9 Fat Grafting to the Breast: Aesthetic and Reconstructive Applications
Summary
Structural fat grafting has assumed an increasingly important role in both cosmetic and reconstructive breast surgery. This chapter serves to highlight the indications for, as well as critical technical aspects of, the performance of the procedure. Clinical examples and outcomes are provided.
Key Teaching Points
Fat must be injected in small aliquots to facilitate graft take.
Injection must be at multiple levels in different directions to encourage revascularization.
The larger the fat globule injected, the less likely the center will revascularize. Large grafts tend to resorb due to central ischemia.
Even layers should be built up to create smooth surface contours.
Fat is best prepared by gentle washing.
Observations
Since the development of implantable devices and the introduction of autologous tissue transfer in aesthetic and reconstructive breast surgery, surgeons have faced the dilemma of how to deal with complications such as contour irregularities, capsular contracture, and minor breast asymmetries. Autologous fat grafting was used as a potential solution to some of these problems in the past, but results were often unpredictable, in large part as a result of resorption, fat necrosis, and cyst formation. In the past two decades technical refinements to the harvesting, preparation, and injection of autologous fat have dramatically improved clinical outcomes. Much of the credit for these advances is attributable to Sydney Coleman, a pioneer in this field, who has provided safer technique and instrumentation to facilitate the safe injection of fat, with results that are more stable, predictable, and long-lasting Although the initial advances were made primarily in facial aesthetic surgery, ultimately applications were developed for the breast, including lipofilling of contour defects around implants and autologous reconstruction. It was soon noted that autologous fat appeared to have a beneficial effect on the adjacent tissue, including irradiated skin flaps, with improvements in texture, contour, and elasticity associated with neovascularization. This further paved the way for a study of the regenerative effects of stem cells harvested within the autologous fat, and this has led to further advances and applications in the use of fat injection for complex reconstructive problems.
With autologous fat injection firmly established in the reconstructive arena in breast surgery, it was inevitable that a role for fat grafting would be incorporated into such aesthetic breast procedures as augmentation mammaplasty and the treatment of developmental breast asymmetries, such as the constricted breast and Poland’s syndrome. The use of fat grafting for breast augmentation is still controversial because of concerns about its impact on mammographic imaging as well as angiogenesis and its potential impact on breast disease. Regardless of its potential limitations, fat injection as a technique in breast surgery is here to stay and has been a dramatic advance for the correction of previously intractable problems as well as an outstanding tool for the correction of minor asymmetries that were very difficult to address simply.
9.1 Biological and Cellular Foundations
Multiple studies confirming the regenerative properties of grafted fat have added a new approach to reconstructive and aesthetic breast surgery. Among the different approaches that have been proposed, those using adipose-derived mesenchymal stem cells (ASCs) appear to be the most successful and promising for the future. A growing body of literature has documented the regenerative potential of the ASCs found in lipoaspirate. The procedures required for harvesting, purifying, and grafting ASCs are relatively straightforward; in vitro cell expansion is not required, and there are few clinical contraindications to the use of fat grafting.
Autologous fat grafting is not a new concept in plastic surgery. Procedures in which fatty tissue was injected were described in the 19th century for therapeutic and cosmetic corrections. However, until relatively recently, little was understood about the biological cellular mechanisms triggered by the transplantation of fat. For many years surgeons rejected the concept of lipofilling, considering it to be a procedure that produced inconsistent results because of resorption, fat necrosis, and the development of oil cysts and granulomas. More recently, the techniques introduced by Coleman for centrifugation and preparation of fatty tissue and his exacting criteria for uniform placement of lipoaspirate have significantly improved the quality and stability of lipofilling outcomes.
In recent years, pioneering clinical studies have focused on purified lipoaspirate transplantation for the treatment of the late side effects of radiation therapy and breast deformities after conservative treatment. These reports were accompanied by immunophenotypic and molecular characterization of lipoaspirates, which allowed researchers to identify and quantify the presence of ASCs and study their potential in comparison with bone marrow–derived stem cells. Ultimately, this has led to a radical change in the way adipose tissue is used in plastic surgery. In particular, it is now understood that the regenerative capability of fat grafting results not from injection of mature adipocytes but of mesenchymal stem cells, which are found in abundance in the vascular-stromal fraction of adipose tissue.
From the perspective of regenerative medicine applied to breast reconstruction and aesthetic surgery, the angiogenic, antifibrotic, and anti-inflammatory capabilities of ASCs have proved very effective in promoting the formation of new adipose tissue for breast filling and reconstruction.
In this chapter, we present our clinical experience with autologous fat grafting for the treatment of breast radiolesions, for total breast reconstruction, and for aesthetic breast procedures. Clearly, ASCs in regenerative medicine are destined to play a major role in breast reconstruction and aesthetic surgery.
9.2 Biological Foundations and Basic Technique
The biological foundations of autologous fat grafting have only recently been investigated to support and explain the clinical outcomes achieved from fat grafting performed in pioneering clinical studies. These studies have demonstrated the remarkable regenerative potential of fat grafting in the repair of ischemic and/or fibrotic tissue.
The biological foundation of such effects has been studied in multipotent stromal cell populations that exhibit properties similar to those found in bone marrow–derived stem cells. Cell biologists have shed light on the nature of the multipotent cell lineage contained in ASCs, which can be induced to myogenic, cardiomyogenic, epithelial, endothelial, and neurogenic differentiation and can express angiogenic, antifibrotic, and anti-inflammatory capabilities.
Although some of the biological mechanisms triggered by autologous fat grafting are still unclear, the current interpretation is that the use of a repeated process of liposuction, purification (centrifugation), and targeted injection of autologous tissue transfers active stem cell niches, including stem cell populations and the surrounding specialized microenvironment, from young, vital tissues to targets where stem cell niches are lacking as a result of pathologic conditions, external agents, or the natural aging process.
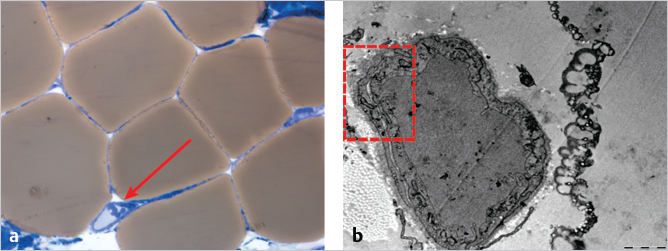
This photomicrograph shows the typical microscopic structure of tissue after fat grafting (▶Fig. 9.1a). The brown areas are mature adipocytes; the light blue area is the stromal component. The red arrow indicates the adipose stem cell niche, consisting of a blood vessel surrounded by adipose stromal cells. This electron photomicrograph shows the ultrastructure of an adipose stem cell niche (▶Fig. 9.1b). At the top left of the depicted vessel (dashed red box), the beginning of vessel membrane duplication (a veil cell) is evident; this event is considered to be related to the start of the biological process of cell formation.
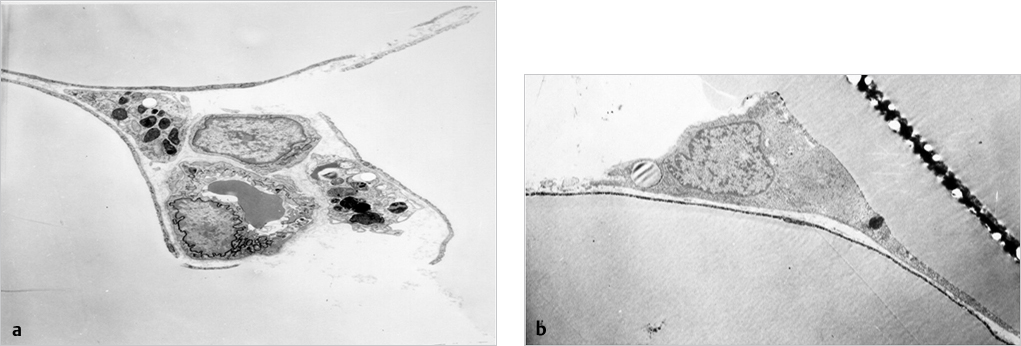
The next stage of transformation is depicted in an electron photomicrograph demonstrating multiple new cell formations around the vessels (▶Fig. 9.2a). Higher magnification demonstrates transformation into preadipocytes exhibiting the typical lipidic droplet (▶Fig. 9.2b).
From a practical standpoint, transferring adipose stem cell niches—the starting point of the regenerative process depicted above—is simple, does not require specific in vitro cell culture, and has few contraindications. Thus fat grafting has the potential for use in treating a broad range of tissue defects as well as pathologic conditions, such as radionecrosis or ulceration.
9.2.1 Operative Technique
Technical Pearl
While fat grafting techniques are relatively simple, they are highly technique and operator dependent, and meticulous attention to the basic principles of fat grafting is essential to success.
Preoperative Planning
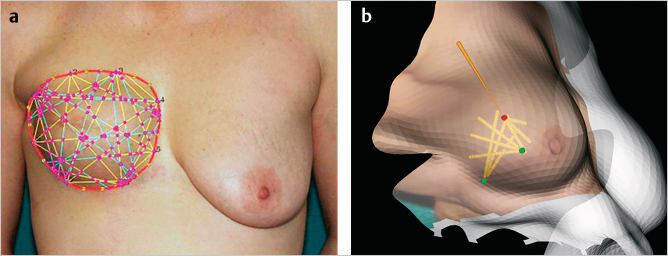
The surgeon must precisely identify the areas to be injected as well as the number and location of entry points and the direction of tissue injection to ensure uniform tissue deposition. Computer-assisted planning based on patient-specific two-dimensional photographs and/or three-dimensional surface and volumetric models can be used to optimize surgical planning and to guide the surgeon intraoperatively.
Images of preoperative planning are designed for optimization of uniform lipoaspirate infiltration (▶Fig. 9.3). On the left is a two-dimensional photograph-based plan; on the right is a three-dimensional surface model–based plan.
Harvesting of Lipoaspirate
As mentioned earlier, the success of autologous fat grafting by injection is highly technique and operator dependent. Fat cells are delicate and prone to damage when subjected to high negative-pressure suction and trauma from sharp harvesting cannulas. Coleman’s basic principles are crucial to a successful outcome.
Basic Principles of Fat Grafting
Fatty tissue is harvested with a blunt cannula.
Low-power suction aspiration is used for harvesting tissue.
The lipoaspirate is processed to remove liquid oil, tumescent fluid, and nonviable cellular components.
Small volumes of concentrated fat are meticulously placed in each tunnel to allow rapid neovascularization.
The miniscule thread of fat is injected as the needle is withdrawn.
Fat harvest donor sites are as follows:
Abdomen.
Outer/inner thighs.
Medial knees.
Flanks.
Buttocks.
Technical Pearl
There is some data to suggest that lipoprotein lipase activity differs from area to area. For example, the enzyme activity is higher in the suprapubic area and lower in the outer thigh suggesting that thigh fat may be less prone to dissolution by lipases than abdominal fat.
Fat Harvest
Adipose tissue is harvested by liposuction. This can be done using either local anesthesia, local anesthesia with the patient under deep sedation, or under general anesthesia if appropriate.
Harvesting Techniques
Syringe suction.
Conventional mechanical suction.
Power-assisted liposuction.
Initially, Coleman’s syringe suction or Tulip’s syringe suction was deemed the most gentle and appropriate techniques for fat harvest. Subsequent research has shown that conventional suction is acceptable and power-assisted liposuction has been shown to achieve as successful a graft take as the supposedly gentler syringe techniques. The donor sites are infiltrated with standard tumescent solution containing 1:1,000,000 epinephrine and 20 mL lidocaine 0.5% per 1,000 mL.
Syringe Suction
Syringe aspiration is performed using Coleman’s instrumentation or the Tulip system. Shown are Coleman’s 15-cm straight and curved cannulas and 23-cm curved and straight cannulas (▶Fig. 9.4).

A 2-mm diameter blunt-tipped, distal orifice cannula connected to a 10-cc Luer-Lok syringe is used for harvesting (▶Fig. 9.5). Fatty tissue is harvested through the incision made for infiltration of the saline solution, but the harvest site usually needs to be enlarged with a number 11 scalpel blade to allow easy access of the 2-mm cannula. Aspiration must be done gently, avoiding too much negative pressure to prevent damage to the delicate adipocytes. Coleman advocates no more than 2 cc of negative pressure within a 10-cc syringe barrel.
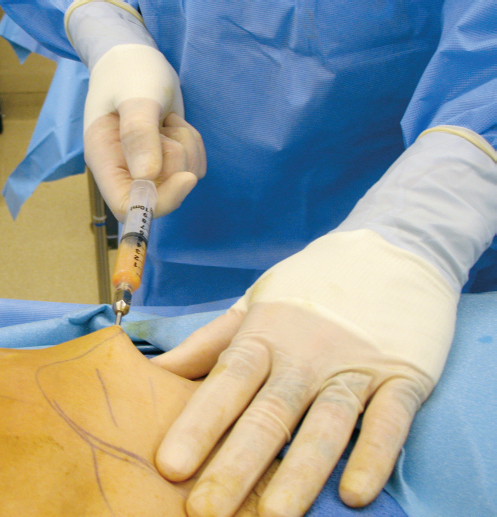
Once harvesting is complete, the cannula is removed from the syringe and a closed syringe cap with a Luer-Lok is applied to the tip of the syringe (▶Fig. 9.6). The plunger is removed, and the syringe is ready to be placed directly into a centrifuge.
Power-Assisted Liposuction
If power-assisted or conventional liposuction is used, the vacuum power should be set at 19 to 21 inches of suction and a 4- to 5-mm turbo cannula can be safely used with pre-tunneling of the donor site using a basket cannula (▶Fig. 9.7). Power assistance allows for a much less tiring harvest for the surgeon and large-volume aspiration can be performed. It does, however, require an in-line trap to capture the harvested fat for processing.

Purification of Lipoaspirate
The processing of fat has received considerable attention over the last 10 years. Coleman’s original description relied on centrifugation (▶Fig. 9.8).

While the procedure produces clean particulate fat, the processing is slow and tedious and large volumes require considerable time expenditure. Subsequent data have suggested that strainer washing or various washing devices produce higher fat graft take rates.
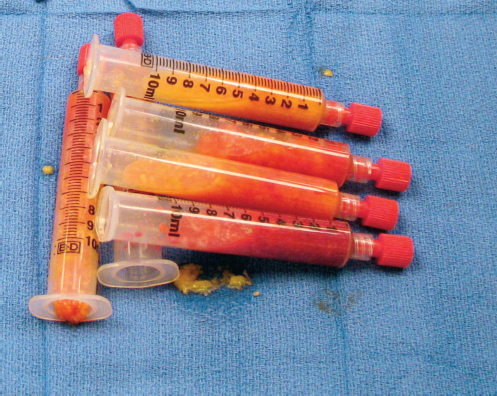
A centrifuge is used to purify the lipoaspirate by separating the liquid components of the aspirate, including oil, triglycerides, and tumescent fluid. If these are not removed initially, they will rapidly resorb after injection, resulting in a lower-volume correction than initially expected. Various centrifugation protocols have been described for optimal purification.
At the end of the centrifugation, three levels are present in the syringe based on their density (▶Fig. 9.9). The top layer consists of oil from ruptured fat cells; the middle layer contains potentially viable fatty tissue; and the bottom layer is composed of blood and residual saline, epinephrine, and lidocaine solution.
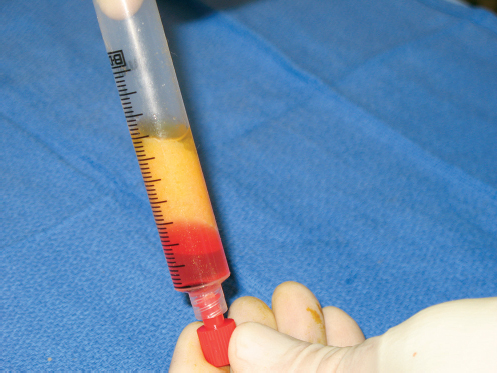
The layers of oil and residual liquid are drained, leaving a solid plug of fat within the syringe barrel that is now ready for injection (▶Fig. 9.10a). Using a Coleman Luer-Lok connector, the fat is transferred into 10-cc syringes; from here it can be transferred to 1- or 10-cc syringes equipped with an infiltration cannula for injection into the operative site (▶Fig. 9.10b).
Gravitational Washing
Gravitational washing is used by several manufacturers, with or without stirring mechanisms. The simplest technique is that used by MicroAire. A large in-line trap collects up to 2 L of fat for washing. Fluid is run into a canister of fat and allowed to settle over a 10- to 15-minute period. The supernatant washing fluid is aspirated leaving cleaned fat floating on its surface (▶Fig. 9.11). The fat can be decanted from the spout at the base of the canister and loaded into 10-cc syringes for injection using blunt-tipped injection needles. The problem with this technique is that, although it is capable of harvesting large volumes of fat, it is time consuming and produces rather wet fat. The high water content results in early volume loss which must be accounted for by over-injection.
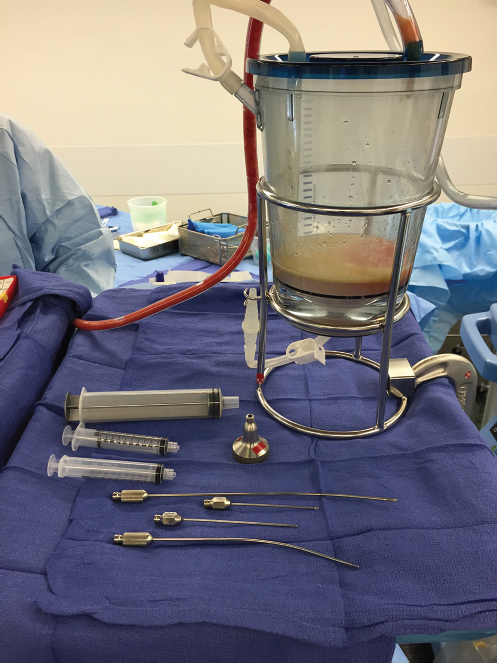
Gravitational Washing with Stirring
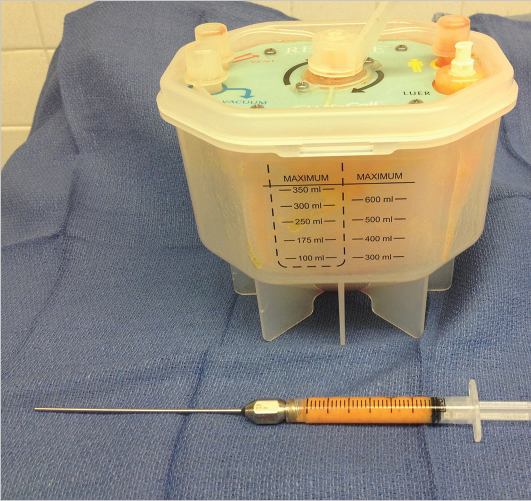
The most widely accepted device currently in use in the United States is the Revolve system by Allergan’s LifeCell. The system involves the use of an in-line trap with a capacity for 375 cc of fat. This is then washed with 1.5 L of warm Ringer’s lactate solution, stirring the mixture with a built-in baffle system that traps strands of connective tissue. Once washing is complete, the fat is aspirated and loaded into 10-cc syringes as usual. The beauty of the system is that it is a closed system which yields incredibly well-washed fat with a lower water content than most of its competitors (▶Fig. 9.12). It is somewhat cumbersome to use at first but is about to be replaced by the next-generation device, which can hold twice the volume (600 cc) and relies on a simple lever system to move between acquisition, washing, and delivery into syringes.
Placement of Lipoaspirate
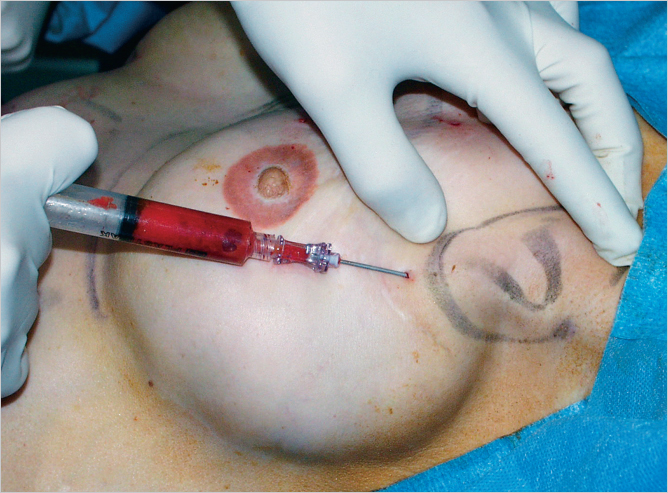
The fat grafting procedure is performed with a local anesthetic and the patient under deep sedation or general anesthesia (▶Fig. 9.13).
Technical Pearl
Blunt needles are used to avoid perforation of veins and arteries, leading to hematoma formation or fat embolism during injection (▶Fig. 9.14). Vasoconstrictive measures are advisable to minimize the incidence of hematomas.

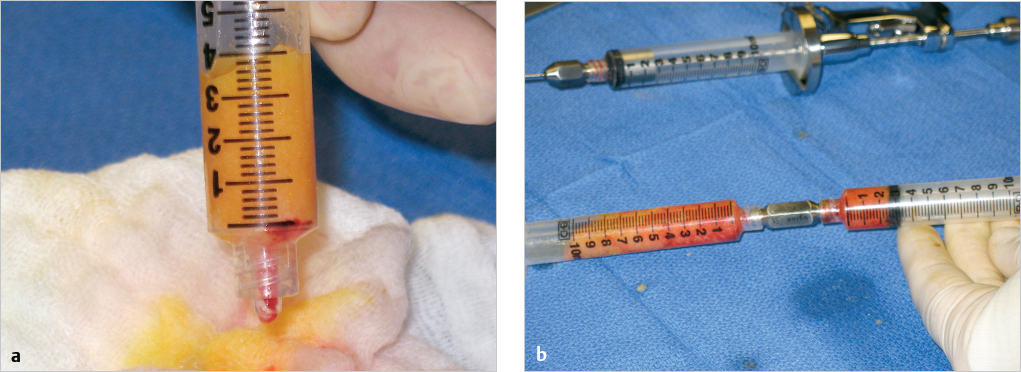
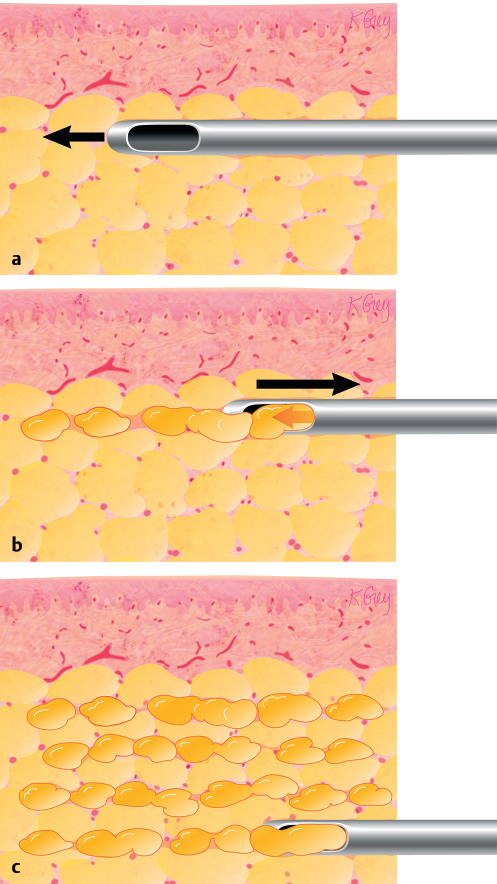
The key to increasing the efficacy or “take” of the graft is to maximize the contact of the lipoaspirate with the host tissues to optimize vascularity. For this reason it is essential to place minute aliquots of lipoaspirate in each tunnel; the miniscule thread of fat is injected as the needle is withdrawn (▶Fig. 9.15a–c). The injector must avoid introducing large quantities of tissue in a single pass, because this can result in the development of oil cysts and/or microcalcifications.
The total quantity of lipoaspirate to be injected must be carefully assessed to avoid problems associated with stiff, tight subcutaneous and glandular tissues and skin contraction. The appropriate quantity should be decided case by case. Care is taken to preserve some elasticity within the treated area at the end of the procedure. Injection sites can be closed with a single suture of 5–0 fast-absorbing catgut.
The number of individual treatment sessions depends on the initial clinical picture and on the patient’s response to the cell therapy. In our experience, three to six treatments have proved sufficient for all our patients. Nevertheless, additional administration of lipoaspirate is not contraindicated.
9.3 Clinical Applications
9.3.1 Fat Grafting for Healing of Chronic Radiation-Induced Lesions
Structural fat grafting was introduced in the clinical practice of regenerative plastic surgery for the healing of radiation-induced chronic ulcers. These problems occur in oncologic patients, who develop late side effects of megavoltage external radiation therapy, particularly when a boost dose has been given to the tumor bed. These radiation-induced lesions do not improve spontaneously and tend to progress as tissues become more atrophic and fibrotic, with potential ulceration.
Complications of radiation therapy are classified according to the LENT-SOMA toxicity scale:
Grade 1—minor symptoms that require no treatment.
Grade 2—moderate symptoms, requiring only conservative treatment.
Grade 3—severe symptoms, which have a significant negative impact on daily activities and require more aggressive treatment.
Grade 4—irreversible functional damage, necessitating major therapeutic intervention.
Grade 5—loss of the organ or death.
Classic symptoms include fibrosis, retraction, and ulceration accompanied by pain and extrusion of an expander or implant. In the worst cases, ulceration may be accompanied by radionecrosis of subcutaneous tissues, muscle, and bone. In LENT-SOMA grade 3 (severe symptoms) and grade 4 cases, radiation-induced lesions are either indolent or progressive, requiring extensive surgery to excise necrotic tissue, followed by flap closure. These procedures are complicated and are associated with high morbidity, including potential delayed healing at the juncture of the flap with adjacent viable but irradiated tissue.
Results from our pilot study used ultrastructural analysis to provide evidence that radiolesions are characterized by microangiopathic etiologic factors, thus making them perfect targets for the angiogenic capabilities of ASCs (▶Table 9.1).
Patient Examples
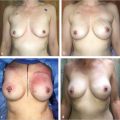
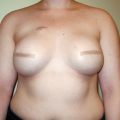
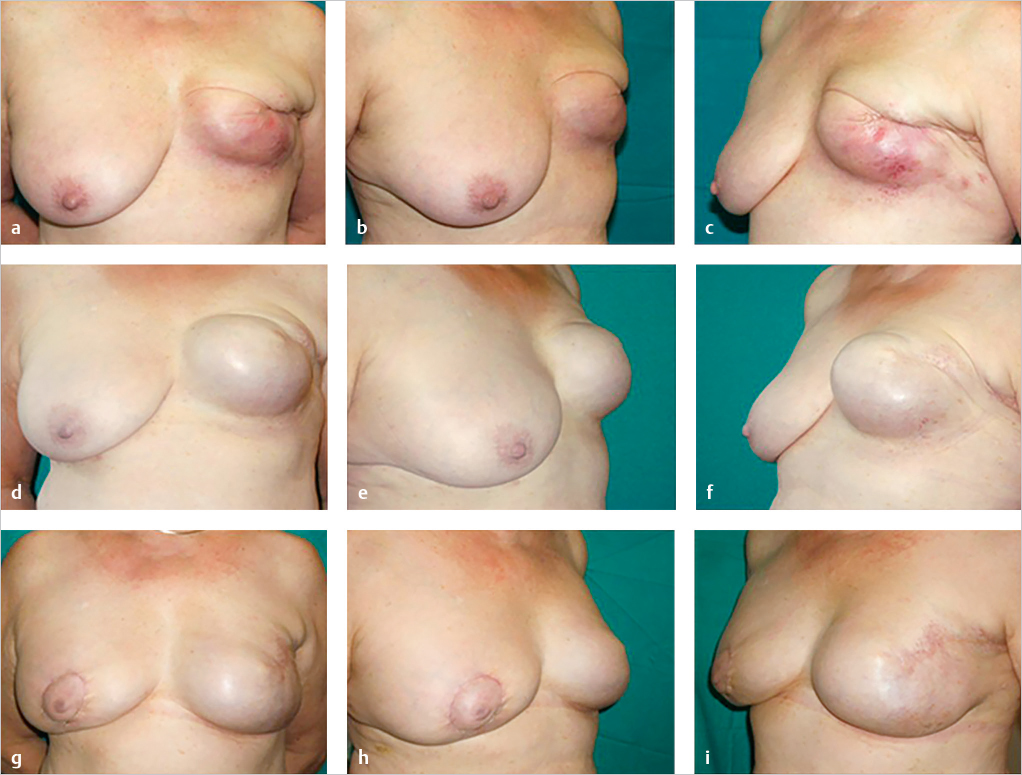
This patient presented with a LENT-SOMA grade 4 lesion of the left breast, with signs of imminent extrusion and thinning of her lower pole mastectomy skin envelope (▶Fig. 9.16). She had undergone radiation therapy after implantation of her breast expander. She was offered autologous fat grafting in an attempt to salvage her deteriorating left breast reconstruction. After four sessions of fat grafting, her clinical picture improved to the point that expansion could be resumed. After successful expansion, an expander–implant exchange was performed for a gel-filled device, accompanied by contralateral right mastopexy. She is shown following these procedures with a successful salvage on the reconstructed side. Her reconstructed breast is soft and naturally shaped.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree