Abstract
The main function of the epidermis is to provide a barrier between the organism and the environment, thereby protecting against harmful substances and water loss. This barrier is the result of a complex terminal differentiation program in which the progeny of epidermal stem cells undergoes a series of molecular and biochemical changes leading to generation of the morphologically different strata of the epidermis, which culminates in the replacement of the plasma membrane with the cornified cell envelope. This chapter reviews key signaling pathways that control this terminal differentiation program. Keratin intermediate filaments, which are expressed by keratinocytes in a differentiation-specific manner, are required to maintain epidermal integrity and have additional roles in the regulation of basic cell functions. Desmosomes, which integrate the keratin cytoskeleton into the cell membrane and establish adhesive points between adjacent cells, are necessary for epidermal stability and stress resistance; these cell–cell junctions also function as signaling centers in epidermal morphogenesis and homeostasis. Inherited skin diseases that are caused by a failure of normal epidermal differentiation are also discussed.
Keywords
adherens junctions, cornified cell envelope, desmosomes, epidermal differentiation, epidermal barrier, keratinocyte adhesion, keratin disorders, keratin intermediate filaments, tight junctions
- ▪
The epidermis functions as a barrier that provides protection against harmful substances and water loss
- ▪
Through a complex terminal differentiation program, the progeny of epidermal stem cells undergo molecular and biochemical changes that generate the morphologically different strata of the epidermis
- ▪
Keratinocyte terminal differentiation culminates in the replacement of the plasma membrane with the cornified cell envelope
- ▪
Keratin intermediate filaments, which are expressed by keratinocytes in a differentiation-specific manner, are required to maintain epidermal integrity and have additional roles in the regulation of basic cell functions
- ▪
Desmosomes, which integrate the keratin cytoskeleton into the cell membrane and establish adhesive points between adjacent cells, are necessary for epidermal stability and stress resistance; these cell–cell junctions also function as signaling centers in epidermal morphogenesis and homeostasis
- ▪
Other cell–cell junctions that are found in keratinocytes include adherens junctions and tight junctions
- ▪
A variety of inherited skin diseases are caused by a failure of normal epidermal differentiation and function
Introduction
The main function of the epidermis is to provide a barrier between the organism and the environment. This barrier is the result of a complex terminal differentiation program in which the progeny of epidermal stem cells undergo a series of molecular and biochemical changes that lead to generation of the morphologically different strata of the epidermis ( Fig. 56.1 ). This chapter reviews key signaling pathways that control this terminal differentiation program, with discussion of inherited skin diseases that are caused by a failure of normal epidermal differentiation.
The Epidermis: Structure and Function
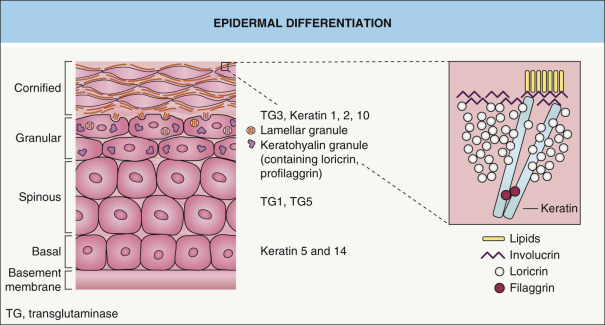
The epidermis, the outermost layer of the skin, functions as a barrier that provides the organism with protection against harmful substances and water loss. The barrier function is established during embryogenesis and maintained in mature epidermis through a continuous program of self-renewal. Under homeostatic conditions, this self-renewal is mediated by epidermal stem cells located in the basal layer of the interfollicular epidermis . Another population of stem cells, located in the bulge region of the hair follicles, contributes to the interfollicular epidermis in response to injury (see Ch. 2 ). Each division of an interfollicular stem cell gives rise to a daughter stem cell and a transit amplifying cell through a process termed asymmetric cell division. Transit amplifying cells undergo a few cell divisions before beginning a terminal differentiation program, which is associated with irreversible withdrawal from the cell cycle. Terminally differentiating keratinocytes detach from the basement membrane and move suprabasally, a process that is mediated by the inactivation of integrins, extracellular matrix receptors that anchor basal cells to the basement membrane (see Ch. 28 ). They are termed spinous keratinocytes when they reach the first suprabasal cell layer, and further differentiation as they move upward leads to the formation of granular keratinocytes.
Keratinocyte terminal differentiation culminates in the replacement of the plasma membrane with the cornified cell envelope (CE), a composite of several covalently cross-linked proteins ( Fig. 56.2 ) , most of which are encoded by genes located in the epidermal differentiation cluster on chromosome 1. Examples of CE components include involucrin, small proline-rich proteins (SPRR), XP-5/late envelope proteins (LEP), loricrin, cystatin, envoplakin, periplakin, elafin, repetin, filaggrin, S100 proteins, keratins, and desmosomal proteins. Note that mutations in some of the genes that encode these proteins can lead to skin disorders (see below). The extracellular surface of the CE is covered by lipids, which form the cornified lipid envelope (CLE) (see Ch. 124 ).
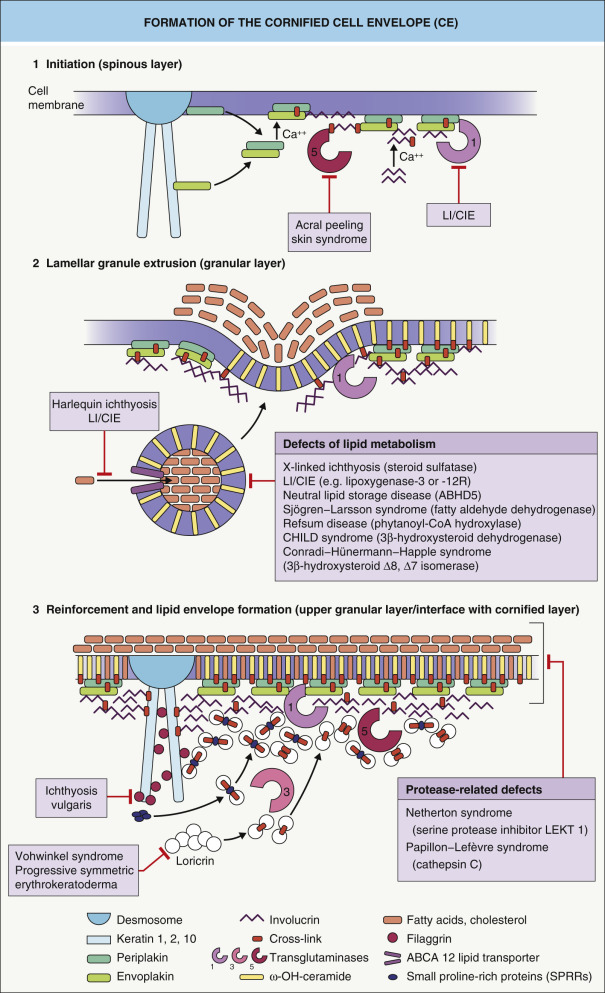
Keratinocytes that have assembled the CE/CLE and lost their nucleus and cytoplasmic organelles are termed corneocytes . These cell remnants constitute the stratum corneum, which serves to cover the body surface of terrestrial mammals. The CE and CLE are major contributors to the cutaneous water barrier. Failure or impairment of the water barrier leads to increased transcutaneous water loss and an increased susceptibility to infections, a major problem in premature infants and in disorders such as Netherton syndrome.
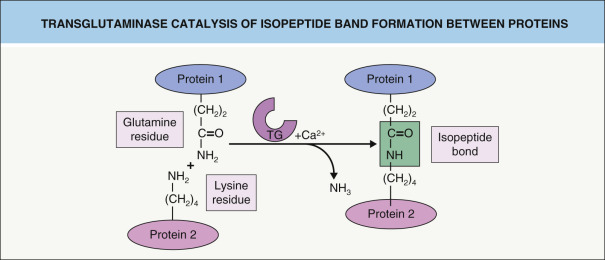
CE assembly begins within the upper spinous and granular cell layers, where proteins are chemically cross-linked, primarily by ε-(γ-glutamyl) lysine isopeptide bonds ( Fig. 56.3 ). This reaction is catalyzed by a class of enzymes termed transglutaminases (TGases). Loss-of-function mutations in the gene that encodes TGase 1 lead to lamellar ichthyosis and congenital ichthyosiform erythroderma, generalized skin disorders resulting from a failure to form proper CEs.
Because the CE is highly resistant to chemical attack, very harsh conditions (e.g. boiling the epidermal cells in buffers containing high concentrations of detergents) are required to isolate and purify CE proteins. One surprising finding is that a loss of individual CE components in knockout mice does not necessarily have a major impact on barrier function. One possible explanation for this unexpected result is the presence of compensatory gene regulatory pathways that maintain barrier function even in the absence of major CE components . Better characterization of these regulatory pathways will provide insights into how the barrier is established and maintained, hopefully leading to the development of new strategies for the treatment of diseases caused by impaired barrier function.
Keratin Intermediate Filaments
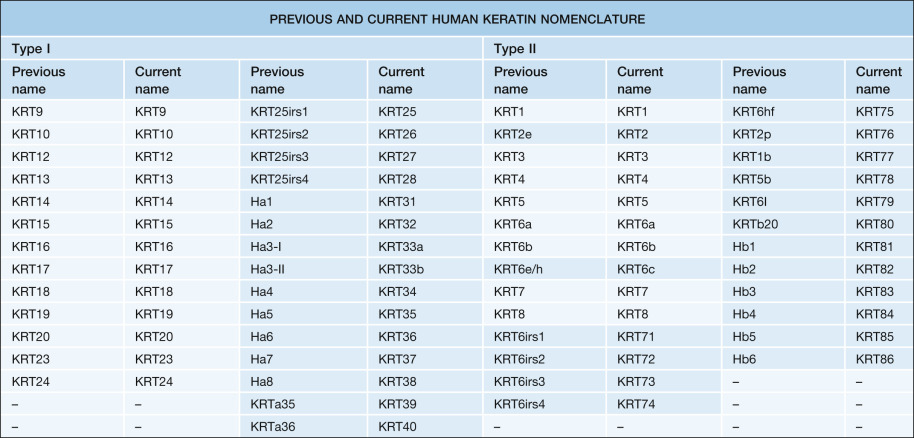
Keratin intermediate filaments provide resilience to keratinocytes, the most abundant cell type in the epidermis. Keratins represent the largest group of intermediate filament proteins ( Table 56.1 ). The current classification system includes 54 human keratin genes ( Table 56.2 ), which can be divided into three categories: (1) epithelial keratin genes; (2) hair keratin genes; and (3) keratin pseudogenes . Based upon their biochemical properties (e.g. isoelectric point, molecular weight), keratins are also classified as either type I (KRT9–KRT28, KRT31–KRT40) or type II (KRT1–KRT8, KRT71–KRT86) (see Table 56.2 ). Type I and type II keratins form obligatory heteropolymers (i.e. pairs composed of one keratin from each group) that become the basic building blocks of epithelial intermediate filaments ( Fig. 56.4 ).
| TYPES OF INTERMEDIATE FILAMENTS | |||||
|---|---|---|---|---|---|
| Type I | Type II | Type III | Type IV | Type V | Others |
| Keratins (acidic) | Keratins (basic) |
|
|
| Filensin Phakinin |
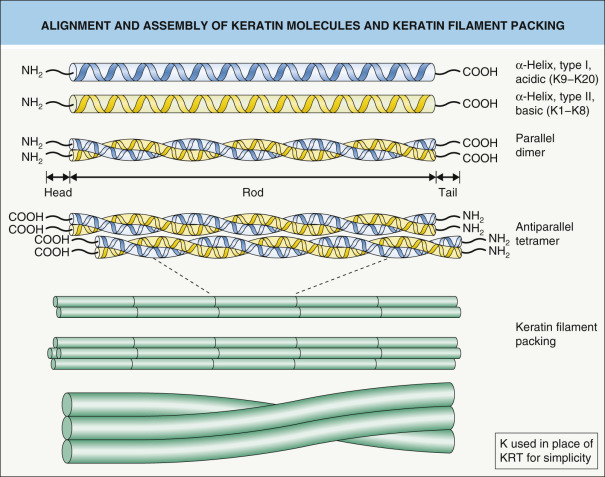
Keratins have a common domain structure that they share with other intermediate filament proteins . The central α-helical rod domain consists of 310 amino acid residues and is divided into four segments (1A, 1B, 2A, 2B), which are interrupted by three non-helical segments of variable lengths, termed linkers ( Fig. 56.5 ). The rod domain is composed of seven-residue amino acid sequence repeats (a-b-c-d-e-f-g) n termed “heptad repeats”, where positions “a” and “d” represent hydrophobic residues that are considered crucial for stabilization of the heterodimer. In the middle of the 2B domain, the heptad pattern is interrupted, giving rise to the “stutter”. This helical segment is highly conserved among intermediate filaments and does not participate in the formation of the coiled-coil dimer that forms the basic building block of intermediate filaments (see Fig. 56.4 ).
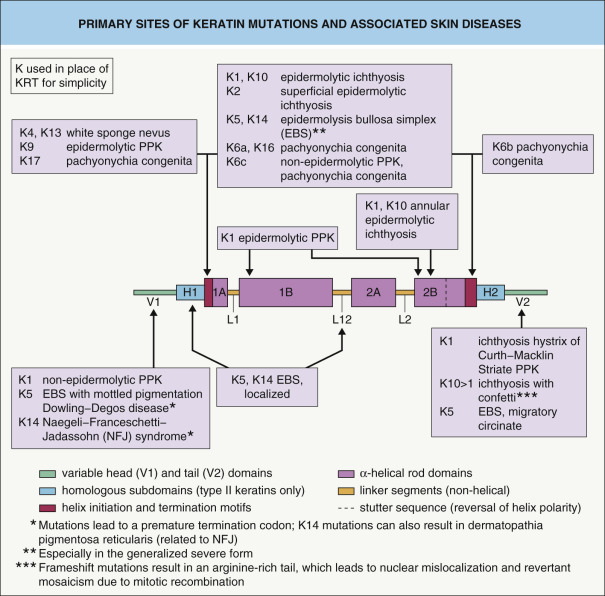
The beginning and end of the α-helical rod domain, referred to as the helix initiation and helix termination motifs, are highly conserved among the different keratins and play a pivotal role in keratin intermediate filament assembly (e.g. filament elongation). These helix boundary peptides represent genetic “hot spots” for mutations in many of the hereditary keratin disorders (see Fig. 56.5 ). The severity and other phenotypic features of genodermatoses caused by keratin defects often correlate with the position of the underlying mutation, with more severe disease resulting from mutations in the helix initiation and termination motifs (see Table 56.4 ).The head and tail regions that flank the rod domain are subdivided into extreme end (E), variable (V), and (in type II keratins) homologous (H) domains. Whereas epithelial keratins possess glycine- and serine-rich head and tail domains, these regions have a high content of cysteine and proline in hair keratins. Variations in the head and tail domains account for much of the diversity among different keratin proteins, which suggests that these domains play an important role in cell type-specific functions.
Keratins and Signaling
Keratins are markers for keratinocyte differentiation and are required to maintain epidermal integrity. However, intermediate filaments may also influence other basic cell functions, such as cell cycle progression, metabolic activity, apoptosis, and migration. The mechanisms by which keratins regulate these processes are not well understood. However, accumulating evidence suggests that intermediate filaments directly interact with and regulate several key cell signaling pathways, including TNF receptor 2-induced apoptosis, Src-mediated migration, and Akt/mTOR-related growth . Interestingly, deletion of the entire murine type II keratin gene cluster, which completely prevented the assembly of keratin intermediate filaments, resulted in an early embryonic lethal phenotype associated with growth retardation and defects in yolk sac hematopoiesis and vasculogenesis . This underscores the importance of keratins to cellular signaling and differentiation of a non-epithelial cell lineage.
Hair Keratins
The mature hair is the differentiation product of trichocytes and is found within the central core of the hair follicle. The anagen (growing) follicle is a complex structure consisting of eight distinct, concentrically arranged cell layers (see Ch. 68 ). The innermost compartment is the hair shaft, which is composed of a medulla, cortex, and one-layered cuticle; the latter serves as a protective coat for the hair shaft. The innermost living cell layer of the hair follicle is the inner root sheath (IRS); it surrounds the growing hair shaft and consists of the IRS cuticle as well as Huxley and Henle layers. The outermost compartment is the outer root sheath (ORS), which is continuous with the interfollicular epidermis. The companion layer is located between the IRS and the ORS.
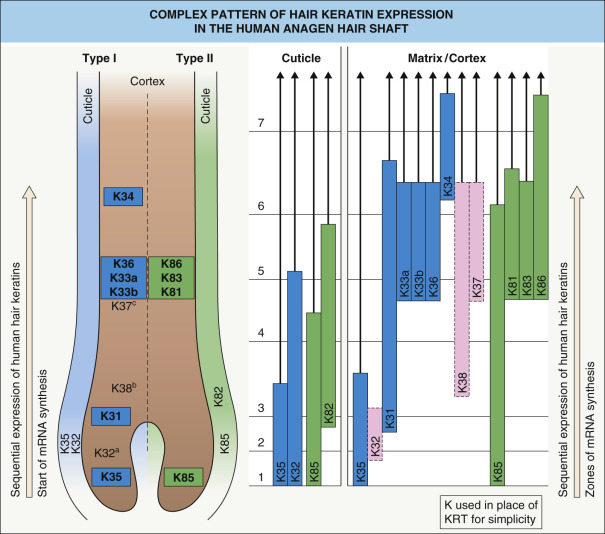
The medulla contains a mixture of epithelial keratins (KRT17, KRT75) and hair keratins (KRT33, KRT34, KRT36, KRT37, KRT81), whereas the cortex contains type I hair keratins (KRT31–KRT38) and type II hair keratins (KRT81, KRT83, KRT85 and KRT86) ( Fig. 56.6 ). Hair keratins KRT32 and KRT35 and their partners KRT82 and KRT85 are found in the cuticle, and the three IRS layers can be detected with antibodies against KRT71, KRT74, and KRT73. The epithelial keratins KRT5 and KRT14 are found throughout the full thickness of the ORS, while expression of KRT6, KRT16, and KRT17 is limited to the isthmus and the lower ORS. Additional keratins expressed in the ORS are KRT15 and KRT19.
Epidermal Differentiation
Epidermal differentiation is a tightly regulated process that involves the transformation of proliferating cells in the basal layer into the dead corneocytes of the stratum corneum. During this process, the keratins that are expressed are highly specific for the state of differentiation ( Fig. 56.7 ). The mitotically active keratinocytes in the basal compartment of the epidermis primarily express the keratin pair KRT5 and KRT14, with less abundant expression of KRT15. In the absence of KRT14, KRT15 can assemble with KRT5, thereby providing mechanical stability to the keratinocyte.
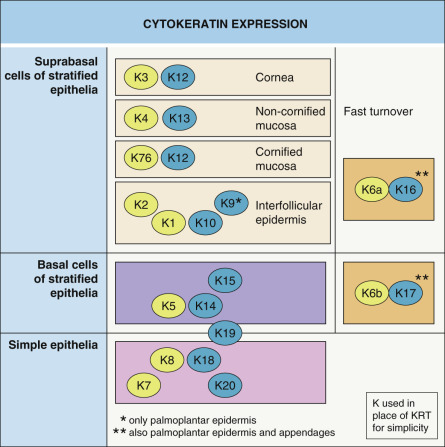
As keratinocytes move suprabasally to the spinous layer, they withdraw from the cell cycle. This process is associated with a down-regulation of KRT5 and KRT14 and an induction of the differentiation-specific keratins, KRT1 and KRT10. Further maturation of spinous keratinocytes into granular keratinocytes results in expression of KRT2, a reinforcement keratin. With further maturation, filaments containing the suprabasal keratins are bundled parallel to the surface and, eventually, keratinocytes lose their cytoplasmic organelles and differentiate into lifeless corneocytes that are shed into the environment.
Interestingly, a number of epidermal keratins have a more restricted anatomic distribution pattern. For example, KRT9 is specifically expressed in the suprabasal cells of palmoplantar skin. KRT6, KRT16, and KRT17 are expressed not only in the palmoplantar epidermis, but also in keratinocytes of the nail bed, hair follicle, and sebaceous and sweat glands. In addition, this group of keratins is rapidly induced by injury and ultraviolet radiation, as well as in hyperproliferative conditions .
Regulatory Pathways Involved in Epidermal Development and Differentiation
The regulatory pathways necessary for normal keratinocyte differentiation include those that: (1) establish and maintain basal keratinocytes; (2) initiate and execute terminal differentiation; and (3) form the stratum corneum. Examples of these pathways are discussed in more detail below.
Genes required for establishing and maintaining basal keratinocytes
The importance of the TP63 gene in epidermal development and differentiation became apparent following the generation of a knockout mouse model . The p63 protein encoded by this gene has at least six different isoforms that activate or repress transcription. Strikingly, p63-null mice fail to initiate epidermal morphogenesis and are born with a single-layered epithelium covering their bodies rather than a stratified epidermis. After birth, exposure of these mice to the environment leads to a rapid death due to dehydration. The failure of p63-deficient epithelial cells to adopt an epidermal fate highlights the critical role of p63 in specifying epidermal lineage.
The p63 protein regulates the transcription of multiple genes in basal keratinocytes. For example, p63 induces expression of epidermal keratins KRT5 and KRT14 while simultaneously repressing expression of KRT18, a keratin expressed in single-layered epithelia. Another key function of p63 is to help maintain the proliferative state of basal keratinocytes by repressing the expression of cell cycle inhibitors.
Genes required for terminal differentiation in mature epidermis
After basal keratinocytes have undergone a few rounds of cell division, they irreversibly withdraw from the cell cycle, move suprabasally, and become spinous keratinocytes. This process is also regulated by p63, in particular its ΔNp63α isoform . In this context, p63 synergizes with Notch signaling to induce expression of KRT1. Simultaneously, p63 mediates the cell cycle exit that is necessary for keratinocyte differentiation. The importance of p63 for normal epidermal development and differentiation is further underscored by the finding that p63 mutations underlie a subset of ectodermal dysplasias, which are characterized by abnormalities in the skin and skin appendages (see Ch. 63 ).
The Notch signaling pathway is also required for the formation of the spinous layer . In mouse models, ablation of Notch signaling resulted in the development of an extremely thin spinous layer, whereas constitutively active Notch signaling resulted in an expansion of the spinous layer. Notch activity promotes terminal differentiation by inducing KRT1 expression and mediating cell cycle withdrawal.
Ca 2+ in epidermal differentiation
In addition to signaling via p63 and Notch, an important trigger of keratinocyte differentiation is an increase in extracellular Ca 2+ concentration . In mature epidermis, there is a gradient of increasing extracellular Ca 2+ concentration from the basal layer to the cornified layer. Increasing the Ca 2+ concentration in the media of cultured keratinocytes can induce a differentiation program indistinguishable from that of keratinocytes in vivo , with successive expression of markers of keratinocyte terminal differentiation .
Several Ca 2+ -responsive proteins in the epidermis have key roles in the formation of the granular cell layer . For example, the protein kinase C (PKC) family of proteins is activated by Ca 2+ signaling and functions specifically in the transition from spinous to granular cells. PKC proteins have a dual role, contributing to the down-regulation of KRT1 and KRT10 expression as well as the induction of markers of granular keratinocytes such as loricrin, filaggrin, and transglutaminases.
In addition to the PKC family, other proteins that undergo conformational changes upon binding to Ca 2+ are expressed in mouse and human epidermis. Of these, the calcium-sensing receptor is specifically expressed in granular keratinocytes. Mice lacking the full-length form of the calcium-sensing receptor fail to properly form a granular layer, while overexpression of the calcium-sensing receptor in basal keratinocytes causes expanded spinous and granular cell layers .
Genes required for terminal differentiation in embryonic epidermis
Whereas the signaling pathways discussed above are critical for the formation of the spinous layer in postnatal skin, the molecular mechanisms that underlie the development of a spinous layer during epidermal morphogenesis appear to be different . One reason for this distinction is that during epidermal morphogenesis, basal keratinocytes do not directly differentiate into spinous keratinocytes. Instead, basal keratinocytes initially differentiate into intermediate keratinocytes that, like spinous cells, express KRT1. However, unlike spinous keratinocytes, intermediate keratinocytes still undergo proliferation. The intermediate cell layer exists only transiently during epidermal morphogenesis, and intermediate keratinocytes ultimately differentiate into spinous and granular keratinocytes, which then undergo further terminal differentiation.
The importance of the intermediate cell layer for normal epidermal development has been demonstrated by mouse models in which intermediate cells fail to mature into spinous and granular cells. Such a block in differentiation occurs in mice lacking expression of inhibitor of κB kinase-α (IKKα), interferon regulatory factor 6 (IRF6), or ovo-like 1 (Ovol1), as well as in mice expressing a mutant form of the 14–3–3σ protein . In all instances, an expanded intermediate cell layer develops, further terminal differentiation is disrupted, and the consequent failure to establish barrier function results in neonatal lethality.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree