6 Multimodality Imaging in the Interdisciplinary Management of Breast Diseases
Summary
Breast cancer is primarily a surgical disease when detected by mammography screening in the preclinical phase as either an in situ or a 1- to 14-mm invasive tumor, still localized to the breast. The current breast cancer terminology reflects the era of large, advanced tumors with a poor outcome. Mammography screening detects breast cancer at earlier phases of development, challenging physicians with an unprecedented large number of nonpalpable breast cancers. Clinical practice has been modified considerably to diagnose and treat these early breast cancers appropriately, but, unfortunately, few changes have been made in the terminology of breast cancer histopathology. The current tumor/node/metastasis (TNM) classification system was created prior to the widespread use of mammography screening and has shortcomings in describing the extent of the early-phase tumors. These shortcomings limit our ability to predict the long-term outcome of individual patients with screen-detected, nonpalpable breast cancers. Since the long-term patient outcome appears to be largely determined by the site of origin of breast cancer, we use a classification system which is based on the apparent anatomical site of origin of breast cancer: the major lactiferous ducts (ductal adenocarcinoma of the breast [DAB]), the terminal ductal lobular units (TDLU) (acinar adenocarcinoma of the breast [AAB]), and the mesenchyme (diffusely invasive breast cancer of mesenchymal origin). The mammographic appearance of each of these breast cancer subtypes reliably predicts the long-term outcome and should be used as imaging biomarkers.
Multifocality and the diffuse nature of breast cancer are independent poor prognostic factors. Breast magnetic resonance imaging is essential for the preoperative assessment of tumor distribution and disease extent for all breast cancer cases. Determining the site of origin of breast cancer by thorough multimodality imaging workup assists in evaluating whether the skin- and nipple-sparing procedure will be suitable for a given patient. Multifocal tumors are poorly suitable for these procedures. The least suitable groups of breast cancers are the DAB subtypes and the diffusely infiltrating tumors of mesenchymal origin.
Correlation of imaging findings with the underlying histology is greatly improved using large format thin and thick section (subgross, three-dimensional) histopathologic techniques.
Custom-tailored management of both palpable and nonpalpable breast cancers is more important than ever before and can best be accomplished through the combination of adequate preoperative mapping of the true extent of the disease and a thorough correlative postoperative histopathologic evaluation employing the large format histopathology technique.
The variety of therapeutic choices and the rapidly improving diagnostic armamentarium emphasize the necessity for close interdisciplinary collaboration for both the preoperative management and postoperative follow-up of cancer patients. The ever-increasing variety of oncoplastic and aesthetic surgery presents a considerable challenge for the imager. On the other hand, the plastic surgeons need to understand the capabilities and limitations of the various imaging tools currently available.
Key Teaching Points
Terminology of breast cancer based on the site of origin.
Unifocal and multifocal in situ acinar adenocarcinoma of the breast (in situ AAB).
Unifocal and multifocal invasive acinar adenocarcinoma of the breast (invasive AAB).
Implications of multifocality.
Flaws inherent to the current tumor/node/metastasis (TNM) classification of breast cancers.
Rationale for multimodality imaging.
Female and male breast cancers localized behind the areola.
Cooper’s ligaments are not ligaments.
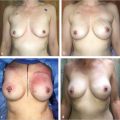
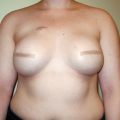
Imaging after breast augmentation.
Imaging of benign changes after TRAM flap surgery.
Radiologic changes after breast reduction.
Breast implant imaging.
Observations
Plastic surgeons and oncological surgeons are seeing increasing numbers of women of all ages who present for both cosmetic and reconstructive breast surgery. Given the increased incidence of breast cancer in the female population as a whole, including younger women, it is imperative that we have an intimate understanding of the radiologic techniques available for breast screening and diagnosis.
Patients who undergo breast surgical procedures may develop complications, which require investigation and diagnosis. Breast reduction and TRAM flap reconstruction may predispose patients to the development of fat necrosis, oil cysts, or calcifications that must be differentiated from malignancy. Procedures involving implants may be associated with calcified capsular contracture, intraluminal gel rupture with or without migration, and architectural distortion that is visible on mammograms. The emerging use of autologous fat grafting to correct deformity in reconstructed breasts as well as for augmenting hypoplastic breasts has increased the need for imaging. Imaging of the fat-injected breast has become a topic of debate.
The last decade has seen the refinement of important diagnostic radiologic techniques, including digital mammography, tomosynthesis, automated breast ultrasound, and breast-specific magnetic resonance imaging (MRI). There is little doubt that these technologies are revolutionizing our ability to diagnose lesions earlier, to detect occult cancers, and determine the true extent of the disease.
The extensive use of skin-sparing mastectomy is widely accepted in properly selected cases and has greatly enhanced our ability to reconstruct a normal-looking breast. The desire to preserve the breast skin envelope may compromise tumor clearance when lesions are close to the breast surface. As will be seen in this chapter, use of the multimodality approach in breast imaging and use of large format histopathologic examination are crucial to avoiding local recurrence in these patients.
Why is the information in this chapter of value to the breast surgeon? Surgeons are given remarkably little didactic training in the interpretation of radiologic images. We perform surgical procedures that have a direct impact on the structure of the breast, yet we have little understanding of the problems in interpretation that these changes may cause for radiologists and pathologists. For example, fat injections may produce oil cysts, calcifications, and architectural distortion, any of which may make interpretation problematic. A poorly integrated biological matrix with seroma formation around it in the dependent portion of the breast after implant reconstruction may mimic the folds of a ruptured implant. These clinical scenarios require close collaboration among the surgeon, pathologist, and radiologist.
This chapter provides a comprehensive primer on interpreting histopathologic and radiologic images of benign and malignant breast pathology pertinent to breast surgeons. The goal is to foster a base of understanding for communication between patients and physicians.
6.1 Background
Breast cancer screening has resulted in the detection of ever-increasing numbers of nonpalpable, in situ, and 1- to 14-mm invasive tumors. The value of early detection has been tested and confirmed in prospective, randomized, controlled screening trials. More recently, comprehensive analysis of mammography service screening has reproduced the results of the randomized controlled trials. These trials have demonstrated a significant decrease in mortality from breast cancer, providing ample proof that breast cancer is not a systemic disease from the outset. Contrary to Fisher’s theory, the success of the screening programs can be explained by the detection and removal of breast cancer before viable metastases have become established, before it has become a systemic disease. Therefore, when detected by mammography screening in the preclinical phase as either an in situ or a 1- to 14-mm invasive tumor, cancer is primarily a surgical disease when it is still localized to the breast. We can conclude from the trial results that the final outcome for breast cancer patients is determined by the point in time during the natural history of breast cancer at which treatment is given. Surgical treatment of breast cancer in its early phase combined with adjuvant treatment in carefully selected cases results in a significantly better outcome than any combination of oncological therapeutic methods at a late stage.
The prerequisite for a significant improvement in the outcome of women with screening-detected breast cancer is that the malignant tissue be completely removed from the breast. For this to succeed, precise preoperative characterization of screening-detected breast cancers has become a crucial step when choosing the therapeutic pathways.
Teaching Points
There is a need to improve the current classification of breast cancer.
The current breast cancer terminology reflects the era of large, advanced tumors with a poor outcome. Mammography screening detects breast cancer at earlier phases of development, challenging physicians with an unprecedented large number of nonpalpable breast cancers. Clinical practice has been modified considerably to diagnose and treat these early breast cancers appropriately, but, unfortunately, few changes have been made in the terminology of breast cancer histopathology.
Also, the current TNM classification system was created prior to the widespread use of mammography screening and unfortunately has shortcomings in describing the extent of the early-phase tumors. These shortcomings limit our ability to predict the long-term outcome of individual patients with screen-detected, nonpalpable breast cancers.
We use a classification system, which is based on the apparent anatomical site of origin of breast cancer: major lactiferous ducts, terminal ductal lobular units (TDLUs) and mesenchyme, since the longterm patient outcome appears to be largely determined by the site of origin of breast cancer.
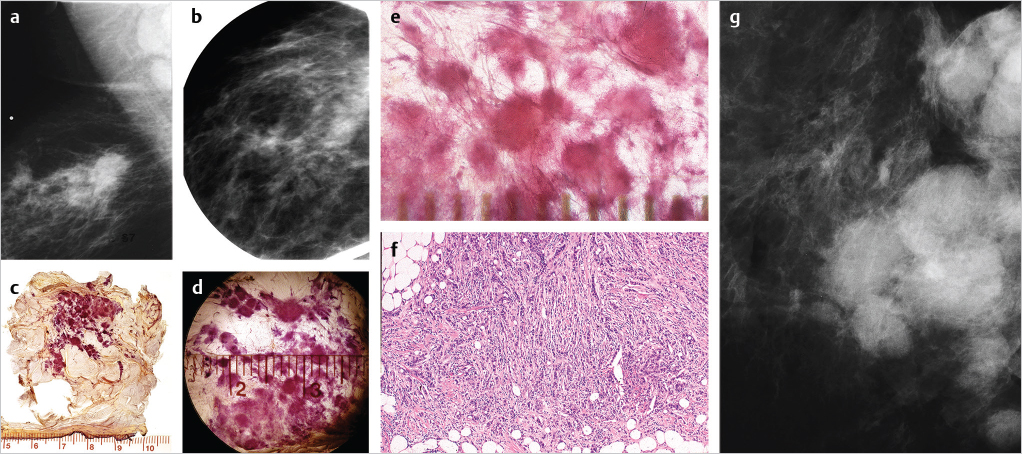
The current terminology tends to impair effective interdisciplinary communication and collaboration in disease management, because the stellate and circular/oval invasive breast cancers—currently termed “invasive ductal carcinoma” actually originate from the acini of breast lobules (TDLUs) (acinar adenocarcinoma of the breast, AAB). When these are detected and treated early, they have an excellent long-term outcome (▶Fig. 6.1). On the other hand, breast cancers, which truly originate from the major lactiferous ducts (ductal adenocarcinoma of the breast, DAB) have a significantly poorer long-term outcome regardless of the use of modern therapeutic regimens (▶Fig. 6.1) (see detailed description in section 6.3.2). The resulting overtreatment of many cancers of apparent acinar origin (AAB) and undertreatment of the cancers of apparent ductal origin (DAB) could be reduced by adopting terminology based on the anatomical site of tumor origin. Large-format thick section (3D) histopathology images are able to unequivocally demonstrate the anatomical structure of the breast in three dimensions as it is altered by the cancer. Examples of each basic type of structural alteration caused by malignancies are shown on ▶Fig. 6.2; their reflection on the mammograms, ultrasound, and breast MRI are shown on many other later figures.
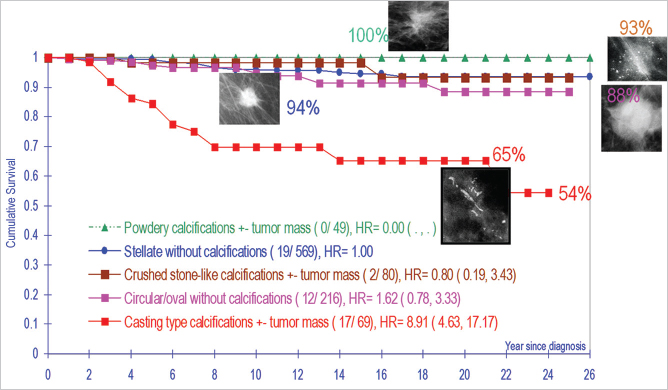
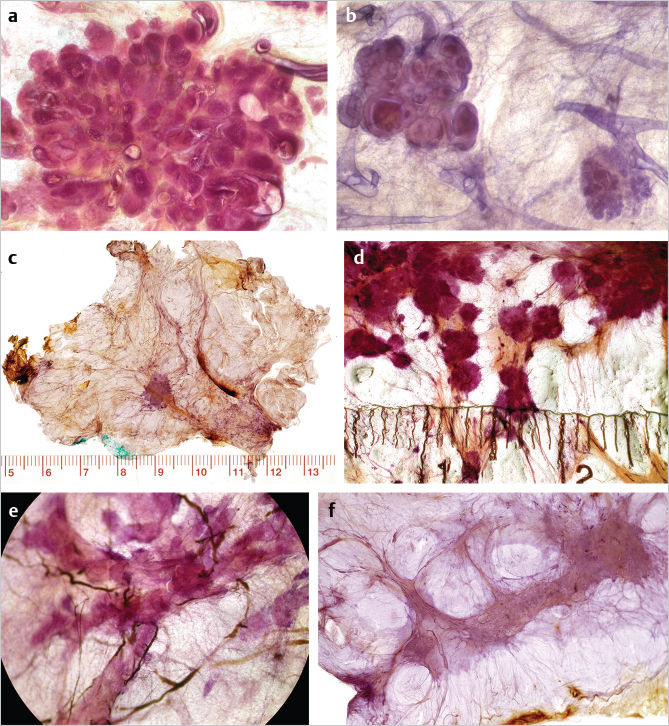
The frequency of unifocal, multifocal, and diffuse breast cancers has been studied using large-format histopathology examination of 1,000 consecutive breast cancers. Approximately 40% of the early invasive breast cancer cases were unifocal and 34% were multifocal. These two groups, comprising 74% of the material, have originated from the TDLUs, and should be termed unifocal AAB and multifocal AAB, respectively. The unifocal 1- to 9-mm stellate or circular invasive AAB cases are primarily surgical diseases with excellent 26-year survival, even without adjuvant therapy. The remaining 26% of the invasive breast malignancies were diffusely invasive breast cancers, of which 20% originate from the major ducts (DAB) (▶Fig. 6.2e). The remaining 6% are conventionally termed “diffusely infiltrating lobular carcinoma,” which we consider to be a diffusely infiltrating breast malignancy of mesenchymal origin (▶Fig. 6.2f). Breast MRI is essential for the preoperative assessment of tumor distribution and disease extent for all breast cancer cases.
The mammographic appearance of each breast cancer subtype reliably predicts the long-term outcome and can thus be used as imaging biomarkers.
6.1.1 Breast Cancers Originating from the Terminal Ductal Lobular Units
In situ Acinar Adenocarcinoma of the Breast
The majority of in situ carcinomas originate within the acini of the TDLUs but are unfortunately still termed “ductal carcinoma in situ” (DCIS). This nomenclature is misleading and anatomically incorrect. As has been pointed out by Jensen et al, “ductal carcinoma in situ of the human breast is of lobular origin. Progressive distension of ductules [acini] with dysplastic and anaplastic cellular elements leads to unfolding and coalescence of the ductules [acini] within the lobule to form larger ovoid structures. Such lesions falsely appear to be small ducts in conventional histology slides.” We confirmed these observations using large format, subgross, three-dimensional (3D) histopathology. Our suggested terminology for in situ carcinomas originating within the TDLUs is grade 1 or 2 unifocal or multifocal in situ AAB, to replace grade 1 and grade 2 “DCIS.” Additionally, the term “DCIS” is also a misnomer when the malignancy actually originates within the major lactiferous ducts (DAB), since this subtype of breast cancer may act through neoductgenesis as an extensive duct forming invasive cancer with a poor outcome.
Unifocal in situ Acinar Adenocarcinoma of the Breast
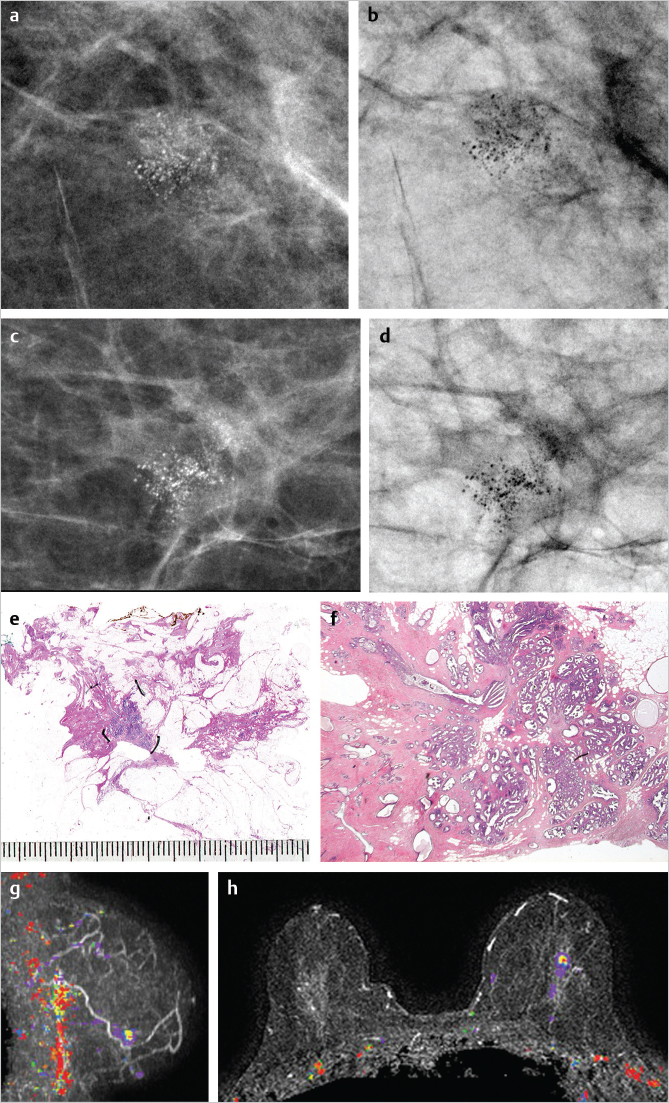
In these cases, large-format histopathology documents unifocal in situ carcinoma localized to a single TDLU. In grade 2 in situ AAB, this is seen on the mammogram as a single cluster of crushed stone-like microcalcifications.
Example
A 64-year-old asymptomatic woman, called back from mammography screening for assessment of the tiny cluster calcification in her left breast (▶Fig. 6.3).
Multifocal in situ Acinar Adenocarcinoma of the Breast
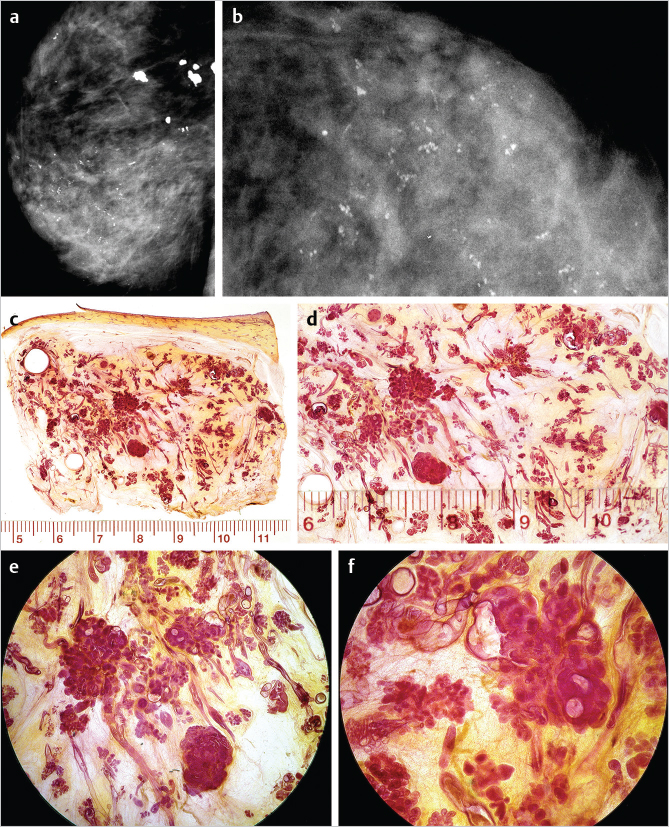
The examples on ▶Fig. 6.4 and ▶Fig. 6.5 represent multifocal grade 2 and grade 1 in situ AABs. The distribution of the calcifications in both grades 1 and 2 appears as multiple clusters on the mammograms, because the malignant process is confined to the TDLUs. The mammographic images shown on ▶Fig. 6.4 and ▶Fig. 6.5 clearly reflect the differences in the respective underlying histopathology: in grade 2 in situ AAB, the calcifications are the result of tumor necrosis within the TDLUs (▶Fig. 6.4), and are seen on the mammogram as multiple cluster, discernable, crushed stone-like microcalcifications. On the other hand, in grade 1 in situ AAB, the cancer cells produce fluid within which psammoma body-like calcifications are formed, seen as powdery calcifications on the mammogram (▶Fig. 6.5).
Example: Multifocal Grade 2 AAB
This 86-year-old woman was referred to mammography for a palpable thickening in her right breast (▶Fig. 6.4).
Example: Multifocal Grade 1 in situ AAB
This 46-year-old asymptomatic woman presented for screening examination. She was called back for assessment of the powdery calcifications distributed throughout much of the right breast (▶Fig. 6.5).
Comment
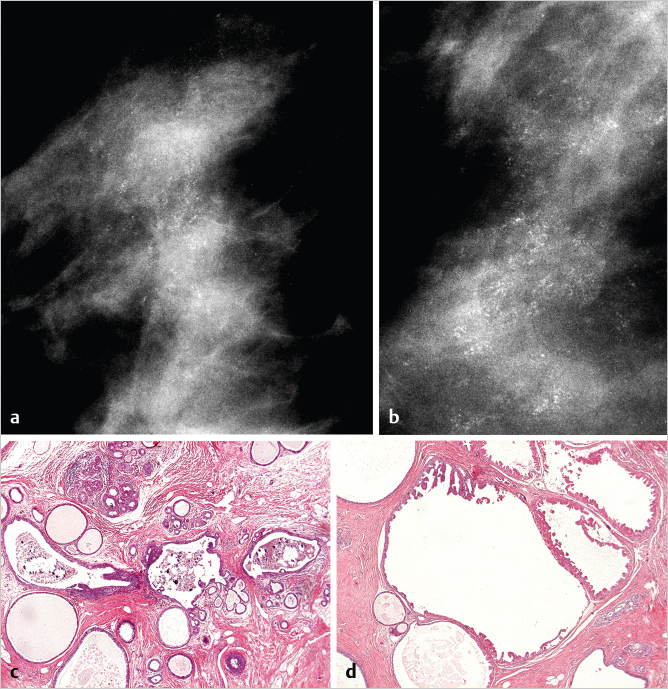
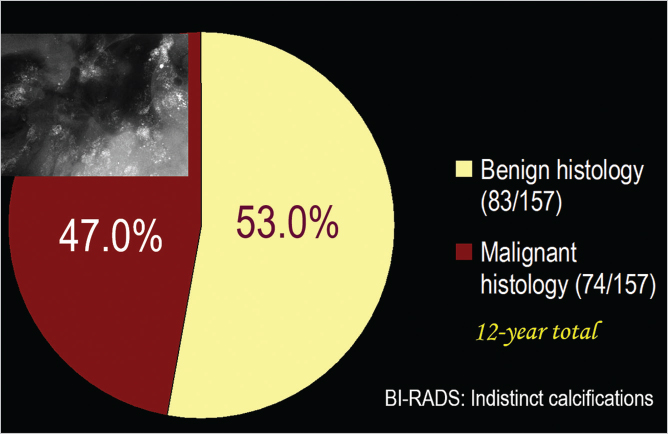
Powdery calcifications on the mammogram reflect the presence of psammoma body-like microcalcifications, which can be associated with either benign sclerosing adenosis (53% of the cases) or grade 1 in situ acinar adenocarcinoma of the breast (AAB) (47% of the cases) (▶Fig. 6.6). The two diseases have an identical mammographic appearance because they both produce the psammoma body-like calcifications. The seemingly harmless grade 1 cancer subtype has the potential to become invasive AAB and dedifferentiate to more aggressive and fatal breast cancers, a process that is more gradual than with grade 2 in situ carcinomas. Most of the 1- to 14-mm invasive breast cancers associated with powdery calcifications are tubular carcinomas or well-differentiated invasive AABs.
Teaching Point
Management of cases with powdery calcifications on the mammogram
Powdery calcifications on the mammogram with no associated tumor mass predict with equal probability the presence of either grade 1 in situ carcinoma or sclerosing adenosis and other benign processes within the lobule (▶Fig. 6.6).
The microcalcifications may be associated with sclerosing adenosis, while the adjacent grade 1 in situ AAB may be occult on the mammogram. Therefore, surgical diagnostic biopsy is the procedure of choice for arriving at the correct diagnosis, because larger-bore needle biopsy may miss the cancer.
Histological examination of the 14-g core biopsy specimen in the case shown on ▶Fig. 6.5 showed only hyperplastic breast changes, but the lumpectomy specimen also contained grade 1 cribriform in situ carcinoma, atypical ductal hyperplasia and a 3 × 3 mm well-differentiated invasive carcinoma, none of which were seen on the mammogram.
The ability of breast MRI to distinguish between the hyperplastic breast change (sclerosing adenosis) and grade 1 in situ carcinoma has not yet been established.
6.1.2 Invasive Breast Cancers of Acinar Origin (Invasive AAB)
The unifocal or multifocal stellate/spiculated or circular/oval-shaped invasive cancers have evolved from the in situ cancers of acinar origin. These invasive tumors are conventionally termed well/moderately/poorly differentiated invasive “ductal” carcinoma NOS (not otherwise specified), tubular cancer, mucinous carcinoma, invasive cribriform carcinoma, basal-like breast carcinoma, the solid form of invasive lobular carcinoma, tubulolobular cancer, etc. Use of the term “invasive ductal carcinoma” to describe cancers that originate from the TDLU (lobules), besides being misleading and anatomically incorrect, may also lead to overtreatment of early-stage breast cancer. Our suggested terminology for invasive breast cancers originating from the TDLUs is well/moderately/poorly differentiated invasive AAB. The combination of the imaging biomarkers (mammographic appearance of the tumor: stellate or circular/oval-shaped) and the histopathologic tumor distribution (unifocal, multifocal) of 1- to 14-mm invasive AAB cases has a powerful prognostic value (▶Fig. 6.1).
Examples of Unifocal and Multifocal Invasive Acinar Adenocarcinoma of the Breast
Unifocal Invasive Acinar Adenocarcinoma of the Breast
The disease process is typically confined to a single invasive lesion, which appears on the mammogram as a stellate or circular/oval-shaped tumor mass.
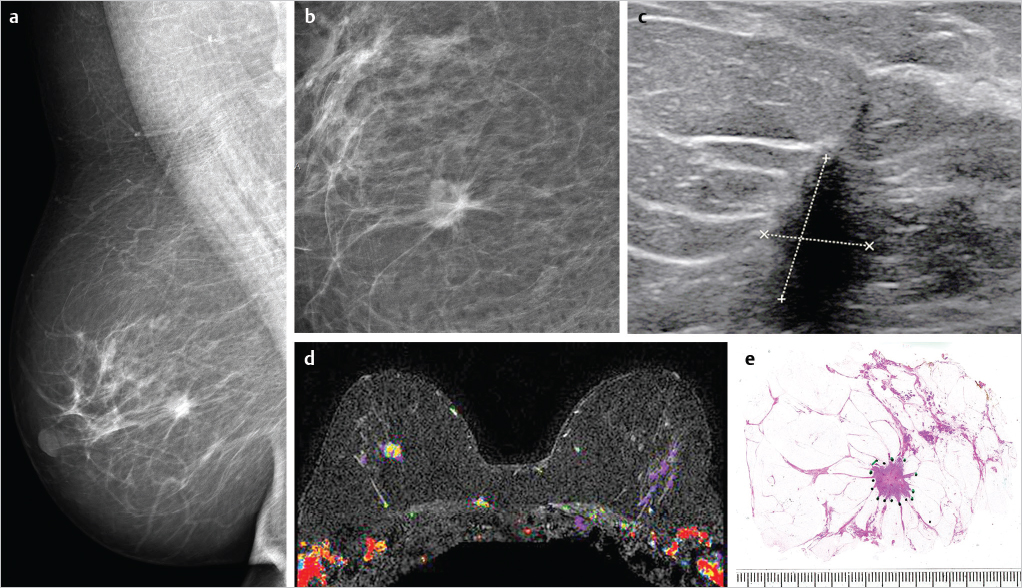
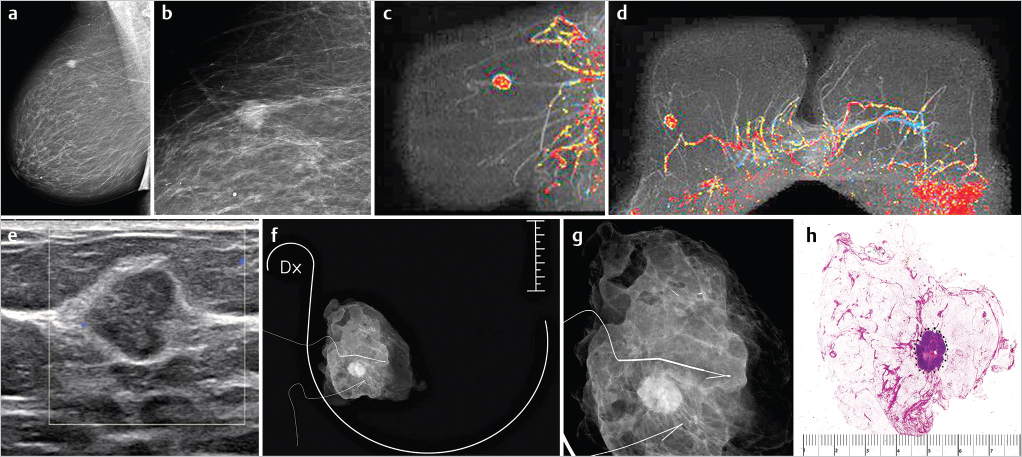
Solitary stellate invasive AAB, multimodality work-up ( ▶Fig. 6.7)
Solitary circular invasive AAB, multimodality work-up ( ▶Fig. 6.8 )
Defining Tumor Size and Multifocality
An unambiguous description of the tumor burden requires the use of two essential terms: the size of the unifocal invasive carcinoma focus and the full extent of the disease within the breast in multifocal (invasive and in situ) cases. Both measurements should be based on integration of imaging and large-format histopathologic findings. The extent of the disease should be defined as the full volume of diseased tissue, including the invasive and in situ tumor foci as well as vessel involvement. The tumor burden will be accurately represented by the size of a single focus in unifocal cancers. Two large studies that examined serial sections found unifocal invasive cancers in 34 and 37% of the cases, respectively. In multifocal carcinomas, the actual tumor burden cannot be adequately described by a single tumor diameter measurement.
Multifocality may consist of the following:
Exclusively AAB in situ carcinoma foci, typically grades 1 and 2. In grade 1 AAB in situ cases, the mammogram shows multiple clusters of powdery calcifications (▶Fig. 6.5); in grade 2 AAB in situ cases, the mammogram shows multiple clusters of crushed stone-like/pleomorphic calcifications (▶Fig. 6.4).
One or more AAB invasive cancer foci associated with AAB in situ carcinoma in a region measuring less than 40 mm. These cases account for approximately 35 to 40% of all breast cancers and most of these will still be suitable for breast-conserving surgery. The mammograms show stellate or circular/oval-shaped tumor masses; the associated AAB in situ carcinoma, when detectable, appears as either powdery or crushed stone-like/pleomorphic calcifications confined to a region less than 40 mm. The in situ carcinoma may be occult at mammography.
Extensive multiple AAB invasive cancer foci (stellate or circular/oval-shaped on the mammogram) with intervening normal tissue.
Extensive multifocal invasive AAB foci (stellate or circular/oval-shaped on the mammogram) with minimal, if any intervening normal breast tissue.
Extensive multiple invasive AAB foci (stellate or circular/oval-shaped on the mammogram) associated with mammographically occult in situ carcinoma.
Comment
Approximately 60% of all breast cancers are either extensively multifocal or diffuse.
Teaching Point
Complete surgical excision requires mapping of disease extent
From the patient’s viewpoint, what matters most is surgically removing the diseased tissue as completely as possible.
Radiotherapy is not a substitute for complete surgical removal of all tumor foci. If radiotherapy were truly able to eliminate all residual tumor tissue, there would be no need for breast surgery.
The use of multimodality imaging techniques, including the mammographic work-up, ultrasound examination, and breast MRI, in combination with percutaneous needle biopsy, can provide a precise preoperative description of the disease extent and localization in most cases. A properly performed and interpreted breast MRI examination is particularly valuable for preoperative tumor mapping.
Finding breast cancers with mammography at an earlier phase allows removal of a small tissue volume only when the tumor is unifocal. Multifocal tumors tend to occupy a larger tissue volume, regardless of how small the individual foci are at the time of detection.
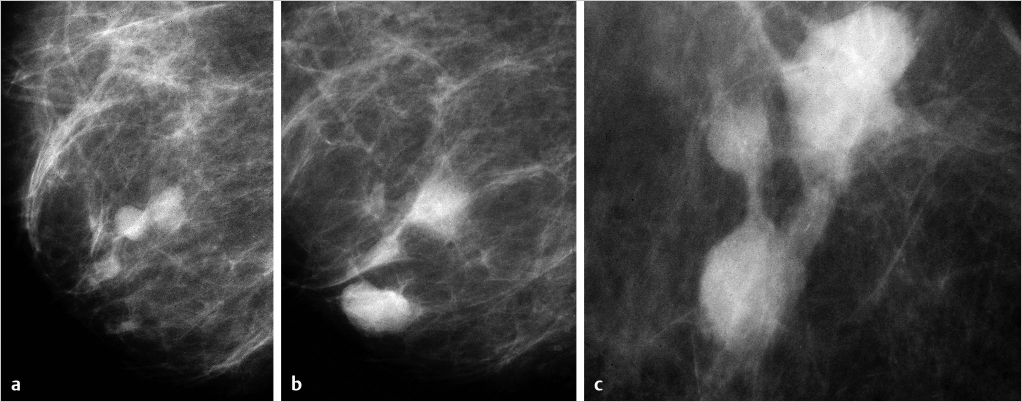
Multifocal Invasive Carcinoma Localized to a Region Measuring Less than 40 mm
The disease process consisting of multiple invasive cancer foci with or without associated in situ carcinoma is confined to a region less than 40 mm. These appear on the mammogram as multiple stellate or circular/oval-shaped tumor masses, with or without associated microcalcifications.
Example
This 82-year-old asymptomatic woman with a vaguely palpable lesion behind the right nipple presented for evaluation (▶Fig. 6.9).
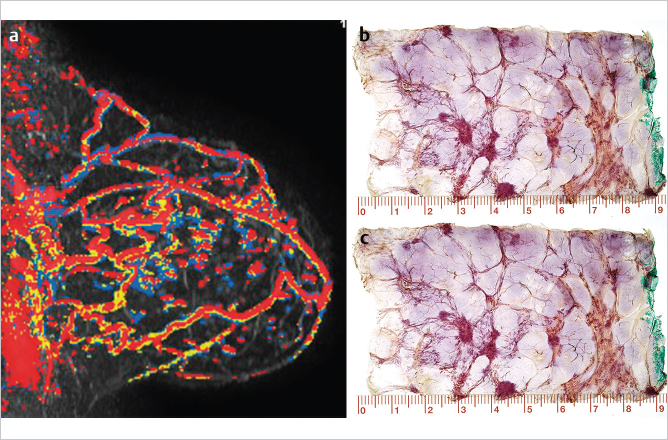
Extensive Multifocal Breast Cancer Consisting of Multiple Invasive AAB Foci with Intervening Normal Tissue
In this subgroup the invasive AAB tumor foci (stellate or circular/oval-shaped) are separated from each other by normal breast parenchyma and spread over a large volume (> 4 cm in diameter).
Example
This 51-year-old woman had been previously treated with mastectomy and chemotherapy for right-sided multifocal metastatic breast cancer. Seventeen months following treatment, she felt a “thickening” and pain in her left breast (▶Fig. 6.10).
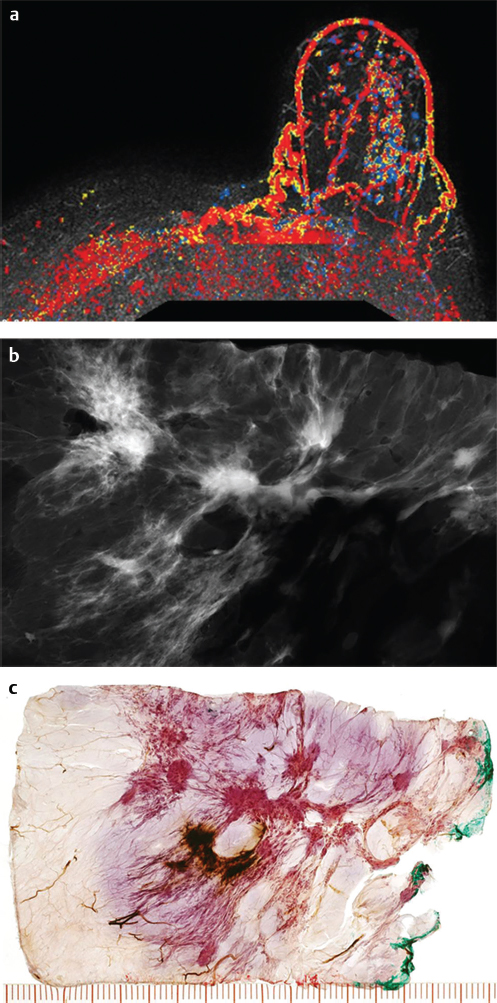
Example: Multifocal Stellate Invasive AAB, Multimodality Work-Up Examples
The suggested new terminology based on the apparent site of origin has direct implications for a better understanding of the value of surgical specimen margin determination. In multifocal in situ (▶Fig. 6.4) and multifocal invasive AAB cases (▶Fig. 6.10, ▶Fig. 6.11) there may be a considerable amount of normal breast tissue separating the individual cancer foci from each other, making the margin determination unreliable when the sampling technique is performed using the small-section histopathology technique. On the other hand, when surgically removing a DAB case with a 1- to 1.5-cm clear circumferential margin, the margin determination is more reliable because of the contiguous growth pattern within the duct and its branches.
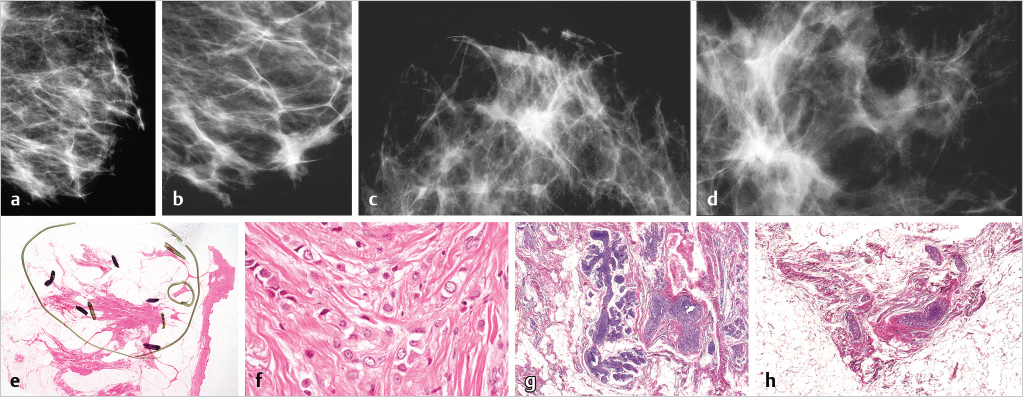
Using the small-section histopathology technique, the resulting incomplete evaluation of the surgical specimen margin has been described as follows: “In a typical 8-cm diameter lumpectomy specimen, assuming four conventional pathology margin sections are removed in a single plane, only 16% of the circumference is examined microscopically.” An example of sampling for margin assessment is illustrated in ▶Fig. 6.12.
Whether the breast malignancy has been completely removed at surgery is a matter of extreme importance to the patient, both physically and psychologically. In addition, subsequent treatment depends on a reliable margin evaluation. This can be accomplished by using the large-format histopathology technique.
The poor sensitivity and poor negative predictive value of margin determination clearly indicate the inadequacies in the current practice in reliably determining the presence or absence of malignancy on the surgical specimen margin. These inadequacies in margin determination, which compromise important decisions on the necessity for further surgical procedures, emphasize the need for improvements to current practice.
In addition to the use of preoperative multimodality imaging, routine implementation of the following procedures could improve the current practice standards for surgical margin determination:
Operative specimen radiography and microfocus magnification radiography of sliced breast specimens.
Large-format histopathology technique as the standard procedure for examining breast cancer specimens.
Radiologic–pathologic correlation of all breast imaging findings.
The current debate on the adequacy of margins appears to be flawed by a nearly complete lack of reliable data on the true status of each patient’s surgical margin in the studies under consideration. Routinely performing an integrated imaging–histopathologic margin evaluation using standardized large-format histopathologic techniques will improve the reliability of margin assessment.
Extensive Multifocal Invasive AAB with Minimal, if Any, Intervening Normal Breast Tissue
When the invasive AAB tumor foci (stellate or circular/oval-shaped) are packed closely together and spread over a large volume (> 4 cm in diameter).
Example
This 65-year-old woman presented with a palpable lump in the upper outer quadrant of her right breast. She died from breast cancer 22 months later (▶Fig. 6.13).
Extensive multiple invasive AAB foci associated with mammographically occult in situ carcinoma
When the invasive AAB tumor foci (stellate or circular/oval-shaped) are spread over a large volume (> 4 cm in diameter) and are associated with mammographically occult extensive in situ AAB.
Example
This 40-year-old woman detected a thickening in her left breast along a surgical scar from her reduction mammoplasty performed 5 years earlier. The mammograms (▶Fig. 6.14) show numerous stellate tumors throughout the lower half of the breast.
Teaching Points
The mammographic tumor appearance/imaging biomarkers of breast cancers arising from the TDLU (AAB) are as follows:
A single cluster of crushed stone-like/pleomorphic calcifications on the mammogram. ▶Fig. 6.3 is an example of a unifocal grade 2 in situ acinar adenocarcinoma of the breast (unifocal grade 2 in situ AAB), indicated by the presence of a single cluster of crushed stone-like calcifications on the mammogram.
Multiple clusters of crushed stone-like/pleomorphic calcifications on the mammogram. ▶Fig. 6.4 is an example a multifocal grade 2 in situ AAB, indicated by the presence of multiple clusters of crushed stone-like calcifications on the mammogram.
Multiple clusters of powdery calcifications on the mammogram. ▶Fig. 6.5a–d is an example of a multifocal grade 1 in situ AAB, indicated by the multiple clusters of powdery calcifications.
Solitary stellate lesion on the mammogram. ▶Fig. 6.7 is an example of solitary stellate invasive acinar adenocarcinoma of the breast (unifocal invasive stellate AAB).
Solitary circular/oval lesion on the mammogram. ▶Fig. 6.8 is an example of a solitary circular invasive acinar adenocarcinoma of the breast (unifocal invasive circular/oval AAB).
Multifocal invasive stellate and/or circular lesions on the mammogram. ▶Fig. 6.9, ▶Fig. 6.10, and ▶Fig. 6.11 are examples of a multifocal invasive AAB.
6.2 Implications of Multifocality
When unifocal and multifocal breast cancers have been compared, investigators have consistently found a higher degree of lymph vessel invasion (LVI) and axillary lymph node metastases in women with multifocal breast cancers. A study by Tot is particularly relevant in that it evaluates the relationship between tumor size and both LVI and macrometastases in the axillary lymph nodes. There appears to be a linear relationship between the lymph node metastases and tumor size, which applies to multifocal and unifocal tumors alike, but the multifocal tumors have approximately twice the risk at each tumor size category. The percentage of cases with LVI was uniformly high for multifocal tumors, irrespective of tumor size category, while the unifocal cancers had a lower frequency that gradually increased with tumor size category (▶Fig. 6.15).
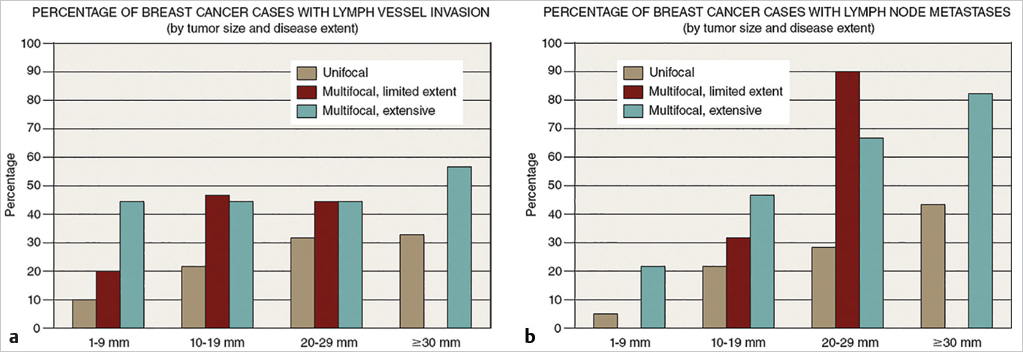
Multifocal and diffuse in situ carcinomas had 13 and 12% recurrence rates, respectively, compared with a 3% local recurrence rate in unifocal carcinomas. These histopathologic tumor features are considered to have important prognostic value and have a considerable impact on treatment planning. Egan’s observation of a much greater annual fatality rate in multifocal versus unifocal breast cancer cases, 15 versus 2.5%, emphasizes the seriousness of multifocality. Thus, the correlation between multifocality and the presence or absence of LVI and lymph node metastases gives added importance to the preoperative detection of multifocality, using all available diagnostic imaging methods, especially breast MRI.
The description of the imaging findings should be correlated with the results of a thorough, large-format histopathologic examination. The combined diagnostic report of each breast cancer case should include the following:
The mammographic appearance (imaging biomarkers) of invasive and in situ components.
Description of the unifocal, multifocal, or diffuse nature of each breast cancer case after a careful imaging work-up.
The histopathologic description of the invasive and/or in situ nature of each finding.
The sizes (mm) of the individual foci and the true extent of the disease process.
The precise location of the diseased tissue within the breast in relation to the skin, the nipple–areola complex and the chest wall.
6.3 Flaws Inherent to the Current TNM Classification of Breast Cancers
The current TNM classification system, which predates the screening era, uses the size of the largest invasive focus as a major descriptive factor and fails to take multifocality into account. In the screening era, when an unprecedented number of in situ and 1- to 14-mm nonpalpable invasive breast cancers are detected, the issues of multifocality and a full preoperative description of the extent of the disease become crucial for adequate surgical removal. There is a pressing need to agree on unambiguous terminology to describe in simple terms the basic tumor characteristics such as tumor size, disease extent (as influenced by multifocality), and tumor location. The current terminology is inadequate, as illustrated by the examples in this chapter.
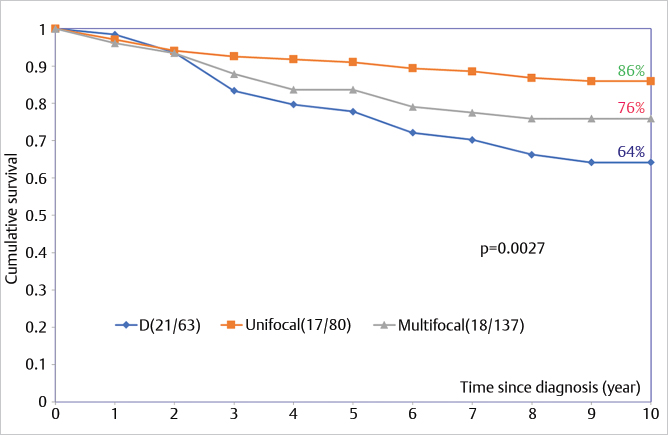
The survival curves in ▶Fig. 6.16 demonstrate that women with multifocal breast cancer have a significantly poorer outcome than women with unifocal breast cancer. Multifocality is a negative prognostic factor, independent of tumor size. The multimodality approach, including breast MRI, offers the best opportunity for establishing the unifocal/multifocal/diffuse nature and describing the true extent of breast cancer.
6.3.1 The Deficiencies Inherent in the TNM Classification System Lead to Several Serious Practical Problems
How do we inform a patient of her tumor size when she has a unifocal “small tumor” (▶Fig. 6.7, ▶Fig. 6.8), or a “small” but multifocal malignancy limited to a small tissue volume (with the largest invasive focus measuring < 10 mm) (▶Fig. 6.9), or when there are many tiny invasive AAB foci, the largest focus measuring less than 10 mm, which spreads over a large volume (▶Fig. 6.10, ▶Fig. 6.11).
How can we explain the significant outcome difference between two cases having an identical tumor size classification but a significantly different tumor burden? One patient with multifocal invasive AAB where the largest tumor focus measured less than 10 mm (demonstrated in ▶Fig. 6.13), died from breast cancer, whereas patients with less than 10 mm unifocal stellate invasive AAB have a 99% 24-year disease-specific survival. Using the TNM classification, all these patients were entered in the Tumor Registry as having a 1-to 9-mm invasive breast cancer.
The tumor burden is certainly greater in the multifocal cancers (▶Fig. 6.9, ▶Fig. 6.10, ▶Fig. 6.11, ▶Fig. 6.13, and ▶Fig. 6.14) than in the unifocal cancers (▶Fig. 6.7, ▶Fig. 6.8). Multifocality is associated with a significantly higher frequency of node and lymph vessel involvement than are unifocal tumors within the same tumor size category (▶Fig. 6.15).
The surgical approach in these examples was determined by the extent of the disease—essential information that is not provided by the TNM classification.
What tumor size should have been reported to the Cancer Registry for each of these patients?
Treatment guidelines are based on data from the Cancer and Death Registries; if multifocal tumors with a poor outcome continue to be placed into the same category as unifocal tumors with an excellent longterm outcome, the therapeutic guidelines will continue to be misleading.
Teaching Points
Imaging Appearance of Diffuse Cancerous Involvement of the Duct System (DAB) and Implications for Management
Detection of the malignant process originating within the preexisting duct system (DAB) is usually delayed because the malignancy does not initially cause mammographically detectable alterations to the structure of the ductal system, even though the entire duct system may have been involved from the beginning. This is in clear contrast to the malignancies developing from the TDLUs (AAB cases), which become detectable already at a much earlier phase of their development.
Strict attention to all the relevant clinical signs (serous or bloody nipple discharge, nipple eczema suggesting Paget’s disease) and the use of multimodal imaging, particularly breast MRI, is needed for the detection and diagnosis of this deceptive and often fatal subgroup of breast cancers.
The high fatality rate (30–40% at 25 years) of the DAB cases appears to be related to their large burden of high-grade tumor cells within the ever-growing newly formed ducts (neoductgenesis). Unfortunately, the current belief is that the extensive disease within the ducts and in the newly formed duct-like structures is a true in situ process termed “DCIS,” but, in reality, this disease often acts as a duct forming invasive carcinoma with a large tumor burden. The term “DCIS” is thus a misnomer, which gives physicians a false sense of security, adversely influencing treatment planning and the prediction of prognosis of patients with DAB.
By the time the DAB cases are detected, they are often associated with one or more AAB tumors of varying sizes. In clinical practice, attention is unfortunately most often given to the small, associated invasive AAB component, which by itself would rarely be fatal.
We emphasize the need for using large-format thin-section and large-format thick-section (3D) histopathology to appreciate the true nature of this subgroup of breast cancers originating from the major ducts.
The six subtypes of DAB differ in their imaging appearance, but have important common characteristic features:
Each subtype occupies a single lobe in one of the two breasts. The entire lobe becomes involved with an ever-increasing number of newly formed ducts.
The main, retroareolar duct can be likened to the trunk of a tree, while the ducts newly formed by neoductgenesis can be likened to an increasing number of branches growing in a disorganized manner.
The outcome of DAB cases is often poor and at odds with the outcome of AAB tumors, given the same TNM status.
The adjuvant treatment regimens introduced during the past few decades have not changed the case fatality rates of breast cancers originating from the major ducts (DAB). The extent of the disease is often underestimated through a failure to appreciate the MRI and mammographic findings. Failure to determine the biomarkers from the DAB itself (the cancer filling the ducts) may cause undertreatment of the DAB.
Combining multimodality imaging with modern, large-format histopathology techniques enables us to classify the breast cancers according to their site of origin. The majority (74%) of breast cancers originate from the TDLUs (AAB) are more easily detected in an early phase and can be more successfully treated, while the DAB subtypes (20%) are diagnostically and therapeutically far more challenging.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree