INTRODUCTION
Chronic venous disorders (CVD) of the lower extremities are common problems caused by venous hypertension. In most cases, CVD are primarily the result of incompetent valves in one or more of the saphenous veins and their primary tributaries. In patients with saphenous vein incompetence, regardless of CVD stage, treatment begins with the elimination of these incompetent pathways. Endovenous thermal ablation (EVTA), using either laser or radiofrequency (RF), is the most common method used to accomplish this. In this chapter we will review EVTA in detail.
The EVTA techniques currently available are endovenous laser ablation (ELA) (multiple wavelengths and various laser fiber kits marketed by multiple vendors, Box 8.1 ) and RF ablation (ClosureFast [CF]) (7 F and a new 6 F version, Medtronic, Minneapolis, MN; Venclose, BD Interventional, Franklin Lakes, NJ; Veineo, F Care Systems, Antwerp, BE) ( Box 8.2 ). The first EVTA tool was radiofrequency ablation or RFA (Closure and later Closure Plus, VNUS medical technologies). EVTA with RFA was extensively investigated and utilized successfully but was recently replaced with CF, which is the most commonly used method. Laser was originally performed with 600-µm bare-tipped fibers. However, manufacturers have developed fibers ranging from 400 to 1000 µm and utilized various tips, including bare, a fiber with a glass cap on the tip surrounded by a metal cylinder around the cap and the distal 2 cm of the fiber, and one- and two-ring light emitting radially firing fibers, the latter being popular in Europe. Currently, there is limited evidence comparing the effectiveness and side effects of the different configurations.
Box 8.1
Endovenous Laser Wavelengths, Commercially Available
-
810 nm no longer produced by AngioDynamics, but available
-
940 nm FlexiPulse, Dornier MedTech America, Inc., USA
-
980 nm Ceralas D980 Biolitec, Inc., USA
-
1320 nm CTEV, CoolTouch, USA
-
1470 nm VenaCure1470, AngioDynamics, USA
-
1470 nm LiteBeam+, Dornier MedTech America, Inc., USA
-
1470 nm TVS 1470 (15 W), Total Vein Systems, USA
-
1470 nm TVS 1470 Nano (7 W), Total Vein Systems, USA
-
1470 and 1940 nm Biolitec AG with ELVesRadial2Ring fiber (internationally available)
Box 8.2
Radiofrequency Devices, Commercially Available
-
ClosureFast (CF) (7 F and a new 6 F version), Medtronic, Minneapolis, MN
-
Venclose, BD Interventional, Franklin Lakes, NJ
-
Veineo, F Care Systems, Antwerp, BE
Both ELA and CF are effective treatments for saphenous incompetence. Both procedures are associated with high success and low complication rates, and most guidelines in the vascular community strongly recommend it based on strong evidence (Grade 1A). The underlying goal for all EVTA procedures is to deliver sufficient thermal energy to the wall of an incompetent vein segment to produce irreversible occlusion, fibrosis, and ultimately disappearance of the vein. The procedures are generally performed in an ambulatory setting with local anesthetic and typically do not require sedation. Following treatment, patients are fully ambulatory, and the recovery time is short.
PATIENT SELECTION
The selection of candidates for EVTA involves a directed history and physical examination as well as a duplex ultrasound (DUS) examination. The details of the clinical and DUS examination have been discussed in other chapters. Indications for endovenous treatment are listed in Box 8.3 , and the contraindications to endovenous treatment are listed in Box 8.4 .
Box 8.3
Endovenous Thermal Ablation Indications
-
I.
Symptoms of venous insufficiency affecting quality of life
-
i.
Aching
-
ii.
Throbbing
-
iii.
Heaviness
-
iv.
Fatigue
-
v.
Restlessness
-
vi.
Night cramps
-
vii.
Pruritis
-
viii.
Spontaneous hemorrhage
-
i.
-
II.
Skin changes associated with chronic venous hypertension
-
i.
Corona phlebectasia, eczema, and pigmentation
-
ii.
Lipodermatosclerosis
-
iii.
Atrophie blanche
-
iv.
Healed or active ulceration
-
v.
Edema
-
vi.
Superficial phlebitis (SVT) in varicose veins
-
i.
-
III.
Cosmetic (restorative) concerns
-
IV.
Anatomical indications
-
i.
Significant reflux documented on duplex ultrasound examination (reflux >0.5 s)
-
ii.
Straight vein segment
-
iii.
Intra- or epifascial vein segment meeting other anatomical criteria that can be pushed away from the skin with tumescent anesthetic
-
iv.
Reflux responsible for venous hypertension leading to the clinical abnormalities
SVT. Superficial venous thrombosis.
INR, International Normalized Ratio.
-
i.
-
V.
Ambulatory patient without contraindication
Box 8.4
Relative Contraindications to Endovenous Thermal Ablation (EVTA)
-
•
Pregnancy or nursing female patients (concerns related to anesthetic use and heated blood effluent which may pass through the placenta to the fetus)
-
•
Obstructed deep venous system inadequate to support venous return after EVTA
-
•
Liver dysfunction or allergy making it impossible to use a local anesthetic (cold saline may be useful as an alternative)
-
•
Allergy to both amide and ester local anesthetics (cold saline may be an alternative)
-
•
Severe uncorrectable coagulopathy (EVTA is anecdotally safe with Warfarin use if INR <2)
-
•
Severe hypercoagulability syndromes (where risk of treatment outweighs potential benefits despite prophylactic anticoagulants)
-
•
Inability to wear compression stockings secondary to inadequate arterial circulation, hypersensitivity to the compressive materials or musculoskeletal or neurological limitations to donning the stocking itself
-
•
Inability to adequately ambulate postprocedure
-
•
Sciatic vein reflux
-
•
Thrombus or synechiae in the vein or tortuous vein making passage of an endovenous device impossible (unless multiple access points are chosen)
Treatment of incompetent superficial truncal veins (saphenous veins) in patients with previous deep vein thrombosis requires a careful assessment of the adequacy of the patent segments of the deep venous system. It also requires a risk stratification as to the risk of postprocedural thrombosis. EVTA is appropriate if the deep system is adequate to support venous drainage and the superficial venous incompetence is responsible for symptoms or significant skin changes. If the patient has any ongoing risk for thrombosis, EVTA may still be appropriate if that risk can be decreased with prophylactic anticoagulants. If saphenous reflux is associated with venous ulcers and an adequate deep venous system is present, EVTA of the causative veins is necessary to hasten the healing of the ulcer and to minimize the risk of a recurrent ulceration.
Treatment of competent enlarged superficial venous segments has no proven medical benefit and should not be performed. In some cases, the enlarged truncal vein may be functioning as a reentry or collateral pathway for another source of reflux or deep vein obstruction.
The use of EVTA to close incompetent perforating veins (IPVs) has been described. Currently, evidence-based and appropriate care indications for use are limited to ablation of pathologic perforator veins (PPVs). Current practice guidelines of the Society for Vascular Surgery and American Venous Forum define PPVs as those near or adjacent to a healed ulcer demonstrating >0.5 seconds of reflux and measuring ≥3.5 mm in diameter. Furthermore, for patients with incompetent pathologic perforators associated with symptomatic residual, recurrent, and rarely primary varicosities, without associated saphenous incompetence, either open or endovascular techniques can be used to treat the perforator veins.
APPLICATIONS
EVTA has been successfully and safely used to ablate the great (GSV) and small saphenous veins (SSV), the anterior and posterior accessory GSV, the superficial accessory saphenous vein, the anterior and posterior circumflex veins of the thigh, as well as thigh extensions of the SSV, including the thigh extension of the SSV known as the Vein of Giacomini. EVTA has been used to treat long straight competent tributary veins outside the superficial fascia if the vein is not touching the dermis, particularly in patients who are obese and in whom either sclerotherapy or microphlebectomy would be difficult, time consuming, or prone to side effects.
EVTA EQUIPMENT
Equipment and supplies common to ELA and CF are listed in Box 8.5 . As an alternative to hand injection, a foot pedal-controlled pump (AngioDynamics or HK Surgical) can be used to infuse the perisaphenous anesthetic solution. Venous access “micropuncture” kits that provide a less traumatic 21-gauge needle to insert a 0.018 in guidewire are useful when accessing small veins. These kits include a 4 or 5 F sheath with a dilator tapered to the 0.018 in guidewire. After the sheath and dilator are inserted, the dilator and 0.018 in guidewire can be removed to allow the placement of a standard 0.035 in guidewire (Cook, AngioDynamics, Vascular Solutions, or Merit Medical). There are also kits that contain a 21-gauge access needle and a 0.018 in guidewire over which a standard catheter can be placed, eliminating the need for a separate venous access kit and transition to a 0.035 in guidewire. These kits are comparable in price to those containing the 0.035 in guidewire.
Box 8.5
Equipment Common to ELA and CF
-
•
Procedure table that can tilt to Trendelenberg and reverse Trendelenberg
-
•
DUS with at least a 7.5 MHz transducer
-
•
Sterile gowns, gloves, masks, drapes, gauze
-
•
Ultrasound gel, sterile ultrasound probe and cord cover
-
•
Antiseptic preparation fluid
-
•
Local anesthetic
-
•
No. 11 or 15 scalpel blade
-
•
18- to 21-gauge needle for percutaneous entry
-
•
21–25 gauge needle for administration of tumescent anesthesia
-
•
Syringes
-
•
Normal saline
-
•
Compression stocking
Additional materials required to perform ELA include the laser generator (available from several vendors), sheath long enough to cross the abnormal venous segment(s) usually included in a kit along with a guidewire and sterile laser fiber. Additional components needed for CF include the VNUS radiofrequency generator, short 6 or 7 F introducer sheaths, CF catheters, and guidewire.
ELA is usually performed by placing a 4 or 5 F sheath into the vein to be treated over a 0.035 in guidewire and then, after inserting a laser fiber into the sheath, withdrawing the sheath to expose the fiber tip. The sheaths are manufactured in multiple lengths, and generally the sheath chosen is as long or longer than the segment(s) to be treated. The fibers are usually premarked to allow the operator to know when the fiber is tip to tip with the end of the sheath, as well as when they extend a fixed distance beyond the tip. In very straight veins a laser fiber can be advanced beyond its sheath to the starting point of ablation, but advancement through the sheath is recommended to avoid passing the fiber through the vein wall.
RF sheaths are either 6 F or 7 F with insertable catheter lengths of either 60 or 100 cm and heating element lengths of 3, 7, or 8 cm. After introduction of its sheath, the CF catheter can be passed into the vein over a 0.025 in guidewire, although the manufacturer has found guidewire use is necessary in only a minority of cases. The catheters have a resistive element at the distal 7 cm that heats to a fixed temperature when activated. The catheters are marked in 6.5 cm intervals to facilitate segmental withdrawal after activation that builds in a 5-mm overlap for each segmental treatment.
TUMESCENT ANESTHESIA
Tumescent anesthetic, when used in phlebology, describes the use of large volumes of diluted anesthetic solutions that are infiltrated into the perivenous space of the veins to be treated. The rationale behind the use of large-volume tumescent anesthesia for EVTA includes: (1) its use as a local anesthetic, (2) its ability to extrinsically compress and empty the vein to maximize the contact of the thermal device and the vein wall for efficient thermal transfer to the vein wall, as well as (3) providing a protective heat sink around the treated vein to minimize heating of adjacent structures.
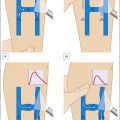
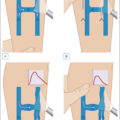
ELA and CF are usually performed with a dilute tumescent anesthetic solution of lidocaine in normal saline (a concentration of 0.1% lidocaine is typically used with an average volume of about 5–10 mL/cm of treated vein) with or without epinephrine, often buffered with sodium bicarbonate. This should be delivered with ultrasound guidance into the perivenous space (saphenous sheath) of the vein to be treated. The solution can be injected either manually or with an infusion pump such that upon completion of the process, the vein is surrounded along its entire treated length with the anesthetic fluid, as demonstrated in Fig. 8.1 .
Tumescent anesthetic should be delivered in copious volumes in the perivenous space of a vein to be treated with endovenous thermal ablation. ( A ) The duplex ultrasound axial image demonstrates an axial US image of the great saphenous vein (GSV) before tumescent anesthetic. ( B ) Demonstrates the hypoechoic halo of tumescent anesthetic fluid after it has been injected around the GSV.
Although the maximum safe dosage of lidocaine using tumescent technique for venous procedures is not well studied, 35 mg/kg with epinephrine has been reported as safe in the plastic surgical literature. However, one should keep in mind the FDA-reviewed inserts accompanying units of lidocaine state a maximum dose of 5 mg/kg without, and 7 mg/kg with epinephrine, with each use.
EVTA TECHNIQUE
The steps common to both ELA and CF are:
-
1.
Perform preprocedural DUS to map the venous segments to be treated. Mark the course of the vein(s) to be treated along with important anatomical landmarks associated with the ablation on the skin, including the proposed venous access site(s) and deep vein junctions. The access site is ideally at the inferior end of the incompetent segment or segments of the treated vein. In most cases, the entire incompetent segment(s) can be treated with one puncture. If microphlebectomy will be performed along with EVTA, the veins to be removed should be marked at this time as well.
-
2.
Prepare the operative tray and equipment. Aside from the thermal ablation device and a venous access kit, only basic supplies such as gauze, a sterilizing solution, sterile barriers, as well as the tumescent solution, with delivery syringes and needle, and an ultrasound probe cover are needed.
-
3.
Position the patient on the procedure table to allow visualization of the veins to be treated. This is generally supine, but the prone position is preferred for treatment of the SSV or Vein of Giacomini. Elevation of the torso of the patient relative to the legs (reverse Trendelenberg or partly sitting position) will prolong venous distention and enhance the likelihood of successful venous access. Carry out sterile preparation and draping of the leg to be treated. Preprocedural antibiotics are not necessary in almost all circumstances, as the procedure is performed sterilely and is considered “clean.”
-
4.
Visualize the access site with DUS.
-
5.
Anesthetize the access site. Nick the skin just large enough to facilitate entry of the sheath through the skin.
-
6.
Insert the access needle into the GSV under ultrasound guidance. Use of a 21 G micropuncture set, as discussed previously, is very popular. Cutdown is rarely needed and used only if percutaneous access fails.
-
7.
Place a guidewire into the vein and confirm intravenous placement with ultrasound.
-
8.
Place the introducer sheath over the wire.
-
9.
Fully advance the short sheath for CF. Position the sheath for ELA to the starting point for ablation. Operators typically advance the ELA sheath beyond the starting point and later withdraw it to bring the laser fiber tip to the ablation starting spot. The movement of withdrawal helps in accurately identifying the tip and in positioning it at the starting point.
-
10.
Remove the wire and its dilator if one is used with the sheath. Check for venous return by aspirating the syringe attached to the sheath and flush. Recognize that the sheath tip may be against the vein wall and may not aspirate freely. Also realize when flushing, microbubbles of air introduced into the vein may produce an acoustic shadow that may limit the ability to see venous detail and device positions.
Additional steps for ELA include:
-
1.
Introduce the laser fiber into the sheath so that the fiber reaches the sheath tip. There is generally a mark on the fiber to show this. Then fix the laser fiber and carefully pull back the sheath to expose about 2 to 3 cm of fiber. One should then withdraw the entire sheath-laser fiber to position the laser to the ablation starting spot.
-
2.
Fine tune the location of the tip of the laser fiber to below the superficial epigastric vein, anterior saphenous vein (ASV) or other large junctional vein for the GSV, and just below the thigh extension junction (or parallel to the skin and just beyond the segment that dives toward the popliteal vein when a thigh extension does not exist) with the SSV for SSV ablations ( Fig. 8.2 ). Others will withdraw the tip of the device to 2 to 3 cm below the deep vein junction in all cases.
Fig. 8.2 ( A ) Great saphenous vein (GSV) and small saphenous vein (SSV). ( B ) Anatomy of the right saphenofemoral junction.
(Modified from a drawing by Pentti Rautio.)
-
3.
Connect the laser fiber to its generator. One can reconfirm that the laser fiber tip is in the correct general location by viewing the visible light that can be delivered into the laser fiber tip and visualized through the skin ( Fig. 8.3 ). This is an additional way to ensure that the tip of the laser is being visualized accurately and that the laser connections were made appropriately, although it is not necessary with experience. If the light is not seen in the expected location the operator should troubleshoot the position of the laser or the connection to the laser to understand why.
Fig. 8.3 Aiming beam of the laser fiber tip visible through the skin during endovenous laser ablation.
-
4.
Administer tumescent anesthesia with ultrasound guidance after the patient has been placed into the Trendelenburg position to help drain the vein.
-
5.
Place appropriate laser safety goggles on everyone in the procedure room and use other appropriate laser safety measures. Connect the laser fiber to the laser and verify proper laser settings. Setting recommendations vary, but as will be discussed, aim to deliver at least 70 to 80 J/cm length of vein treated.
-
6.
Set the laser to continuous mode and select the power to be used. Power settings vary for each operator, and there is limited evidence to support specific settings. Reverify the placement of the laser tip with ultrasound before activating the laser generator ( Fig. 8.4 ).
Fig. 8.4 Longitudinal (sagittal) ultrasound image of the saphenofemoral junction demonstrating a 400-µm laser fiber tip in the great saphenous vein (GSV) just below the superficial epigastric vein (SEV). FV , Femoral vein.
-
7.
Activate the laser and withdraw the fiber and sheath at the speed that is dependent on the amount of energy you wish to deliver at the power setting selected with the laser in continuous mode. Be sure an adequate amount of energy is deposited in the upper 10 cm of the treated veins to ensure closure of the segment of the vein where failure most commonly occurs.
-
8.
Stop laser energy delivery at the distal aspect of the vein and place the laser in standby mode.
-
9.
Remove the fiber/sheath from the vein. Be sure the entire fiber is removed to exclude the possibility of a fracture of the device intravascularly.
-
10.
Record the watts, laser on-time, total joules delivered, length of the segment treated, and amount of tumescent administered. Calculate the withdrawal rate and joules delivered per cm to ensure the targets for successful ablation have been met.
Additional steps for ClosureFast (CF) are as follows:
-
1.
Introduce the 6 F or 7 F ClosureFast catheter through its sheath either bare or over a 0.025 in guidewire and position its tip to the desired starting position 2 cm below the deep junction. Connect the CF catheter to the RF generator.
-
2.
Deliver tumescent anesthesia and place the patient in the Trendelenburg position as discussed previously.
-
3.
Withdraw the sheath so that one of the 6.5-cm marks on the catheter is located at the point the catheter exits the sheath.
-
4.
Activate the generator and perform two 20-second heating cycles at the preset temperature of 120°C. Withdraw the catheter 6.5 cm and repeat the heating cycle until the entire vein is ablated. The second 20-second cycle is recommended by the manufacturer at the first treatment site, although some operators will perform second cycles at all levels or selectively at levels with aneurysmal segments or near large tributaries, such as an incompetent perforator to ensure thorough ablation. Similarly, some operators may begin the ablation with three cycles for larger diameter veins.
-
5.
When the amount of vein left to treat is <7 cm long, marks will be visible on the catheter to alert the operator. The sheath should be withdrawn at this point to allow the heating element to extend beyond the sheath to ensure vein treatment as well as to avoid heating the sheath or the skin. Cease treatment when the catheter tip enters the introducer sheath or exits the vein.
-
6.
Remove the CF catheter and sheath after the final segment is treated.
-
7.
Record the parameters, duration of treatment, and length of segment treated ( Fig. 8.5 ).