Introduction
Skin substitutes are an important tool in the management of burn injuries. They provide a means to cover and protect wounds while promoting healing and reducing the risk of infection. In modern burn care, physiologic wound closure is the recognized clinical goal in the treatment of burn injuries, and best outcomes are achieved by optimizing epidermal and dermal function during the healing process. Reestablishment of an epidermal barrier provides reduction in infection, increased fluid retention, electrolyte and temperature homeostasis, and requisite stabilization of the patient that ultimately saves lives. Dermal reconstitution provides the flexibility, strength, heat dissipation and conservation, lubrication, and sensation that is essential for best skin function. Understanding burn wound depth and healing potential allows for selection of appropriate methods of skin reconstruction, which may include use of skin substitutes to accelerate, augment, or temporize the healing process of burn injuries.
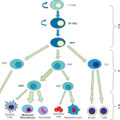
Currently, more than 70 skin substitute products are available ( Fig. 14.1 ). These can be generalized as epidermal cells and constructs, dermal scaffolds, and full skin substitutes. Each type has its own benefits, indications, and mechanisms of action, and the optimal choice will depend on the specific characteristics of the patient and the wound.
Categories of skin substitutes and examples.
Epidermal substitutes may be biologic or synthetic dressings that act as a temporary cover for partial-thickness wounds. Therapy of these wounds is mainly directed at adequate coverage to prevent further fluid loss, provide a barrier against bacterial colonization, and provide pain control.
Epidermal analogues consist of epidermal cells, usually of the patient’s own skin, and are used to achieve epidermal closure. These skin substitutes can be applied in different ways, such as cell suspension, cultured sheet, or skin transplant. These are specifically indicated in larger-area burn wounds when donor areas for the harvesting of split-thickness skin grafts (STSGs) are insufficient.
Skin analogues, such as biologic tissues, are also frequently used as temporary cover for excised deep dermal or full-thickness wounds to preserve and prepare the wound bed for the appropriate grafting technique.
Dermal scaffolds are composed of synthetic or biologic materials and are used to provide permanent structural support to the wound bed. These are typically used for full-thickness burns and are meant to prevent contraction of the wound. In general, after revascularization and incorporation into the wound, these materials are ultimately remodeled by the body of the recipient patient to form a neodermis, which has mechanical properties resembling the dermis. They can be applied in a one-step procedure, together with a STSG. This treatment modality is mainly used when there is a well-vascularized wound bed with limited bacterial load and minimal exposure of tendons and bone tissue. When these criteria are not fulfilled, a two-step procedure may provide a better treatment approach. In a two-step procedure, first the dermal construct is applied to the wound bed and allowed to vascularize during a 2- to 3-week period. After that, the STSG is placed.
Full-skin substitutes consist of an epidermal and a dermal component procured from allogeneic, xenogeneic, or autologous sources; these are applied in different phases of wound healing.
Autologous full-skin substitutes are used to permanently replace the entire thickness of the skin, usually in full-thickness burn injuries. Although making significant recent advancements, autologous full-skin substitutes are still largely in developmental phases and have not yet achieved widespread use.
In recent years, subcutaneous fat and its role in skin function and repair has received more attention. Absence of subcutaneous fat in burn scars can lead to adherent scars and associated functional problems. The subcutis acts as a sliding layer between the skin and underlying structures, such as tendon, muscles, and bone. Damage to this layer can lead to problems in movement and function, stiffness, and pain. Full skin regeneration is therefore not complete without repair of the subcutis, and subcutaneous fat has a functional role in skin function. Lipofilling or fat-transplantation techniques were originally developed in cosmetic surgery but are now gaining interest in scar treatment and reconstructive surgery. More research on adipose tissue–derived stem cells and their role in skin regeneration has increased this even further. The position of fat grafting in burn care is currently not well-defined but gaining attention.
This chapter focuses on epidermal, dermal, and full-skin substitutes and highlights the use of these materials in modern burn care. Best outcomes are achieved by understanding the benefits, limitations, and costs of these technologies and by choosing the most appropriate option based on the specific needs of the patient, the burn injury, and the resources of the system in which care is being delivered.
Epidermal cells and constructs
Management of large (>40% total body surface area [TBSA]) full-thickness burns is a challenge, but resourceful surgical planning for the coverage of these large burns has led to refinement of some early innovative procedures.
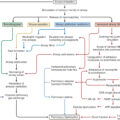
Initially, thin autologous skin transplants will be used for wound coverage. These can be widely expanded, meshed STSGs or small skin islands produced according to the Meek micrograft procedure. Further treatment modalities are provided in combining these transplants with cultured epidermal autografts (CEAs) and autologous skin cell suspension (ASCS) ( Fig. 14.2 ). Successful treatment begins with the early development of a detailed surgical plan that includes standardized techniques and approaches, such as milestone timelines to treat and manage these patients. This approach fosters team participation in the plan and understanding of standard procedures and any deviation.
An example timeline showing the major milestones from patient admission to cultured epidermal autograft (CEA) application. CEA biopsies are obtained at the first or second excision and sent for processing. The interval of approximately 3 weeks for delivery of the CEA allows for allograft/dermal substitute placement and engraftment, and coverage and vascularization of the underlying dermis. Excision and debridement procedures generally begin within 48 hours of admission with continuing debridement and complete coverage of burn areas with allograft within 3 to 4 days. The patient will undergo additional multiple excisions of nonviable tissue, and replacement of nonadherent allograft as needed. Excisions are compartmentalized to specific locations (e.g., posterior surfaces on one day, hands/feet on a different day) to minimize the surgical burden with the goal of allograft engraftment and a well-vascularized wound bed. If during this time the wound beds are well vascularized and the CEAs are not ready, treatment proceeds with wide mesh or Meek autografts with autologous skin cell suspension. When the CEAs become available, continued closure is achieved quicker. %TBSA , Percentage total body surface area; STSG , split-thickness skin graft.
Split-thickness skin graft
STSG remains the standard of care for wound coverage of burns; however, given the lack of donor sites, manual limits of STSG expansion rates greater than 6:1, and difficulty in handling, there is a potential to leave significant areas of the wound uncovered.
Meek
The Meek micrografting method was introduced in 1958 by Cicero Parker Meek. Modifications of the technique in 1993 facilitated the achievement of true and reliable expansion rates of autografts up to 9:1. , In addition, grafts using the Meek technique have been shown to have superior graft take compared with other skin-grafting techniques in an equine animal study. Expansion ratios of 3:1, 4:1, 6:1, and 9:1 require approximately half of the donor site area compared with meshing.
Cultured epidermal autografts
CEAs consist of cultured epidermal cells harvested from a small full-thickness skin biopsy taken from an unaffected area on the burn patient. A single, full-thickness biopsy measuring 2 cm 2 can be expanded in area up to 10,000 times.
After culturing in the laboratory, CEA consists of rectangular sheets of proliferative keratinocytes ranging from two to eight cell layers thick. , These keratinocytes play an important role in epidermal restoration after injury through proliferation and reepithelialization, replacement of the damaged extracellular matrix (ECM), and restoration of tissue structure and function.
The first use of CEA was conducted by O’Connor & Gallico. Early take rates were poor secondary to CEA fragility, unfamiliarity with CEA technique, and inadequate wound preparation.
Long-term results of CEA demonstrated fragile skin with easy blister formation, which was found to be related to poor regeneration of the basement membrane and lack of anchoring filaments that link epidermis to dermis. It was not until Cuono et al. introduced the “allodermis composite grafting technique” in 1987 and Langdon et al. showed keratinocytes placed on allodermis in 1988 that they were better able to reconstitute normal epidermal and dermal architecture where successful CEA application was reproducible. Sood et al. and Hickerson et al. also reported over 70% take rates and over 90% survival rates when CEA was used in conjunction with allodermis for patients with major burns.
Sood et al. also reported good CEA take when placed over a 6:1 wide mesh STSG. The use of meshed STSG placed under CEA has been reported to minimize shear forces and hasten graft take. Similarly, Gardien et al. reported slightly faster healing and better long-term scar quality for nonconfluent cultured epidermal cells placed on top of widely meshed STSG. Different stages of wound bed preparation and CEA application are shown in Fig. 14.3 .
Different phases of wound treatment of a patient with large areas of full-thickness burns. (A) Widely meshed autograft. (B) Meek autografts. (C) Debrided wound bed, placing of AlloDerm. (D) Engrafted cadaver graft, well-vascularized wound bed. (E) Cultured epidermal autograft (CEA) takedown. (F) Airing/drying out period of CEA.
Cell sprays
Application of autologous epidermal cells can also be achieved by onsite harvesting of a small number of epidermal cells and spraying them onto the debrided wound surface. An ASCS device (RECELL; Avita Medical, Valencia, CA) was developed from the work of Stoner and Wood. It is processed from a thin (0.006–0.0008 inch) autograft that can cover up to 80 cm 2 of treatment area from every 1 cm 2 of donor skin needed. It is a point-of-care product and can be applied immediately to an excised wound bed. With the significant decrease in donor site size and point-of-care application, ASCS offers clinical advantages in closure time and optimizing patient outcomes. ASCS also induces rapid epidermal regeneration, achieving reepithelialization to heal burns and STSG donor sites. Holmes et al. reported widely meshed STSG and ASCS are effective in achieving short- and long-term healing of mixed-depth burns while significantly decreasing donor skin use.
From clinical experience and reported data, CEA and ASCS application over a dermal scaffold provides strength and durability to the final closure and will yield better long-term results. As STSG is the standard of care in burn-wound closure, a widely expanded autograft has become the preferred base for CEA and ASCS use in large full-thickness burns.
Practical guideline for use of CEA and ASCS in large-area burns
The approach to large burns and use of the wide mesh STSG and Meek micrograft, in conjunction with CEA or ASCS, requires standardization. Identification of appropriate patients for potential placement of CEA and the establishment of a broad treatment plan are important first steps. Patients with over 30% TBSA are considered for CEA placement because conversion of superficial partial-thickness to deep partial-thickness or full-thickness burns may occur during hospitalization, thereby increasing the percentage of TBSA and requiring additional intervention. In addition, the location and availability of donor sites is taken into consideration when planning coverage of these large full-thickness burns.
From experience, CEA and ASCS application over a dermal scaffold provides strength and durability to the final closure and will yield better long-term results. In addition, a clear plan and strategy must be developed early as the process for each different matrix (wide meshed STSG, Meek micrograft, or allograft) involves different amounts of preparation time relative to the application of CEA. The best CEA results are obtained with an autograft matrix (wide meshed STSG or Meek). However, an allograft dermis or dermal substitute can be used if donor sites are limited, especially in patients with over 80% TBSA. It is not uncommon to use a combination of dermal matrices on a patient.
The rapid progression of technology and advances in burn care have allowed us to successfully manage large burns. Although these can be intimidating, it may also be the only option in successfully treating patients with large burns. Therefore the best course when employing these options is to have a team and standardized approach to care. Fig. 14.4 depicts different aspects of postoperative care.
A timeline showing the major milestones from patient culture epidermal autograft (CEA) application to patient discharge. (Timeline photographs are shown in Fig. 14.3 .) Postoperative (postop) care of CEAs is just as important as the preparation and application, and this is essentially performed in dedicated burn centers. All team members are vital to a successful outcome. Nursing staff are fully trained and experienced in managing these burn wounds before and after CEA application. It is crucial staff understand this process thoroughly. Initial dressings for all CEA postop patients are silver nitrate wring-out (i.e., surgical pad soaked in silver nitrate solution and squeezed until excess liquid is removed) until final culture results are available. Antibiotic topical therapy is directed by culture results taken at the time of CEA application; culture results are usually returned within 1 to 3 days. Pressure ulcer preventive measures are taken by the use of mattresses that are programmed to produce alternating pressure redistribution and low air loss. On postop day 1, the outer dressings are removed, and the CEAs are open to air and allowed to dry for at least 8 hours. This will increase to 12 hours depending on patient tolerance. The patient is turned side to side to facilitate the dryout of the CEA coverage and wound bed, with shorter periods at first, then longer periods. The duration on a side depends on the patient’s tolerance while trying to maximize the drying times. The CEAs are meticulously examined by the team daily with potential areas of infection noted and windowed out to prevent further loss of surrounding CEAs. Patients remain supine or lateral. Patients are not placed in prone position. Removal of the gauze backing, or takedown, generally occurs in the operating room, 7 to 10 days post-CEA application, and allows for the evaluation of initial CEA take by assessment of the percentage of engrafted areas of CEA (i.e., areas that have developed epidermis). A nonadherent dressing is applied followed by gauze dressing soaked in antimicrobial and wrung out before being applied to CEA. Physical and occupational therapy is started soon after takedown.
Dermal constructs
Allogeneic tissues
Virtually all of the characteristics of normal skin not related to barrier function are provided by the dermis. These characteristics include flexibility, strength, heat dissipation and conservation, lubrication, and sensation.
Human allograft skin is generally used as a STSG after being procured from organ donors. When used in a viable fresh or cryopreserved state, it vascularizes and remains the gold standard of temporary wound closures. It can be refrigerated up to 7 days, but it can be stored for extended periods when cryopreserved. It is also used in a nonviable state after preservation in glycerol or after lyophilization. Viable split-thickness allograft provides durable biologic cover until it is rejected by the host, usually within 3 or 4 weeks. Prolongation of allograft survival through the use of antirejection drugs has been considered, but it is not generally practiced for fear that antirejection drugs will increase the risk of infection. , Allograft skin in glycerol is used as a membranous dressing on partial-thickness burns, especially scald burns in children.
Allograft skin is also effectively used in combination with meshed autograft in patients with large burns. The interstices of the meshed graft are immediately closed by the overlying unexpanded allograft possibly reducing metabolic stress and local wound inflammation. Limitations of the use of human allograft skin include limited availability and the need for modern tissue banking techniques and adoption of regulations that warrant the safety and quality of banked skin.
Human amniotic membrane is used in many parts of the world as a relatively cheap and temporary dressing for superficial wounds. , Amniotic membrane is generally procured fresh and used after brief refrigerated storage. , It can also be used in a nonviable state after preservation with glycerol. Beneficial effects of amniotic membrane on partial-thickness burns have been described in a randomized controlled trial by Mostaque et al., with improved clinical outcomes such as time to epithelialization, length of stay, pain, and frequency of dressing changes. The principal concern with amnion is the difficulty in the screening of the donor material and the potential risk of undetected disease transmission.
A dermal analog that is (in structure at least) closest to the original dermis is freeze-dried allogenic dermis. This material is intended to be combined with a thin epithelial autograft at the time of initial wound closure. It is marketed as AlloDerm (LifeCell Corporation [now Acelity], San Antonio, TX). , Split-thickness allograft skin is obtained from deceased human donors through tissue banks after proper screening for transmissible diseases. Using hypertonic saline, the epithelial elements of the grafts are removed. The remaining dermis is treated in a detergent to inactivate any viruses, and the device is freeze-dried. The process is intended to provide a nonantigenic dermal scaffold, leaving basement membrane proteins (particularly laminin and type IV and VII collagen) intact. The material is rehydrated immediately before placement on wounds with overlying ultrathin epithelial autograft. Clinical experience with this material in burn surgery is limited, but early experiences have been favorable. Nevertheless, these products now seem to be used more in breast reconstructive surgery than in acute burns. A similar product, based on allogeneic dermis preserved in glycerol, is Glyaderm (Euro Tissue Bank, Beverwijk, The Netherlands). Also here, antigenicity is reduced by washing out the cells and preserving the original ECM. Initial studies on burn patients have been performed and reported favorable results.
Nonallogeneic tissues
Various animal skins and tissues have been used for many years to provide temporary coverage of wounds, and several types of intact skin substitutes from nonhuman sources have emerged over the last decade.
Acellular fish skin xenografts, such as Kerecis Omega3 (Kerecis, Isafjordur, Iceland) may represent an effective, potentially low-cost alternative in treatment of superficial and partial-thickness burns.
Acellular fish skin contains collagen, fibrin, proteoglycans, and glycosaminoglycans; it therefore acts as a dermal substitute. In comparison to mammalian acellular grafts (e.g., conventional porcine or bovine), risks of disease transmission of these grafts may be reduced and may be produced under a simpler sterilization process while retaining an omega-3 fat source.
Pigs hold great promise as a therapeutic source as they share many physical and structural similarities with humans, such as similar hair coverage, papillary dermis, and rete ridges to those of human skin. Additionally, pigs are domesticated animals with favorable breeding characteristics and well-established animal husbandry practices sufficient to achieve specific agricultural and food industry–prescribed standards. ,
Conventional (wild-type) porcine xenografts commonly consist of decellularized, reconstituted sheets of homogenized dermis fashioned into sheets and meshed, such as EZ-Derm or Medi-Skin. They are used to achieve temporary, superficial wound coverage, provide pain control, and act as scaffolds for dermal regeneration therapies. Such conventional xenografts do not retain the metabolically active, naturally present cell populations and patent, native vasculature, and thus do not permit graft reperfusion, capillary ingrowth, or vessel-to-vessel connections. Consequently, the skin graft becomes ejected rather than immunologically rejected in a matter of days and requires frequent changing. Split-thickness, wild-type porcine skin has been used clinically worldwide in a variety of conditions (e.g., fresh, after brief refrigeration, after cryopreservation, after glycerol preservation).
Another biologic tissue that is used in wound healing is OASIS (Cook Medical Corporation, UK), an acellular ECM derived from porcine small intestinal submucosa. OASIS functions as a scaffold to promote the migration, adhesion, and cell proliferation of epidermal keratinocytes and fibroblasts around the wound site and is gradually integrated in new dermislike tissue.
Dermal scaffolds
As an alternative to biologic tissues providing an alternative to dermis, dermal scaffolds have been developed to replace dermal tissue in large and deep wounds. These scaffolds are generally composed of isolated and purified biologic molecules, such as collagen, hyaluronan, or a combination of collagen and other ECM molecules such as elastin, elastin derivatives, or glycosaminoglycans.
The first dermal substitute used clinically was Integra (IntegraLife, Plainsboro, NJ) and has been used worldwide for several years to treat burns and in reconstructive surgery. This material was developed in the early 1980s by a biomaterials research team led by surgeon John Burke from the Massachusetts General Hospital and materials scientist Ioannis Yannas from the Massachusetts Institute of Technology. The research team had the goal of developing a wound covering that would provide both a temporary vapor and a bacterial barrier when placed on excised burn wounds, while providing a scaffold for later dermal regeneration. The silicone outer layer serves as a temporary epidermis and is removed after vascular ingrowth into the dermal replacement is completed (usually within 2–3 weeks). The inner layer of this material is a 1- to 2-mm-thick combination of collagen fibers isolated from bovine tissue and the glycosaminoglycan chondroitin 6-sulfate. To manufacture the device, glycosaminoglycan and collagen fibers are precipitated and then freeze-dried and cross-linked by glutaraldehyde. The outer layer of the membrane is a 0.009-inch (0.23-mm) polysiloxane polymer and is permeable to water vapor like normal epithelium. This membrane is intended to be placed on freshly excised full-thickness burns, and the outer silicone membrane is replaced with a thin epithelial autograft 2 to 3 weeks later. Clinical reports in patients with large burns have been generally favorable. One risk factor is bacterial colonization or infection underneath the membrane. If this occurs, local removal of the membrane and cleansing are required, which may impact final clinical outcomes in terms of scar formation. Integra has also been found to be useful in selected burn reconstruction operations.
Integra is generally applied in a two-stage operation: first debridement and application of the substitute, followed by removal of the silicone layer 2 to 3 weeks later and replaced by grafting of a STSG. Some substitutes, however, can be applied in a one-step procedure. The first dermal substitute that was applied in this manner was Matriderm (Medskin Solutions, Billerbeck, Germany). Matriderm consists of bovine collagen fibers mixed with 3% elastin hydrolysate, purified, and freeze-dried into a dry scaffold that can be immediately placed on the wound and can be prewetted with a saline solution ( Fig. 14.5 ). Clinical results have indicated a delayed graft take, reflecting the interpositioning of the unvascularized scaffold between the wound bed and the graft. However, this was compensated by a better outcome in terms of scar quality as long as 12 years later. Other scaffolds in this same category include Permacol (Tissue Science Laboratories, Hampshire, UK), a porcine dermal collagen implant mainly indicated for hernia and abdominal wall repair; PELNAC (Eurosurgical, UK), a bilayer product that consists of an atelocollagen sponge layer, derived from porcine tendons, and an outer silicone film; and Hyalomatrix (Medline, United States), which consists of a bilayer of an esterified hyaluronan beneath a silicone membrane. , The scaffold delivers hyaluronan to the wound bed, and the silicone membrane acts as a temporary epidermal barrier. Most of these materials are now provided in variants that can either be used in a one- or two-stage procedure, and even injectable variants are available for scar revision procedures.
(A) Flame burn in a 52-year-old patient, postburn day 16. (B) Debridement of wound and application of Matriderm dermal substitute. (C) Split-skin mesh graft, ratio 1.5:1 enlarged. (D) Result after 6 weeks.
Application in a one-stage procedure consists of wound debridement, careful hemostasis, placement of the dermal scaffold, and immediate coverage with a STSG. This is generally performed for large and deep wounds with a well-vascularized wound bed and no signs of wound-bed colonization or infection. A two-stage procedure consists of wound debridement, hemostasis, and placement of the dermal scaffold in combination with a silicone membrane as temporary epithelial coverage. Generally, vascularization is observed for 2 to 3 weeks, after which the silicone layer is removed, and a split-thickness autograft is placed on top of the dermal scaffold. A two-staged procedure is indicated for the treatment of large, deep wounds, which are less well-vascularized or contain poorly vascularized structures, such as exposed bone or tendon, or in the case of insufficient suitable donor sites for the autograft.
In both procedures, topical negative pressure (TNP) can be applied in combination with the dermal scaffold or as wound-bed preparation before application of the scaffold. The function of TNP is twofold: mechanical fixation of the scaffold or scaffold plus autograft and as a stimulation of vascularization. Potentially, TNP application could also reduce the bacterial load, although reports on this aspect are less conclusive. ,
Recently, synthetic dermal substitutes have been successfully introduced for clinical application. Advantages of synthetic materials include reduced risk of disease transmission and cross-species immune reaction, off-the-shelf availability, potential low-cost production, and less susceptibility to bacterial colonization or infection. Whereas previous efforts to introduce synthetic polymers as dermal substitutes suffered from drawbacks, such as a high inflammatory response and foreign body reactions, this new therapy seems to have clinical promise. ,
Novosorb Biodegradable Temporizing Matrix (BTM) (Polynovo Ltd., Port Melbourne, Victoria, Australia) consists of a polyurethane foam combined with a nonbiodegradable polyurethane seal and has been clinically evaluated in a multicenter clinical study. Novosorb BTM was applied in a two-stage procedure, and the reported time between initial application and final autografting was 32 days. During this period, infections were reported in 39% of cases. Although comparative scar quality data are currently still lacking, autograft take was reported to be high (82%).
Cellular dermal substitutes
Dermal scaffolds seeded with allogeneic dermal fibroblasts are described in literature, such as Dermagraft (Organogenesis, United States), which consists of a polyglactin mesh seeded with neonatal dermal fibroblasts. This is US Food and Drug Administration (FDA) approved for treatment of chronic diabetic foot ulcers. The therapeutic mechanism of action is thought to be based mainly on the excretion of growth factors and cytokines.
New early-stage developments in this field involve the use of stem cells, fetal cells, adipose tissue–derived cells, 3D-printing technology, , and nanotechnology using nanocomposite polymers. However, clinical safety and effectivity still need to be evaluated.
Full-skin substitutes
An ideal skin replacement would provide immediate replacement of both dermal and epidermal layers. Thus combining epithelial cells with a dermal analogue in the laboratory seems logical; however, the choice for the best suitable cell source is not an easy task. Cells of allogeneic origin generally do not survive for more than 2 to 3 weeks. Tissue-engineered delivery systems that contain cultured autologous keratinocytes with or without fibroblasts are not suitable for initial placement on severe and extensive deep partial- and full-thickness burn wounds because of the inherent time required for manufacturing.
The first commercially available and FDA-approved full-skin substitute was Apligraf (Organogenesis, Canton, MA). The product consists of human foreskin-derived neonatal fibroblasts in a bovine type-I collagen matrix over which human foreskin-derived neonatal epidermal keratinocytes are cultured. Although the allogeneic cells ultimately are rejected, cytokine and growth factor production have been shown to be beneficial for chronic wounds such as venous leg ulcers and diabetic foot ulcers. However, for definite wound closure, a source of keratinocytes needs to be present.
OrCel (Fortcell Bioscience, Englewood Cliffs, NJ) contains a layer of cultured human allogeneic neonatal dermal fibroblasts and a separate layer of cultured human allogeneic neonatal epidermal keratinocytes on a type-1 bovine collagen sponge to provide physiologic levels of growth factors and cytokines. ,
StrataGraft (Mallinckrodt, Hazelwood, MO) is a bioengineered, allogeneic cell construct and xenotransplantation product containing cultured keratinocytes (NIKS) and dermal fibroblasts in murine collagen matrix. The fully stratified epidermis contains metabolically active cells that produce and secrete growth factors and cytokines and is approved in the United States for the treatment of deep partial-thickness burns that contain residual, intact dermal elements with a shelf life of 12 months.
Novel developments in xenotransplantation
At present, no skin substitute or FDA-approved therapy exists that replaces all the characteristics of healthy human skin and provides complete and durable wound closure in full-thickness burn wounds after a single application. Xenotransplantation of vital skin transplants from genetically engineered, designated pathogen-free (DPF) porcine donors is a promising solution to this unmet medical need.
Previously, use of conventional wild-type porcine xenografts resulted in antibody-mediated, hyperacute rejection and rapid necrosis because of an intense complement cascade that damages the graft endothelium resulting in graft ischemia. , The creation of “humanized” porcine donors via germline genetic alterations, which consequently lack expression of antigenic wild-type extracellular moieties, significantly reduces this adverse immune response. This improvement now enables engineered, porcine xenogeneic therapies to be clinically useful in applications beyond those of conventional, wild-type–derived xenograft predecessors.
realSKIN (Alexis Bio, United States) is a bilayered, split-thickness, membranous skin xenotransplant sourced from genetically engineered DPF porcine donors. It contains vital (i.e., nonterminally sterilized) epidermal and dermal skin tissue layers and patent, native vasculature; it also consists of metabolically active keratinocytes, melanocytes, fibroblasts, and other essential living cells and bioactive compounds, including cytokines, growth factors, and other metabolites. The inclusion of the complete milieu of healthy living skin distinguishes a xenotransplant skin from a conventional xenograft and allows for the rapid revascularization and prolonged graft survival, reepithelialization, and durable wound closure. However, natural tissue remodeling and cellular apoptotic cycles eliminate the presence of localized and systemic porcine cells within 90 days. Patient enrollment in a US FDA phase-3 clinical trial of realSKIN began in 2023 to demonstrate durable wound closure within 90 days of treatment after a single application; if approved, it would become the first live biotherapeutic product from an intentionally genetically altered animal.
Full-skin substitutes with autologous cells
The most recent addition to the field is the development of skin substitutes comprised of a dermal scaffold seeded with autologous dermal cells in combination with an epidermal layer consisting of autologous epidermal cells. These products are advanced therapy medicinal products (ATMP), and production is heavily regulated (e.g., EU Regulation 1394/2007/EC, FDA Regulation Title 21 CFR Parts 600–680 [Requirements for biological products] and Title 21 CFR 1271 [Biologicals and medical devices]).
Several products are under development based on a collagen scaffold seeded with a patient’s own cells. NovaDerm (Regenicin, Las Vegas, NV), originally developed by Dr. Steven Boyce and colleagues, was shown to retain skin quality and elasticity for many years postsurgery. Also, denovoSkin (Cutiss, Zurich, Switzerland), an autologous bioengineered human skin graft, is presently being tested in clinical trials. This full-skin substitute, which uses the patient’s own cells, may lead to better scar quality than conventional treatment modalities such as STSGs and dermal substitutes. In Canada, Dagher et al. continue to advance the self-assembled skin substitute, but further developments will be needed to overcome current drawbacks (e.g., the long culture time of 3–5 weeks, during which temporary coverage of excised wound areas is required).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree