This article is a review of cost considerations and outcomes pertaining to the use of acellular dermal matrices (ADMs) in reconstructive and aesthetic breast surgery. The history of the use of ADMs in breast surgery and all case series describing outcomes and use of ADM in breast reconstructive and aesthetic surgery are reviewed. Weighted averages for clinically relevant outcomes for reconstructive and aesthetic breast surgery are provided. Cost considerations of ADM use in breast surgery are described and as an example, a single institution’s experience with implementation of ADM into a preexisting breast surgery program, is used.
- •
A formal cost evaluation of the use of ADMs in breast surgery requires knowledge of the cost of the product, the cost of the surgery being performed, the cost of potential outcomes, costs of patient time expended for the intervention, costs associated with care giving, economic costs borne by employers, and costs borne by the rest of society.
- •
A true cost-utility analysis of the use of ADM in breast surgery requires high quality outcomes studies and standardized health utility states which are currently lacking from the literature.
- •
Preliminary cost minimization analysis in a Canadian center shows that conversion of traditional 2-stage total submuscular alloplastic reconstruction to 1-stage alloplastic reconstruction using ADM results in cost savings as a result of elimination of the cost of the tissue expander and second-stage surgery.
- •
On short-term follow-up, addition of ADMs to alloplastic breast reconstruction (1-stage and 2-stage outcomes combined) seems to result in favorable outcomes for capsular contracture, late revision, and total implant loss compared with traditional 2-stage reconstruction. Rates of mastectomy flap necrosis, seroma, and infection are slightly higher than those seen in the Allergan and Mentor core studies for two-stage non-ADM reconstruction.
- •
In the setting of aesthetic surgery ADMs may prove cost-effective if a decreased subsequent revision rate with use of ADM at the first revision surgery can be proven.

Economic analyses in surgery are required to show the relative benefits of a new or alternative intervention. The use of autologous dermal grafts in breast reconstructive surgery has been described since 1979. Because of variability in revascularization and donor site morbidity, acellular dermal matrices (ADMs) were developed. ADMs have shown promise for use in both aesthetic and reconstructive breast surgery, but concerns about their use remain because of the significant costs associated with these products.
Economic analysis in surgical literature
The use of ADMs in breast surgery is relatively new. Since 2001, there have been 27 published series outlining surgical technique and preliminary outcomes in patients undergoing breast reconstruction and 9 series describing the use of ADMs in aesthetic breast surgery ( Tables 1 and 2 ). One formal meta-analysis, 1 systematic review, and 2 cost analyses have been published (see Table 1 ).
| Author, Location | Title | Journal | Year | Sample Size | 1-Stage vs 2-Stage | Sample Population | Design | Surgical Technique |
|---|---|---|---|---|---|---|---|---|
| Antony et al, United States | ADM implantation in 153 immediate two-stage tissue expander breast reconstructions: determining the incidence and significant predictors of complications | Plast Reconstr Surg | 2010 | 96 patients, 153 breasts | 2-stage Immediate | Median age: 44.5 y (range 28–79 y). 30 breasts received preoperative or postoperative radiation | RCS | Standard a via inframammary fold incision. No elevation of SA |
| Ashikari et al, United States | Subcutaneous mastectomy and immediate reconstruction for prevention of breast cancer for high-risk patients | Breast Cancer | 2008 | 65 patients, 130 breasts | 1-stage Immediate | Mean age 44 y (range: 25–64 y) All prophylactic | RCS | Standard |
| Austen et al, United States | A simplified technique for single stage breast reconstruction | New England Surg Soc Meeting abstract | 2006 | 25 patients, 35 breasts | 1-stage Immediate | Mean age: 47 y Radiation data not included | RCS | Standard with SA elevation |
| Becker et al, United States | AlloDerm ® vs DermaMatrix ® in immediate expander-based breast reconstruction: a preliminary comparison of complication profiles and material compliance | Plast Reconstr Surg | 2009 | 30 patients, 50 breasts | 2-stage Immediate | Mean age: 52 y Radiation data not included | RCS | Standard with prepectoralis fascia preserved |
| Bindingnavele et al, United States | Use of ADM and tissue expansion in postmastectomy breast reconstruction | J Plast Reconstr Aesth Surg | 2007 | 41 patients, 65 breasts | 2-stage Immediate | Mean age: 50 y (range: 31–69 y) 5 patients radiated postoperatively | RCS | Standard with no SA or RA fascia elevation |
| Breuing & Warren, United States | Immediate bilateral breast reconstruction with implants and inferolateral AlloDerm ® slings | Ann Plast Surg | 2005 | 10 patients, 20 breasts | 1-stage Immediate | N/D | RCS/ Technique | Standard |
| Breuing & Colwell, United States | Inferolateral AlloDerm ® hammock for implant coverage in breast reconstruction | Ann Plast Surg | 2007 | 43 patients, 67 breasts | Mix: 1-stage, 2-stage, delayed, immediate Revisional | Mean age: 46 y | RCS | Standard with no SA or RA fascia elevation |
| Chen et al, United States | A novel cost-saving approach to the use of ADM (AlloDerm ® ) in postmastectomy breast and nipple reconstructions | Plast Reconstr Surg | 2010 | 13 patients, 23 breasts | 2-stage | Mean age: 55 y | RCS | Cost-saving approach with banking of AlloDerm ® for later use in nipple reconstruction |
| Chun et al, United States | Implant-based breast reconstruction using ADM and the risk of postoperative complications | Plast Reconstr Surg | 2010 | 269 breasts | 1-stage 2-stage Immediate | Mean age: 46 y | RCS | Standard |
| De Blacam et al, United States | Cost analysis of implant-based breast reconstruction with acellular dermal matrix | Ann Plast Surg | 2011 | N/A | N/A | N/A | Cost analysis | N/A |
| Derderian et al, United States | Wise-pattern breast reconstruction modification using AlloDerm ® and a vascularized dermal-subcutaneous pedicle | Ann Plast Surg | 2009 | 20 patients | 1-stage Immediate | Mean age: N/D 5 patients radiated preoperatively | RCS | Standard with variable SA elevation for lateral implant coverage |
| Ellsworth et al, United States | Breast implant salvage with the use of acellular dermal matrix after partial mastectomy | Plast Reconstr Surg | 2010 | 1 patient | 1-stage | Age: 60 y Previous bilateral subglandular augmentation with right-sided breast cancer Postreconstruction radiation | Case report | AlloDerm ® used to reinforce lateral capsule |
| Gamboa-Bobadilla, United States | Implant breast reconstruction using ADM | Ann Plast Surg | 2006 | 11 patients, 13 breasts | 1-stage Immediate and delayed | Median age: 58 y (range: 41–68 y) | RCS | Standard |
| Jansen & Macadam, Canada | The use of AlloDerm ® in postmastectomy alloplastic breast reconstruction: Part I. A systematic review | Plast Reconstr Surg | 2011 | N/A | 1-stage | N/A | Systematic review | N/A |
| Jansen & Macadam, Canada | The use of AlloDerm ® in postmastectomy alloplastic breast reconstruction: Part II. A cost analysis | Plast Reconstr Surg | 2011 | N/A | N/A | N/A | Cost analysis | N/A |
| Lanier et al, United States | The effect of ADM use on complication rates in tissue expander/implant breast reconstruction | Ann Plast Surg | 2010 | 75 patients ADM group 52 patients non-ADM group | 2-stage Immediate | Mean age: 51 y | RCS | Standard |
| Liu et al, United States | Postoperative complications in prosthesis-based breast reconstruction using ADM | Plast Reconstr Surg | 2011 | 192 patients, 266 breasts | 1-stage 2-stage Immediate | N/D | RCS | Standard |
| Losken, United States | Early results using sterilized ADM (neoform) in postmastectomy tissue expander breast reconstruction | Plast Reconstr Surg | 2009 | 22 patients, 31 breasts | 2-stage Immediate | Mean age: 48 y | PCS | Standard |
| Margulies et al, United States | Total skin-sparing mastectomy without preservation of the nipple-areola complex | Am J Surg | 2005 | 31 patients, 50 breasts | 2-stage Immediate | Mean age: 42 y | RCS | Standard |
| Nahabedian, United States | AlloDerm ® performance in the setting of prosthetic breast surgery, infection, and irradiation | Plast Reconstr Surg | 2009 | 76 patients, 100 breasts | Mix: revision reconstruction, revision augmentation | Mean age: 46 y | RCS | Standard |
| Namnoum, United States | Expander/implant reconstruction with AlloDerm ® : recent experience | Plast Reconstr Surg | 2009 | 20 patients, 29 breasts | 2-stage Immediate | Mean age: N/D Radiation data not included | RCS | Standard with no SA elevation |
| Newman et al, United States | The true incidence of near-term postoperative complications in prosthetic breast reconstruction using human ADMs: a meta-analysis | Aesth Plast Surg | 2011 | 789 breasts | 1-stage 2-stage Immediate | N/D | Meta-analysis | 12% overall infection rate on meta-analysis of 12 studies |
| Parikh, United States | Immediate breast reconstruction with tissue expanders and alloderm | Plast Reconstr Surg | 2006 | 43 patients, 58 breasts | 2-stage | N/D | RCS | Standard |
| Preminger et al, United States | The influence of AlloDerm ® on expander dynamics and complications in the setting of immediate tissue expander/implant reconstruction: a matched-cohort study | Ann Plast Surg | 2008 | 45 patients | 2-stage Immediate | Mean age: N/D 6 of 90 patients from both cohorts were radiated preoperatively | RCS matched for TE, RTx, and Dx | Standard with no SA or RA fascia elevation |
| Salzberg, United States | An 8-year experience of direct-to-implant immediate breast reconstruction using human ADM (AlloDerm ® ) | Plast Reconstr Surg | 2011 | 260 patients, 466 breasts | 1-stage Immediate | Mean age: N/D 21 breasts were radiated postoperatively | RCS | Standard |
| Salzberg, United States | Nonexpansive immediate breast reconstruction using human acellular tissue matrix graft (AlloDerm ® ) | Ann Plast Surg | 2006 | 49 patients, 76 breasts | 1-stage Immediate | Mean age: N/D Radiation data not included | RCS | Standard |
| Sbitany et al, United States | Acellular dermis-assisted prosthetic breast reconstruction vs complete submuscular coverage: a head-to-head comparison of outcomes | Plast Reconstr Surg | 2009 | 50 patients, 92 breasts | 2-stage | Mean age 48 y (SD: 8.6) 6 breasts received postoperative radiation | RCS | Standard |
| Spear et al, United States | Acellular dermis-assisted breast reconstruction | Aesth Plast Surg | 2008 | 43 patients, 58 breasts | 2-stage Immediate | Mean age at stage 1: 50 y (range 36–66 y) 3 breasts were radiated preoperatively 8 breasts were radiated postoperatively | RCS | Standard with no SA, RA, or pectoralis minor elevation |
| Topol et al, United States | Immediate single-stage breast reconstruction using implants and human ADM with adjustment of the lower pole of the breast to reduce unwanted lift | Ann Plast Surg | 2008 | 23 patients, 35 breasts | 1-stage Immediate | Mean age: N/D Radiation data not included | PCS | Standard with no SA elevation |
| Uflacker & Janis, United States | The ADM dermal matrix in the correction of visible parasternal deformities after breast reconstruction | Plast Reconstr Surg | 2010 | 1 patient | N/A | Age: 38 y Visible implants in the upper pole | Case report | AlloDerm ® used in upper pole to camouflage implant visibility |
| Zienowicz & Karacaoglu, United States | Implant-based breast reconstruction with allograft | Plast Reconstr Surg | 2007 | 24 patients, 30 breasts | 1-stage Immediate | Mean age: 47 y (SD: 8) Radiation data not included | PCS | Standard with SA elevation 100% adjustable Spectrum implant |
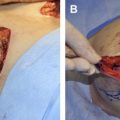
a Standard technique: after the completion of the skin-sparing mastectomies, the inferolateral origin of the pectoralis major muscle is elevated off the anterior chest wall. Using blunt and electrocautery dissection, a subpectoral pocket is developed to the extent of the previously marked perimeter of the breasts. After satisfactory creation of a subpectoral pocket, an appropriately sized piece of ADM is rehydrated in normal saline according to the manufacturer’s recommendations. The ADM is sewn along the chest wall at the level of the previously marked inframammary fold and curved laterally and cephalad along the lateral border of the breast perimeter to recreate the natural curvilinear origins of the inferolateral aspect of the detached pectoralis muscle and breast mound unit.
| Author, Location | Title | Journal | Year | Sample Size | Design | Cost | Technique |
|---|---|---|---|---|---|---|---|
| Baxter, United States | Intracapsular allogenic dermal grafts for breast implant-related problems | Plast Reconstr Surg | 2003 | 10 patients | RCS/Technique paper | N/D | Use of ADM to correct symmastia, bottoming out and rippling in both aesthetic and reconstructive cases |
| Brown et al, United States | Preventing the “bottoming out” and “star-gazing” phenomena in inferior pedicle breast reduction with an acellular dermal matrix internal brassiere | Aesth Plast Surg | 2010 | 27 patients | RCS | N/D | Use of ADM to prevent bottoming out of the breast after Wise-pattern breast reduction |
| Curtis et al, United States | Use of AlloDerm ® for correction of symmastia | Plast Reconstr Surg | 2010 | 1 patient | Case report | N/D | Use of medial C-shaped slings of AlloDerm ® bilaterally to correct symmastia |
| Colewell & Breuing, United States | Improving shape and symmetry in mastopexy with autologous or cadaveric dermal slings | Ann Plast Surg | 2008 | 10 patients | RCS | N/D | Use of an internal ADM sling to support the breast in mastopexy |
| Duncan, United States | Correction of implant rippling using allograft dermis | Aesth Surg J | 2001 | 34 patients | RCS | N/D | Segmental capsulectomy in the area underlying the rippling to produce a vascularized recipient site for allograft. Allograft was sutured along the inferolateral pectoral muscle edge, placement of implant, and suturing of allograft to the inframammary crease |
| Hartzell et al, United States | The use of human acellular dermal matrix for the correction of secondary deformities after breast augmentation: results and costs | Plast Reconstr Surg | 2010 | 23 patients, 38 breasts | RCS | Average: $3536–$4856 per breast | Use of human ADM to correct surface irregularities and implant malposition |
| Maxwell & Gabriel, United States | Use of the acellular dermal matrix in revisionary aesthetic breast surgery | Aesth Surg J | 2009 | 78 patients | RCS | N/D | Use in revision augmentation and revision augmentation mastopexy |
| Mofid & Singh, United States | Pocket conversion made easy: a simple technique using alloderm to convert subglandular breast implants to the dual-plane position | Aesth Surg J | 2009 | 10 patients, 20 breasts | RCS | N/D | Revision breast augmentation with conversion of subglandular implants to the dual plane position using AlloDerm ® |
| Spear et al, United States | Acellular dermal matrix for the treatment and prevention of implant-associated breast deformities | Plast Reconstr Surg | 2011 | 52 patients, 77 breasts | RCS | N/D | Use of AlloDerm ® to correct surface irregularities and implant malposition and to treat capsular contracture |
Economic evaluation in the surgical literature is rare. A systematic review of the number of published cost-utility studies over a 6-year period revealed 649 publications in the medical literature compared with 57 in the surgical literature. These investigators state: “the lack of formal economic evaluation in the surgical literature likely stems from the complexity of the subject and (with few exceptions) the lack of surgeon training in clinical research methodology.” In current health care systems, surgeons should make decisions based not only on patient outcomes but also on the cost of a particular surgical intervention. Cost analysis allows for comparison of costs of competing medical interventions and their corresponding health outcomes states. Such analyses can aid health policy decision makers in weighing the benefits and risks of a new surgical technique and to decide whether the benefits provided by a new procedure warrant potential additional costs.
Four different types of economic evaluation are used in the medical literature :
- 1.
Cost-minimization analysis
- 2.
Cost-effectiveness analysis
- 3.
Cost-utility analysis
- 4.
Cost-benefit analysis.
Cost-minimization analysis is used to compare 2 alternatives that have comparable outcomes but different costs. In this type of analysis, cost in dollars is the only reported metric.
A cost-effectiveness analysis reports outcomes in physical units, such as lives saved or successful surgeries or cost per outcome.
A cost-utility analysis is similar to the cost-effectiveness analysis, but reports outcomes in cost per quality-adjusted life-year (QALY). A QALY is a value determined by the number of extended life-years in a particular health (utility) state attributable to a particular intervention, in which the utility is the preference of an individual or society for the health state.
A cost-benefit analysis attaches a value to the cost-effectiveness of an intervention by asking health care consumers what they would be willing to pay for a certain health outcome.
The use of ADM in breast surgery is relatively new, with the first reports published in 2001 and 2003. Because of its relative newness in breast surgery, utilities for health states in patients who have had breast surgery living with and without ADM have not been standardized. A formal cost-utility analysis adhering to the guidelines set out by the Panel on Cost-Effectiveness in Health and Medicine comparing breast surgery (aesthetic and reconstructive) with and without the use of ADM is therefore difficult. Only 2 studies have been published that have evaluated the cost of implementation of ADM into breast surgery. Jansen’s cost-minimization analysis compared traditional 2-stage tissue expander/implant breast reconstruction with 1-stage breast reconstruction using ADM. Outcomes were estimated from a previous systematic review. De Blacam’s cost-minimization analysis compared 2-stage tissue expander/implant reconstruction, 1-stage reconstruction using ADM, and 2-stage tissue expander/implant reconstruction using ADM. Outcomes were estimated from weighted averages from a literature review. Inherent limitations of this type of cost analysis include the variability in reporting of the included articles. The probabilities of clinically relevant outcomes after breast surgery using ADMs are difficult to estimate because all published studies have been case series, the largest including 260 patients who had had single-stage breast reconstruction. Ideally, outcome probabilities should be estimated from an average of results from several high-quality studies.
Development of ADM use in breast surgery
ADMs have been used successfully across multiple specialties and for varied applications including the resurfacing of burn injuries, gingival recession, abdominal hernia repair, aesthetic facial contouring, eyelid reconstruction, tympanic membrane reconstruction, and dural replacement.
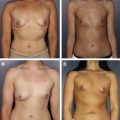
In 2001, the first report of ADM (AlloDerm ® , LifeCell Corporation, Branchburg, NJ, USA) use was documented in aesthetic breast surgery in a series of 34 patients with surface irregularities after breast augmentation. Applications in this field expanded to include correction of symmastia, implant malposition, and capsular atrophy.
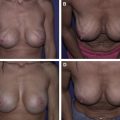
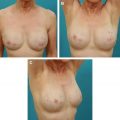
In 2005, Breuing and Warren reported the first use of an ADM in breast reconstruction. In their series, 10 patients underwent bilateral mastectomy with immediate reconstruction using an implant and inferior AlloDerm ® sling. This series marked the introduction of ADM-assisted single-stage breast reconstruction. Benefits of ADM use in single-stage breast reconstruction such as improved lower pole projection, additional implant coverage, and maximization of the breast skin envelope in immediate reconstruction were soon recognized, and in 2007 Bindingnavele and colleagues reported the first series of patients to undergo 2-stage reconstruction with AlloDerm ® . Purported benefits of this application include decreased postoperative pain, expedited expansion, and elimination of elevation of the serratus anterior lateral muscle sling. Use of ADM in breast reconstruction surgery evolved from the initial applications in breast aesthetic surgery and has now become a standard option in alloplastic reconstructive surgery.
ADM Products Expanding
Since these early reports, ADM use in breast reconstructive and aesthetic surgery has become common. The introduction of human-derived AlloDerm to the ADM market in 1994 was followed by development of ADMs from both allogenic and xenogenic sources. The most commonly used and studied matrix is AlloDerm ® , but recently developed products including FlexHD ® (Ethicon [Johnson & Johnson], San Angelo, TX, USA), DermaMatrix ® (Synthes, Inc., West Chester, PA, USA), AlloMax™ (Davol, A BARD Company, Warwick, RI, USA), SurgiMend ® (TEI Biosciences, Boston, MA, USA), and Strattice™ (LifeCell Corporation, Branchburg, NJ, USA) are now being used for the same breast-related indications. Each product differs in terms of source, shelf life, rehydration time, sterility, and cost.
Advantages and Disadvantages
Increased surgeon awareness, perceived benefits, and early favorable results fuel the demand for ADM by patients undergoing breast surgery. Steady surgical procedure volumes continue to support demand for the use of these products. However, the potential advantages must be weighed against the increased costs. Budgetary concerns among health care facilities may lead to pressure on companies to lower product pricing or to negotiate discounts on single items as well as bundled product purchases to curb costs. This situation may offset part of the added cost of these products, but as usage gains momentum it is necessary for surgeons to complete rigorous assessment of outcomes to support the common perception that ADM use in breast surgery decreases the need for revisional surgery and thus may prove cost-effective.
Scientific considerations in use of ADM s in breast reconstruction
Biocompatible dermal matrices are produced from allogenic and xenogenic sources by removal of the epidermis and all of the cells of the dermis. The most extensively studied ADM that has been used in breast surgery is AlloDerm ® . AlloDerm ® is processed via a proprietary deoxycholate/freeze-drying process in which components including types IV and VII collagen, elastin, and laminin are preserved within the residual dermal matrix. The ideal ADM lacks immunogenic epitopes and, therefore, evades rejection, absorption, or extrusion. In addition, collagen-dermal scaffolds have the potential to be revascularized, repopulated by fibroblasts, and replaced by autogenous collagen.
Revascularization and Encapsulation
Numerous histologic analyses have shown the potential for human-derived and xenogenic-derived ADM to become incorporated into the host with evidence of revascularization. There is also evidence to suggest that some ADMs resist encapsulation, which may be attributable to reduced numbers of myofibroblasts and inflammatory cytokines at the ADM/host interface. It is postulated that the host may not recognize human-derived ADM as foreign, and therefore reacts with a muted inflammatory response. This hypothesis may be reflected clinically by reduced capsular contracture rates after ADM-assisted breast surgery. Several series have supported this concept, with low rates of capsular contracture on early follow-up.
Postoperative Breast Irradiation
Few clinical studies have examined the use of ADM in the setting of previous or postoperative breast irradiation. Expanding indications have resulted in increasing numbers of patients who have had breast reconstruction requiring radiotherapy. Multiple investigators have reported higher complication rates, including capsular contracture, infection, and reconstructive failure in tissue expander/implant reconstruction associated with either prereconstruction or postreconstruction radiation. Experimental studies have shown favorable results in terms of the ability of ADMs to become vascularized after application to an irradiated bed or after radiation of the ADM itself. Komorowska-Timek and colleagues have specifically studied the behavior of ADM around implants in an irradiated, animal model. They found decreased inflammation and pseudoepithelium formation in the presence of an ADM.
Costs of Complications
Differences in capsular contracture rates and outcomes in the setting of irradiation further influence the cost-benefit of use of ADMs in breast surgery. Clinical series examining the effect of radiation in the setting of ADM-assisted breast surgery are scarce and results are conflicting. Antony and colleagues have shown favorable results in the setting of irradiation; however, Spear and colleagues have shown a higher rate of complications. Further basic science and clinical studies are required to evaluate the effects of ADM on capsule formation and behavior in the setting of radiation.
Cost considerations of ADM s
ADMs come at a significant additional line item cost to the health care system. Introduction of ADMs into practice requires a consideration as to whether the benefits provided are worth spending the limited resources available at a particular institution. This type of evaluation can be conducted from several different viewpoints, such as that of the hospital, the primary payer, or society. The Panel on Cost-Effectiveness in Health and Medicine recommends a societal perspective when conducting cost analyses. This perspective takes into account everyone affected by a particular intervention, whether or not people affected are those who received the intervention.
Proof of Clinical Effectiveness
Before performing an economic analysis, the clinical effectiveness of a new intervention should be proven. Pooling results from multiple randomized controlled trials (RCTs) is the gold standard when evaluating outcomes. Because the number of RCTs comparing surgical interventions is low, surgeons must use data from observational studies with a lower level of evidence and then decide if outcomes from such studies are generalizable to their own practice. In the case of breast surgery, several potential outcomes can be measured, including short-term and long-term complications, aesthetic outcome, patient satisfaction, and patient-reported quality of life. The estimation of costs and effects relies on appropriate measurements. The price of a particular intervention or surgery differs between locations, and thus a particular analyst at 1 institution may reach a different conclusion on cost-effectiveness compared with an analyst in a different country. The estimated cost of a procedure in the United States (multiple third-party payers) is likely not applicable to a surgeon in Canada with 1 surgical fee (1 party payer), which may be a fraction of the equivalent surgical fee in the United States. In addition, when using the societal perspective the cost or charge of a surgical intervention, resource costs such as nursing time and investigations, as well as the cost to other people affected by the intervention must all be taken into account.
Formal Cost Evaluation
A formal cost evaluation of the use of ADMs in breast surgery requires knowledge of the cost of the product ( Table 3 ), the cost of the surgery being performed, the cost of potential outcomes, costs of patient time expended for the intervention, costs associated with care giving, economic costs borne by employers, and costs borne by the rest of society, also known as friction costs, associated with absenteeism. In addition, it is essential to determine the probabilities of all relevant outcomes and whether outcomes with the use of ADMs differ from outcomes with the alternative techniques. With only 27 published series on the use of ADMs in breast reconstructive surgery and 9 published series in breast aesthetic surgery, it is difficult to know with certainty if outcomes using ADMs are better, worse, or equivalent to outcomes without the use of ADMs. Formal RCTs evaluating surgery with and without ADMs are required to make this determination. A summary of current published outcomes with use of ADMs in both reconstructive and aesthetic breast surgery follows.