12 Imaging for Perforator Flap Breast Reconstruction
Julie V. Vasile
Magnetic resonance angiography (MRA) is an extremely useful preoperative imaging test for evaluation of the vasculature of donor tissue to be used in autologous breast reconstruction. MRA has sufficient spacial resolution to reliably visualize 1-mm perforating vessels and to accurately locate vessels in reference to a patient’s anatomic landmarks without exposing the patient to ionizing radiation or iodinated contrast. The use of a blood pool contrast agent and the lack of radiation exposure allow multiple studies of multiple anatomic regions in one examination. This chapter discusses the MRA protocol developed with the radiologists at our center and provides examples that illustrate the utility of MRA in perforator flap breast reconstruction.
Preoperative Imaging
Preoperative anatomic imaging of the vasculature markedly enhances the ability of the surgeon to devise a surgical strategy before going to the operating room. Prior to the era of preoperative perforator imaging, a surgeon had little knowledge of the patient’s vascular anatomy until surgery was well underway. As a result, perforator selection could be a complicated decision process taking place in the operating room at the expense of operating time and prolonged anesthesia time.
Doppler
As technology has advanced, surgeons have favored varying modalities for preoperative imaging. Initially, a handheld Doppler ultrasound was solely used to attempt to locate perforating vessels. A Doppler ultrasound is portable and simple to use but cannot differentiate perforating vessels from superficial and deep axial vessels, or robust perforators from minuscule ones, cannot accurately determine the location where perforators exit the fascia, and cannot provide information on the anatomic course of a vessel.1,2 In comparison, color Duplex sonography provides more detailed information about the anatomy of the vessels, but requires highly trained technicians with knowledge of perforator anatomy, and it is time-consuming to perform.2 The technique’s most crucial drawback is an inability to produce anatomic images in a format that a surgeon can easily and independently view.
Computed Tomographic Angiography
Computed tomographic angiography (CTA) is a modality that can demonstrate vessel anatomy, assess vessel caliber, accurately locate perforators, and produce anatomic images in a format that a surgeon can easily and independently view. Although CTA can be performed quickly in as little as 15 minutes,1,2 patients must be exposed to ionizing radiation. Radiation exposure precludes multiple imaging studies in one examination. Articles in the medical literature and the lay press warn that physicians may be exposing patients to excessive and potentially unnecessary radiation.3–6 Patients with breast cancer may have a heightened concern about any modality that can potentially increase the risk of developing a second cancer and may perceive the risks of radiation exposure even more negatively. Patients with breast cancer gene (BRCA) mutations, which confer an increased risk of developing both breast and ovarian cancer, are especially concerned about receiving radiation to the abdomen. An addition drawback of CTA is that the iodinated contrast that is needed to enhance vessels for CTA can be associated with a small but real risk of anaphylaxis and nephrotoxicity.7,8
Magnetic Resonance Angiography
Magnetic resonance imaging (MRI) uses a magnetic field to uniformly align the spin of hydrogen atoms in tissue. The subsequent application of a radiofrequency pulse results in the release of energy as hydrogen atoms return to their relaxed state. MRI coils detect the released energy, and computer software processes the data into anatomic images. Exposure to a magnetic field or radiofrequency pulse with MRI has not been linked to the development of cancer.9 A paramagnetic contrast agent (gadoliniumcontaining) is injected to enhance vessel imaging our group reported demonstrated that MRA accurately locates perforating vessel branches and shows vessel anatomy in a format that is easily viewed by a surgeon.10–13 However, because MRI does not use radiation, this modality has an important advantage over CTA, in that it enables obtaining multiple series of images of the patient in the supine and prone position.
The disadvantages of MRA is that it is contraindicated in patients with a cardiac pacemaker and in very claustrophobic patients. Most patients with claustrophobia can tolerate an MRI if an anxiolytic is prescribed. Continuing advances in MRA have decreased the procedure time for a single donor site to as little as 20 minutes, and decreased the actual acquisition scan time to 20 seconds.11,13–15 However, the examination time could be as long as 40 minutes for multiple donor-site studies.
Magnetic Resonance Angiography Contrast Agents
Gadolinium-containing contrast agents used for MRA have several distinct advantages over iodinated contrast agents used for CTA. The incidence of an acute allergic reaction to iodinated contrast is 3%, which is orders of magnitude higher than the 0.07% incidence of allergic reaction to gadolinium contrast.7,16 Unlike gadolinium contrast agents, iodinated CT contrast agents can induce renal insufficiency even in patients with normal renal function.8,17 Gadolinium contrast agents can potentially induce nephrogenic systemic fibrosis (NSF), also called nephrogenic fibrosing dermopathy. However, reports of NSF have been limited to patients with impaired renal function.18–20 Patients with an acute kidney injury or chronic severe renal disease (glomerular filtration rate < 30 mL/min/1.73 m2) are considered most at risk.18 NSF is a very rare disease, with 380 cases reported worldwide.19,20 Although patients undergoing elective microsurgical free flap are generally healthy and thus are not at significant risk of developing NSF, a creatinine level is drawn preoperatively in patients with a history of hypertension, diabetes, renal disease, or any other indication that renal function may be impaired.
Advances in gadolinium contrast agents with blood pool contrast agents have resulted in a decreased amount of contrast required and improved MR images, and have increased even further the number of donor sites that can be imaged in one study. The MRA protocol developed with the radiologists at our center uses 10 mL of gadofosveset trisodium, a blood pool MRI contrast agent. Prior to using this blood pool agent, 20 mL (instead of 10 mL) of gadolinium contrast (gadobenate dimeglumine) was required. Gadofosveset trisodium is a gadolinium chelate that reversibly binds to serum albumin with 90% binding fraction, and effectively stays within the blood pool with a redistribution half-life of 28 minutes.21 It also demonstrates greater T1 relaxivity that enables administration of a fourfold lower molecular dose while still conferring greater vascular enhancement compared with most other gadolinium chelates. This virtually eliminates the risk of NSF.22
Gadofosveset improves the vessel-to-muscle contrast ratio and vessel sharpness, mainly due to preferential enhancement of vessels compared with muscle derived from the blood pool distribution of gadofosveset.23 This results in significantly improved images of the intramuscular course of perforating vessels, which gives valuable information for choosing the best perforating vessel, avoiding transection of muscle, deciding on the feasibility of a flap with more than one perforator, and planning for double flaps to make one breast.
Because blood pool contrast agents are bound to albumin, with a redistribution half life of 28 minutes, there is a significantly increased amount of time to acquire images.24 This affords the opportunity to assess many donor sites for autologous breast reconstruction in a single examination, with the patient turning from the prone to supine positions to image the posterior (thigh, buttock, back) and anterior (abdomen) donor sites, respectively. In addition, flap volume estimates can be more accurately determined at both anterior and posterior donor sites because the imaging is acquired with the patient in the supine and prone positions, respectively, so that the tissue is not compressed. For example, buttock flap volumes are calculated with the patient in the prone position and abdominal deep inferior epigastric perforator (DIEP) flap volumes are calculated with the patient in the supine position. Once the surgeon has acquired the imaging findings regarding the vessels and flap volume at each donor site, the selection of the most suitable flap donor sites can be discussed with the patient. Moreover, a patient who is found not to be a candidate for an abdominal perforator flap based on imaging findings, who suddenly changes her donor-site preference, or who has a flap failure and requires another perforator flap reconstruction will not require further studies.
Magnetic Resonance Angiography Protocol
Magnetic resonance angiography is performed on a longbore, self-shielded 1.5-tesla (T) scanner (GE Signa 14.0, Waukesha, WI) using an eight-channel phased array coil. The field of view is individualized, but usually extends from 5 cm above the umbilicus to the upper thigh, and transversely is set to match the width of the patient. After acquiring a three-plane localizer, axial and coronal T2-weighted, single-shot, fast spin echo images are acquired to screen for unexpected pathology and to help characterize any lesions detected on postgadolinium scans. Because most of these patients have a history of breast cancer, metastatic disease is detected occasionally. This sequence is also helpful in confirming the central position of the umbilicus in the prone position. A transverse preand postcontrast arterial-phase three-dimensional (3D) liver accelerated volume acquisition (LAVA) sequence is acquired with the following imaging parameters: TR/TE/ flip = 3.9/1.9/15; bandwidth = 125 kHz; slice thickness = 3 mm reconstructed at 1.5-mm intervals using twofold zero interpolation (ZIP 2); matrix = 512 x 128-256; parallel acceleration factor = 2. Precontrast imaging is important to determine the adequacy of fat suppression. Central frequency and shim field of view can be adjusted as necessary to ensure effective fat suppression over the subcutaneous tissues of interest if Dixon fat-water separation is not available.
The arterial-phase imaging is bolus tracked by automated triggering (SmartPrep, GE Healthcare), and scanning is initiated after the contrast arrives in the suprarenal aorta; 10 mL of gadofosveset trisodium blood pool MRI contrast agent is injected, followed by 20 mL of normal saline at a rate of 1 mL/s. Hand injection is preferred, especially if there is a tenuous intravenous (IV) line, because approximately one third of patients may experience some sensation at the injection site or in the pelvis related to the ionic contrast agent.24 The k-space is mapped sequentially, with the absolute center of k-space collected in the middle of the scan, which is ~ 20 seconds after bolus detection, for a 35-second scan duration and a 5-second pause for breath holding instruction. This is important to provide time for the contrast to reach and fill perforating arteries. However, only the largest perforator arterial/vein bundles are adequately seen on this sequence. This is followed by equilibrium phase transverse 3D LAVA at higher resolution without parallel imaging using the following parameters: TR/TE/flip = 4/1.9/15; matrix = 512 x 512 x (172-240); bandwidth = 125 kHz; slice thickness = 3 mm reconstructed at 1.5-mm intervals using ZIP2 slice interpolation. Phase encoding is set to the right-left direction. This is the primary sequence utilized to generate reconstructions and create reports and also serves as a reference for the plastic surgeons. It is acquired with free breathing and typically requires 3- to 5-minute acquisition duration with 0.9 x 0.9 x 3 mm acquired voxel dimension and 0.9 x 0.9 x 1.5 mm reconstructed voxel dimensions. Thereafter, a lower resolution coronal and sagittal plane LAVA is acquired with acquisition matrix of 512 x 256 and 512 x 224, respectively, in a single breath hold and parallel acceleration factor of 2 to evaluate internal organs.
The planned donor site is imaged first, followed by single high spatial resolution equilibrium phase imaging of other potential donor sites using the free-breathing 3D LAVA sequence described above. A typical complete perforator flap MR examination, including abdomen, buttocks, and upper thigh, can take 45 minutes.
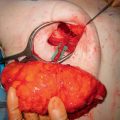
After screening axial and coronal single-shot fast spin echo images for unexpected pathologies, the arterialphase images are reviewed to determine the number of perforators available and to look for any enhancing lesions. High spatial resolution equilibrium phase images are used for final perforator evaluation, as perforators are best visualized on these images. The equilibrium phase series is loaded on a computer workstation (GE Advantage Windows 4.4, Milwaukee, WI) for postprocessing. Coronal, sagittal, and surface-rendered reformatted images are generated. The reference point and each candidate perforator artery/ vein bundle are identified. The diameter and perforator exit location at the point where the vessel pierces the superficial fascia and enters into subcutaneous fat are noted. The cephalad/caudal and right/left distances of each perforator exit site relative to the reference point are calculated to create a perforator location coordinate. The intramuscular course and length of each perforator is measured to predict the vascular pedicle length. Finally, a predicted flap volume is calculated on the same workstation assuming an elliptical geometry on a slice-by-slice basis.
Coordinates identifying the location of the perforating arteries on the axial images are superimposed and displayed on volume-rendered 3D reconstructed images and coronal 3D minimum intensity projection (MIP) images. These images are especially helpful to locate the perforator vessels during preoperative surface marking and then intraoperatively.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree