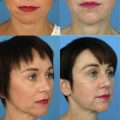
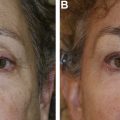
Ptosis surgery is performed via an anterior/external or posterior/internal approach, primarily defined by the eyelid elevator muscle surgically addressed: the levator complex anteriorly or Muller muscle posteriorly. Posterior ptosis surgery via Muller muscle conjunctival resection is an excellent first choice for cases of mild to moderate ptosis with good levator function, as it is predictable, provides a reliable cosmetic outcome, requires no patient cooperation during surgery, portends a lower rate of reoperation, and rarely leads to lagophthalmos and/or eyelid retraction postoperatively. External levator resection is preferred in patients with severe ocular surface/cicatricial conjunctival disease, shortened fornices, and lesser levator function.
Key points
- •
There are 2 major approaches to ptosis surgery, anterior/external and posterior/internal, primarily defined by the respective eyelid elevators surgically addressed: the levator complex anteriorly and Muller muscle posteriorly.
- •
Posterior approach Muller muscle conjunctival resection (MMCR) surgery is an excellent first choice for most cases of mild to moderate ptosis with good levator function because it is predictable, provides a reliable cosmetic outcome, requires no patient cooperation during surgery, is associated with a lower rate of reoperation, and rarely leads to lagophthalmos and/or eyelid retraction.
- •
There are cases in which an anterior approach external levator resection (ELR) is preferred, including severe ocular surface/cicatricial conjunctival disease, shortened fornices, and in patients with poor levator function.
- •
There are circumstances in which the authors prefer MMCR and others may traditionally favor ELR. These include patients with mild congenital ptosis and good levator function, contact lens–associated ptosis, glaucoma with superior filtering blebs, and myasthenia gravis.
Introduction
Blepharoptosis is one of the most common problems presenting to the ophthalmic plastic surgeon. Most ptosis surgery involves some combination of resecting and advancing or plicating the eyelid retractors (the levator muscle/aponeurosis or Mueller muscle) with the intention of elevating the upper eyelid margin. Excluded from this paradigm are bypass procedures, such as frontalis suspension, which is not a focal topic of this discussion. Although ptosis surgery may conceptually appear straightforward, it requires a detailed knowledge of relevant eyelid anatomy, understanding of the pathophysiology of the cause of ptosis, and careful surgical planning to execute effectively. Even with these considerations, ptosis surgery can be frustratingly unpredictable, signifying that major gaps exist in our knowledge as it relates to ptosis physiology, mechanisms of repair, and the biological healing process.
Current surgical management of ptosis is typically by 1 of 2 approaches, anterior and posterior, mostly defined by the respective eyelid elevators surgically addressed: the levator anteriorly and Muller muscle posteriorly. The anterior/external approach involves advancement or resection of the levator aponeurosis, and for consistency in this discussion this will be referred to as an external levator resection (ELR). Posterior/internal approach ptosis surgery involves resection of the Muller muscle and conjunctiva, and for consistency this will be referred to as Muller muscle conjunctival resection (MMCR). It should be noted that neither of these surgeries is likely pure, and in each case some manipulation of the other elevator is expected. It is also important to differentiate the conceptual approach from surgical access. The incision does not define the approach. Techniques in which a posterior incision is used to plicate the levator are considered anterior approaches. Vice versa, methods in which an anterior incision is used to plicate the Muller muscle would be considered posterior approaches. We refer to anterior and posterior as they relate to the elevators primarily involved in the surgery, not the position of the incision or surgical access. Thus, whether the skin or conjunctiva is incised is not particularly relevant for this discussion of anterior and posterior ptosis surgery.
The decision of whether to perform anterior or posterior ptosis surgery in many ways ultimately depends on surgeon preference, although there are circumstances in which one approach may be preferred over the other. In this article, we discuss relevant anatomy, pathophysiology, surgical techniques, and our decision-making process for selecting the appropriate ptosis surgery.
Anatomic considerations
The 2 primary retractors of the upper eyelid are the levator palpebrae superioris (LPS) muscle and Muller muscle. The frontalis muscle is often considered a secondary retractor of the eyelid; however, recent investigations suggest that the mechanical action of the frontalis (elevating the brow) does not transmit to the eyelid under normal circumstances. The frontalis is thus not considered an eyelid elevator for this discussion.
The LPS is a skeletal muscle, originating from the lesser wing of the sphenoid bone superolateral to the optic canal. , Its muscular portion is approximately 36 mm long and fans out as it courses anteriorly from a width of 4 mm posteriorly to 18 mm at the level of the superior transverse (Whitnall) ligament. The Whitnall ligament consists of a band of fibrous tissue extending from the lacrimal gland fossa laterally to the trochlea medially. Whitnall’s ligament is believed to function as a pulley for the LPS, allowing the muscle to change direction.
Anterior to the Whitnall ligament, the LPS courses inferiorly as a fibrous aponeurosis. Microscopically, the transition from LPS to aponeurosis is gradual with muscular fibers present in diminishing amounts as it travels distally into the eyelid. The levator aponeurosis expands medially to form the medial horn, which coalesces with the posterior portion of the medial canthal tendon and inserts onto the posterior lacrimal crest. Similarly, a lateral horn is formed laterally, which divides the lacrimal gland into orbital and palpebral lobes and attaches to the lateral orbital (Whitnall) tubercle. , ,
The levator aponeurosis has been classically thought to insert inferiorly onto the lower half of the anterior surface of the tarsus in white eyelids. Histopathological evidence suggests though that this insertion may be more diffuse and variable, effectively anywhere along the tarsus. This is particularly evident in Asian eyelid configurations. Fibers of the aponeurosis additionally extend anteriorly through the orbicularis oculi muscle and insert onto the skin, which is believed to form the eyelid crease. ,
The Muller muscle is a smooth muscle. Its origin is found within the Whitnall ligament, where the LPS undergoes muscle-aponeurosis transition. Histologic studies demonstrate interweaving smooth and striated muscle fibers at this position. , The Muller muscle is approximately 8 to 12 mm in length and courses deep to the levator aponeurosis, in what some consider to be a potential “postaponeurotic” space. Distally, there is some controversy regarding the insertion of the Muller muscle. Classically it is described as inserting onto the superior border of the tarsus. Other investigations have suggested that it may insert more inferiorly onto the anterior surface of the tarsus. Some have further proposed that the Muller muscle terminates above the tarsus within a complex network of elastic and collagen fibers involving the levator aponeurosis, Muller muscle, and orbicularis. , It is likely that this insertion is variable across populations, and may even change over time. Interestingly, the LPS-Muller configuration is somewhat unique in the body, with the skeletal LPS and smooth Muller muscle being arranged in series without a fixed anchor point.
The Muller muscle receives sympathetic innervation, a finding that has been confirmed clinically by its involvement in Horner syndrome, experimentally by immunostaining of sympathetic receptors, and pharmacologically by using sympathetic agonists and antagonists. Although it is known to be sympathetically innervated, the precise nervous pathway within the orbit is poorly understood. It is likely that innervation is derived from multiple sympathetic roots. The Muller muscle is responsible for approximately 3 mm of upper eyelid excursion. This has been confirmed experimentally in healthy subjects where from a neutral position, the eyelid can be lowered 1.5 mm with sympathetic antagonists and raised 1.5 mm with agonists.
There is some controversy regarding the function of the Muller muscle. Some suggest it is a transmitter of the levator aponeurosis action, whereas others propose it is a stretch receptor for the LPS, providing a primarily proprioceptive role. The most widely accepted hypotheses suggest that it functions to maintain tonic eyelid position, allowing for responses based on sympathetic stimulation, or that it maintains a 3 mm “fine tuning” role in upper eyelid position. It is additionally possible (and plausible) that the Muller muscle is multifunctional.
Pathophysiology
The determination of eyelid position likely involves a comprehensive maintenance system with afferent inputs, central processing, and efferent outputs. Dysfunction anywhere in this system may lead to ptosis, and ptosis surgery is theorized to modify this system, leading to a re-setting of static eyelid position.
Potential afferent inputs to this system include vision, touch sensation, and proprioception. Although it is well-documented that functional visual field impairment due to ptosis can be reversible with surgery, visual stimuli may not be a critical component in the development of ptosis, as patients with anophthalmic sockets can demonstrate ptosis and respond to both anterior and posterior ptosis surgery. Simple touch sensation also may be a less relevant input, as it is well known that an anesthetic eye, for instance after topical proparacaine use, is not reliably associated with ptosis. Proprioceptive stimuli are of keen interest as potential vital inputs ; however, much of the evidence for this pathway is speculative at this point. Further research is needed to determine the influence of these various afferent factors.
The flow of afferent information is also incompletely understood. Presumably these signals follow the trigeminal system. The trigeminal sensory pathway passes through the trigeminal ganglion, synapses in the main sensory trigeminal nucleus, decussates, and travels along the contralateral ventral trigeminal lemniscus before synapsing in the ventral posteromedial (VPM) nucleus of the thalamus. The proprioceptive signals from the extraocular muscles and eyelid elevators presumably follow this pathway; however, animal data suggest that at least some of the extraocular muscle proprioceptive input travels directly to the lateral geniculate nucleus with retinal ganglion cells and even with motor nerves, including the oculomotor nerve. From the thalamus, these signals travel diffusely throughout the brain to visual, sensory, and balance centers. Indirect evidence of eyelid position signals traveling through the thalamus can be drawn from studies in which thalamic injury (infarct, hemorrhage) can be associated with ptosis. These afferent systems are relatively poorly understood; however, they clearly play a role in eyelid position homeostasis.
Output systems are more completely characterized. Motor innervation to the LPS via the superior division of the third cranial nerve is well-established. , Muller muscle innervation is part of the sympathetic system. There is some controversy as to the precise anatomic projection of efferent intraorbital pathways to the Muller muscle, and the motor control could perhaps have input from other related extraorbital systems. However anatomically transmitted, much evidence of sympathetic innervation exists.
The integration and coordination of the afferent and efferent signals is the most sparsely investigated and consequently most poorly understood aspect of eyelid position physiology. Some have proposed a reflex arc involving afferent stretch receptors in the Muller muscle and efferent output via the LPS and frontalis muscles. , This work is theoretic, yet perhaps it does provide some framework for conversion of afferent to efferent signals, though without central processing.
There is, however, much emerging evidence of integration capacity, and thus central processing, in the eyelid position maintenance system. For instance, eyelid position is relatively preserved when gravitational forces are altered. We examined photographs of astronauts both on Earth and in space and found zero gravity does not significantly change marginal reflex distance 1 (MRD1). We have also shown that in healthy individuals, changing postural position was also associated with relative preservation of eyelid position. In addition, reducing protracting forces in facial palsy is in most cases associated with maintenance of normal eyelid position, although it should be noted that more severe facial weakness is associated with a breakdown in this system and eyelid position abnormalities. , These results together suggest there is adaptive capacity within the eyelid position maintenance system. However, at present the capacity, functionality, and neural basis for this system are each very poorly characterized. Clearly, more research in this area is needed to more accurately understand the determinants of eyelid position, particularly as it relates to central processing and proprioception.
Mechanisms of repair
External Levator Resection
ELR surgery was developed based on the principle that the primary defect in ptosis is a disinsertion or lengthening (thinning) of the levator aponeurosis. Surgery is directed at the presumed root cause of the disorder and involves reinserting (with or without shortening) the levator aponeurosis to the tarsus. This is supported histopathologically, as one study demonstrated aponeurotic defects in most patients with involutional ptosis.
However, various pieces of information have challenged this assertion over time. Some believe the high incidence of levator aponeurosis disinsertion is iatrogenic, and with careful dissection its incidence can be decreased dramatically. Others have demonstrated fatty infiltration of the LPS with an attenuated aponeurosis but no disinsertion or dehiscence in patients with ptosis, suggesting again that the incidence of disinsertion is lower than originally suspected. , Additional studies noting decreased levator function in patients affected by involutional ptosis suggest an alternative myogenic rather than aponeurotic etiology for involutional ptosis. Further, as described in the preceding sections, it is quite likely that there are more complicated pathophysiologic processes involved in at least some proportion of patients with ptosis. Simple mechanical theories appear to incompletely explain the pathophysiology of ptosis generally and the mechanisms of ELR efficacy specifically.
Overall, although it is widely believed that mechanical advancement/reinsertion of the levator aponeurosis onto the tarsus is the mechanism of eyelid elevation in ELR, the system may in fact be more complicated. There is emerging evidence that the surgery may have more nuanced effects on eyelid position.
Muller Muscle Conjunctival Resection
The mechanism for MMCR surgery is even less clear. It was originally proposed that resection/plication of the Muller muscle would raise the eyelid an equal amount as would phenylephrine applied in the preoperative setting. However, phenylephrine response is not entirely predictive of surgical change. This suggests that the mechanism is more complicated than mechanically replicating the configuration of a contracted muscle. Further supporting this notion, histologic studies have demonstrated little association between the amount of Muller muscle resected or the quantity of adrenergic receptors and degree of eyelid elevation.
In response to this unequal relationship between Muller muscle resection and eyelid elevation, some have proposed that plication of the levator aponeurosis is an essential aspect of the mechanism for MMCR surgery. This is supported by histologic identification of levator fibers in MMCR specimens. , However, there are observations that challenge this explanation as well. Mechanical levator plication does not explain the variability in response to similar tissue resection lengths. For instance, the standard deviation of change in MRD1 for 1 mm of resection length is close to 1.0 mm, indicating high variability. In addition, there is little empiric evidence of a relationship between resection length and change in MRD1 when preoperative MRD1 is taken into account. , Further, MMCR surgery appears to have less effect on eyelid contour compared with ELR, and with similar levator-related mechanisms, one would expect the contour effects to be the same.
Finally, the progression of MRD1 change after ELR and MMCR surgery is also different, suggesting alternative mechanisms. In patients who undergo ELR, change in MRD1 evident by 1 week postoperatively is stable at 3 months, and this is independent of the degree of swelling. However, patients who undergo MMCR experience an increase in MRD1 from 1-week to 3-month follow-up, although there is no difference in final MRD1 result between the groups. These studies suggest that the 2 surgeries work by different mechanisms, albeit with some overlap.
Although there is ample evidence as to the deficiencies of mechanical Muller or levator-specific mechanisms, there is unfortunately little evidence for alternative theories. The unequal evidence for any one theory can suggest 2 overlapping possibilities. The first is that multiple pathophysiologic processes are at play, and thus in some but not all cases mechanical mechanisms are managed appropriately and primarily responsible for efficacy. Alternatively, mechanical processes may be only marginally involved, in so much as they stimulate more adaptive neural processing mechanisms that reset eyelid position. These systems may be functional or dysfunctional, creating variability in the response. This dichotomy would be akin to the mechanical and sensory theories of strabismus. Emerging research will ideally provide better answers for this question in the future. Presently, we can use practical experience to guide best practices, accepting some variability in outcome.
Surgical technique
External Levator Resection
Anterior ptosis surgery was first described by Everbusch in 1883, but gained popularity after Jones and colleagues demonstrated disinsertion of the levator aponeurosis in 1975. The surgery involves advancement/reattachment of the levator aponeurosis onto the tarsus, typically via an external upper eyelid skin incision.
Steps of procedure
- 1.
Mark the upper eyelid crease.
- 2.
Local anesthesia with or without conscious sedation. Conservative use of local anesthesia may preserve levator function and allow for accurate assessment of eyelid position intraoperatively.
- 3.
Incise skin.
- 4.
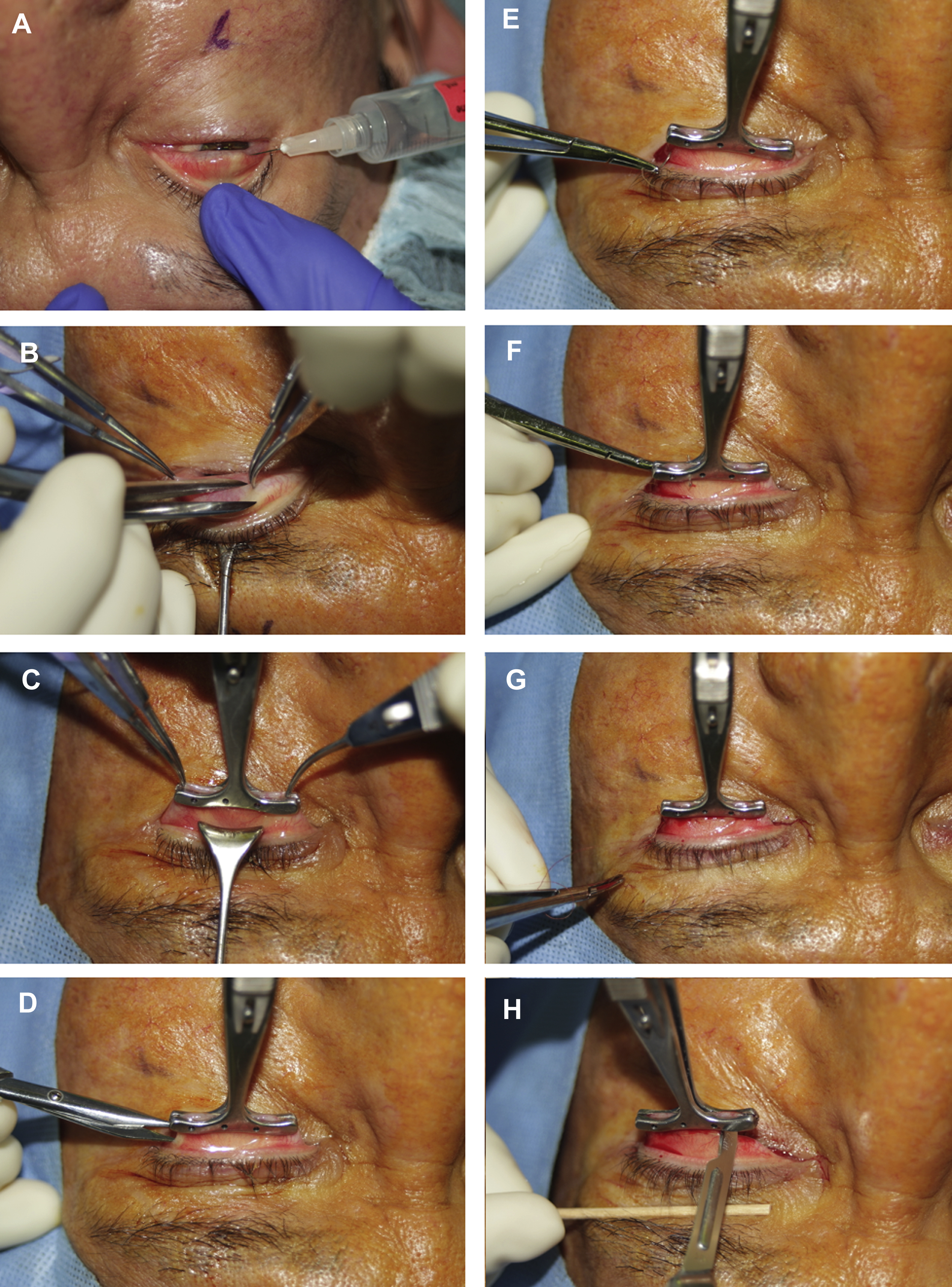
Traverse orbicularis muscle ( Fig. 1 A) and identify levator aponeurosis ( Fig. 1 B) and tarsus.
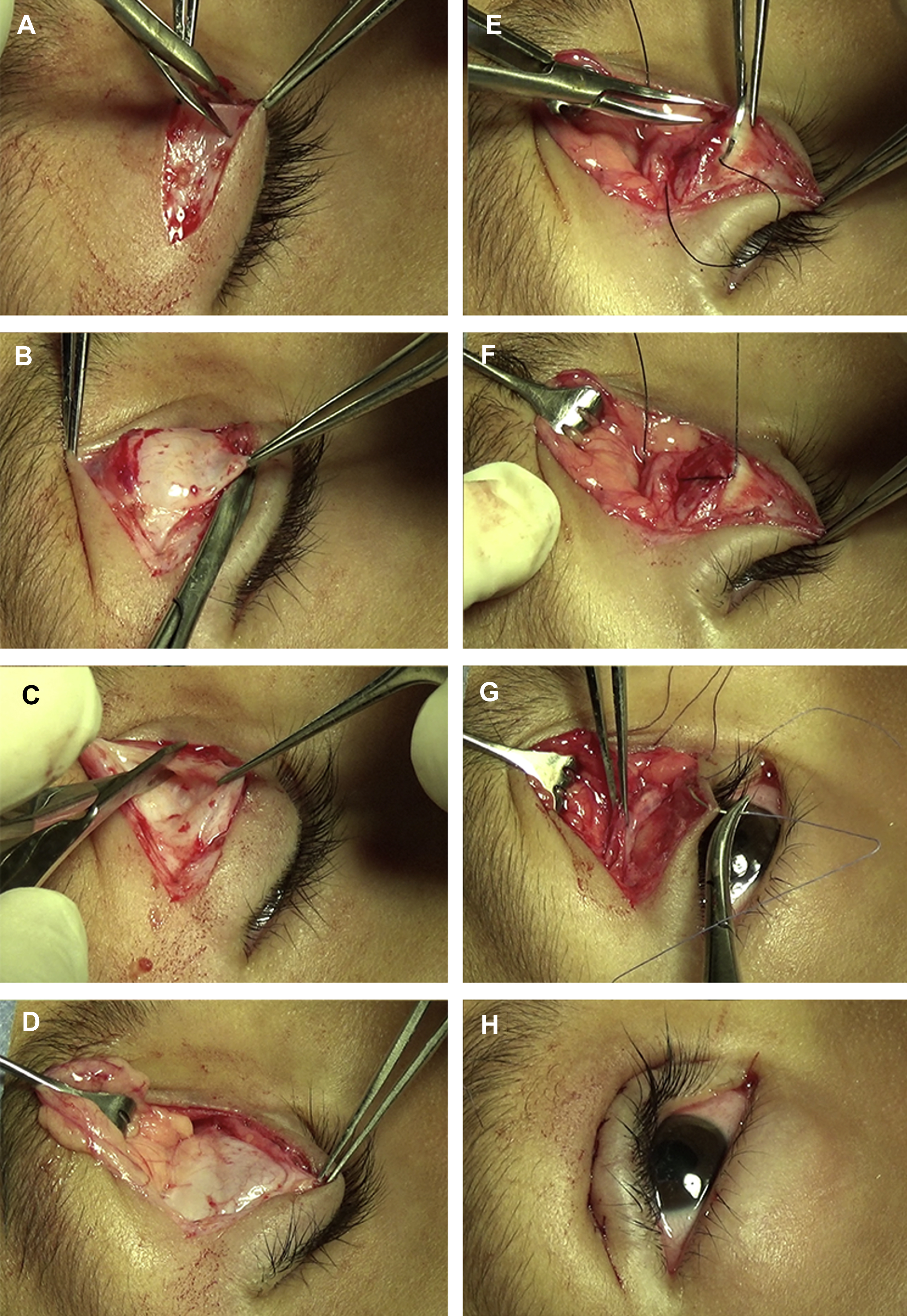
Fig. 1
Steps of ELR procedure. ( A ) The orbicularis oculi muscle is incised, and ( B ) levator aponeurosis identified. The levator is dissected free from the septum ( C ) and pre-aponeurotic fat pads ( D ) above. ( E ) A suture is passed partial thickness through the tarsus at the desired point of maximal elevation, then through the posterior surface of the levator ( F ). ( G , H ) The skin is closed, with incorporation of levator aponeurosis if eyelid-crease formation is desired.
- 5.
Dissect the levator free from the superior border of the tarsus.
- 6.
At this point, many dissect the levator free of the Muller muscle below and/or the septum ( Fig. 1 C) and pre-aponeurotic fat pads ( Fig. 1 D) above.
- 7.
Determine point of maximal eyelid elevation and pass a double-armed suture partial thickness through the tarsus at this location ( Fig. 1 E), then through the posterior surface of the levator ( Fig. 1 F) and tie on a slip knot.
- 8.
Sit the patient up vertically and assess eyelid position and contour. Suture may need to be adjusted. A second suture may need to be placed to attain optimal lid height and contour.
- 9.
Tie suture ends permanently and cut.
- 10.
Close skin ( Fig. 1 G,H).
Variations in technique
It should be noted that huge variability in the steps of this procedure exist in the community. A wide range of modifications have been proposed from changing the size and location of the skin incision, , to the order of dissection along the anterior or posterior portions of the levator (or both), even including whether to disinsert the levator at all or to include the horns in a wide dissection. The number, placement, and configuration of sutures have also been varied in as many ways as would be conceptually possible. There is no standard procedure for ELR surgery and no convincing evidence that one incision, dissection, or suture technique is better (or worse) than another.
More significant procedural modifications also have been described. Intraoperative evaluation of the eyelid in the sitting position with patient cooperation has been shown to more accurately predict postoperative eyelid position, supporting the use of upright patient positioning and suture adjustment intraoperatively. The use of adjustable sutures has been reported to decrease the reoperation rate to 5% to 18%.
To predict surgical outcome and decrease reoperation rates, there have been attempts to modify the amount of aponeurosis advancement via the creation of nomograms. Various approaches have been described. The levator function technique assumes that the eyelid height set during surgery will elevate or fall a predictable amount after surgery based on preoperative levator function. In the MRD technique, the preoperative MRD1 dictates the amount the levator is advanced. This approach has been refined to a 2:1 aponeurosis resection to expected eyelid elevation algorithm, suggesting that for every 2 mm of resection, 1 mm of eyelid elevation is to be expected. This has been further modified by using a spring scale to apply a constant force on the aponeurosis to account for its expansile nature. , None of these nomograms have gained wide popularity for a number of reasons, including the variability in defining the “end of the levator” and empiric experience revealing extensive variability in the response. These nomograms simply do not demonstrate the type of consistency required for practical utility and have thus not been widely adopted.
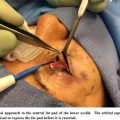
Muller Muscle Conjunctival Resection
Since it was originally described by Putterman and Urist in 1975, MMCR surgery has gained acceptance and popularity, particularly among younger surgeons in the United States, although less so internationally. , The basic premise of the surgery involves excision of a tissue flap containing the Muller muscle, conjunctiva, and some levator aponeurosis fibers.
Steps of procedure
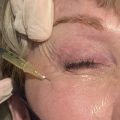
- 1.
Local anesthesia with or without conscious sedation. The posterior eyelid can be anesthetized by everting the eyelid and injecting directly into the Muller muscle plane ( Fig. 2 A).