Abstract
Extracellular matrices (ECMs) consist of highly organized networks of collagens, elastin, glycoproteins, and proteoglycans. In addition to establishing tissue architecture and providing mechanical strength, ECMs are biologically active, regulating cellular behavior, fate, and communications. The functions of the ECM are adapted to meet the specific needs of each tissue, with tissue-specific molecular composition that is fine-tuned by intricate post-translational modifications. Genetic defects that lead to a disorganized and dysfunctional ECM underlie a broad spectrum of heritable diseases with cutaneous manifestations. This chapter describes the molecular composition of the dermal ECM and provides an overview of genetic ECM disorders affecting the skin as well as potential molecular therapies.
Keywords
Extracellular matrices, collagens, extrafibrillar matrix, glysosaminoglycans, proteoglycans, elastic fibers, elastin
- ▪
Extracellular matrices (ECMs) represent specifically organized networks of collagens, elastin, glycoproteins, and proteoglycans that have distinct structural roles and specific functional properties in all tissues
- ▪
ECMs are biologically active, interact with cells, and regulate their functions during development, regeneration, and normal tissue turnover
- ▪
Mutations in ECM genes cause a broad spectrum of human diseases, from Ehlers–Danlos syndrome to epidermolysis bullosa, and components of the ECM are targeted in autoimmune diseases, e.g. bullous pemphigoid, bullous systemic lupus erythematosus, lichen sclerosus
- ▪
The collagen family contains 28 different subtypes. All collagens consist of three polypeptide α-chains, which are folded into a triple helix. In each chain, every third amino acid is glycine (Gly), and thus the sequence can be expressed as (Gly-X-Y) n . A hallmark of collagens is the presence of hydroxyproline (Hyp) in the Y position of this repeat sequence. Collagens are expressed in all tissues of the human body, and distinct sets of collagens co-polymerize into highly organized suprastructures, e.g. fibrils and filaments, in a tissue-specific manner
- ▪
Elastin provides tissues with elasticity. Elastin monomers contain repetitive hydrophobic sequences and are highly cross-linked. The cross-links between several individual molecules provide both elasticity and insolubility to elastic fibers, which can be stretched by 100% or more and still return to their original form. The elastic fibers in the dermis also contain a microfibrillar component which attaches the fibers to surrounding structures.
Introduction
The different types of extracellular matrix (ECM) represent specifically organized assemblies of the matrix macromolecules listed in Table 95.1 . These macromolecules have characteristic patterns of aggregation into insoluble suprastructures with a high degree of order at successive hierarchic levels . Each of these structures is tissue-specific and adapted to the particular needs of a given tissue. The major constituents are often similar in functionally diverse ECMs. However, different types of quantitatively minor molecular components associate with major elements in tissue-specific suprastructural arrays determined by their relative compositions. The matrix suprastructures may be likened to alloys, each having metallurgic properties that differ from each other and those of the pure metals ( Fig. 95.1 ).
| COMPONENTS OF THE EXTRACELLULAR MATRIX | |
|---|---|
| Collagens (28 types) | Laminins (15 types) |
| Elastin | Proteoglycans ( Table 95.3 ) |
| Fibrillins (3 types) | Glycoproteins ( Table 95.4 ) |
| LTBPs (4 types) | Integrins |
| Fibulins * (7 types) | Enzymes that modify ECM assemblies |
* Fibulins are believed to function as intra-molecular bridges that stabilize ECM structural networks (e.g. elastic fibers, microfibrils, basement membrane structures).

Individual ECM macromolecules are usually oligomers composed of one or several polypeptides. Intimate contacts between the subunits are formed by coiled-coil structures, such as the collagen triple helix or supercoiled α-helices comprised of three or more polypeptides. In addition, large matrix macromolecules can be regarded as linear sequences of structural modules that are similar in a large variety of proteins . The modules can be recognized by several cellular receptors, but receptor clustering will be determined in a tissue-specific manner and the response may be different in different tissues.
Our knowledge of matrix macromolecules has expanded dramatically in recent years due to advances in molecular genetics and proteomics. A multitude of molecules have been characterized and their expression, regulation, tissue specificity, and functions discerned . The assembled ECM structures are generally adhesive, enabling the attachment of tissue-specific cells, leukocytes, tumor cells, and even microorganisms. Through integrin-mediated interactions with cells, matrix molecules control cell proliferation, differentiation, and migration, especially during development and regenerative processes. Without contact with the ECM, many cells undergo a form of apoptosis known as anoikis . Furthermore, the ECM can function as a reservoir of information; certain proteoglycans and proteins bind growth factors (e.g. transforming growth factor [TGF]-β), releasing and activating them as needed to control cellular functions . To date, mutations in >50 different genes encoding ECM molecules have been found to underlie heritable disorders in humans and mice.
Structure and Function of the Extracellular Matrix
Collagens
The collagen family of proteins plays an important role in maintaining the integrity of most tissues. The family currently includes 28 proteins formally defined as collagens ( Table 95.2 ). They contain at least 45 distinct polypeptide chains, each encoded by a different gene, and more than 15 other proteins have a collagen-like domain (e.g. macrophage scavenger receptor 1 and 2, ectodysplasin, pulmonary surfactant proteins).
| THE COLLAGEN FAMILY OF PROTEINS | |||
|---|---|---|---|
| Type | Chains | Gene | Tissue distribution |
| Fibril-forming collagens | |||
| Collagen I | α 1 (I), α 2 (I) | COL1A1, COL1A2 | Skin, most ECM |
| Collagen II | α 1 (II) | COL2A1 | Cartilage, vitreous humor |
| Collagen III | α 1 (III) | COL3A1 | Skin (including fetal skin), lung, vasculature |
| Collagen V | α 1 (V), α 2 (V), α 3 (V) | COL5A1, COL5A2, COL5A3 | Skin, with collagen I heterotypic fibrils |
| Collagen XI | α 1 (XI), α 2 (XI), α 3 (XI) | COL11A1, COL11A2, COL11A3 | With collagen II heterotypic fibrils |
| Collagen XXIV | α 1 (XXIV) | COL24A1 | Developing bone and cornea |
| Collagen XXVII | α 1 (XXVII) | COL27A1 | Cartilage, eye, ear, lung |
| FACITs | |||
| Collagen IX | α 1 (IX), α 2 (IX), α 3 (IX) | COL9A1, COL9A2, COL9A3 | With collagen II heterotypic fibrils |
| Collagen XII | α 1 (XII) | COL12A1 | Skin, tissues containing collagen I |
| Collagen XIV | α 1 (XIV) | COL14A1 | Skin, tissues containing collagen I |
| Collagen XVI | α 1 (XVI) | COL16A1 | Skin, many tissues |
| Collagen XIX | α 1 (XIX) | COL19A1 | Basement membranes, fetal muscle |
| Collagen XX | α 1 (XX) | COL20A1 | Skin, cornea, cartilage, tendon |
| Collagen XXI | α 1 (XXI) | COL21A1 | Many tissues, including skin |
| Collagen XXII | α 1 (XXII) | COL22A1 | Tissue junctions (including between the anagen hair follicle and dermis) |
| Basement membrane collagen | |||
| Collagen IV | α 1 (IV), α 2 (IV), α 3 (IV), α 4 (IV), α 5 (IV), α 6 (IV) | COL4A1, COL4A2, COL4A3, COL4A4, COL4A5, COL4A6 | All basement membranes, isoforms vary Skin: α 1 (IV), α 2 (IV), α 5 (IV), and α 6 (IV) |
| Microfibrillar collagen | |||
| Collagen VI | α 1 (VI), α 2 (VI), α 3 (VI) | COL6A1, COL6A2, COL6A3, COL6A5,COL6A6 | Skin, other microfibril-containing tissues |
| Network-forming collagens | |||
| Collagen VIII | α 1 (VIII), α 2 (VIII) | COL8A1, COL8A2 | Skin, subendothelial matrices |
| Collagen X | α 1 (X) | COL10A1 | Hypertrophic cartilage |
| Anchoring fibril collagen | |||
| Collagen VII | α 1 (VII) | COL7A1 | Skin, mucous membranes, cornea |
| Transmembrane collagens | |||
| Collagen XIII | α 1 (XIII) | COL13A1 | Skin, many tissues |
| Collagen XVII | α 1 (XVII) | COL17A1 | Skin, mucous membranes, cornea |
| Collagen XXIII | α 1 (XXIII) | COL23A1 | Lung, cornea, brain, skin, tendon, kidney |
| Collagen XXV | α 1 (XXV) | COL25A1 | Brain, neurons |
| Multiplexins | |||
| Collagen XV | α 1 (XV) | COL15A1 | Many tissues; parent molecule of restin † |
| Collagen XVIII | α 1 (XVIII) | COL18A1 | Many tissues, including skin, subendothelial matrices; parent molecule of endostatin † |
| Other collagens | |||
| Collagen XXVI | α 1 (XXVI) | COL26A1 | Testis, ovary |
| Collagen XXVIII | α 1 (XXVIII) | COL28A1 | Schwann cells; fetal skin and calvaria |
Collagen triple helix
All collagens consist of three polypeptide chains, known as α-chains, which are folded into a triple helix. In some collagens, the α-chains are identical (homotrimers), while others contain two or three different α-chains (heterotrimers). In the collagenous repeat of each polypeptide chain, every third amino acid is glycine (Gly), and the sequence of an α-chain can be expressed as (Gly-X-Y) n , where X and Y represent other amino acids and n varies according to the length of the repeat. A high number of proline (Pro) and hydroxyproline (Hyp) residues are in the X and Y positions, respectively, and hydrogen bonds between the hydroxyl groups of Hyp contribute to the stability of the helix. The prototype collagen (type I) has an uninterrupted Gly-X-Y repeat sequence that is almost 1000 amino acid residues in length; this forms a rigid, rodlike structure with a diameter of 1.5 nm and length of 300 nm. In some collagens, the (Gly-X-Y) n repeats are interrupted by one or more amino acids. The interruptions may be numerous and longer than the (Gly-X-Y) n repeats, and they provide the molecule with flexibility, which is important for the specific functions of a given collagen type .
Biosynthesis of collagens
Collagen biosynthesis involves a number of post-translational modifications ( Fig. 95.2 ). Some collagens are first synthesized as procollagens that have propeptide extensions at their N-terminus, C-terminus, or both. The main intracellular steps in collagen biosynthesis include the following:
- •
cleavage of signal peptides
- •
hydroxylation of certain Pro and lysine (Lys) residues to 4-Hyp, 3-Hyp, and hydroxylysine (Hyl)
- •
glycosylation of some of the Hyl residues to galactosyl-Hyl and glucosylgalactosyl-Hyl
- •
glycosylation of certain asparagine residues
- •
association of the α-chains in a specific manner
- •
formation of intra- and interchain disulfide bonds
- •
folding of the triple helix.
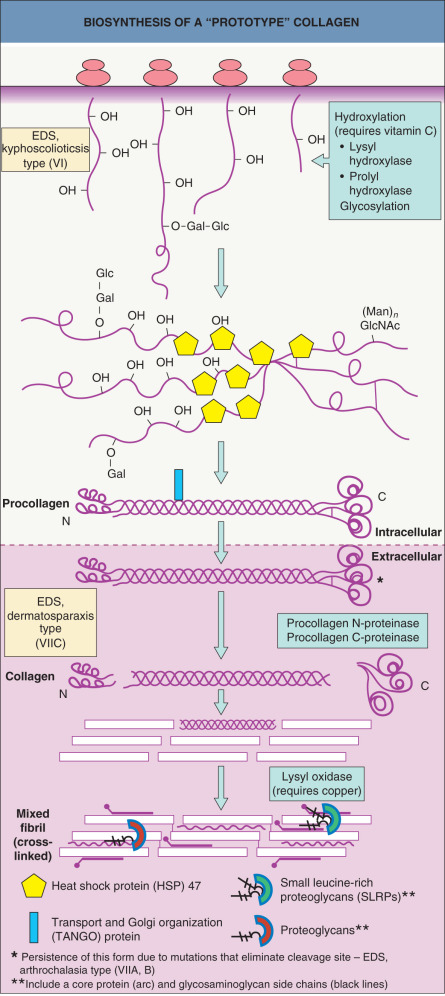
The specific enzymes involved in the biosynthesis of collagens include prolyl-4-hydroxylase and prolyl-3-hydroxylase, which hydroxylate Pro residues to Hyp, and three lysyl hydroxylases, which hydroxylate Lys residues to Hyl. These enzymes require O 2 , Fe 2+ , α-ketoglutarate and ascorbate as cofactors for the reactions. In the rough endoplasmic reticulum, glycosyltransferases add glucosylgalactosyl disaccharides onto the α-chains. The same intracellular enzymes modify all collagen chains , whereas extracellular processing enzymes have a higher substrate specificity. Procollagen I N-proteinase cleaves the N-propeptide of procollagens I and II. This enzyme is a member of the disintegrin and metalloproteinase (ADAM) proteinase family and is also designated as ADAM with t hrombo s pondin type 1 motif (ADAMTS)-2. Procollagen C-proteinases that cleave the C-propeptide of collagens I, II, III, V, and VII belong to the astacin family and include bone morphogenetic protein-1 (BMP-1) and tolloid-like proteins; meprins in this family can cleave both N- and C-termini of collagens .
Cross-linking between collagen molecules involves the ε-amino groups of Lys and Hyl and is catalyzed by lysyl oxidase, a copper-requiring enzyme ; this process can be regulated by proteoglycans . Another enzyme that catalyzes cross-linking of some collagens is tissue transglutaminase. Collagen VII-containing anchoring fibrils in the skin appear to be transaminated, and collagen VII serves as a substrate for tissue transglutaminase in vitro .
The collagen family of proteins
The length and continuity of the triple helical domains vary among the collagen types. For practical purposes, the collagens have been divided into groups according to their ability to form supramolecular aggregates, which are depicted in Fig. 95.3 . The types and tissue distributions of the collagens in each group are listed in Table 95.2 , and more detailed information is available in reviews .
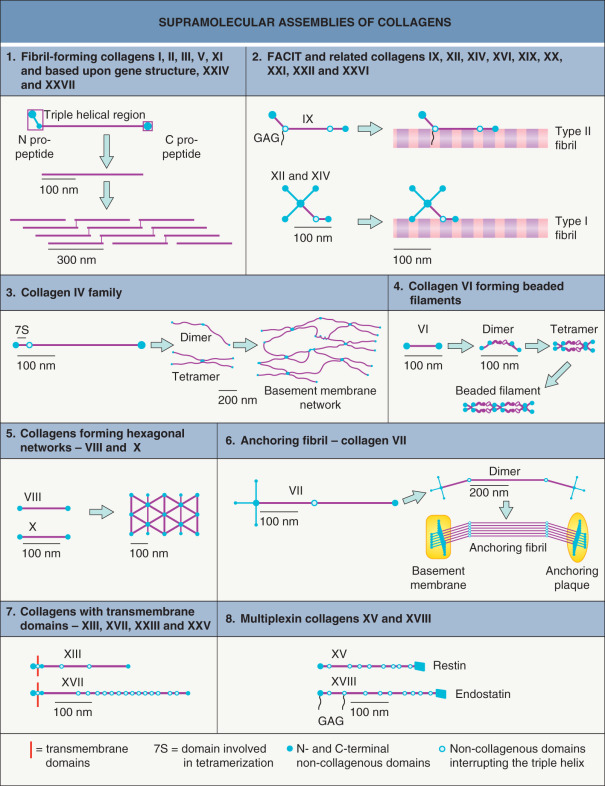
Collagens of the skin
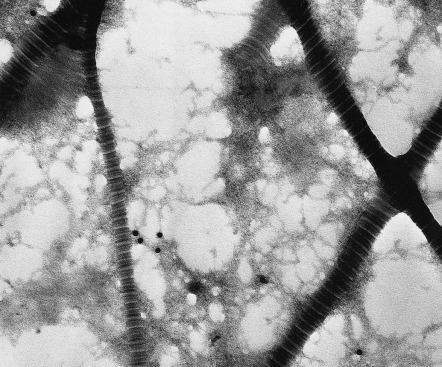
Collagens account for 75% of the dry weight and 20–30% of the volume of the dermis. Different collagens polymerize into distinct suprastructures and have specific functions in the dermis as well as in epidermal and vascular basement membranes. “Pure” collagen fibrils do not exist; these fibrils are always mixtures of several collagens and other molecules, e.g. proteoglycans . Classic, ultrastructurally recognizable, cross-banded fibrils in the dermis contain collagens I, III, V, XII, and XIV. The characteristic cross-banding ( Fig. 95.4 ) with a periodicity of 64 nm results from precise lateral packing of the different collagens within the fibrils (see Fig. 95.3 ). Collagen I is the major component of the fibrils, and the amount of other collagens varies. For example, during embryonic development and wound repair, the relative content of collagen III increases. Collagen VI, a highly glycosylated and disulfide-bonded collagen, is a component of almost all tissues, including the skin. In vitro , it polymerizes to form beaded filaments (see Fig. 95.3 ), but in vivo in the dermis, the ultrastructure of the collagen VI fibrils is reminiscent of microfibrils.
The collagen IV molecules in different basement membranes contain six genetically distinct but structurally homologous α-chains, which form three major networks – α 1 /α 2 , α 3 /α 4 /α 5 , and α 1 /α 2 /α 5 /α 6 . The chain composition is determined by the carboxy-terminal non-collagenous (NC1) domains; covalent interactions between these domains link α-chains to each other. In the skin, the α 1 /α 2 -containing collagen IV network dominates within the dermal–epidermal junction (see Ch. 28 ), but the α 1 /α 2 /α 5 /α 6 -containing network is also thought to be present .
Two collagens are essential for the cohesion of the epidermis and dermis (see Ch. 28 ). Collagen VII is the major, if not sole, component of the anchoring fibrils that attach the basement membrane to the dermal ECM. Collagen XVII (also known as bullous pemphigoid antigen 2) is a component of the anchoring filaments that bind the basal keratinocytes to the lamina densa of the basement membrane. It is a transmembrane collagen in type II orientation with a long extracellular C-terminal region containing a multiply-interrupted triple helix referred to as collagenous domains 1–15 (see Fig. 31.9 ). The ectodomains can be shed from the cell surface by transmembrane proteases , a process important for regulation of cell adhesion and migration. Basal keratinocytes also express other transmembrane collagens such as type XIII, which is a component of focal contacts, and type XXIII.
The vascular basement membranes in the skin contain yet other collagens – namely, collagens VIII and XVIII. Collagen VIII builds hexagonal networks below the endothelial basement membranes (see Fig. 95.3 ) and, thus, structurally strengthens the vascular wall. Collagen XVIII is localized at the dermal side of vascular basement membranes. It is present in both epidermal and vascular basement membranes. Its C-terminal fragment, endostatin, is proteolytically released from the collagen molecule and has independent antiangiogenic and anti-scarring activities .
Most collagens in the skin are products of dermal fibroblasts. Exceptions include: (1) collagen XVII, a surface component of epidermal keratinocytes; (2) collagen VII, which can be synthesized by both keratinocytes and fibroblasts; and (3) collagens VIII and XVIII, which can also be produced by endothelial cells. Several genetic and acquired diseases are associated with abnormalities of skin collagens or the enzymes that process them (see Tables 95.5 & 95.6 and below).
Elastic Fibers
The elasticity of many tissues, including the skin, depends upon the structure of elastic fibers, which have variable compositions. A characteristic property of these fibers is that they can be stretched by 100% or more and still return to their original form. The main components of elastic fibers are elastin and microfibrils.
Elastin
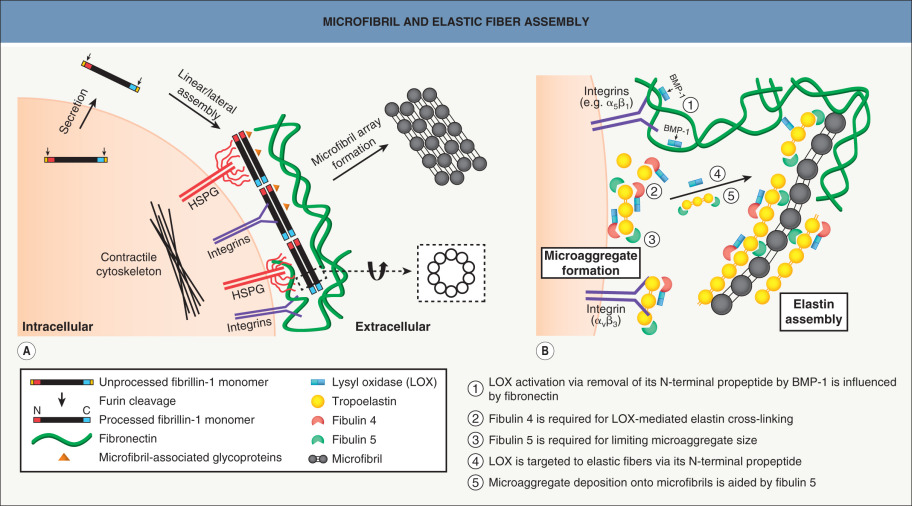
Elastin is a highly cross-linked protein . Its monomer, tropoelastin, exists in several tissue-specific splice variants. Ala- and Lys-rich repeats form critical cross-linking domains within a background of hydrophobic amino acid repeats. Lysyl oxidase, the same copper-dependent enzyme that catalyzes collagen cross-linking (see above), catalyzes the formation of desmosine cross-links between elastin molecules, which account for the elasticity and insolubility of the elastic fibers. Elastin makes up ~90% of mature elastic fibers. During fiber assembly, tropoelastin both self-assembles and interacts with fibulins 4 and 5, fibrillin 1, and microfibril-associated glycoprotein-1 (MAGP-1). A model for this spatially and temporally regulated process is shown in Fig. 95.5 .

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree