1 Anatomy, Physiology, and Aesthetics
Summary
The anatomy and physiology of the breast are outlined in this chapter. The anatomy is presented as applied anatomy, with clinical application outlined in detail.
Key Topics
Skin
Parenchyma
Fascia
Cooper’s ligaments
Blood supply
Nerve supply
Lymphatics
Nipple–areola complex
Musculature
Nipple–areolar position
Symmetry
Clinical application of breast aesthetics
1.1 Observations
Competence in surgery, regardless of the discipline, depends on a detailed understanding of anatomy and physiology, an appreciation for aesthetics, and a mastery of surgical skills. While surgeons enjoy the technical aspects of operating, there is little doubt that an intimate knowledge of anatomy is critical to achieving consistent and outstanding results. Fifty years ago, a basic knowledge of classical anatomy stood any surgeon in good stead. Today’s complex breast surgery procedures do not afford that luxury. They stress both form and function and frequently incorporate muscle and musculofascial flaps—often with microvascular transfer. This requires an intimate knowledge of functional anatomy including an awareness of locoregional perforators. The growing emphasis on minimally invasive approaches requires detailed anatomical knowledge, as does the increasing use of multiplanar surgical exposures within the breast and the multiplicity of vascular pedicles used in breast reduction and breast reconstruction surgery. These mandate an intimate understanding of vascular anatomy and gross morphologic anatomy. In addition, the advent of perforator free tissue transfer underscores our need to comprehend vascular anatomy in a far more precise manner than ever before.
Knowledge of anatomy is also essential for patient safety, to preserve function and sensation, and to prevent complications from reoperative breast surgery. Today, the widespread acceptance of breast conservation therapy to treat breast cancer has created a large volume of patients requiring secondary reconstruction for breast deformity following lumpectomy and radiation therapy, or for mastectomy reconstruction after local recurrence with failed breast conservation. These women’s scarred breasts present an anatomical conundrum. The anatomical and vascular changes resulting from previous operations must be carefully considered in any surgical plan. Breast radiation increases the challenge, since tissues are affected at a molecular level, impairing the way tissues move and heal. Furthermore, the expanded use of three-dimensional imaging in the form of computed tomography scans, magnetic resonance imaging scans, and various forms of infrared and laser imaging of flap blood supply have mandated a deeper understanding of flap anatomy.
Aesthetic judgment is also essential to successful breast surgery. In the 21st century, aesthetic goals and patient expectations for cosmetic results have become accepted norms in determining surgical outcomes and in evaluating the quality of results obtained. In the early years of breast reconstruction, attainment of a breast mound was synonymous with a successful outcome. Today that is no longer the case; a surgeon is expected to create a breast that is as close to normal in shape and consistency as possible. These aesthetic norms are even more compelling in the realm of aesthetic surgery, where many patients are seeking perfection. Therefore, a surgeon performing either cosmetic or reconstructive breast surgery needs to develop a keen aesthetic eye to successfully execute present-day procedures and to meet the exacting demands of the future.
1.2 Embryology
Embryologically, the breast develops from the linear ectodermal ridge or mammary ridge, which extends from the axilla to the groin. By the sixth week of fetal development, a so-called milk line appears; this may give rise to varying degrees of breast and nipple development at any point along its length, if the natural involution of the majority of the mammary ridge does not take place.
1.2.1 Embryology
See ▶Fig. 1.1.
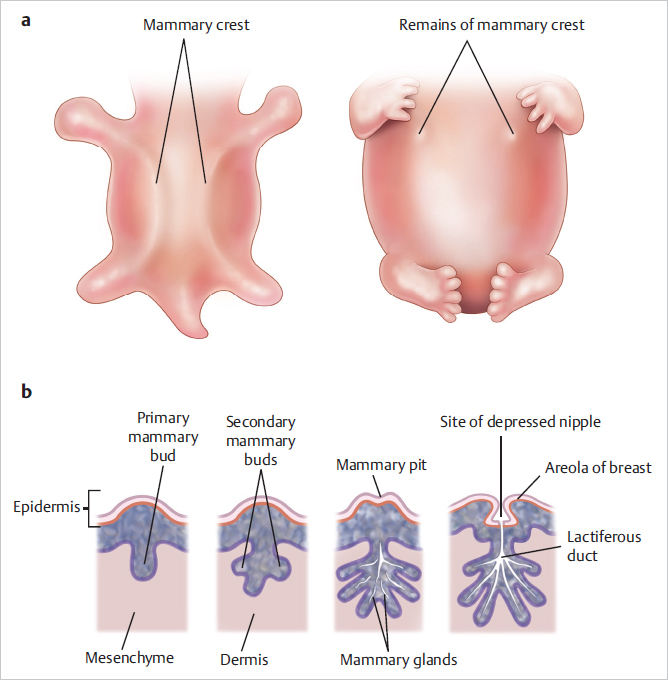
During fourth to sixth week, breast-specific progenitor cells appear.
During fifth to sixth week, epithelial proliferation on thorax to create mammary ridges (“milk line” along which nipple or breast development may occur if long-term regression of the mammary ridge does not take place inferior to the breast itself).
By 16th week in utero, the ectodermal ridge penetrates the underlying mesoderm, sending approximately 20 ingrowths radially into the developing breast. Each ectodermal bud deepens in its growth pattern and ultimately canalizes into the lactiferous ducts of the established breast, triggered by mesenchymal regulatory factors. The lactiferous duct lining is bilaminar. The innermost layer is secretory, while the basal layer differentiates into myoepithelial cells. Lactiferous duct development is probably triggered by placental hormones entering the fetal circulation.
By 20th week, the areola has begun to develop.
By 28th week, the acini develop around the terminal tips of the lactiferous system, while the ducts concentrate superficially into a pit in the skin of the embryonic chest wall. Mesodermal proliferation in the area gives rise to the circular and longitudinal smooth muscle fibers of the nipple–areola complex. The areola is ectodermal in origin and contains apocrine glands, enlarged versions of which become Montgomery’s tubercles. The mature breast contains approximately 20 lobules each with a primary ductal drainage system. Because of overlap between the drainage system and the lobes, these cannot be readily identified clinically as distinct anatomical units. Support for the breast comes from both the skin envelope as well as the fibrous suspensory ligaments of Astley Cooper (Cooper’s ligaments) anchoring the breast to the pectoralis major fascia. These structures are mesenchymal in origin.
Third trimester: The epidermis in the developing nipple dimples to form a mammary pit. Lactiferous ducts converge on this area to form retroareolar ampullae. Smooth muscle fibers developed both longitudinally and in a circular fashion to create nipple projection in the mature breast.
Clinical Pearl
Division of Cooper’s ligaments during mastectomy allows for considerable expansion of the remaining breast skin envelope. This provides an opportunity for a surgeon to enhance reconstructive breast volume beyond that of the patient’s original breast volume at the time of the mastectomy without having to resort to tissue expansion.
Immediately after birth following term gestation, an infant’s breasts may exhibit some activity and can even secrete milk because of the high levels of hormones circulating in the mother. This is not seen in preterm neonates.
A supernumerary nipple is common in the inframammary area and may be confused with a nevus. It can function during pregnancy and lactation and may also be the site of a breast carcinoma (▶Fig. 1.2).
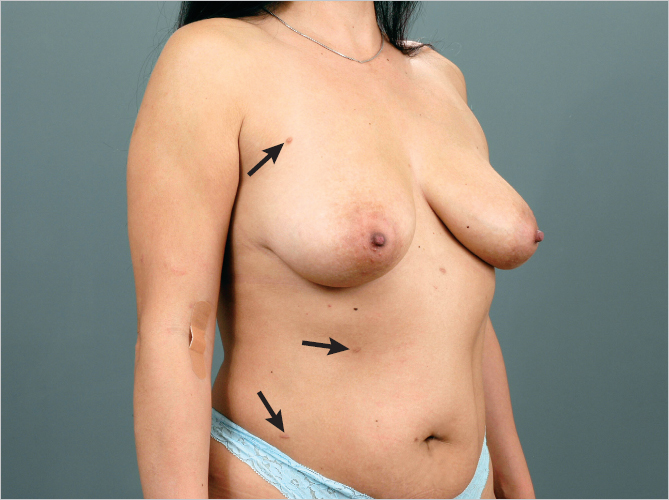
An axillary breast can present as axillary fullness. It too can be the site of breast cancer. Axillary breasts are unsightly and distressing to the patient; supernumerary nipples may also occur in this location (▶Fig. 1.3).
1.3 Infancy to Puberty
During breast-feeding, decreasing levels of maternal estrogen stimulate the neonatal opportunity to produce prolactin which may result in mild breast enlargement and even lactation in the infant.
At 3 to 4 months’ postdelivery, a surge in the infants’ estradiol level may stimulate further breast growth especially in female children.
Between birth and 2 years of age, the breast undergoes morphologic and functional changes. By 2 years of age, the breast is typically quiescent.
After the newborn period, however, breast activity subsides until puberty. Today puberty is occurring earlier in many young women, sometimes as early as 7 to 9 years of age. This early onset can speed the development of breast problems in girls who are emotionally and psychologically unprepared to cope with them or their necessary treatment. These young women and their parents should be advised to delay surgical intervention as long as possible, ideally until growth ceases, so that the patient will be better psychologically prepared, and potentially to avoid the need for additional operations.
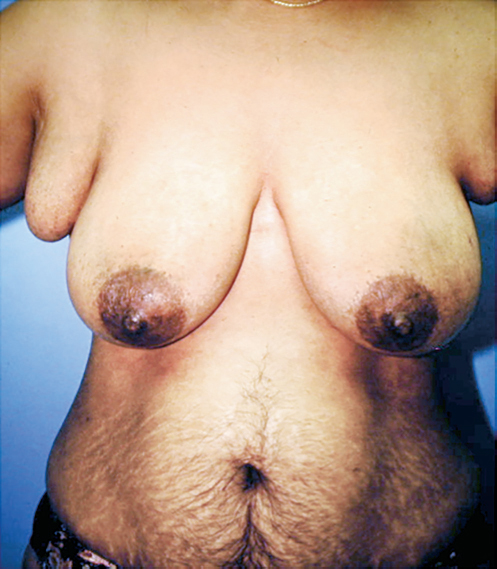
Before the onset of puberty, the nipple is located at the fourth intercostal space in both males and females. When a breast bud first appears, it is palpable just under the nipple–areola as a localized small, firm breast disk. Breast buds can be tender and sensitive, but this is a normal condition and should not be a source of concern. I have seen a number of partial breast deformities and even complete absence of a breast as a result of biopsy or excision of a normal breast bud because the parents and physicians feared that this was a tumor (▶Fig. 1.4).
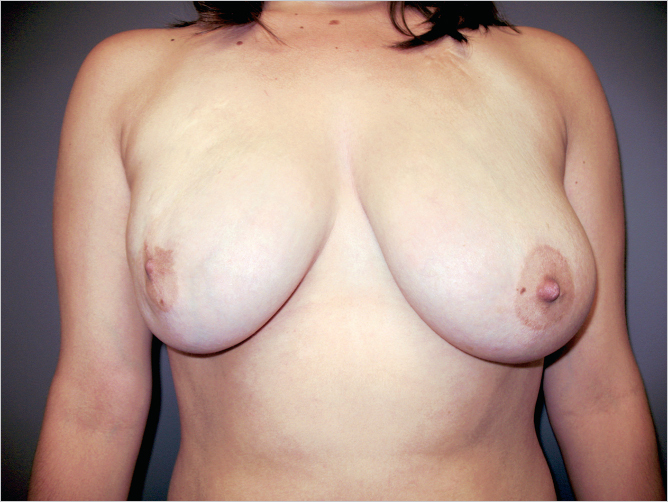
In addition, surgical placement of a chest tube in infancy may create abnormal breast bud development in puberty as illustrated by the patient in whom a right-sided chest tube caused significant breast growth impairment (▶Fig. 1.4).
1.4 Pubertal Breast Development
The pubertal surge of estrogen drives breast development between the ages of 8 and 14.
The response to estrogen is dependent on pituitary growth hormone and its stimulation of insulin-like growth factor-1 (IGF-1).
1.4.1 Tanner Described Six Stages of Female Breast Development
Tanner stage 1: Elevation of the papilla in preadolescence, preceding stromal development and the development of pubic hair by 6 months.
Tanner stages 2 and 3: Early stromal proliferation characterized by the beginnings of an ill-defined breast mound. Asymmetry is common at this time.
Tanner stage 4: Typically seen in the early adolescents, a secondary breast mound begins to differentiate with the development of the true breast shape.
Tanner stage 5: The areola recesses onto the breast mound diffusing around its periphery with lots of separation of contour. The stage is typically complete by age 15.
1.4.2 Cellular Changes within the Pubertal Breast
At puberty, the breast contains pluripotent stem cells and lineage-restricted progenitor cells.
Fatty deposition and stromal proliferation precede a subsequent ductal elongation and dichotomous ductal branching influenced by estrogen.
The bilaminar ductal lining develops, the inner layer being secretory with the outer layer consisting of myoepithelial cells.
Some degree of alveolar proliferation occurs but pregnancy is the main driving force for this process.
1.4.3 Lobular Development
Type 1: Short terminal ducts ending in alveoli containing secretory cells.
Types 2 and 3: Terminal ducts subdividing into several ductules with increasing numbers of alveoli.
Type 4: (Seen only in women who have undergone pregnancy and lactation) consisting of terminal duct branching into ductules were much increased numbers of alveoli.
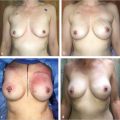
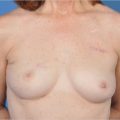
The female breasts begin to develop between the ages of 8.5 and 13.5 years. Failure to develop any breast tissue by age 14 should be investigated. At this time, the breast bud develops into a conical structure with some projection. Although both breasts usually grow simultaneously at puberty, one can develop a few months before the other. Temporary developmental differences or permanent asymmetries may occur, and these are first noticeable shortly after breast development begins. During this phase of breast development, the breast is highly susceptible to changes in body weight. Excessive weight loss as seen in anorexia nervosa can cause shrinkage of the breast with wrinkling of the skin termed “instant senility.” If the breast buds have been incised, such as during a thoracotomy, girls can develop significant deformities during puberty. When one breast begins developing weeks to months before the other, patience is advised until the other breast starts to grow. Adolescents are most concerned and sensitive psychologically about their breast development. Although some mild breast asymmetries are normal and should be expected, when they are marked and the patient and parents become concerned, surgical correction may be requested. Generally, it is best to delay correction of breast asymmetry until development is complete, usually when the girl is 14 to 16 years old, when full vertical height has been attained, and when progressively larger brassiere sizes are no longer needed. When asymmetrical breasts are treated at this early age, the patient and parents must be informed that various factors, such as weight loss or gain, pregnancy and lactation, endogenous or exogenous hormones, and aging may produce additional breast asymmetry over time, and that will require further operations.
Certain types of asymmetry will not improve with time. In cases of unilateral hypoplasia or aplasia and Poland’s syndrome, the asymmetry can actually increase with time, often developing early and requiring operative correction. When the patient has a constricted breast with an inframammary fold that originates more laterally and higher on the chest wall or a tubular protrusion of the central breast and nipple–areola, these are permanent conditions that will not be remedied by time.
Breast hypertrophy can also develop in young women just after the onset of puberty. Basically, two manifestations of hypertrophy concern the teenager and her parents: giant juvenile hypertrophy and, more frequently, teenage breast hypertrophy. In my experience, extensive endocrine work-ups do not reveal abnormalities in these young patients with breast hypertrophy. Hormonal therapy has not proved helpful, and surgical reduction of their large breasts is the only realistic option for these patients. Where possible, I prefer to delay breast reduction until age 18, at which time most young women’s breasts have attained stability. The patients are also more mature by this time and are able to make a more informed decision about the impact of scarring on their breasts.
1.5 Physiology
The breast undergoes three major phases of development: the initial developmental phase, menstrual phase, and menopausal phase.
1.5.1 Initial Developmental Phase
The initial phase occurs between birth and puberty. At this time, the breast stroma and glandular tissue increase gradually, while the breast ducts elongate and canalize in preparation for milk storage later in life. Numerous hormones influence the developing breast; these include estrogen, progesterone, prolactin, insulin, thyroxine, and growth hormone. The pituitary-based release actors, follicle-stimulating hormone and luteinizing hormone, trigger the release of estrogen and progesterone, which stimulate breast development at puberty and during pregnancy. Progesterone strongly affects the terminal glandular component, while prolactin triggers the differentiation of alveolar epithelial cells into milk-producing cells.
1.5.2 Menstrual Phase
During the menstrual cycle, breast size and consistency vary. In the resting phase, the ducts are lined with a bilaminar layer of cuboidal cells, while the terminal ductal alveoli have a single cuboidal cell lining. The major draining ducts are lined with pseudostratified columnar epithelium. Stromal tissue increases in density as the periphery of the breast is approached. A high number of fibroblasts, plasma cells, lymphocytes, mast cells, and histiocytes are present in the unstimulated breast.
Proliferative phase of the menstrual cycle (first week): Estrogen levels rise between days 3 and 7. Intraductal epithelial proliferation occurs producing two to three layers of polygonal B cells that almost obscure the lumen of the ducts. Stromal density increases, as do the numbers of fibroblasts and plasma cells.
Luteal phase (second week): Progesterone release from the stimulated follicles results in further epithelial proliferation, myoepithelial cells in the basal layer become more prominent, and B cells increase in number. Columnar A cells with a high RNA content become visible, while inflammatory cells are less obvious.
Secretory phase (third week): Estrogen, progesterone, and prolactin all influence the breast. Steroid hormones trigger lipid droplet formation in the alveoli and this, combined with water retention, is responsible for the 15 to 30% increase in breast volume seen premenstrually. The increase in water retention within the stroma appears to be related to an estrogen-induced histamine release affecting the stromal microcirculation. Prolactin stimulates an increase in ribosomes and RNA content within luminal epithelial cells, while the stroma becomes less dense with edema and venous congestion. This can become a major source of discomfort.
Withdrawal phase (fourth week): This is associated with estrogen and progesterone decline. Although luminal secretions are transiently retained, the stroma reverts to its more densely compacted nonedematous state.
1.5.3 Menopausal Phase
During menopause, there is a decline in estrogen associated with a rise in progesterone. Glandular involution occurs, with partial regression of glandular epithelium and decreased vascularity and cellularity of the stroma. Postmenopausally, fat infiltration into the breast occurs with increased elasticity and reduced vascularity. Lymphocytic infiltration around the ductal remnants may occur.
Clinical Pearl
Postmenopausal fatty infiltration into the breast may increase breast volume, manifesting as a secondary growth spurt in older women. This may precipitate a request for breast reduction in later life.
Breast enlargement is usually complete by the late teens but may continue into early adulthood. It may be associated with physical abnormalities, including hypertrophy, gigantomastia, asymmetry, and ptosis. Excessive weight gain may contribute to massive breast enlargement. Some women experience breast atrophy after menopause, whereas others develop significant secondary breast enlargement with fatty infiltration of the breast, as mentioned earlier. Excessive breast growth may precipitate functional symptoms causing patient discomfort and embarrassment.
Mammary hypertrophy appears to be related to abnormal end-organ responsiveness to estrogens. Jabs and colleagues demonstrated that women with hypertrophy have a normal number of estrogen receptors and usually have normal estrogen levels suggestive of end-organ hypersensitivity. The breast stroma increases (both fat and fibrous elements), while the glandular component remains fairly low. Eliasen showed that benign ductal hyperplasia is a common component of hypertrophied breasts, but these patients do not seem to have an increased cancer risk unless obesity is associated as a risk factor. Juvenile hypertrophy, first described as early as 1670 by Durston, manifests in late childhood or early adolescence. Gigantomastia, defined as breast size requiring a resection of at least 1,800 g per side, usually begins when the individual is in early adolescence and is independent of body weight.
Clinical Pearl
Patients with gigantomastia are highly symptomatic and require radical surgery as spontaneous involution is a rare occurrence. Ongoing breast development after a reduction procedure is a potential risk that may require further surgery at a later age.
Unilateral enlargement should be carefully evaluated to exclude the presence of a giant fibroadenoma, cystosarcoma phyllodes, juvenile hamartoma, juvenile hypertrophy, hematoma, and of course, the rare but occasional breast cancer in adolescence. Attempts to suppress breast development with hormone manipulation have been ineffective. Baker has shown tamoxifen to positively affect some young girls with juvenile hypertrophy, but the adverse effects of long-term tamoxifen therapy, especially in a young patient, preclude its safe use.
1.6 Anatomical Structures
1.6.1 Skin
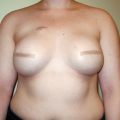
As a skin appendage, the breast is closely related to the skin, and its quality, thickness, and elasticity affect breast appearance. The skin enveloping the breast is subject to hormonal, expansible, weight, gravitational, and aging influences. After breast development, the skin can become stretched, especially in its lower pole and in areas of relative fullness, with thinning skin and loss of elasticity. Striae, actual tears and separations of the thinned dermis with thinning of the epidermis, are often noted in the supra-areolar and periareolar regions in skin that has stretched rapidly or extensively or after weight loss or postpartum involution. The thickness and elasticity of the breast skin are important in obtaining immediate and lasting surgical results. When the skin is thin and inelastic with striae, it will not provide optimal support for the weight of the breast or the addition of an implant over the long term. In these situations, it is likely that normal postoperative stretching after a mastopexy will precipitate the recurrence of ptosis earlier than is usually expected. In such a case, some other method for supporting the breast is needed combined with resection of breast parenchyma in the lower pole to minimize the pull of gravity. The nipple–areolar skin has a thinner dermis than the remainder of the breast skin. Central breast skin thinness and absence of elastic containment for the breast parenchyma are associated with a constricted breast and can lead to the development of a central breast protrusion, the tubular and tuberous breast.
Blood Supply to the Skin
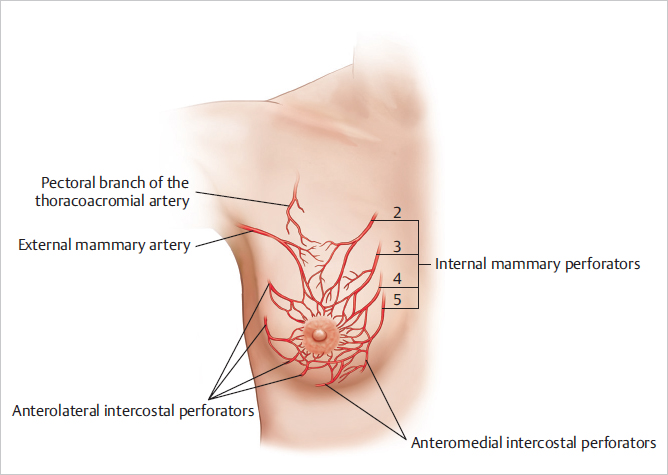
The blood supply to the breast skin originates primarily from the subdermal plexus with communications to the underlying anteromedial intercostal and chest wall breast parenchymal perforators and the subjacent breast parenchyma. Ricbourg identified a cutaneoglandular plexus which connects to the intraglandular anastomotic plexus and retroglandular plexus. When flaps of breast skin are elevated during surgery, the viability of these flaps is determined by their length, the quality and presence of the subdermal plexus, the quality of the microcirculation, and the presence of specific anastomotic inflow vessels such as the medial breast perforators. Skin incisions can interrupt the subdermal blood supply and diminish or interrupt blood flow, leading to flap necrosis. The blood supply to the skin of the central breast area focuses on the nipple–areola, and the blood supply from the subdermal plexus around the nipple–areola is enhanced in this region (▶Fig. 1.5).
Nerve Supply to the Skin
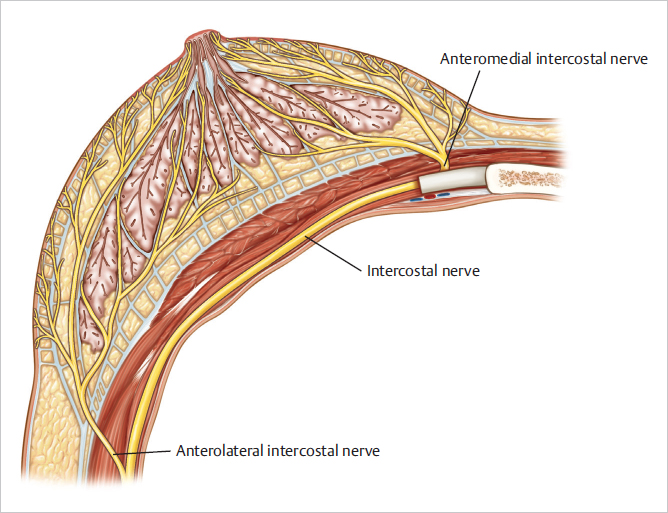
The sensation of the breast skin is segmental and derives from the dermatomes of breast development. The innervation of the central breast and the nipple–areola is primarily from the T3–T5 branches of the anterolateral and anteromedial intercostal nerves. Any breast reduction technique selected should preserve at least one of these branches to ensure that breast sensation is maintained. The lower fibers of the cervical plexus also contribute to the innervation of the important upper breast area. Skin-sparing mastectomy and free nipple-graft reduction techniques generally preserve more sensation. Skin incisions interrupt sensory nerves and thus decrease sensation. Nipple–areolar sensation should be documented before any operation that could potentially diminish it is undertaken.
Injury to the segmental nerves in the anteromedial and anterolateral regions during breast procedures can result in areas of hypesthesia or actual anesthesia. When there is partial injury to the nerves, the reinnervation is accompanied by paresthesias, dysesthesias, and increased sensitivity of the involved areas. I have found that this hypersensitivity is diminished when the patient uses sensory reeducation with frequent massage of the affected areas (▶Fig. 1.6).
1.6.2 Breast Parenchyma
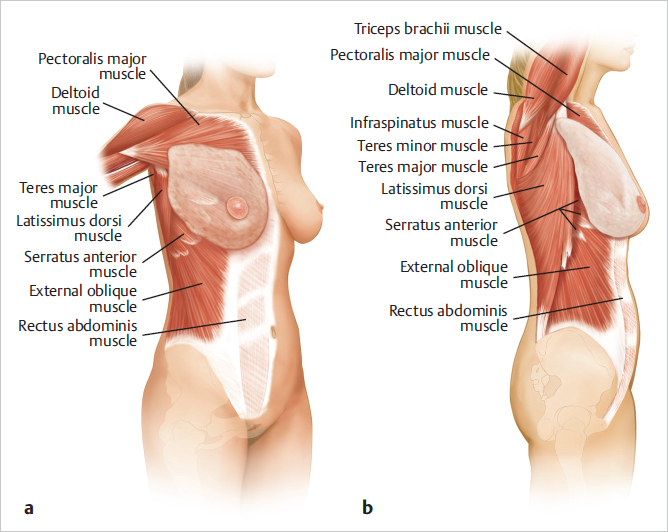
Originating from the ectoderm, the breast is basically a specialized skin gland with breast parenchyma extending up to the subdermal level. Breast parenchyma in the adult covers a large area of the anterior chest wall, extending upward to just below the clavicle and downward to below the usually well-defined inframammary crease. Medially, some breast tissue (the cleavage) reaches to the midline and even over the sternum, and laterally, it extends lateral to the lateral breast fold and to or beyond the edge of the latissimus dorsi muscle. Breast parenchyma also reaches into the axilla; the axillary tail of the breast actually penetrates through axillary pectoral fascial foramina, and breast parenchyma is present in the axillary fat pad (▶Fig. 1.7).
Studies have demonstrated the limitations of surgical procedures designed to completely remove all breast parenchyma. Despite the thoroughness of surgical ablation, it is estimated that 2 to 4% of the total breast mass is left behind after mastectomy. As a consequence, there is always a slight risk of neoplasia arising in these preserved remnants.
Breast shape and contour are influenced by the volume of breast parenchyma, the amount and location of the subcutaneous fat and intraparenchymal fat, the bony contour of the chest wall, its muscular covering and thickness, and the tightness and elastic quality of the skin. The fascial attachments of the breast to the underlying chest wall also influence breast appearance.
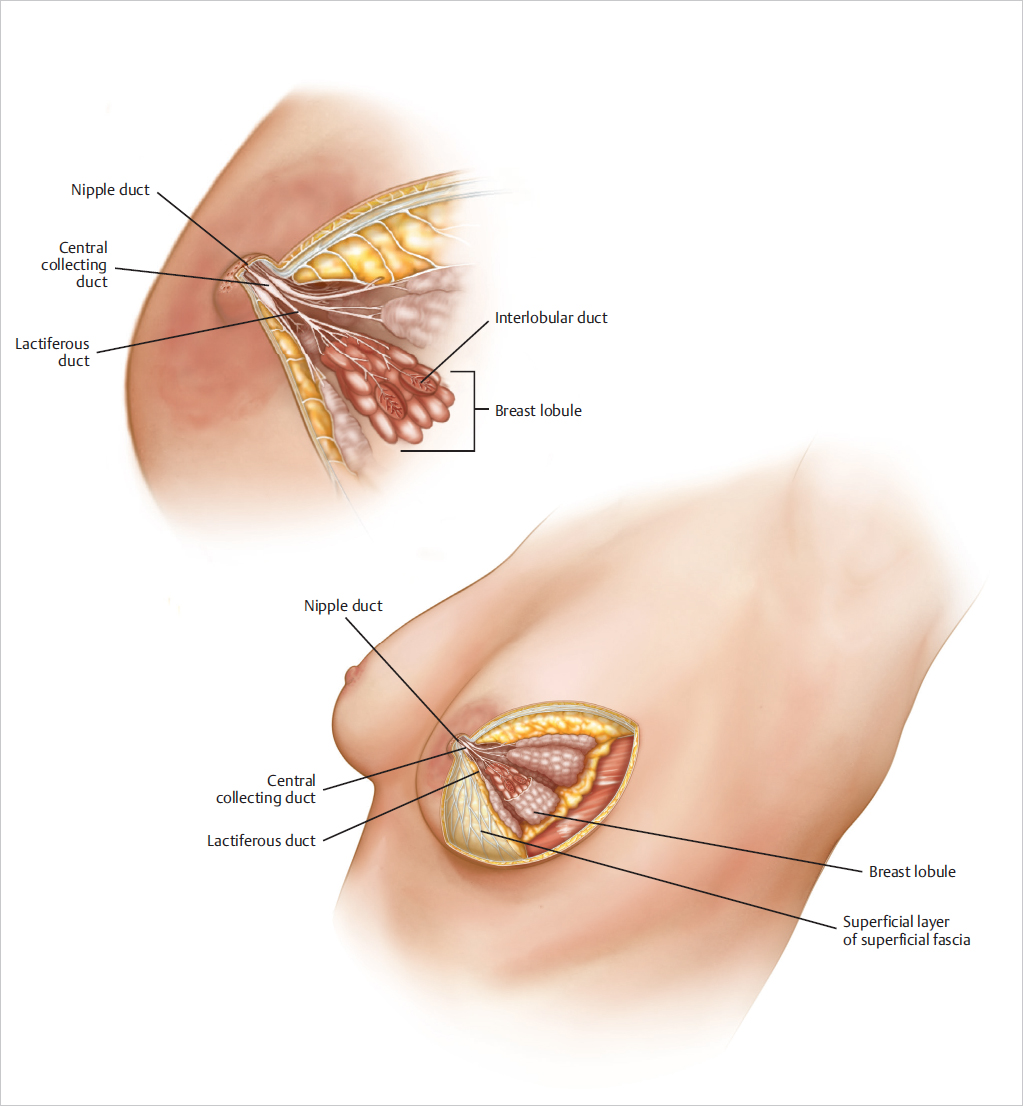
Glandular Architecture
The actual glandular, secretory, milk-producing component of the breast is located within a connective and fatty tissue stroma. These glandular and stromal tissues respond to systemic hormonal and genetic influences. Approximately 20 main lactiferous ducts connect and drain the individual breast lobules to the nipple. The breast lobules are distributed radially about the breast; each of these breast lobules is composed of hundreds of potentially secretory acini, and each has a separate interlobular duct connecting to the lactiferous ducts. The concept of lumpectomy in breast conservation is based on the removal of the affected breast lobule together with a cuff of peripheral normal breast tissue.
The nipple is a focused part of the parenchyma from a functional and sensory perspective. Contained within the nipple are special lactiferous ducts that act as conduits for glandular secretions.
The breast, lactiferous ducts, and breast lobules normally contain bacteria, because the ducts provide a conduit for bacteria to enter the breast through the nipple. Bacteria can usually be cultured from the breast tissue, most commonly Staphylococcus epidermidis. The presence of bacteria helps to explain the incidence of infection after mastectomy and the presence of S. epidermidis in cultures of most breast capsules surrounding breast implants. Several studies concerning the causes of capsular contracture around breast implants implicate infection as a possible causative factor. It is this premise that has led to the widespread acceptance of the use of a triple-antibiotic solution lavage of the breast implant pocket during breast augmentation. Additionally, I recommend systemic antibacterial therapy when breast implants are used or the breast parenchyma is divided. Subpectoral implant positioning probably disturbs fewer ducts than subglandular implant placement does, because it does not separate the parenchyma from the pectoralis major fascia (▶Fig. 1.8).
Fat Content
The breast is a hormone-responsive end organ. The degree of steroid hormone metabolism depends to some extent on the fat content of the breast. The quantity of fat within the breast varies; this fat is responsible for most of the bulk, contour, softness, consistency, and shape of the breast. Fat is selectively deposited within the breast and is influenced by genetic and hormonal factors. The percentage of body fat also influences the amount of breast fat, although some women seem to selectively deposit more fat within their breasts. A breast with considerable fat content feels soft, whereas a breast with a predominance of stromal and glandular components is often firm, irregular, and nodular. Patients who undergo a significant weight loss often notice a change in the consistency of their breasts as they begin to feel the nodularity of the normal breast tissue that previously was undetectable or not palpable within the fatty tissue. This is particularly true of massive weight loss patients. Women who experience cyclic fibrocystic changes notice that this condition diminishes with the onset of menopause, and their breasts often are less lumpy. When estrogen is administered after menopause, the breast parenchyma can proliferate and nodularity can persist. The breast fat component increases as the glandular components subside after lactation or atrophy after menopause.
Breasts of postmenopausal women have more fat and are easier to evaluate on mammography. Liposuction of the breasts in these women lowers the fat content of the parenchyma, and their breasts appear more dense on mammography. Preoperatively, it is difficult to predict the amount of fat that can be removed. Before 40 years of age, women’s breasts are more glandular and dense and contain less fat. It is generally harder to detect the early signs of breast cancer in these women. Breast liposuction can reduce the fat content of the parenchyma.
Recently there has been increased interest in the use of fat injection for breast augmentation of the normal but hypoplastic female breast. Although emerging data are increasingly suggesting that the procedure is safe, there are concerns in some quarters that changing the fat content of a young, hormonally active breast may disturb the end-organ responsiveness to circulating steroid hormones. This raises the question of whether such procedures could possibly contribute to the development of breast cancer in later life. Furthermore, fat injection in sizable volumes has been demonstrated to cause the formation of oil cysts and calcifications, including rim calcification around the oil cysts that may result in unnecessary breast biopsies being performed to exclude the possibility of neoplasia. The medicolegal implications of these findings warrant caution at this time.
1.6.3 Fascia
The breast develops and is contained within supporting layers of superficial fascia. Specifically, the superficial layer of this superficial fascia is located near the dermis and is not always distinct from it. The superficial layer of the superficial fascia is the outer layer covering the breast parenchyma and is most clearly defined in thin patients whose skin is mobile over a more fibrous parenchyma. A variable amount of subcutaneous fat is present between the dermis and the actual breast parenchyma. In some thin individuals the breast parenchyma is not separated from the subdermis by a fat layer, whereas in others the subcutaneous layer is well established.
Some identifiable breast tissue is located in the subcutaneous tissue and can be identified microscopically after a mastectomy, even with thin skin flaps remaining. Therefore, at this level, sharp dissection is necessary during mastectomy (▶Fig. 1.9).
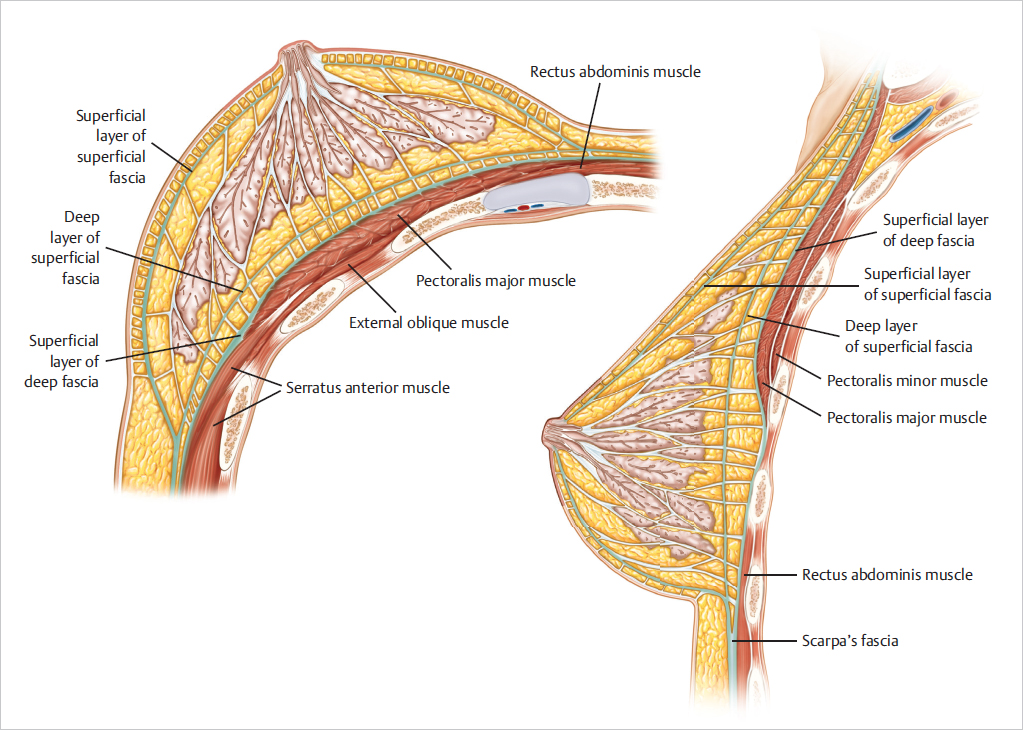
The subdermal plexus of blood vessels that supplies the breast skin is situated on the deep surface of the dermis in close approximation to the subcutaneous fat and the outer layer of the superficial fascia. This layer of subdermal blood vessels must be left intact to maintain breast flap viability. Incisions into the dermis undersurface can devascularize the breast flaps. This is even more important with the skin-sparing mastectomy technique, which requires longer flaps and has an increased incidence of skin flap necrosis. When striae are present, the dermis is thinned, and the subdermal plexus lies under the thinned dermis and the epidermis. These layers and anatomical components of the superficial breast area have distinct implications for breast flap survival, the completeness of mastectomies, and the techniques used for breast reconstruction, especially for immediate breast implant coverage.
More distinct is the superficial fascia’s deep layer on the deep posterior surface of the breast; it is identifiable on the deep surface of the breast when the breast is elevated during a subfascial augmentation mammaplasty. A loose areolar area is interposed between the deep layer of the superficial fascia and the deep fascial layer that covers the pectoralis major, the adjacent rectus abdominis, the serratus anterior, and the external oblique muscles.
The superficial layer of the deep muscle fascia covers the outer layer of musculature of the chest wall and overlays the outer surface of the pectoralis major, the medial serratus anterior, the upper portions of the rectus abdominis, and the external oblique muscle in the lower central breast. This fascia is better defined, more substantial, firmer, and less elastic over the lower breast area than in the upper breast area over the pectoralis major muscle. It is thinner over the muscular portions of the pectoralis major and serratus anterior. The lower fascia is inelastic; if not released properly during subpectoral dissections for submuscular implants, it can constrict an underlying implant or resist expansion and descent of an implant, contributing to an unacceptable result (▶Fig. 1.10).
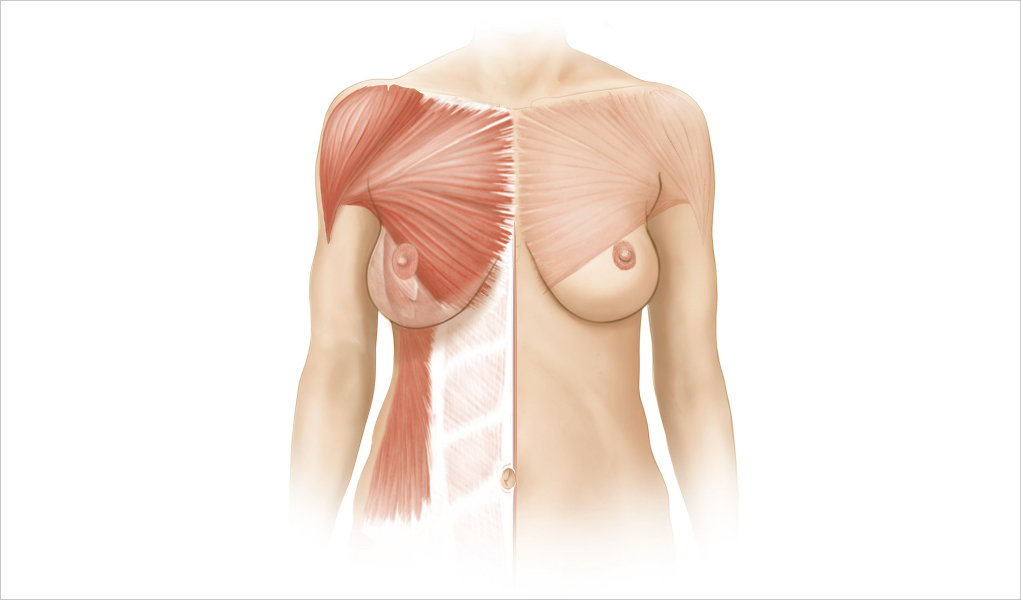
The posterior surface of the breast parenchyma is covered by the deep layer of the superficial fascia. This layer is separate and distinct from the superficial layer of the deep fascia that covers the pectoralis major muscle and the serratus anterior muscle. This deep layer of the superficial fascia is penetrated by fibrous attachments and support (Cooper’s ligaments) as well as vessels, nerves, and lymphatics extending from the deeper structures to the breast parenchyma. Some microscopic portions of breast parenchyma, including microscopic breast ducts, can also be identified extending through the outer layer of the deep fascia and within the subjacent muscles. The deep layer of the superficial fascia is better defined when the breast parenchyma is mobile over the superficial layer of the deep fascia. This is most often seen in the ptotic or hypertrophic breast. When the breast is excessively mobile, the connecting fascial attachments between these two layers become attenuated and appear as areolar tissue. It is this plane that is separated, and the breast parenchyma elevated for subglandular breast augmentation. When there is loss of support and elasticity of the breast attachments to the deep fascia, methods used to increase support and maintain a corrected ptotic breast by suturing the breast parenchyma to the fascia at a higher level have not been documented to be successful over time. When there is poor skin elasticity, poor deep support, and breast ptosis, one strategy to improve this condition is to transfer the glandular tissue by rotating the lower breast parenchyma to the upper breast region and suturing it to minimize the upper breast flatness.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree