Introduction
Advances in resuscitation and the critical care of burn-injured patients have greatly improved survival in well-resourced settings. In organized emergency, trauma, and burn care systems, death from burn shock and its sequelae has become uncommon. Timely and functional recovery of the burn-injured patient hinges on accurate and early wound assessment and management. Early excision with immediate or staged reconstruction that is individualized to each patient (e.g., physiologic condition, injury mechanism, wound characteristics, anatomic/functional area) has been among the greatest advances in burn care to date. Before early excision and reconstruction, burns were treated by daily washing, removal of loose dead tissue, and application of some sort of topical nostrum until wounds healed or granulated, often with significant wound contraction. Superficial dermal burns (i.e., second-degree, superficial partial-thickness wounds) healed within 2 weeks, and deep dermal burns (i.e., second-degree, deep partial-thickness wounds) healed over 3 or more weeks if infection was prevented. Full-thickness burns (i.e., third degree or deeper) lost their eschar in 2 to 6 weeks by bacterial enzyme production, autolysis, and daily bedside debridement. Burns that healed within 2 to 3 weeks generally did so without severe hypertrophic scarring or functional impairment, although dyspigmentation was variable. Burns that required longer than 3 weeks to heal often resulted in severe hypertrophic scars and functional impairment. Split-thickness skin grafts were applied after 3 weeks of healing, and a greater than 50% graft survival rate was acceptable. Repeated grafting eventually closed the wound. The prolonged and intense inflammatory response led to severe hypertrophic scars, contractures, and considerable physical and psychological disability. In low- and middle-income counties, where 90% of the world’s burn injuries occur, and burn care providers do not have access to safe anesthesia, blood for transfusion, skin substitutes, and critical care capabilities, this expectant or open wound management remains standard of care for larger burn injuries. ,
State-of-the-art burn care now involves early excision and reconstruction for wounds that would otherwise not heal within 2 to 3 weeks. The definition of early is contested, but it is generally considered to be excision within 5 to 7 days of injury. Knowing which burn wounds benefit from early excision and reconstruction is the art of burn surgery and requires understanding of skin biology and the common and unique pathophysiologic changes caused by thermal, chemical, and electrical injuries. Despite ongoing efforts to objectively assess wound depth, the standard technique for determining burn depth remains clinical assessment by an experienced burn provider.
Pathophysiology of the burn wound
Normal skin biology
Skin has several vital functions. It protects against fluid and electrolyte loss, infection, radiation, and toxins, and it provides thermal regulation. Skin contact provides clues to the surrounding environment through perception of touch, temperature, and pain. In addition, skin appearance is a major determinant of identity and body image and affects interpersonal interactions, for better or worse. The largest organ in the human body, skin is comprised of two layers: the epidermis and the dermis. Epidermal thickness varies among different body parts: from 0.05 mm on the eyelids to over 1 mm on the soles. Most skin thickness comes from the dermis, which varies with age, gender, and body location.
Epidermis derives from ectoderm; the principal cell is the keratinocyte, but epidermis also contains melanocytes, Langerhans cells, Merkel cells, and immunoinflammatory cells (e.g., lymphocytes, macrophages). Keratinocytes begin their division and differentiation at the stratum basale and differentiate progressively outward over 2 to 4 weeks through the stratum spinosum, stratum granulosum, stratum lucidum, and stratum corneum, at which point they are flattened, anuclear, cornified structures. Melanocytes produce melanin pigment essential for protection against ultraviolet radiation, and Langerhans cells and other immunoinflammatory cells perform phagocytosis and antigen presentation to prevent or mitigate local and systemic infection. In a wound with an injured and/or absent epidermal basal layer, keratinocytes proliferate and migrate from the wound edges and epidermal appendages (i.e., hair follicles, sweat and sebaceous glands) to achieve epithelialization. Melanocytes differentiate and migrate more slowly and less predictably, frequently leading to temporary or permanent dyspigmentation. ,
Epidermal projections (i.e., rete ridges) interdigitate with dermal projections (i.e., papillae) at the basement membrane zone, which connects the epidermis and dermis via keratinocyte-derived collagen VII anchoring fibrils, critical structures that stabilize the epidermal-dermal junction and provide resistance to mechanical shear. Anchoring fibrils take several months to mature during wound healing ; resultantly, minor shearing forces frequently cause blistering and epidermal loss.
Dermis derives from the mesoderm and is comprised of superficial papillary and deeper reticular layers that are separated by capillary networks (i.e., superficial papillary, reticular, and subcutaneous plexuses) that deliver necessary nutrients to dermal cellular structures. There is an abundant extracellular matrix, comprised primarily of collagen and elastin fibers, that provides the dermal structure and its mechanical functions; organized collagen fiber orientation provides tensile strength, and cross-linked tropoelastin and microfibrils form elastin that imparts cutaneous recoil (i.e., pliability). Glycosaminoglycans and proteoglycans, such as hyaluronic acid and chondroitin sulfate, attract water to maintain matrix hydration, provide absorption, and regulate cellular crosstalk by binding and releasing inflammatory mediators. Protein turnover, accounting for the high plasticity of skin, increases with mechanical stress and responses to injury. After wounding, microvascular endothelial cells mediate local and systemic inflammatory responses and coagulation-fibrinolysis cycles. Sensory nerves, which traverse the dermis into the epidermis, also play a significant role after injury as they mediate pain and itching, modulate inflammation, and appear to influence the remodeling phase of wound healing. The dermis, like other mesoderm-derived structures, heals not by regeneration but by fibrosis and scarring.
Pathophysiologic changes after thermal injury
Applied heat at the cellular level causes denaturation of proteins and loss of plasma membrane integrity. Temperature and duration of contact have a synergistic effect; cell necrosis occurs after 1 second of exposure at 156°F (69°C) and after 1 hour at 113°F (45°C). Following a flame or scald burn, necrosis occurs at the center of the injury and becomes progressively less severe at the periphery. The necrotic to healthy tissue transition is more abrupt in wounds caused by contact with hot objects and friction. Jackson’s 1953 description of the three zones of thermal injury remains our conceptual understanding of the burn wound ( Fig. 9.1 ). The zone of coagulation is at the center of the wound where no viable cells remain. A mix of viable and nonviable cells, capillary vasoconstriction, repeated microvascular thrombosis and fibrinolysis, and tissue ischemia characterizes the surrounding zone of stasis. This at-risk zone analogous to the penumbra of brain tissue at the margins of a stroke may progress to necrosis with edema, hypoperfusion, desiccation, and/or infection. Systemic factors such as advanced age, diabetes, and other chronic illnesses increase risk for wound progression. Approximately half of the cells in the zone of stasis are undergoing apoptosis or necrosis as a result of oxidative stress, ongoing inflammation, and decreased blood flow due to edema and sustained microvascular thrombosis. Efforts to enhance wound healing have focused on prevention of progressive necrosis in the zone of stasis because medical care has little impact on outcome in the zone of coagulation. Protection of this sensitive area is achieved with accurate fluid resuscitation (i.e., no under- or overresuscitation), deresuscitation/diuresis as soon as appropriate, avoidance of vasoconstriction, mitigation of edema, and prevention of infection. Optimal wound care consists of nondesiccating dressings, topical antimicrobials, edema management (i.e., compression, elevation, active motion), and regular monitoring. At the periphery of the burn wound, the zone of hyperemia contains viable epidermis cells with vasodilation mediated by local inflammatory mediators. Tissue in this zone almost invariably recovers unless complicated by edema, infection, prolonged vasoconstriction, and/or hypoperfusion.
Jackson’s zones of burn injury in partial- and full-thickness wounds.
(From Quizlet.com, Burns.)
Interest in cooling of the wound to minimize the extent of injury can be traced to antiquity , ; but firm evidence of its efficacy is lacking. Cooling immediately after injury should not supersede other priorities in the evaluation of the injured patient. The optimal temperature and duration of cooling are unknown, and excessive or prolonged cooling may be harmful in that it promotes vasoconstriction and systemic hypothermia ; the latter is consistently associated with poor outcome. Generally limit cooling the injury with tap water to 30 minutes for only minor burns (i.e., ≤10% total body surface area [TBSA]). In areas with fewer resources and limited burn surgical expertise, minimizing wound progression through immediate cooling is typically more prioritized. Modalities to improve dermal perfusion and block secondary injury from released inflammatory mediators have also garnered much interest. Whereas potential efficacy for many pharmacologic agents (e.g., heparin, steroidal and nonsteroidal antiinflammatory agents, thromboxane inhibitors, epidermal growth factor) has been reported, all remain investigational because none has demonstrated clinical effectiveness.
Assessment of burn depth
Clinical examination
Burn injury may involve one or both layers of the skin, and may extend into the subcutaneous fat, muscle/tendon, and even bony structures. Burns involving only the layers of the epidermis (first-degree burns) are erythematous and painful but do not form blisters and maintain much of the skin functions (e.g., mechanical, fluid, and infection barrier). Most sunburns fit this category of superficial, epidermal injury. Within 3 to 4 days, the dead cells/layers of the epidermis slough and are replaced by regenerating keratinocytes from the uninjured stratum basale.
Superficial dermal burns (superficial second-degree burns) extend into the papillary dermis and characteristically form blisters. Blistering may not occur immediately after injury, and burns initially perceived to be superficial may subsequently be diagnosed as dermal burns. The wound bed underlying a blister on a superficial partial-thickness burn is pink, wet, blanches after pressure, and is hypersensitive to touch ( Fig. 9.2 ). Debriding the blister is often painful due to the currents of air passing over the wound and exposed nerve endings already sensitized by local inflammatory mediators. These wounds blanch with pressure because of vasodilation and increased blood flow in the dermis compared to normal skin. With appropriate wound care, superficial dermal burns usually heal within 2 weeks and carry a low risk of scarring; they do not require excision and reconstruction. Use of dressings and/or temporary epidermal substitutes while the wound epithelializes is generally sufficient.
Superficial partial-thickness burn wound.
(From Tiny Hearts Education.)
Deep dermal burns (deep second-degree burns) extend into the reticular dermis and generally take 3 or more weeks to heal. They blister, but the underlying wound surface appears mottled pink, white, and/or discolored immediately following the injury ( Fig. 9.3 ). When pressure is applied to the burn, capillaries refill slowly or not at all. The wound is often less sensitive to pinpricks than the surrounding normal skin. By postburn day 2, the wound may be white and dry. As a rule, most deep partial-thickness burns that have not healed by 3 weeks should be excised and reconstructed (e.g., split-thickness autograft). For select patients (e.g., wound location and size, patient preference, low-anesthesia risk-to-benefit ratio), wound preparation to preserve healthy dermis and suspension epidermal autografting is also an option. Although the wound may close more quickly, the long-term impact of this technique on scarring and function remains unknown.
Deep partial-thickness burn wound.
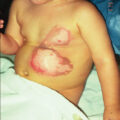
Full-thickness burns (third-degree burns or deeper) involve the entire cutaneous layer and may extend into the superficial subcutaneous tissue. Their appearance may be charred, leathery, dry, firm, discolored, and depressed when compared to adjoining normal skin. Noncharred full-thickness burns can be deceptive. Like deep dermal burns, they may be mottled and discolored in appearance (e.g., white, red) ( Fig. 9.4 ). However, they do not blanch with pressure and are uniformly dry. In some cases, the burn may appear somewhat translucent with deeper clotted vessels visible. These wounds are insensate to light touch and pinprick. Some full-thickness burns, particularly immersion scalds or bake injuries (i.e., burns caused by convective heat), may have a red appearance and can be confused with a superficial dermal burn by an inexperienced observer. Most full-thickness burns should undergo early excision and reconstruction to minimize risks of infection, hypertrophic scarring, and reduced function and to expedite patient recovery. Deeper burns that involve subcutaneous fat (fourth-degree burns), muscle (fifth-degree burns), and bone (sixth-degree burns) typically require surgical management unless they are very small or in very high-risk patients.
Full-thickness wound on the left flank.
Partial-thickness scald burns are usually managed nonoperatively (i.e., without excision) for up to 10 to 14 days or are carefully prepared and treated with suspension epidermal autografts. If the wound progresses to being full thickness or does not appear to be capable of healing within 3 weeks, then excision and reconstruction or commitment to later scar management should be considered. Expectant management of superficial partial-thickness burns with good wound care for 2 weeks postinjury has been associated with excision of significantly smaller areas or avoidance of surgery entirely. , ,
The most difficult management decision involves the assessment and care for intermediate partial-thickness burns. The determining factor as to whether these burns heal in 3 weeks may be less than a millimeter of viable dermis with its regenerative appendages. These burns are aptly called indeterminate burns because their healing potential becomes evident with serial assessments over several days after injury. Histologic studies suggest that burn injury is a dynamic process that peaks around 3 days postinjury. Initial evaluation by an experienced surgeon as to whether an indeterminate dermal burn will heal in 3 weeks is only 50% to 70% accurate.
Although developed to improve the timeliness of early eschar removal and reduce the collateral damage associated with tangential excision, chemical debridement with proteolytic enzymes can serve as a triage tool and adjunct to routine wound assessment for indeterminant partial-thickness wounds. The challenge of accurately assessing the potential for wounds to heal within 2 to 3 weeks with sufficiently low risk of hypertrophic scarring is related to our inability to determine viable dermis. Chemical or enzymatic debridement allows nonsurgical removal of nonviable dermis and reveals healthy tissue. Reassessment of the wound after chemical debridement allows a more accurate estimation of healing potential. However, the appearance of the wound after enzymatic debridement is different than nondebrided burn wounds and requires acquired expertise ( Fig. 9.5 ). When used as an adjunct to serial examination, enzymatic debridement may facilitate earlier diagnosis of wounds that require excision and reconstruction.
Appearance of wound before and after enzymatic debridement.
Note that, after enzymatic debridement, the white tissue represents viable dermis (i.e., partial-thickness burn).
Adjuncts to clinical evaluation
Search for technologies to obtain a more precise and timely method to diagnose burn depth surged when the benefits of early excision and reconstruction were recognized. Multiple modalities have been trialed, including thermography, photometry, optical coherence tomography, nuclear imaging, pulse-echo ultrasound, and laser Doppler and speckle contrast imaging ( Fig. 9.6 ). , , These techniques take advantage of the ability to detect dead cells or denatured collagen (e.g., biopsy, ultrasound, vital dyes), , wound color (e.g., light reflectance), physical changes such as edema (e.g., magnetic resonance imaging), and altered blood flow (e.g., indocyanine green or fluorescein, laser imaging, thermography). , Unfortunately, none of these techniques has proven superior and practical in a clinical setting to serial examination by an experienced burn provider. For now, these techniques remain adjunct, rather than a substitute, for clinical assessment.
Wound evaluation with laser speckle contrast imaging.
(From Mirdell R, Farnebo S, Sjöberg F, et al. Interobserver reliability of laser speckle contrast imaging in the assessment of burns. Burns . 2019;45[6]:1325-1335.)
Mechanisms of thermal injury
Flash and flame burns
Flash and flame burn injuries represent between 10% and 80% of burn injuries in different populations depending on energy, poverty, access to safe cooking, lighting and heating appliances, implementation and enforcement of building codes, and workplace safety standards. Explosions of natural gas, propane, gasoline, and other flammable liquids cause intense heat for a very brief time, causing flash burns. Flash burns often result from use of a flammable liquid as a fire accelerant (e.g., camp, trash, and brush fires). For the most part, flash burns injure dermal layers in proportion to the amount and kind of fuel that explodes. Gasoline, especially, has highly flammable vapors that are 3 to 4 times denser than air. At room temperature, gasoline vapors diffuse above ground and accumulate in enclosed spaces. Clothing, unless it ignites, is protective in flash burns. Thus a flash burn-injury distribution typically involves exposed skin, with the deepest areas facing the source of ignition. If a flash burn causes clothing or hair to catch on fire, then the injury will be more typical of a flame burn. Whereas flash burns often heal without extensive skin grafting, they may cover large areas and may be associated with thermal injury to the upper airway.
Flame burns invariably generate deep dermal or full-thickness injury because of exposure to intense heat even if only for a short period of time. Although the incidence of injuries from house fires has decreased with the widespread presence of functional smoke detectors/alarms, self-extinguishing cigarettes, residential sprinkler systems, and flame-retardant fabrics, continued smoking, improper use of flammable liquids, and clothing ignited from stoves or heaters still contribute to major morbidity and mortality globally. People whose bedding or clothes have been on fire usually sustain some full-thickness burns. People injured in structure fires with prolonged exposure to flames or heat due to immobility, intoxication, or confusion caused by hypoxemia or systemic toxicity (e.g., carbon monoxide, cyanide) are also prone to sustain deeper burns. Loss of consciousness exposes people to convective heat inside a burning room. This type of bake injury may deceptively appear shallow with intact epithelium to the inexperienced observer but is truly a full-thickness burn.
Scald burns
Hot liquid scalds are the next most common cause of burn injuries. Despite health promotion and injury prevention initiatives, the epidemiology and incidence of scalds worldwide has changed very little. The depth of scald injury depends on the liquid-specific heat, viscosity, and temperature as well as skin thickness and duration of contact. Water at 140°F (60°C) creates a deep dermal burn in 3 seconds but will cause the same injury in 1 second at 156°F (69°C). , Freshly brewed coffee from an automatic percolator is generally about 180°F (82°C). Once in the pot, coffee temperature approximates 160°F (70°C). Boiling water often causes a deep dermal burn unless the duration of contact is very short. Soups and sauces, which have a higher viscosity due to solids and oil, remain in contact longer with the skin and invariably cause deep dermal burns. Exposed areas tend to incur shallower burns because clothing (e.g., diapers, socks) retains the hot liquid in contact with the skin for a longer period. Consequently, scald injuries are often a mosaic of superficial, indeterminate, and deep dermal burns. A common example is a toddler who reaches above head level and pulls down a cup of coffee or tea or a bowl of noodles; the face bears a superficial burn, the trunk burn is of indeterminate thickness, and skin under the diaper has a deep dermal burn.
Immersion scalds are often deep because of prolonged skin exposure even though the water temperature is not as high as it is with spill scalds. These injuries more frequently occur among individuals who do not perceive pain normally (e.g., people with peripheral neuropathy or spinal cord injury) or who are not able to escape (e.g., young children, elderly, people with physical and/or cognitive disabilities). The latter are vulnerable to nonaccidental injuries. Circumferential extremity injuries (e.g., stocking distribution) and symmetric burns to buttocks and perineum with sparing of seated and flexor areas represent injury patterns that should raise suspicion of nonaccidental trauma ( Fig. 9.7 ). Allegations of abuse should include expert burn wound assessment by a team that includes experienced burn provider(s), pediatrician(s)/geriatrician(s), and suspected child abuse and neglect program member(s) who are familiar with burn injury etiologies and patterns and use validated maltreatment risk assessment tools.