higher risk of burn injuries. Laitakari et al. observed that burn injuries in this age group are most often witnessed and take place while the infant is being held at the same time as the hot item. Scalds are the most common type of burn in this age group, followed by contact with household objects. Purcell et al. observed that factors that increase infant burn mortality risk include %TBSA burn, flame burn, and lack of operative intervention; Lorch et al. found that increased severity of burn, concern for abuse, and burn to the hand are associated with increased chances of admission.
When deep debridement or surgical intervention is indicated in the treatment of burns in infants, general anesthesia is essential. In fact, due to their developmental stage, infants are unable to cooperate with procedures that are painful or prolonged, and local or regional anesthesia alone is typically insufficient to ensure adequate analgesia and immobility. General anesthesia not only facilitates a controlled surgical environment but also minimizes physiological stress and potential psychological trauma. Careful anesthetic management is crucial, given the unique vulnerabilities of this age group, including immature organ systems, limited physiological reserves, and increased sensitivity to fluid shifts and blood loss.
General anesthesia in pediatric patients is common, such that 15% of children in the United States undergo general anesthesia before 3 years of age. Some data are available in the current medical literature regarding the vulnerability of a young developing brain exposed to anesthetics and potential neurotoxic substances. Furthermore, warnings about using general anesthetics and sedation drugs in young children have been published by the US Food and Drug Administration in 2016 and 2018.
However, although general anesthetics have the potential to induce neurotoxicity, very little clinical evidence exists to support this. In fact, there is strong evidence that brief exposure is not associated with any long-term risk in humans, and the risk is greater with multiple exposures.
Initial evaluation
The initial management of children affected by burn injuries is of paramount importance. A retrospective study of children with burn injuries 50% TBSA or greater who were admitted to Shriners Hospitals for Children–Northern California showed that most children who died as a result of their massive burn injury died within the first month of admission. Prognosis of burned child is heavily influenced by how they are treated in the first 24 hours.
First, the burn patient needs to be removed from the source of thermal injury expeditiously and clothing and jewelry removed immediately. The first-aid principles in the management of burns are analgesia, cooling, and covering. After the burning process is stopped, the burns should be cooled for 20 minutes with cool running water. , Appropriate cooling therapy can reduce local thermal damage, decrease edema, inhibit the release of oxygen-free radicals, reduce inflammation, and effectively improve local wound microcirculation.
The patient should be kept warm by covering with a sterile (if available) or clean sheet or blanket. If the burn is chemical, the patient should be removed from the chemical immediately, and the burn s hould be irrigated with copious amounts of water for at least 30 minutes. If the chemical is powder, it should be brushed off before irrigation.
The immediate focus of management of pediatric burn patients includes resuscitation and stabilization, fluid management, and pain control.
Burn patients should be assumed to be trauma patients, and any potential life-threatening injuries should be identified and treated. All large burns (>10% TBSA) should undergo Advanced Trauma Life Support evaluation if traumatic injury is suspected. The airway should be assessed first. If inhalation injury is suspected, then 100% oxygen should be administered. In addition, arterial blood gas and carboxyhemoglobin levels should be measured. Pulse oximetry readings will be falsely normal in patients with elevated carboxyhemoglobin levels because carboxyhemoglobin is read as oxyhemoglobin by the pulse oximeter.
Tachypnea, stridor, and hoarseness indicate an impending airway narrowing caused by inhalation injury or edema, and immediate intubation should be considered. A full-thickness circumferential chest burn can interfere with ventilation. Chest expansion should be observed to ensure adequate air movement. If the patient is on a ventilator, airway pressure and Pa co 2 should be monitored. If ventilation is compromised, escharotomy of the chest should be considered to improve ventilation.
Cooling and covering the burn have an analgesic effect, but pharmacologic therapy should be given to alleviate the pain. The most common medications used for this purpose are acetaminophen, ibuprofen, hydrocodone, fentanyl, and morphine.
Blood pressure measurement may be difficult in patients with burned extremities. These patients may require an arterial line to monitor their blood pressure. A radial arterial line in pediatric patients with extremity burns may be difficult to obtain. A femoral arterial line may be more reliable and easier to secure.
A urinary drainage catheter is placed to monitor urine output. Placement of a nasogastric tube is recommended in patients with major burns as they can develop gastric ileus. Accurate and prompt determination of burn size is fundamental for the proper management of burn injury.
Burn intensive care unit (ICU) admission should be considered for patients with major burn injuries (>10% for children). Other criteria for ICU admission are inhalational injury with obvious or potential airway involvement, altered mental status or shock, circumferential burns compromising respiration or limb perfusion, impending or known compartment syndrome of limbs or abdomen, evidence of evolving organ dysfunction, or associated major trauma.
Resuscitation
In pediatric burns, fluid resuscitation is needed for burns greater than or equal to 15% TBSA compared with burns greater than or equal to 20% TBSA for adults. There is a systemic capillary leak after a major burn, which increases with the burn size. Delays in commencement of resuscitation of burned patients result in worse outcome ( Fig. 29.3 ). Therefore it is vital that IV access is obtained early. Because of the small circulating volume in children, resuscitation should be started immediately; even the short delays increase the risk of shock. Peripheral IV access is preferred, and it may go through the burned skin if necessary. An IV access should be well secured. Children with large burns should have two large-bore IV lines. The presence of two IV lines provides a safety margin if one stops functioning. When a peripheral IV access is not available because of severe extremity burns, a central venous line is placed.
Time to intravenous access in survivors and nonsurvivors.
Mortality increases with delays in starting an intravenous line and instituting volume resuscitation.
When a vascular access is unobtainable, the intraosseous route is a viable option. Fluid volumes in excess of 100 mL/hour can be administered directly into the bone marrow. Intramedullary access can be utilized in the proximal tibia. A 16- to 18-gauge bone marrow aspiration needle, spinal needle, or commercially available intraosseous needle can be used to cannulate the bone marrow compartment. Although previously advocated only for children younger than 3 years of age, intraosseous fluid administration can be safely performed in all pediatric age groups. The proximal anterior tibia, medial malleolus, anterior iliac crest, and distal femur are preferred sites for intraosseous infusion. The needle should be introduced into the bone, avoiding the epiphysis, either perpendicular to the bone or at a 60-degree angle, with the bevel facing the greater length of the bone ( Fig. 29.4 ). The needle has been properly inserted when bone marrow can be freely aspirated. Fluid should be allowed to infuse by gravity drip. The use of pumps should be discouraged in case the needle becomes dislodged from the marrow compartment.
Intraosseous line placement in the proximal tibia (A) and distal femur (B).
(From Fleisher G, Ludwig S, eds. Textbook of Pediatric Emergency Medicine . 2nd ed. Baltimore: Williams & Wilkins; 1988:268.)
Fluid losses are proportionally greater in children owing to their small body weight-to-body surface area ratio. Normal blood volume in children is approximately 80 mL/kg body weight and in neonates 85 to 90 mL/kg, compared with an adult whose normal blood volume is 70 mL/kg. The commonly used rule of nines, useful in adults and adequate in adolescents, does not accurately reflect the burned body surface area of children under 15 years of age ( Fig. 29.5 ). The standard relationships between body surface area and weight in adults do not apply to children because infants possess a larger cranial surface area and a smaller surface area in the extremities than adults.
The rule of nines altered for the anthropomorphic differences of infancy and childhood.
Depending on the burn center where the patient is treated, there are differences in the TBSA burn size at which fluid resuscitation is initiated, coefficients related to the Parkland formula, criteria to initiate dextrose-containing fluids, and urine output goals. The initial fluid rates for patients with visibly large burns are determined by patient age (≤5 years old: 125 mL lactated Ringers [LR]/hour; 6–13 years old: 250 mL LR/hour; ≥14 years: 500 mL LR/hour).
Formulas can be used as a starting point, but further fluid therapy should be adjusted to the actual need of an individual patient. The American Burn Association (ABA) recommends calculation of resuscitation volume for the first 24 hours as 2 mL/kg/%TBSA for adults and 3 mL/kg/%TBSA for children less than 14 years of age. In addition, it is recommended that children less than 30 kg should also receive 5% dextrose in LR at standard maintenance rate.
Fluid resuscitation of burn patients is commonly initiated using modified Brooke or Parkland formula, but the use in children of the most common resuscitation formulas (initially designed for the adult population) can easily result in suboptimal resuscitation ( Table 29.1 ).
Table 29.1
Resuscitation by the Parkland Formula Only Compared with Maintenance Fluid Requirements Alone.
|
CALCULATED NEEDS
|
REPLACEMENT BURN LOSS | ||||
|---|---|---|---|---|---|
| Example | % Burn | Resuscitation | Maintenance | mL | mL/kg/% |
| 1 yr old | 15 | 600 | 800 | −200 | −1.33 |
| 10 kg | 30 | 1200 | 800 | 400 | 1.33 |
| 0.48 m 2 BSA | 60 | 2400 | 800 | 1600 | 2.67 |
| 90 | 3600 | 800 | 2800 | 3.11 | |
| 4 yr old | 15 | 990 | 1200 | −210 | −0.85 |
| 16.5 kg | 30 | 1980 | 1200 | 780 | 1.58 |
| 0.68 m 2 BSA | 60 | 3900 | 1200 | 2760 | 2.79 |
| 90 | 5940 | 1200 | 4940 | 3.33 | |
| 12 yr old | 15 | 2400 | 2250 | 1150 | 1.92 |
| 40 kg | 30 | 4800 | 2550 | 2550 | 2.12 |
| 1.13 m 2 BSA | 60 | 9600 | 2250 | 7350 | 3.06 |
| 90 | 14,400 | 2250 | 12,150 | 3.38 | |
BSA, Body surface area.
Pediatric burn patients should therefore be resuscitated using formulas based on body surface area, which can be calculated from height and weight using a standard nomogram ( Fig. 29.6 ) or formulas ( Table 29.2 ). The commonly used resuscitation formula in pediatric patients calls for the administration of 5000 mL/m 2 TBSA burned plus 2000 mL/m 2 TBSA for maintenance fluid given over the first 24 hours after burn, with half the volume administered during the initial 8 hours and the second half given over the following 16 hours. Early and appropriate fluid resuscitation of burn injury is critical to prevent the consequences of burn shock. The subsequent 24 hours and for the rest of the time the burn wound is open, the requirement is 3750 mL/m 2 TBSA burned or remaining open area plus 1500 mL/m 2 TBSA for maintenance fluid. The fluid requirement decreases as a patient achieves more wound healing. As in the adult patient, resuscitation formulas offer a guidance for the amount of fluid necessary for replacing lost volume, and the amount of fluid should be titrated according to the patient’s response. Hyponatremia is a frequently observed complication in pediatric patients in the first 48 hours after burn. Hypernatremia has been identified as an independent predictor of mortality in adult burn patients, , and is associated with poor outcomes in critically ill burn patients. Risk factors for hypernatremia in burn patients are inhalation injury, mechanical ventilation, and IV fosfomycin and colistimethate sodium. Furthermore, hypernatremia was associated with longer length of hospital stay and higher mortality and is considered to be markedly disadvantageous for the survival of skin grafts. ,
Standard nomogram for the determination of body surface area based on height and weight.
The example depicted is for a child of 100 cm in height and 23 kg in weight.
(From Eichelberger MR, ed. Pediatric Trauma: Prevention, Acute Care and Rehabilitation . St. Louis: Mosby Year Book; 1993:572.)
Table 29.2
Formulas for Calculating Body Surface Area (BSA).
| Dubois formula | BSA (m 2 ) = height (cm) 0.725 × weight (kg) 0.425 × 0.007184 |
| Jacobson formula | BSA (m 2 ) = [height (cm) + weight (kg) − 60]/100 |
Potassium losses are usually replaced with oral potassium phosphate rather than potassium chloride, as hypophosphatemia is frequent in this population. , Serum phosphate concentrations in pediatric burn patients are associated with the magnitude of inflammatory response. Hypophosphatemia can cause cardiac, hematologic, immunologic, and neuromuscular dysfunction and it was associated with decreased probability of ICU discharge and increased time on mechanical ventilation.
Calcium and magnesium losses also must be supplemented. A recent study observed that major burns (>20% TBSA) are associated with increased serum levels of parathyroid hormone and decreased serum levels of vitamin D, but serum levels of calcium are not affected by major burns. IV resuscitation fluid should be isotonic. LR solution is the most commonly used resuscitation solution for the first 24 hours after burn. Children less than 1 year of age should also receive a separate maintenance dextrose fluid solution to prevent hypoglycemia because their glycogen stores are limited.
Assessment of resuscitation
The routine clinical signs of hypovolemia in adult burn patients, such as low blood pressure and decreased urine output, are late manifestations of shock in the pediatric patient, and tachycardia is ubiquitous. , , Because of their cardiopulmonary physiologic reserve, pediatric patients do not show overt signs of hypovolemia until a decrease of at least 25% of the total blood volume occurs; at this point, hemodynamic decompensation occurs abruptly. Changes in distal extremity color, capillary refill, pulse pressure, and mental status reflect volume status. Capillary refill is a good indicator of volume status in pediatric patients. Decreased capillary refill should warn a clinician of imminent cardiovascular collapse. Measurements of arterial pH, base deficit, and lactic acid are of particular importance in this age group, reflecting decreased tissue perfusion. Improvements in base deficit or lactic acid demonstrate successful resuscitation.
Normal blood pressure range varies according to age, with a systolic blood pressure of 100 mmHg or less considered normal in patients younger than 9 years ( Table 29.3 ). Renal compensatory mechanisms for hypovolemia (e.g., tubular concentration) are not well developed in young children, contributing to hypovolemia with sustained urine production despite reduced intravascular volume.
Table 29.3
Normal Pediatric Vital Signs.
| Age | Minimum Heart Rate (beats/min) | Systolic Blood Pressure (mmHg) | Respirations (breaths/min) | Minimal Hemoglobin (g/dL) | Minimal Hematocrit (%) |
|---|---|---|---|---|---|
| <2 yr | 100–160 | 86–106 | 30–40 | 11.0 | 33.0 |
| 2–5 yr | 80–140 | 89–112 | 20–30 | 11.0 | 33.0 |
| 6–9 yr | 70–120 | 97–115 | 18–25 | 11.5 | 34–35 |
| 9–12 yr | 70–115 | 102–120 | 18–25 | 11.5 | 34–35 |
| >12 yr | 60–110 | 90 | 12–20 | 12.0 | 36.0 |
An indwelling urinary drainage catheter is essential for burns greater than 20% during resuscitation. During the early phase of resuscitation, urine output should be assessed hourly, and the resuscitation fluid adjusted appropriately. Fluid administration should be titrated to achieve a urine output of 1 mL/kg/hour in children and 2 mL/kg/hour in infants. If needed, initial fluid boluses should be administered in amounts appropriate to the size of the child and should be less than 25% of the total blood volume (20 mL/kg). Overresuscitation is a common occurrence because many burn centers routinely administer fluids greater than predicted by the Parkland formula ; this must be avoided because it can lead to congestive heart failure, pulmonary edema, abdominal and extremity compartment syndromes, and cerebral edema in burn patients. In children, cardiac output depends mainly on the heart rate caused by the low compliance of the heart, which limits increase in stroke volume. In addition, the heart is more susceptible to volume overload. Transthoracic echocardiograms should be used early to assess cardiac function in patients who are not responding to conventional therapy. Children are particularly prone to the development of edema from both vasogenic and hydrostatic sources. Vasogenic edema occurs in the early postburn period when vascular integrity is impaired. The maintenance of intravascular osmotic pressures reduces the likelihood of edema development. In difficult resuscitations, colloids such as albumin can be used. Albumin can be expected to remain in the intravascular space if administered more than 8 hours after burn.
Deresuscitation should be considered in patients with clear evidence of fluid overload. Fluid restriction and diuretics should be considered in patients with fluid overload after the resuscitation.
Evaluation and management of airways
Airway evaluation and management must be given priority. It is important to stabilize the airway first. Look for signs of inhalational injury (such as soot in nares or mouth) or respiratory distress. Mucosal swelling after an inhalation injury will cause stridor and/or hoarseness and respiratory distress. Children are more prone to obstruction caused by the smaller aperture of their trachea. Airway edema causes disproportionate increments in resistance with concomitant reduction of the cross-sectional area. Inhalation injury can lead to delayed airway edema after administration of massive resuscitation fluids.
Potential hemorrhage and edema make emergency intubation difficult. Hence, early intubation should be considered when a long transfer is anticipated, severe inhalation injury is present, or a patient has a large burn that likely will develop airway edema secondary to the large amount of fluid resuscitation. Concurrent placement of an endotracheal tube (ETT) over the bronchoscope should be considered at the time of bronchoscopy if bronchoscopy is performed for inhalation injury. A readily available estimate of airway diameter is the width of the patient’s little finger, an age-based formula (age + 16)/4, or the use of Broselow tape.
The ETT must be well secured. In a child, burn wounds and moist dressings can make securing ETT difficult. One successful approach is to attach the ETT with tape around the back of the head, both above and below the ears. An additional piece of tape over the top of the head, secured to the tape behind the head, will prevent accidental extubation in most children. In addition, commercially available ETT holders exist; however, securing the tubes with tape strips of sufficient length and width has proved superior to two different commercial tube holders.
Inhalation injury
Inhalation injury is one of the main contributors of burn mortality. , The mortality rate of children with isolated thermal burns is 1% to 2% but increases to approximately 40% in the presence of severe inhalation injury. , Inhalation injury independently increases the mortality in pediatric patients with a massive burn injury. Expectedly, inhalation injury is associated with longer hospital stay and increased risk of pneumonia. Carbon monoxide poisoning coupled with hypoxia is the most frequent cause of death caused by inhalation injury. Any patient with a flame-related injury, particularly if confined in a closed space, should be evaluated for inhalation injury. If inhalation injury is suspected, arterial blood gas and carboxyhemoglobin levels should be obtained, and the patient should be placed on 100% oxygen. Signs of potential inhalation injury include facial burns, singed nasal vibrissae, carbonaceous sputum, abnormal mental status (agitation or stupor), respiratory distress (dyspnea, stridor, hoarseness, wheezing), or an elevated carboxyhemoglobin level of greater than 10%, especially in a closed-space fire. The initial carboxyhemoglobin level should be calculated from the time the admission level is drawn, back to the time of the burn injury. A carboxyhemoglobin level of greater than 60% has a greater than 50% chance of mortality. Diagnosis is made by visualization of the airway with fiberoptic bronchoscopy ( Table 29.4 ).
Table 29.4
Carbon Monoxide Poisoning.
| Carboxyhemoglobin (%) | Symptoms |
|---|---|
| 0–10 | Normal |
| 10–20 | Headache, confusion |
| 20–40 | Disorientation, fatigue, nausea, visual changes |
| 40–60 | Hallucination, combativeness, convulsion, coma, shock state |
| 60–70 | Coma, convulsions, weak respiration and pulse |
| 70–80 | Decreasing respiration and stopping |
| 80–90 | Death in <1 h |
| 90–100 | Death within a few minutes |
Treatment modalities for inhalation injury include airway maintenance, secretion clearance, and pharmacologic management ( Table 29.5 ). Further care is mainly supportive and includes ventilator support as needed, vigorous pulmonary toilet, humidification of inspired air, and antibiotics for documented infection. A combination of nebulized heparin and N-acetylcysteine has been shown to decrease the duration of mechanical ventilation after inhalation injury.
Table 29.5
Airway Maintenance, Clearance, and Pharmacologic Management.
| Turn side to side | Every 2 h |
| Sitting or rocked in chair | As soon as physiologically stable |
| Ambulation | Early |
| Chest physiotherapy | Every 2 h |
| Suctioning and lavage (nasal/oral tracheal) | Every 2 h |
| Bronchodilators | Every 2 h |
| Aerosolized heparin/acetylcysteine | Every 2 h alternating |
| Heparin 5000–10,000 units with 3 mL NS | Every 4 h |
| Alternated with acetylcysteine 20% 3 mL | Every 4 h |
NS, Normal Saline
Hypermetabolism
Profound hypermetabolism is a classic feature of children with large burn injury. No other pathologic state produces as dramatic an effect on the metabolic rate as burn injury, defined as an elevated resting energy expenditure (REE) above 110% of predicted REE. Hypermetabolism slows wound healing, prolongs generalized weakness, and leads to loss of lean body mass.
Very early alterations at the mitochondrial level largely explain the hypermetabolic response, with a diminished coupling of oxygen consumption and adenosine triphosphate production.
The hypermetabolic response increases with increasing burn size and can last up to 24 months. There is an upregulation of catabolic agents, such as catecholamine, cortisol, and glucagon, which induces a hyperdynamic cardiovascular response; elevated oxygen consumption; increased energy expenditure; proteolysis, lipolysis, and glycogenolysis; loss of lean body mass; delayed wound healing; and immune suppression.
Pharmacologic agents have been used to attenuate hypermetabolism in burn injury. To minimize loss of lean body mass, several therapeutic agents have been used in pediatric burn patients: anabolic hormones, such as recombinant human growth hormone, insulin, and insulin-like growth factor 1; anabolic steroids, such as testosterone and synthetic analogue oxandrolone; and adrenergic antagonists, such as propranolol.
Thermoregulation
Approximately 2 to 5 days after major burn injury, a hyperdynamic and hypermetabolic state develops. Hypothalamic dysregulation induced by various inflammatory cytokines causes elevation of core body temperature after a major burn even in the absence of infection. Burn patients strive for temperatures of around 38°C. Low temperature is more likely indicative of overwhelming sepsis or exhausted physiologic capabilities. The augmented heat loss secondary to epidermal loss after a major burn makes conventional methods of heat conservation inappropriate in burn patients. In addition, low muscle mass in young children and their limited shivering capacity are factors that increase the risk of hypothermia in this population.
Every effort should be made to reduce heat loss. Environmental temperature should be maintained at 30°C to 33°C to reduce energy demands and evaporative water losses. Bathing and wound care should be performed expeditiously with avoidance of unnecessary environmental exposure.
Hypothermia produces numerous consequences. The heart is particularly sensitive to temperature, and ventricular arrhythmias are not uncommon. Hypothermia also increases the susceptibility of the myocardium to changes in electrolyte concentrations. The oxyhemoglobin dissociation curve is shifted to the left by decreased body temperature, impairing peripheral oxygenation. In extreme cases, hypothermia produces central nervous system and respiratory depression, coagulopathy, and loss of peripheral vasomotor tone.
Nutritional support
Nutritional support is an essential part of management of acute burn patients. Early enteral nutrition is used to accomplish nutritional support of the hypermetabolic response in patients with severe burns. When enteral nutrition starts early, it preserves gut mucosal integrity, improves intestinal blood flow and motility, can abate the hypermetabolic response to burn, decreases underfeeding risk, and reduces ICU length of stay. Shahi et al. recommend protocols to institute feeding for patients with burns of more than 10% TBSA within 4 hours of admission in pediatric patients. Patients with smaller burns should receive a high-protein, high-calorie diet to support their metabolic response. Large burn injuries result in a hypermetabolic state, with increases in energy expenditure up to 130% to 140% above predicted values and can result in protein catabolism and weight loss for up to 2 years after injury. Those with a large burn that covers more than 30% TBSA benefit from enteral feedings to supplement their diet. Enteral feedings can be given through a nasoduodenal or nasojejunal feeding tube, bypassing the stomach and enteral nutrition initiated within a few hours of admission. Most children will tolerate enteral feedings as early as 1 to 2 hours after burn. Several studies have demonstrated the efficacy of early alimentation and its additional salutary effects. Several formulas are available to estimate caloric requirements in burn patients. Because caloric demands are related to burn size, caloric support should be given in amounts calculated based on total and burned body surface areas in burned children. A series of different formulas based on body surface areas has been developed to meet the differing requirements of the various age groups. The Curreri formula has likewise been amended to reflect the differing demands of the pediatric group ( Table 29.6 ).
Table 29.6
Two common formulas used to calculate daily caloric needs of pediatric burn patients. TBSA total body surface area.
|
Galveston formula |
||
| 0–1 year | 2100 kcal/m 2 body surface area + 1000 kcal/m 2 TBSA | Focuses on maintaining body weight |
| 1–11 year | 1800 kcal/m 2 body surface area) + 1300 kcal/m 2 TBSA | |
| 12–18 years | 1500 kcal/m 2 body surface area + 1500 kcal/m 2 TBSA | |
| Modified Curreri formula (Curreri junior) | ||
| <1 year | Recommended dietary allowance (25 kcal × weight in kg) +15kcal × TBSA | Commonly overestimates caloric needs |
| 1–3 years | Recommended dietary allowance (25 kcal × weight in kg) +25kcal × TBSA | |
| 4-15 years | Recommended dietary allowance (25 kcal × weight in kg) + 40kcal × TBSA | |
Growth delay
Growth delay is a common concern in the pediatric burn population and can have a significant impact on the long-term health and well-being of children who have suffered burn injuries. , A burn injury can cause a number of physical and psychological changes in a child, including changes in hormone levels, alterations in metabolism, and changes in the immune system. These changes can result in growth delay and stunted development, which can have a lasting impact on the child’s overall health and quality of life. One of the most significant factors that contribute to growth delay is the disruption of the body’s normal metabolic processes caused by the burn injury. Burns cause the body to release large amounts of stress hormones, which can disrupt the normal functioning of the body’s metabolic processes, leading to a reduction in growth and development.
Another factor that can contribute to growth delay in the pediatric burn population is the impact of the burn injury on the child’s immune system. Burn injuries can weaken the immune system, making the child more susceptible to infections.
There are several strategies that can be used to help prevent or mitigate growth delay in the pediatric burn population. One of the most important strategies is providing adequate nutritional support to children who have suffered burn injuries. This can help ensure that they receive the nutrients and energy they need to support growth and development.
Increased protein degradation has been reported up to 9 months after major burns. Growth delay and osteopenia can persist up to 2 years in children. Despite adequate nutritional support, children with greater than 50% TBSA burn have a linear growth delay of height and weight and a decrease in maximal exercise capacity during the first year after burn, which slowly resolves to near normal distribution by 3 years after burn. The causes of this growth delay are still not completely understood. It has been demonstrated that body composition of severely burned children significantly improved in the second year compared with the first year after injury. Administration of recombinant human growth hormone, propranolol, and oxandrolone has been shown to ameliorate growth delay secondary to burn injury in children. , ,
Oxandrolone improves burn injuries through stimulating anabolic and reducing catabolic processes; β-blocker attenuates cardiac work and REE after severe thermal injury and improves fat metabolism and insulin sensitivity.
Management of burn wound
The key elements of burn wound management include cleansing, debridement, application of topical antimicrobial agents, dressing changes, and surgical treatment. The final aim is wound closure. Prompt and permanent wound closure after burn injuries is fundamental for patient recovery.
Cleansing can be performed with soap and water, and debridement can be initially performed using gentle mechanical techniques. Topical antimicrobials are the most commonly used treatment in partial-thickness burns, but one drawback of using topical antimicrobials in burn wounds is the pain associated with dressing changes, especially in children. Immediate application of silver-based dressings is one of the treatment options for partial-thickness burns, primarily burns covering less than 30% TBSA. Several silver-based dressings are available for partial-thickness burns that can be left on for several days, thus decreasing the number of dressing changes and the associated pain.
One of the most important steps in the management of burn patients is the early surgical excision of the deep partial- and full-thickness burn wounds after resuscitation is complete. This practice, along with improvements in fluid resuscitation and the general care of burn patients, has reduced the incidence of sepsis in burn patients. Before early excision and grafting, third-degree burns were treated by removing small amounts of eschar at a time followed by grafting. Commonly, eschar was allowed to separate through lysis by bacterial enzymes, which led to a high incidence of invasive infection, wound sepsis, increased length of hospital stay, and increased mortality. Currently, massive excision can be managed in children and contributes to shortened length of hospital stay and reduced mortality. Early excision of massive burns in the first 24 hours is safe and effective.
In extensive burns, skin substitutes such as allograft, xenograft, biodegradable temporizing matrix, and Integra can be used to cover the wound until donor sites are available for procurement.
Dermal and skin substitutes can be used as temporary coverage for severe burns. Integra, a bilaminar skin substitute, can be used instead of homografts as temporary coverage. The Silastic superficial layer can be removed after 3 weeks, and a thin autograft is placed on top. The entire wound can be covered with Integra, which is subsequently autografted when donor sites are available.
It may be difficult clinically to determine the precise depth of scald burns during the early postburn period because the wounds may be indeterminate and contain a mixture of superficial and deep partial-thickness burns and sometimes even full-thickness burns. Indeterminate-depth scald burns covering less than 20% TBSA in young children are best managed with delayed surgical intervention instead of early excision. Unless the wound is clearly full thickness, the scald burn is conservatively managed for approximately 2 weeks to allow the wound to heal or demarcate. This delayed surgery results in a smaller area of wound being excised and less blood loss.
Large scald burns can be treated with allograft or xenograft, which significantly reduces the pain involved with dressing change and wound care. Scald burns covering more than 20% TBSA and of indeterminate depth were randomized to treatment with allograft skin vs. topical antimicrobial therapy. Treatment with allograft skin led to faster healing and less pain. In another study, patients with greater than 40% TBSA burn were randomized to treatment with allograft skin or topical antimicrobials. Patients who received allograft skin had a significantly shorter length of hospital stay.
Blisters are characteristic findings of second-degree superficial burns. Varied opinions for the management of burn blisters are available in the literature. One option is to puncture it in a sterile way, keep the overlying epidermis as a biologic cover, and cover with dressing. Fluid in the blister is ultrafiltrate of the plasma, which is rich in proteins such as immunoglobulins, various cytokines, prostaglandins, and interleukins. This fluid is proinflammatory, and the evidence regarding its effect on wound healing is varied.
Split-thickness autograft skin is the gold standard for closure of extensive/deep burns, but because donor site availability may be insufficient in massive burns, alternatives have been evaluated for safety and efficacy of wound closure.
Furthermore, Palmieri observed that split-thickness skin grafts (STSGs) close the wound, but on the other hand they leave patients with visible scars, dry skin, pruritus, pain, pigmentation alterations, and changes in sensation. New potential options are emerging for application in extensive burn wounds.
Autologous engineered skin substitutes are available for repair or regeneration of excised, full-thickness burns. These include cultured epidermal autografts, acellular dermal substitutes consisting of collagen and glycosaminoglycans, and other cell therapy options. , Although they are effective ways to cover large burns where donor sites are limited, they may not be the most cost-effective approaches. ,
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree