4 Deep Inferior Epigastric Perforator Flap for Breast Reconstruction
Robert J. Allen, Sr., Constance M. Chen, and Maria M. LoTempio
Carl Hartrampf’s contribution of the transverse rectus abdominis musculocutaneous (TRAM) flap in the early 1980s was a great advance in autogenous breast reconstruction. He popularized a technique that could be performed by all plastic surgeons to reconstruct a breast without a prosthesis. However, as the procedure became more popular, the donor-site morbidity became more obvious. Agreeing that the ideal donor tissue in the parous female lies transversely across the lower abdomen, investigations began in the late 1980s to utilize this coveted donor area without sacrifice of the rectus abdominis muscles. An early free flap from the 1970s was the superficial inferior epigastric artery (SIEA) flap, usually oriented vertically. By injecting fresh abdominoplastic specimens with radiopaque dye and methylene blue, it was determined that a transverse abdominoplasty flap could be transferred. Seven breast reconstructions with the SIEA flap were performed at Louisiana State University Medical Center by the senior author in 1989 and 1990.1 The chief problem with the SIEA flap was the variable vascular anatomy, with the artery being either absent or very small in a significant percentage of patients.
Given its limitations, the SIEA flap for breast reconstruction was temporarily abandoned, and investigations of the perforators of the deep inferior epigastric vessels began. Following vascular injection studies, the first deep inferior epigastric perforator (DIEP) flap for breast reconstruction was performed in 1992.2 Plastic surgeons now had a technique that could be done in any patient who was a candidate for a TRAM flap but without muscle sacrifice.3
The DIEP flap steadily gained acceptance worldwide until it became the gold standard for autogenous breast reconstruction. The main drawbacks with the DIEP technique is the requirement of microsurgical expertise and the extended duration of the procedure. The success rate with free tissue transfer is higher than that with any other technique. In over 2,000 DIEP flap breast reconstructions, our success rate has been over 99%. The advent of preoperative imaging with computed tomography angiography (CTA) or magnetic resonance angiography (MRA) in the past decade has contributed to reduced operating room (OR) times. Unilateral DIEP flap breast reconstructions are now routinely completed in under 4 hours, and bilateral DIEP flap breast reconstructions are completed in about 4 hours.
In 2013, a total of 232,340 women in the United States received a breast cancer diagnosis, and an additional 64,640 women received a diagnosis of in-situ breast cancer. The overwhelming number of women who receive a breast cancer diagnosis will go on to live a long and cancer-free life. Of women who undergo surgical treatment, two thirds elect to undergo breastconserving therapy (BCT) and one-third elect to undergo mastectomy. For most women, the crucial element that drives their choice is trying to balance their concern about cosmetic result with their fear of recurrence. Thus, the ideal solution for most women faced with a breast cancer diagnosis would be to reduce the risk of breast cancer while preserving breast appearance.
In 2013, a total of 95,589 women in the United States underwent breast reconstruction, a number that is almost quadruple that of 15 years earlier. Eighty percent of these breast reconstructions were done with tissue expanders and implants, and 20% were done with autologous tissue. Interestingly, a 2009 University of Michigan study demonstrated that although there is no difference in short-term (<5 years) patient satisfaction after implant-based or autologous tissue breast reconstruction, over the long term (>9 years) there is significantly less patient satisfaction after implant-based breast reconstruction, whereas patient satisfaction after autologous tissue breast reconstruction remains consistently high.
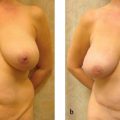
Of all the types of autologous tissue breast reconstruction, the current gold standard is perforator flap breast reconstruction. In particular, the DIEP flap breast reconstruction has achieved popularity as a consistent and reliablemethodof creating along-lastingandnatural-appearing breast with minimal patient morbidity.4,5 By using the skin and fat of the abdominal tissue to re-create the breast mound, the DIEP flap achieves the plastic surgery principle of “replacing like with like.”
Indications
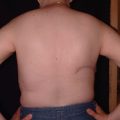
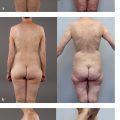
The best candidates for DIEP flap breast reconstruction are women with adequate abdominal fat and no major medical comorbidities who have not previously undergone autologous tissue breast reconstruction. In particular, women who have undergone multiple pregnancies often have experienced a “tissue expander” effect on their abdominal skin and fat that renders their abdomen an ideal donor site for DIEP flap breast reconstruction. The abdominal fat must incorporate an adequate blood supply from the deep inferior epigastric vessels, however. Thus, women who have undergone abdominoplasty are not candidates for DIEP flap breast reconstruction, as the perforators from the deep inferior epigastric vessels will have been taken. Likewise, women who have undergone gynecologic exenterations or extensive intra-abdominal procedures should also obtain preoperative imaging prior to a DIEP flap breast reconstruction to ensure that the perforating vessels from the deep inferior epigastric vessel are still intact. Liposuction is not an absolute contraindication to DIEP flap breast reconstruction, but very thin women with no abdominal fat may not have enough subcutaneous tissue to enable the surgeon to re-create a satisfactory breast mound. If a woman does not have adequate abdominal fat for a DIEP flap breast reconstruction, she may still be able to undergo autologous tissue breast reconstruction using the thigh, back, or buttock as donor sites.
Another common indication for DIEP flap breast reconstruction is failed implant-based breast reconstruction.6 About 20% of our breast reconstruction patients are women who have experienced problems such as infection, capsular contracture, implant displacement, or other such problems after implant-based breastreconstruction. Particularly in women who have had to undergo radiation therapy as an adjunct to their breast cancer treatment, autologous tissue breast reconstruction can create a new breast mound that looks and feels natural while using the woman’s own tissues.
Besides abdominoplasty, the primary contraindications to DIEP flap breast reconstruction are severe medical comorbidities. For example, significant heart disease, pulmonary disease, or other systemic disease may preclude any reconstructive effort. Breast reconstruction is an elective operation and, despite its significant psychological benefits, it should not endanger the welfare of the patient. Other risk factors in the medical history that should influence decision making include coagulopathies, autoimmune disease, obesity, previous history of abdominal surgery, and prior radiation therapy. Additionally, smoking has been found to be significantly associated with overall complications. Active smokers should be counseled to abstain from tobacco and nicotine use for a minimum of 4 weeks prior to any reconstructive procedure. For potential postmastectomy radiotherapy patients, it may be prudent to place a tissue expander as a skin spacer or to delay reconstruction to avoid the complications that can result from irradiating a free flap.
Preoperative Planning
All patients require an extended preoperative consultation to delineate goals, explain alternatives, discuss expectations, and review potential complications. In doing so, the patient becomes actively involved in the decision-making process and participates in the selection of a reconstructive technique.
During the initial consultation, potential patients are informed that DIEP flap breast reconstruction entails three stages. The first stage involves the actual microsurgical breast reconstruction with the DIEP flap. (In a unilateral breast reconstruction, a symmetrizing procedure on the contralateral breast such as mastopexy, augmentation, or reduction may be performed either at the first or second stage.) The second stage involves nipple reconstruction, and if nipple-sparing mastectomy has been done, any revision to the donor site, breast flap, or the contralateral breast in a unilateral reconstruction. For example, any excess tissue at the donor site or fat necrosis in the breast flap may be excised in this stage. The third stage involves tattooing of the nipple-areolar complex if nipple reconstruction is needed. The first stage is an inpatient procedure that requires an average hospital stay of 3 to 4 days. The second stage is performed as an outpatient procedure in the hospital or in the office. The third stage is always performed in the office. Increasingly, most patients are candidates for nipple-sparing mastectomy and immediate DIEP flap breast reconstruction. In these patients, one stage is possible, but a second stage is often done to achieve the optimal results.
Suitable patients who elect DIEP flap breast reconstruction undergo routine presurgical testing, including appropriate laboratory and diagnostic studies and anesthesia consultation. Blood typing and screening is not necessary. Preoperative imaging, either CTA or MRA, is obtained to delineate the topography of the deep inferior epigastric perforators.7 Preoperative imaging reduces our operative time significantly, and it also contributes greatly to patient safety the surgeon can be confident about exactly which perforating vessels will be the largest and most robust to use for the DIEP flap. The dissection can be planned preoperatively, and no time is wasted guessing which perforating vessels to use while the patient is under general anesthesia. In addition, imaging facilitates preoperative mapping of the recipient vessels and helps provide an estimate of the flap volume from the DIEP flap.
Anatomy
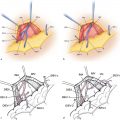
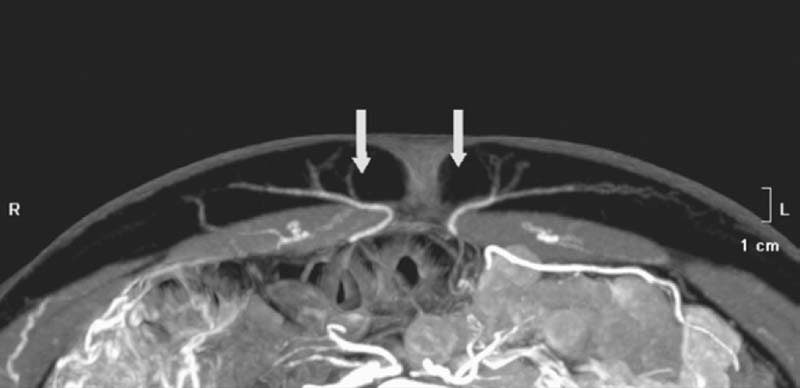
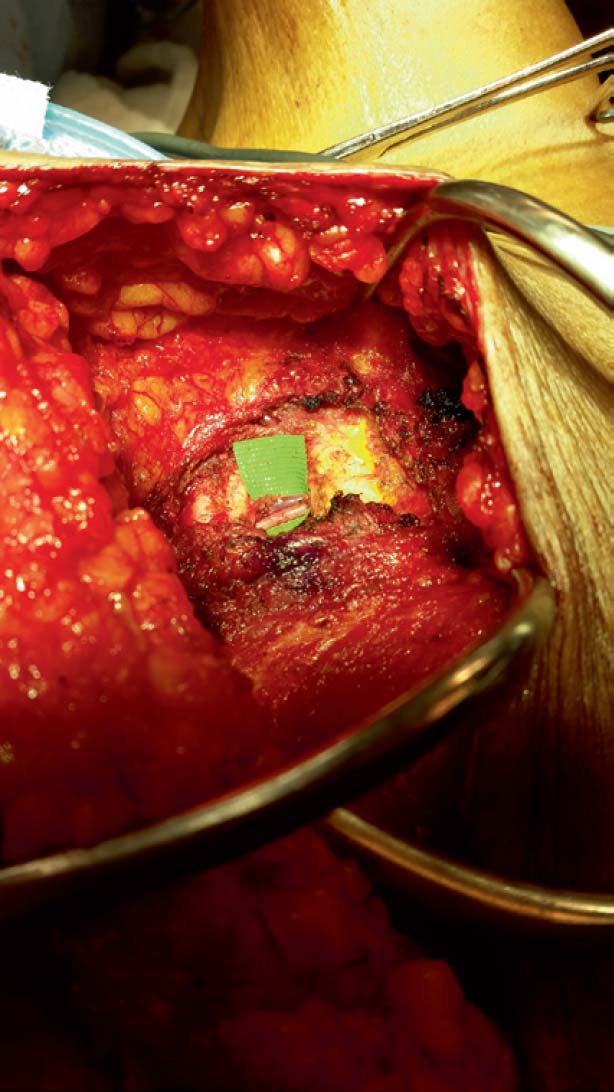
Detailed knowledge of the deep inferior epigastric vasculature and its perforator topography is required for successful execution of the DIEP flap. Accurate knowledge of the perforator topography may be obtained via preoperative imaging—either CTA or MRA. The largest perforator vessels may be chosen preoperatively and then mapped on an X-Y axis. A large perforator trumps the location. Prior to the era of preoperative imaging, the flap was based on one perforator 20% of the time and usually used the lateral row.8 With preoperative imaging, one perforator flap is used 70% of the time, and the medial row is used in 75% of cases. In 8% of patients, a septocutaneous or paramuscular perforator may be found in which the dominant perforator vessel travels around the medial edge of the rectus abdominis muscle with no intramuscular course at all ( Fig. 4.1 ). This completely eliminates any intramuscular dissection and damage to the musculature, and the best way to identify a septocutaneous perforator is via preoperative imaging ( Fig. 4.2 ). Often, the best perforator vessels are around the umbilicus or slightly above the umbilicus. In these situations, The DIEP flap is designed to incorporate the largest and most robust perforator vessels as seen on preoperative imaging.
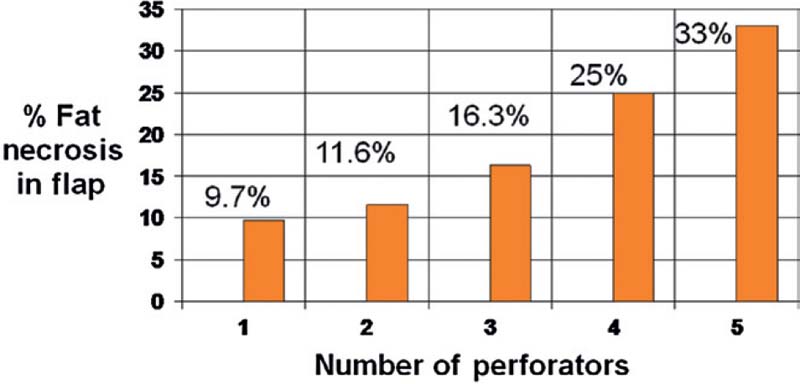
Interestingly, our 10-year review of DIEP flaps prior to the era of preoperative imaging has demonstrated that the incidence of any complication increases with the number of perforator vessels used ( Fig. 4.3 ).8 For example, univariate analysis of partial flap loss indicated significance of any complication with the use of five perforators (p < 0.05). Both univariate and multivariate analysis demonstrated significantly fewer complications with the use of one perforator (p < 0.05). For fat necrosis, there was a steady trend of increased incidence of fat necrosis with increasing number of perforators (p < 0.05). Thus, it is generally best to harvest one perforator for a DIEP flap, as long as the single perforator chosen is large and the best perforator. Ideally, this is delineated on preoperative imaging and confirmed intraoperatively.
Surgical Technique
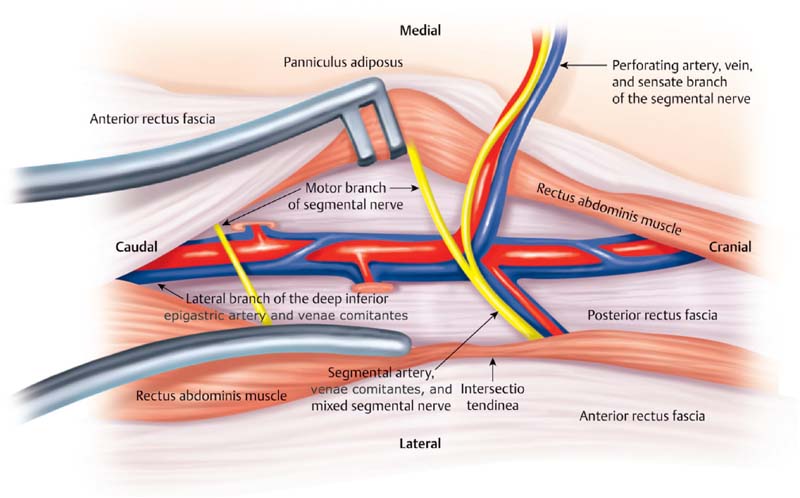
The DIEP flap is based on the perforating vessels that originate from the deep inferior epigastric system. These perforators travel through or around the rectus abdominis muscle, pierce the anterior rectus sheath, and supply the overlying skin and fat ( Fig. 4.4 ). During DIEP flap surgery, the lower abdominal soft tissue is mobilized on one or more perforators in preparation for free tissue transfer. Because the rectus abdominis muscle is not harvested, the pedicle length is substantially longer than in a TRAM flap. The longer pedicle length makes the DIEP flap easier to position in the chest wall and also simplifies the microsurgical anastomosis. In addition, less dissection is required inferiorly toward the external iliac vessels.
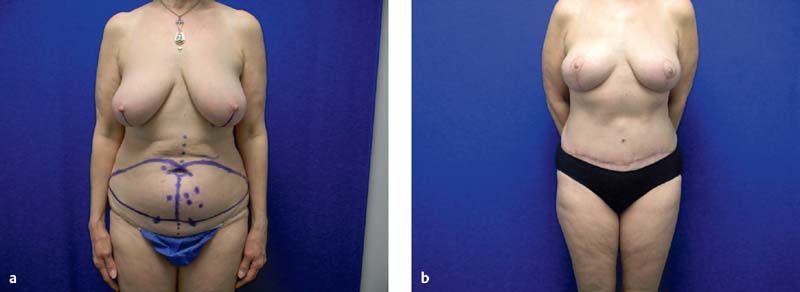
Patients are marked preoperatively in the standing position for anatomic landmarks on the breast and abdomen ( Figs. 4.5 and 4.6 ). On the anterior chest wall, this includes the boundaries of the existing breast mound. On the lower abdomen, marks are placed at the midline, at the perimeter of the umbilicus, and on the proposed outline of an elliptical flap in the hypogastrium. With the patient in the supine position, the key perforators are then marked according to the findings of the CTA or MRA. A handheld Doppler can also be used to confirm the position of key vessels. The upper mark of this proposed flap is placed above the umbilicus if needed to incorporate paraumbilical perforators. The lower mark is placed in a suprapubic crease, and may be shifted superiorly during surgery to ensure less tension in the abdominal closure. The abdominal scar can be lowered 7 cm at the second stage.
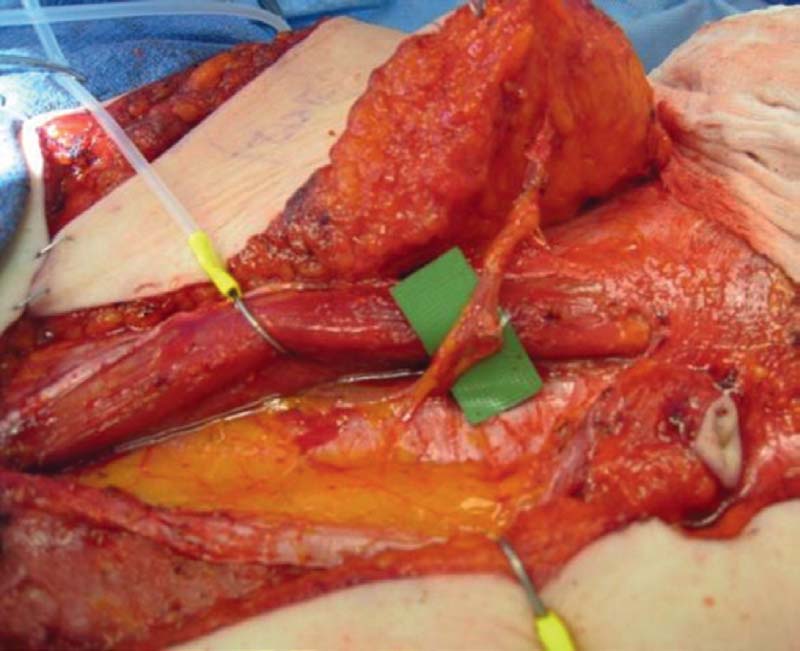
The patient is positioned supine on the operating table with the arms at 60 degrees or tucked by her side, depending on surgeon preference. Tuck the arms unless the breast surgeon needs to have the arms extended for a sentinel lymph node biopsy and possible axillary dissection. Simultaneous abdominal flap harvest and mastectomy, followed by preparation of the chest wall vasculature is performed via a two-team approach. The internal mammary system is used as a recipient site. The vessels are usually approached at the second interspace between the second and third costal cartilages. Some or all of the third costal cartilage can be removed if needed, but in patients with appropriate anatomy the anastomosis can be achieved without removing any rib at all ( Fig. 4.7 ). The diameter of the internal mammary vessels is largest in the second intercostal space. When technically feasible, a rib-sparing technique is used to preserve the pectoralis muscle and prevent a chest wall deformity. Occasionally, the internal mammary system may have adequately sized perforators that can be used for microanastomosis, thus precluding the need to dissect through the rib space to the deeper internal mammary vessels. However, in nipple-sparing mastectomies these perforators should be preserved to provide needed blood supply and sensation to the nipple-areola complex.
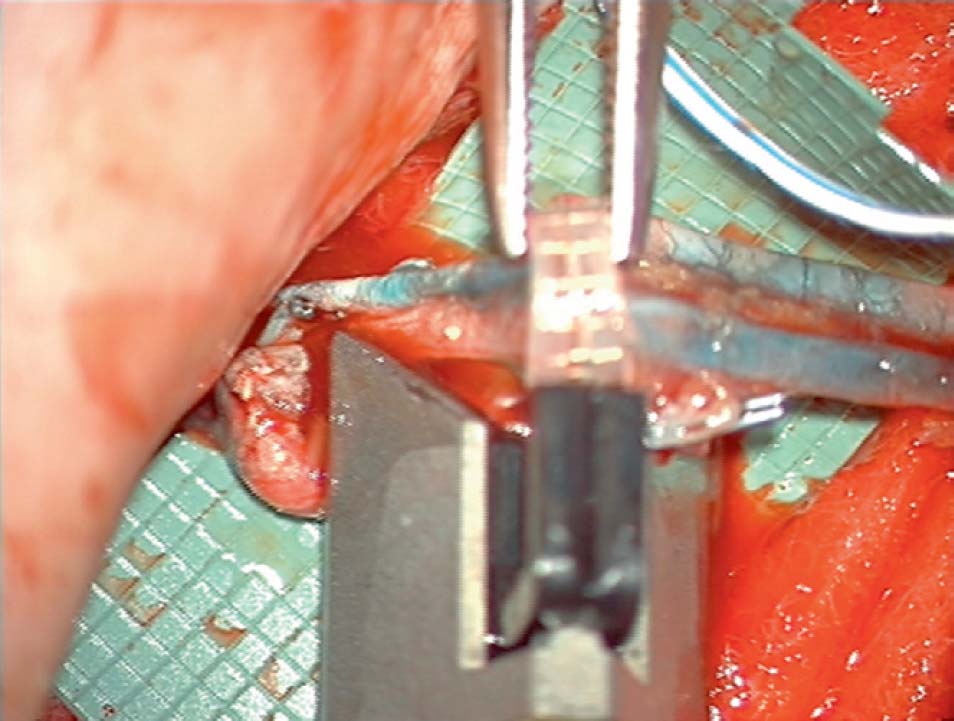
Increasing numbers of microsurgeons who routinely perform perforator flap breast reconstruction identify the main perforators of the deep inferior epigastric vasculature preoperatively via CTA or MRA. This enables them to obtain precise information on perforator diameter and three-dimensional course. A handheld Doppler probe is used to confirm the vessels that they choose preoperatively, and then identify them with direct visualization during flap harvest and perforator isolation.
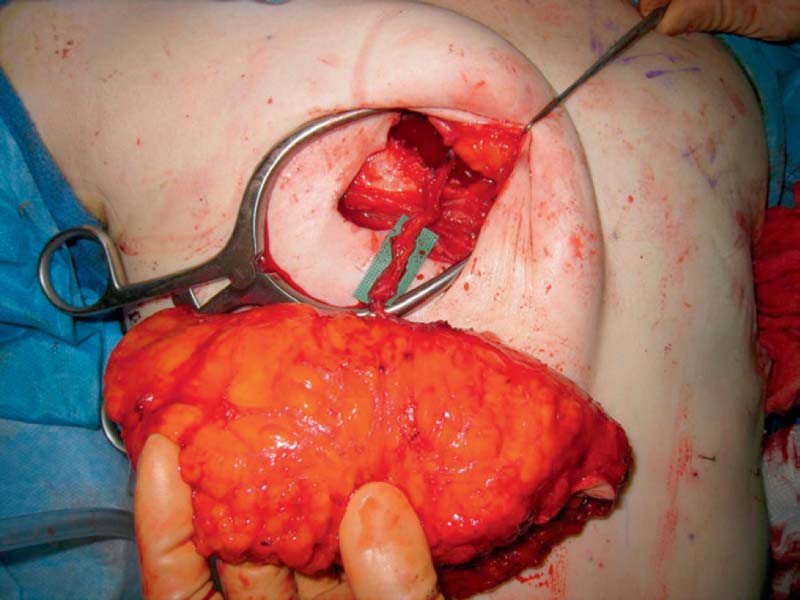
Harvesting of the DIEP flap begins with an incision at the upper marked line, dissection through the full thickness of the abdominal wall to the anterior fascia, and elevation of a superior abdominal wall flap. If the perforator has already been identified via preoperative imaging, then the dissection can continue directly to the chosen perforator. In a bilateral DIEP flap, the midline incision is made first. If the perforator is a medial row or a septocutaneous paramuscular perforator, then the chosen perforator is isolated prior to elevating the lateral edges of the abdominal flap. Once the largest perforator has been identified, the dissection proceeds efficiently with elevation of the skin-fat flap from the underlying rectus fascia. The remainder of the perforators are divided. Care is taken when making the inferior incision to look for superficial inferior epigastric vessels. In particular, it is helpful to preserve the superficial inferior epigastric vein in case the flap has a superficial-dominant drainage system.
Isolation of the DIEP flap proceeds under loupe magnification, and begins by opening the anterior rectus sheath around the selected perforator. Some surgeons incorporate a 1- to 2-mm cuff of the sheath around each perforator, but no fascia is needed. Using a Westcott microsurgical scissors or bipolar electrocautery, the perforator is followed through the rectus muscle, carefully clipping or coagulating all small side branches. If a second or third perforator has been selected, particular attention must be paid to maintaining its continuity in line with the first. Preoperative imaging is very helpful in determining the vascular course.
As dissection proceeds, the muscle is split along the direction of its fibers, preserving all intercostal nerves that do not lie between two perforators. A divided nerve between two perforators should be repaired. The dissection is continued inferiorly until the main trunk of the deep inferior epigastric vessels are reached. An optimal pedicle length is 8 to 10 cm.
The DIEP flap is then harvested by individually dividing the deep inferior epigastric artery and its one or two venae comitantes. It is helpful to mark the anterior surface of the vessels with methylene blue prior to vessel division. In addition, the smaller vena comitantes can be clipped, leaving the larger vena comitantes and artery open for anastomosis. The flap is weighed at this time to determine its comparability to the mastectomy specimen or implant. It is then transferred to the anterior chest wall and temporarily secured in position. This can be done by suturing the flap to the chest wall or by wrapping the flap in a moist lap sponge that is stapled to the chest wall.
The operating microscope is positioned at the recipient site and the pedicle prepared for microanastomosis, taking care to avoid twisting of the vessels. An end-to-end venous anastomosis is completed with a microvascular coupling device ( Fig. 4.8 ). The end-to-end arterial anastomosis is hand-sewn with 9-0 nylon sutures. Some microsurgeons use a Doppler flow-through venous coupler or implant a venous or arterial Doppler cuff at the completion of microsurgery for postoperative monitoring. Preferably, leave a skin island for postoperative monitoring.
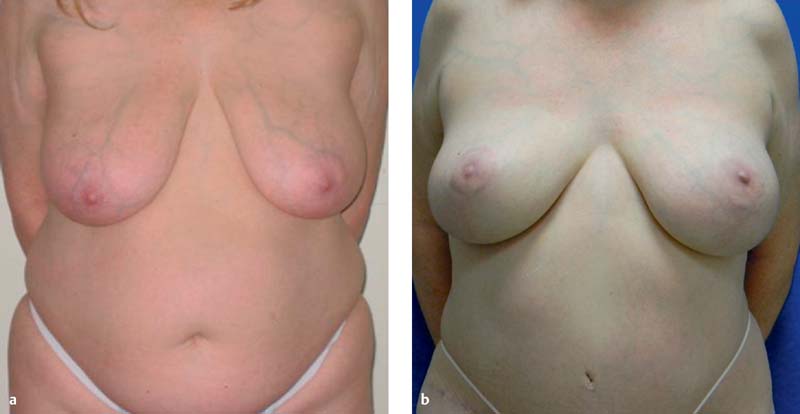
The DIEP flap is then debulked, contoured, and inset to achieve the desired breast size and shape ( Fig. 4.9 ). Areas underlying the native mastectomy flaps are de- epithelialized in situ. The pedicle position is secured and protected with deep absorbable suture. Peripheral absorbable sutures inset the flap in the appropriate position. A closed suction drain is placed and brought out through the lateral edge of the skin closure or through a separate stab incision. After the flap has been shaped and inset, the skin is closed with subcutaneous absorbable sutures.
Ideally, closure of the abdomen begins as soon as the flap(s) are harvested and finished by the time the flaps are inset. The anterior rectus sheath is closed with two-layer running barbed suture. Because no fascia has been harvested, mesh is not used. Any diastasis is repaired with running suture as well. The remainder of the abdominal closure is in layered fashion, using interrupted 2-0 absorbable suture or running barbed suture in Scarpa’s fascia, and running subcuticular skin closure. Two closed suction drains are placed in the abdomen and brought out through the lateral incision. The umbilicus is then brought through the abdominal wall at a predetermined site after de-epithelializing an elliptical patch of skin. It is secured in place using 4-0 half-buried mattress sutures.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree