30 Fat Grafting in the Breast-Conserving Therapy Deformity
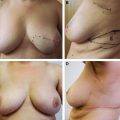
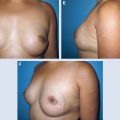
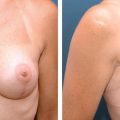
Breast-conserving therapy (BCT) offers patients with early-stage breast cancer a valuable option for local control of the tumor and potentially acceptable cosmetic results. Several studies have shown that the oncologic safety of a lumpectomy or quadrantectomy followed by radiotherapy is comparable to that of a mastectomy, with the advantage of being less mutilating. 1 – 6 In recent years, however, disappointing cosmetic results have increased because of poor attention to cosmesis and the widespread use of BCT for women with large breasts or for those who require large excisions for tumor removal. Despite the development of oncoplastic techniques, 7 , 8 more unsatisfied patients present to plastic surgeons for further reconstruction because of persistent deformities. BCT deformities have been classified according to Berrino et al 9 as postquadrantectomy deformities and have been reclassified by Clough et al 10 as types I through III (see Chapter 24). The conventional approach to the correction of BCT deformities is surgery; however, this often requires complex flap procedures, which are further complicated by the poor response of irradiated tissue to surgery, leading to a relatively low success rate.
It is important to consider the two main causes of BCT deformities and what is required to correct them: (1) shape modifications caused by fibrosis, scar contracture, and radiotherapy and (2) volume modifications resulting from the resection of glandular and subcutaneous tissue.
When these two causes are considered, the therapeutic approach to BCT deformities should also involve the concept of tissue regeneration. Shape modifications can be resolved if fibrotic tissue is restored to normal tissue; volume modifications can be achieved by adding volume and creating new, healthy tissue at the site where the tumor and appropriate margins were surgically excised.
Adipose Tissue: Regenerate Factors
The use of adipose tissue as a filler material in plastic surgery has a long history. Since the nineteenth century, fat grafting has been described and proposed for therapeutic and cosmetic purposes. 11 Only recently has progress in molecular and cell biology allowed further insight into the scientific basis of fat transfer and the emerging evidence of the regenerative potential of fat grafts. 12 – 15 Although still controversial, strong evidence suggests that adipose tissue plasticity is attributable to multipotent stromal cell populations capable of differentiating into mature adipocytes with multilineage properties. Recent studies have shown that the adipose stromal vascular cell fraction (SVF) contains regenerative precursor cells with proangiogenic capabilities. 16 – 18 Adipose stromal cells were reported to secrete angiogenic and antiapoptotic factors 19 , 20 that allow differentiation into endothelial cells and their incorporation into vessels, 21 thus promoting neovascularization in ischemic tissues.
Transplanted adipose tissue (lipoaspirate) offers the potential to restore volume after BCT because of its ability to promote the generation of mature adipocytes. In addition, the observed neoangiogenic properties of the stromal cell fraction contained in lipoaspirates provide neovascularization factors in fibrotic tissue to promote shape recovery.
Cytologic Characterization of Lipoaspirates
In a recent clinical pilot study, 22 we reported striking results with repeated fat grafts for the treatment of chronic radiation-induced lesions. To understand the mechanisms responsible for such impressive regeneration capabilities of fat injection, we applied cytologic techniques to characterized lipoaspirates to quantitatively assess the presence and density of the mesenchymal stem cell (MSC) fraction in the transplanted tissue and to evaluate differentiation potentialities. The analysis was performed according to current standard methods and procedures.
Phase One: Isolation of the Stromal Vascular Cell Fraction
Sterile Hanks’ balanced salt solution was used to wash 40 cc of lipoaspirate. After washing, the extracellular matrix was digested at 37° C in the same salt solution with 1 mg/ml collagenase type I and 2% bovine serum albumin (BSA). After incubation, the digestion enzyme was neutralized, and centrifugation at 1200 rpm for 10 minutes was performed to obtain a high-density SVF pellet. This was resuspended in 160 mM NH4Cl and incubated at room temperature for 10 minutes to lyse contaminating red blood cells. After incubation, the SVF was collected by centrifugation and filtered through a 70 µm nylon mesh to remove cellular debris.
Phase Two: MSC Expansion
To obtain a homogeneous MSC population, the SVF was incubated for 72 hours at a concentration of 1 × 105 cells/cm2 with a high glucose concentration, GlutaMAX-I, 15% heat-inactivated fetal calf serum (FCS), 100 U/ml penicillin, and 100 µg/ml streptomycin. The cells underwent a second incubation period of 2 to 3 weeks in specific flasks (BD Falcon). Cells were cultured at a temperature of 37° C in a 5% CO2 atmosphere.
Phase Three: MSC Immunophenotypic Analysis
MSCs were identified according to their immunophenotype using monoclonal antibodies specific for CD105 (endoglin), CD73, CD106 (VCAM-1), CD29, CD44, and CD90. In addition, we assessed the lack of endothelial cell (with anti-CD31 antibodies) and hematopoietic cell (with anti-CD45, anti-CD14, anti-CD11c, and anti-CD34 antibodies) marker expression. The immunophenotypic analysis was performed by incubating 100 cc of cell suspension with the specific antibody for 30 minutes. At least 10,000 events were analyzed using flow cytometry. The results showed that the immunophenotype of adipose tissue–derived and in vitro–expanded MSCs were positive for surface CD105, CD73, CD29, CD44, and CD90, and therefore were similar to bone marrow–derived stem cells. They were negative for endothelial (CD31) and hematopoietic (CD45, CD14, and CD34) markers. A significant quantity of clonogenic CD105-positive cells was documented (1.02 × 103 CFU-F [colony forming units–fibroblast]) could be obtained with a single lipoaspirate.
Phase Four: MSC Differentiation Assay
MSCs were tested for their ability to differentiate into adipocytes, osteoblasts, and chondrocytes. Differentiation was obtained after culturing MSCs with specific media (adipogenic, osteogenic, or chondrogenic, depending on how the cells were to be processed) for 2 weeks. More than 90% of the cells differentiated, depending on the time left in culture with the differentiating agent.
Clinical Results of Fat Grafting
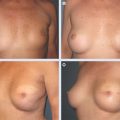
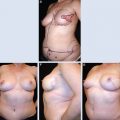
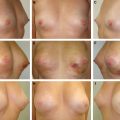
This clinical pilot study focused on the treatment of radiolesions and showed very significant improvements in all 20 participants. 22 The clinical evaluation was associated with ultrastructural analysis of the tissue within the pathologic site, with up to 36 months of follow-up. The analysis showed clear signs of gradual tissue regeneration. The final results showed very well–hydrated tissue, a significant density of mature and normal adipocytes, and newly formed microvascular networks with normal ultrastructure. These results encouraged us to evaluate the use of lipoaspirate autologous transplant for the treatment of BCT deformities, with the hypothesis that the promotion of tissue regeneration processes and neovascularization would lead to increased volume expansion and a more normal shape, respectively.
Surgical Technique
Surgical Plan
Harvest the lipoaspirate.
Purify the lipoaspirate.
Place the lipoaspirate.
No specific patient preparation or surgical planning procedures are required. It is preferable to wait at least 1 year after breast irradiation before initiating reconstruction. The deformity may range from a minor contour irregularity with healthy skin coverage (preferable) to significant fibrosis with scar contracture (more difficult). More complex deformities require more procedures, which need to be discussed preoperatively with the patient. Preoperative cancer surveillance and breast imaging are important before reconstructing any BCT deformity.
The targeted area, the number and location of entry points, and the direction of tissue injection tunnels are defined preoperatively. The goals are to achieve maximum uniformity of distribution and to limit significant overlaps and gaps in tissue deposition.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree