11 Venous Salvage Procedures in the Deep Inferior Epigastric Perforator Flap for Breast Reconstruction
Thomas Constantinescu and Ming-Huei Cheng
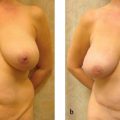
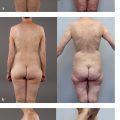
The deep inferior epigastric perforator (DIEP) flap is presently the standard for autologous breast reconstruction. The major advantage of the DIEP flap is decreased donorsite morbidity, as the function of the rectus abdominis muscle is preserved. In a pedicled or free transverse rectus abdominis musculocutaneous (TRAM) flap, this muscle may need to be partially or completely sacrificed. The major disadvantage of the DIEP flap is its associated venous congestion.1–6 Studies show that venous congestion can limit reliable flap dimensions to a larger extent than arterial inflow.1,7 As well, the superficial venous system is interrupted in harvest, and is not always compensated well by the deep venous system,7 which can result in venous congestion. Some authors advocate primary anastomosis of both venous systems, with excellent survival rates.8
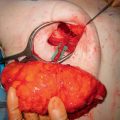
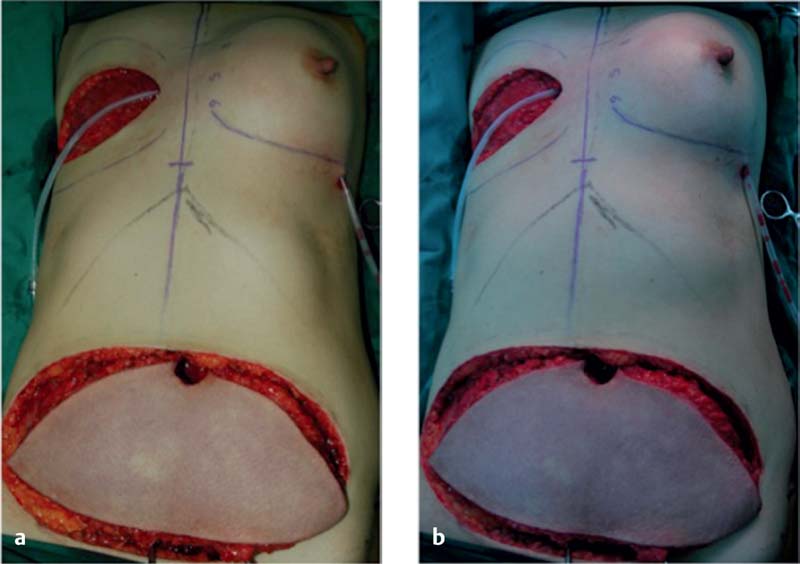
Venous congestion is defined as a noncompensated mismatch in a flap’s ability to drain the blood it requires for adequate perfusion of its entirety. Common etiologies can include an insufficient venous network for the given flap volume, tension at inset, thrombus, and pedicle compression. The incidence of venous congestion in DIEP flaps is reported to be 4 to 20%.4,9,10 The early sign of venous congestion is flap hyperemia, as the venous system begins to be overwhelmed by arterial inflow. As this discrepancy is not alleviated and persists, capillaries begin to burst as arterial pressure exceeds normal capillary pressure, and the flap begins to look purple. Skin survival is largely compromised at this point, but there is the potential for flap survival and eventual reepithelialization with successful salvage. In our series,10 venous congestion developed about one third (31%) of the time before pedicle division ( Fig. 11.1 ), and about two thirds (69%) of the time after microsurgical anastomosis ( Fig. 11.2 ), including postoperatively ( Fig. 11.3 ).
Key Anatomy Points
There are important steps in flap harvest that can help prevent venous congestion, as well as help treat it once it is established. The potential venous drainage of the DIEP flap is through the deep system via the deep inferior epigastric vein (DIEV) or veins accompanying the arterial pedicle, and through the superficial system via the superficial inferior epigastric vein (SIEV), located above Scarpa’s fascia, 4 to 6 cm lateral to the midline.
Attention must be paid to the SIEV early in flap harvest when it is encountered. After making the inferior incision, the vein should be carefully identified in the subcutaneous fat. In a virgin field, it can be dissected inferiorly to a length of 7 to 10 cm with a diameter of 1 to 3 mm.10 As the rest of the flap dissection proceeds, the surgeon should take care to identify sizable perforators for use, and to protect the pedicle, including the DIEVs. The maximum length of DIEVs should be harvested, in case additional length is needed for augmentation, or a DIEV would be required as a vein graft to bridge the SIEV to a recipient vein.
Preoperative Management
As with any surgery, patient selection is key. In taking the medical history, it is important to note prior low abdominal scars, whether midline, paramedian, or transverse, in case the SIEV needs to be used. Theoretically, prior scars can also contribute to “delay” of the flap and improved blood supply. A previous transverse cesarean section can sometimes injure both SIEVs, negating their use as lifeboats. In addition, knowing the availability of other potential recipient vessels, such as the thoracodorsal vessels, will assist with intraoperative decision making. Finally, it is important to gather information preoperatively about any clotting issues that may preclude free flap reconstruction.
The patient is asked to sign a specialized informed consent form that specifically includes consent for vein grafting, additional donor-site incisions, and scars in the event that additional recipient veins need to be dissected. We inform the patient that if complications occur the recovery time and the time by which the breast reconstruction will be completed will be delayed. Finally, we routinely discuss the need for potential transfusion perioperatively, especially if needed for nonmicrosurgical decongestion of the flap.
Intraoperative Management
The importance of early diagnosis cannot be overstated. Hypervigilance and early recognition of flap compromise is essential for the microsurgeon, especially because ischemia critically affects transferred tissue. Diagnosis of venous congestion begins intraoperatively. In a study by Dr. Cheng’s group,10 all patients with venous congestion showed intraoperative signs of it ( Fig. 11.4a ). Examination is aided by removing the operative lights from the field ( Fig. 11.4b ). Damaged pedicles during harvest must be recognized and repaired. Veins that drain well must be chosen for anastomosis. This step is especially critical when large DIEP flaps are used, with large angiosomes responsible for draining of substantial amounts of tissue, in the contralateral medial or even contralateral lateral zone.11 Once the pedicle is skeletonized but not divided, examining the flap for early signs of venous congestion can help alert the surgeon to any need for venous salvage procedures. A step-by-step sequential solution to venous congestion is presented in this chapter.
Microsurgical Salvage
The key issue in assessing venous congestion is the etiology, which is necessary to determine in order to decide on the appropriate treatment. Unless flap compromise is due to a hematologic problem, intraoperative physical exam is crucial to reveal the cause of flap congestion. Because flap congestion is almost always a surgical problem, it requires a surgical solution. The best time to provide the surgical solution is intraoperatively, during the initial surgery. The complication rate is significantly improved with intraoperative salvage versus postoperative reexploration.10
Alleviation of Compression
Early hematoma, leading to venous compression and compromise can be detected intraoperatively. Even with closure over drains, hematoma can develop and must be treated. Pedicle compression can occur easily in this setting, when wedged between a heavy DIEP flap and a rigid chest wall, or when limited by tight radiated skin envelope in a delayed reconstruction.
Flap Trimming
Undue tension can cause flap compromise if the flap is too large for the breast footprint and the remaining mastectomy skin flaps. Such a flap should be trimmed and inset again in tension-free fashion. The penumbra zone of the flap can be evaluated clinically or with SPY technology (Novadaq, Bonita Springs, FL). If a portion of the congested flap can be discarded, especially if it is part of zone IV or the contralateral lateral zone, the treatment is excision, which will reduce venous load and may ameliorate circulation of the entire flap.
Pedicle Repair and Repositioning
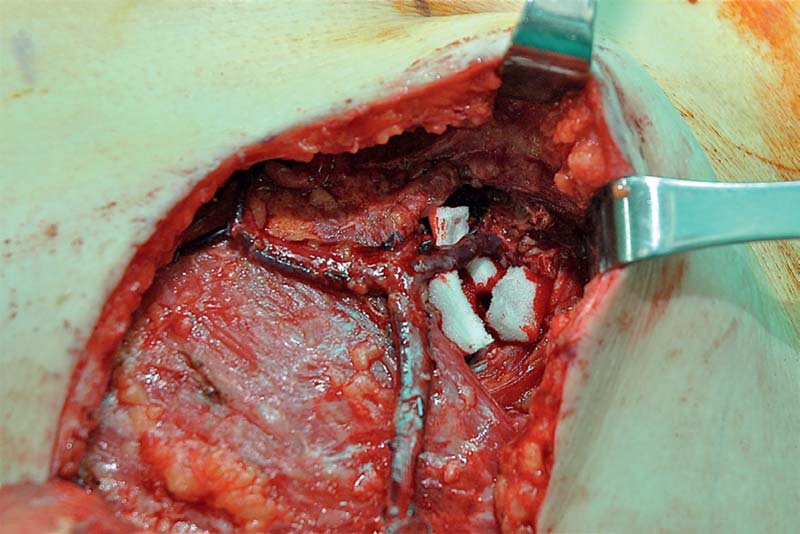
The anastomosis, although the weakest part of the vessel when it was divided, is often the most rigid part of it after suturing and especially after the stenting effect of coupling. Thus, sometimes the cause of venous failure is far from the anastomosis, as in pedicle injury or kinking. With anastomosis to the internal mammary vessels, a kink can occur at the junction of the pectoralis major and rib periosteum, regardless of whether a rib is removed or not ( Fig. 11.5 ). The search for a sharp bend in pedicle should include the entire length of the pedicle, as repeated reanastomosis will not treat an unrecognized kink or pedicle injury. Such misdirected efforts will only lead to further ischemia, unresolved problems, and narrowing of the salvage window.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree