Key points
- •
Describe the skin morphology.
- •
Intrinsic aging of the skin.
- •
Extrinsic aging of the skin.
- •
Mechanisms of repair of photodamage.
Morphology of skin
Most of the changes we associate with aged skin are due to chronic sun-damage. Chronological aging produces subtle changes in the skin that are often unnoticed upon histopathological examination, and hard to determine upon gross examination of skin. Before embarking on a description of the changes associated with chronic photoaging or chronological aging, one must understand the normal composition of skin.
Morphology of skin
Most of the changes we associate with aged skin are due to chronic sun-damage. Chronological aging produces subtle changes in the skin that are often unnoticed upon histopathological examination, and hard to determine upon gross examination of skin. Before embarking on a description of the changes associated with chronic photoaging or chronological aging, one must understand the normal composition of skin.
The epidermis
In contrast to the relatively hypo-cellular dermis, the epidermis is composed mostly of cells, tightly packed with the objective of keeping what is outside out, and what is inside in, all while communicating with the outside world. The basal keratinocytes create a parade of skin cells that move outward and gradually die to create the stratum corneum that forms the barrier to the outside world. One only needs to experience an ablative laser to appreciate the function of an intact epidermis. An intact epidermis prevents infection, prevents fluid loss and protects us from a variety of environmental insults. Keratinocytes are derived from the ectoderm and comprise the majority of the epidermis. Their main product is the protein keratin, a fibrillar protein that forms the outer layer of the superficial stratum corneum. Interspersed between the keratinocytes are melanocytes, one for every 10 basal keratinocytes, which are derived from the neural crest. These cells produce melanin pigment, a chemical that functions to protect us from solar radiation, and which functions as a free-radical scavenger. Melanin is of interest in that it absorbs maximally in the ultraviolet spectrum, but continues to absorb throughout the visible spectrum into the infrared. Melanin is transferred to keratinocytes via melanosomes, protecting the epidermis from damage due to ultraviolet radiation. Langerhans cells are the other major cell type present in the epidermis. They are typically found above the basal layer of the epidermis, comprise approximately 5% of the cells in the epidermis and are bone marrow derived cells that participate in antigen presentation and immune functions.
The junction between the dermis and epidermis is a very complex structure that serves to allow nutrients to diffuse from the dermis to the avascular epidermis, and permit communication between these two active skin compartments. The basement membrane zone provides a fertile footing for the basal keratinocytes and melanocytes to reside upon, and anchors the epidermis to the dermis, all while allowing nutrients, cytokines and other messengers including cells to traverse this barrier while it remains intact. Various extracellular matrix components such as collagens, glycosaminoglycans and other components make up the basement membrane zone. Communication is two-way, with the dermis providing sustenance to the epidermis, while the epidermis influences the superficial dermis, ensuring that the adjacent dermis provides a good base to support the overlying epidermis.
Adnexal structures punctuate the dermis and epidermis and consist of eccrine sweat glands, apocine glands, and hair follicles. These structures traverse the epidermis through the dermis and often into the underlying fat. They supply a source of epidermal cells to repopulate an area following wounding, including wounding by laser treatment. Melanocytes that live deep in the hair follicles are often protected from insults that impact the overlying epidermis, including laser treatment. Hair follicles cycle from an active growth phase to a resting phase, evidencing the stem cells that reside there.
The dermis
The dermis is derived from the mesoderm, with the exception of the nerve cells. Fibroblasts are the principal resident dermal cells, which manufacture most of the extracellular matrix material comprising the dermis. There is an extensive dermal vasculature which is divided into two main plexuses, one superficial and one deep. The vascular endothelial cells are quite competent at secreting cytokines that affect the adjacent dermis, and they permit circulating cells to take up residence in the vicinity of blood vessels further increasing their influence. Mast cells take up residence alongside blood vessels and direct wound healing and formation of extracellular matrix. In addition to blood vessels, the dermis is rich in nerves, some of which extend up into the epidermis. Nerves secrete numerous cytokines and affect the formation of extracellular matrix and cell function.
The fibroblast is the key cell that secretes the non-water materials that comprise the bulk of the dermis: the extracellular matrix. By far the most abundant extracellular matrix component is collagen, specifically type I collagen. Proteins and complex sugars are arranged into complex structures that form the bulk of the dermis. The main components of the dermis are proteins such as collagens and proteins comprising elastic fibers, as well as complex sugars such as hyaluronic acid and hybrids of proteins and sugars, the glycosaminoglycans.
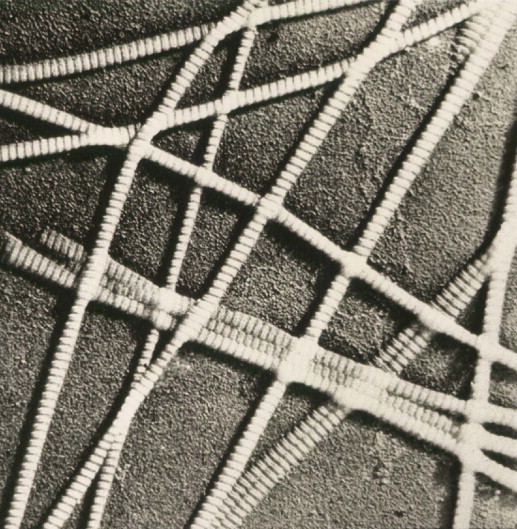
The fibrillar collagens form the majority of the dermal matrix and add strength and bulk to the skin, allowing it to resist outside forces ( Figure 1.1 ). Collagen is derived from the Greek ( kolla , glue; gennao , to produce; collagen, glue former), because when boiled collagen forms glue. The fibrillar collagens, types I and III are the most abundant collagen types in skin. Type I collagen forms 80% of the dry weight of the dermis, and is a major structural components all over the body. Type I collagen is comprised of polypeptide chains which are arranged in a triple helix. Two of the chains making up type I collagen are identical while the third chain is distinct. These are named the α1(I) chains, while the remaining chain is termed the α2(I) chain. Collagen production involves steps that take place intracellularly as well as in the extracellular space. Collagen is produced and then excreted in the extracellular space containing extensions which are non-helical and consist of N-terminal and C-terminal domains.
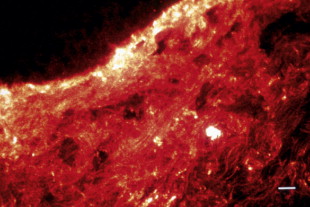
Collagen molecules are aligned very specifically, thus producing a banding pattern which can be seen by electron microscopy ( Figure 1.2 ). The bands occur at 68 nm intervals, and this is characteristic of collagen. These banding patterns occur due to the amino acid arrangement within the collagen molecule, and are thus a function of the molecular make-up of the collagen molecules. Every third amino acid in the collagen molecule is glycine, resulting in the Gly-X-Y sequence of the collagen molecule. The Gly moiety refers to glycine, while the X and Y positions are often occupied by the amino acids proline and hydroxyproline. At first, collagen is stabilized by non-covalent bonds or by disulfide bonds. Cross-links are then formed as the result of deamination of lysyl and hydroxylysyl residues.
Type III collagen is also called fetal collagen because it is abundant in fetal tissues. It is the second most abundant type of collagen in human skin. Although type III collagen makes up about half the total collagen in fetal skin, adult skin has a ratio of 6 : 1 type I to type III collagen. Type III collagen is also quite abundant in extensible tissues such as the gastrointestinal tract and arteries. Unlike type I collagen, which is composed of two different chains, type III collagen is composed of three identical α1(III) chains. Type III collagen is also arranged in a triple helical configuration.
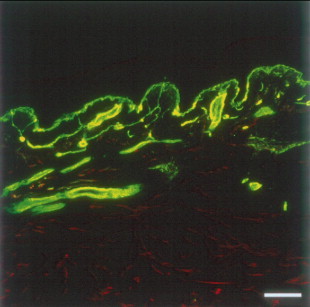
Type IV collagen is a major component of the complex basement membrane zone separating the epidermis and dermis ( Figure 1.3 ). It contains many regions that interrupt the triple helical configuration, and this may confer flexibility to type IV collagen. Like type I collagen, type IV collagen is composed of two types of collagen chains, two α1(IV) chains, and one α2(IV) chain. Type VI collagen has a very short triple helical segment with very large amino- and carboxy-terminal domains. Type VI collagen is composed of three separate polypeptide chains called the α1(VI), α2(VI), and α3(VI) chains.