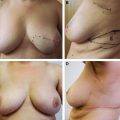
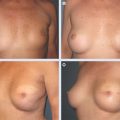
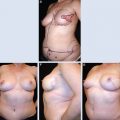
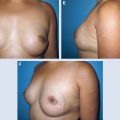
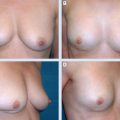
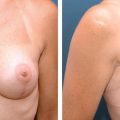
29 Breast Lumpectomy Reconstruction With External Vacuum Expansion and Autologous Fat Grafting
More than 1.5 million women worldwide are diagnosed with breast cancer every year, and in the United States, approximately 231,840 women are diagnosed annually. 1 , 2 Although about 35% to 40% of these women will undergo total mastectomy, 3 – 5 the majority of patients (60% to 65%) are offered the option of breast-conserving therapy (BCT) with partial mastectomy and radiation. Of women who undergo mastectomy, 25.8% elect to have their breast reconstructed with an implant-based technique and 12% with autologous flap reconstruction. 6 However, most women who opt for BCT and radiation do so in hopes of avoiding reconstruction surgery. Unfortunately, many partial mastectomy surgeries produce significant deformities that still require reconstruction.
In this chapter we describe the workup, patient selection, and techniques for reconstructing the breast after lumpectomy using external tissue expansion (ETE) to prepare the recipient area and fat grafting to replace the missing tissue. We also elaborate on the principles and techniques of large-volume autologous fat grafting, especially those pertinent to grafting scarred and irradiated tissue. We review the ideal patient for lumpectomy reconstruction using ETE and fat grafting.
Minimally invasive fat grafting is another option to reconstruct partial defects, and to optimize its success, the defect should be prepared with ETE.
ETE stretches the scar and increases its vascularity to generate a favorable recipient fibrovascular scaffold for fat grafting.
Within a few days to several weeks, under the effect of cyclic mechanical forces, ETE creates a three-dimensional, vascularized, fibrous scaffold. The skin envelope, subcutaneous fat, residual parenchymal tissue, and—to a lesser extent, because it is stiffer—the scar tissue expand to generate new stromal, connective, and vascular tissue. 7 – 13 This is effectively tissue engineering. ETE induces the in situ creation of a vascularized biologic scaffold that is optimized to be seeded with microdroplets of judiciously transferred fat grafts. 14
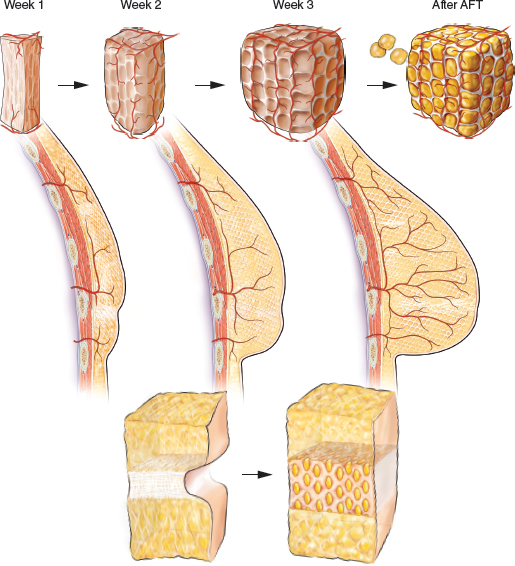
These images show how the tissue scaffold is created with ETE, showing the generation of new stromal and vascularized tissues. Notice how after fat grafting the small holes that are made in the expanded scaffold percutaneously are filled with small droplets of grafted fat. The grafted fat particles occupy the small spaces created and undergo revascularization. Ultimately, when the graft “takes,” the volume deficit and contour irregularities are corrected.
The combination of ETE and fat grafting has been shown to be a safe, effective, and minimally invasive outpatient method for reconstructing the breast after a partial breast mastectomy. 15 – 17
Principles of Fat Graft Survival
There are two fundamental factors that can limit the survival of free fat grafts: (1) restoration of the blood supply, and (2) adequate recipient site capacity.
Restoration of the Blood Supply
No free graft can survive without a blood supply. Angiogenesis, the process responsible for neovascularization, can only cause growth of vessels about 1 mm inside the grafted tissue before the deeper parts die from ischemia. Stem cells in the graft are most resistant to ischemia and are able to survive at that depth to regenerate cells to replace the more superficial fat cells lost to ischemia. 18 Restoration of the blood supply therefore depends on a graft-to-recipient interface that cannot be much wider than 1 mm. Cells positioned farther from the blood supply are doomed to necrose from ischemia. Multiple experimental models have confirmed that free grafts larger than 2 mm across (1 mm per side), even pure stem cell grafts, will invariably have a central necrotic focus. This means fat droplets larger than 10 µl will invariably have some central necrosis and for a 1 cc droplet, only its outermost 40% shell will survive. Fortunately, the body is able to scavenge the central necrotic debris, leaving only the larger necrotic cysts to persist after a few months. Therefore, ideally, grafted fat should be delivered as a fine mist of tiny 10 µl droplets or 2 mm wide microribbons that remain separated by recipient tissue and that do not coalesce into larger blobs. 13
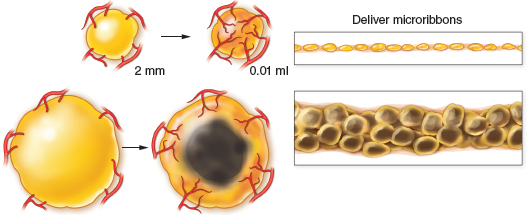
Large-volume three-dimensional avascular graft survival is shown in terms of the fundamental principle of the graft-to-recipient interface, and the droplet size restriction principle. It is important to evenly spray 0.01 ml microdroplets that do not coalesce into large lakes of fat that will ultimately undergo fat necrosis.
Recipient Site Capacity
As the surgeon inserts tiny individual droplets (or microribbons) into the recipient site, the area has to expand to accommodate the added tissue. However, even if these droplets are meticulously inserted without any coalescence, there is a limit to how much the recipient site is capable of expanding before the filling pressure increases to levels that choke capillary circulation. Beyond this limit, grafting is counterproductive, and more grafting will lead to more significant necrosis. This limit to expansion is determined by the mechanical compliance of the recipient site.
The lumpectomy site must be prepared to best accept the amount of fat graft required for the reconstruction.
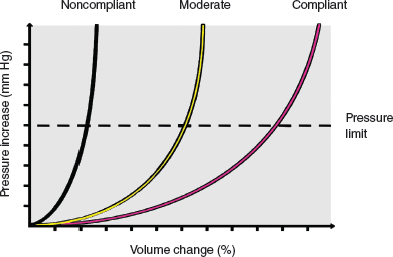
Normal subcutaneous fat is the body’s natural buffer for excess fluid and is the most compliant recipient tissue. Studies have shown that it can accommodate up to a 20% volume change without a significant pressure increase. From 20% to 30%, every percentage point leads to 0.5 mm Hg pressure rise in interstitial tissue pressure, a tolerable increase. With a 30% to 40% volume increase, every percentage point results in a 1 mm Hg pressure rise, an increase that is somewhat less tolerable. Then from 40% to 50%, the pressure increment becomes steeper, and unless the tissue of the site has been preexpanded, no recipient site can accommodate more than a 50% volume increase. The compliance curve is not linear; as fatty tissue is added, compliance becomes stiffer and stiffer, and smaller increments lead to a major pressure increase. It is important to avoid grafting beyond that last drop that can tilt the pressure to the necrosis zone. Unfortunately, partial mastectomy defects, especially regions that have been irradiated, are much less compliant and much less tolerant of overgrafting. In practical terms, the recipient site volume and its mechanical compliance determine the amount of graft that can be tolerated. 13
The pressure compliance curve of various tissue recipient beds is shown. Noncompliant, moderate, and compliant beds show how the percent volume change can increase with minimal increases in pressure gradients in more compliant recipient sites. In this chart, the more compliant curve reflects breast preexpansion using an external tissue expansion system.
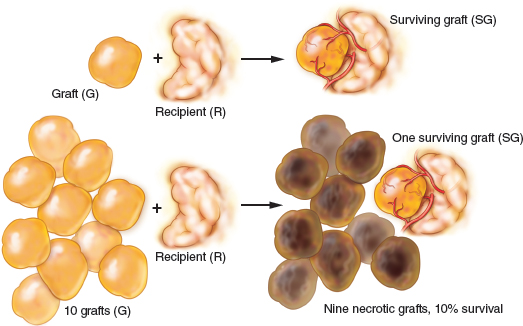
It is also important to recognize the stoichiometry of graft-to-recipient interaction, where 1 graft (G) plus 1 recipient site (R) leads to 100% survival; however, if we place 10 grafts to 1 recipient site, we end up with 1 surviving graft (SG) and 9 necrotic grafts, for a 10% survival rate.
Every graft G (the smallest fat droplet unit) needs to interact with a specific recipient site R (the smallest unit of the fibrovascular network) to yield a surviving GR complex. Adding more Gs in the face of a fixed number of Rs is counterproductive. It will not result in more GRs—only more necrotic Rs; not more augmentation, only a lower percentage graft survival. Herein lies the fallacy of percentage graft survival.
Assuming an ideal grafting technique and that every G successfully reacts with an R, grafting 10 Gs when there are only 5 Rs will at the very best lead to 5 GRs; the remaining Gs will eventually die, for a 50% graft survival. Similarly, grafting only 5 G could lead to a 100% survival; this constitutes the same effective augmentation, but double the percentage survival with less necrosis. On the other hand, brutally grafting 20 G of the same graft in the same 5 G recipient bed will probably not lead to 25% survival, but total necrosis, because the compliance of the recipient site would be unable to accommodate the grafted tissue.
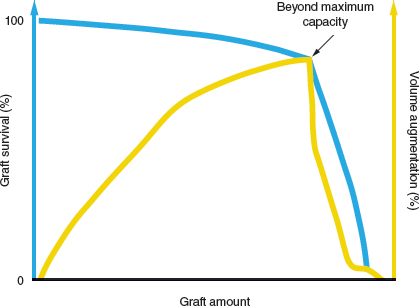
These two superimposed graphs show graft survival (light blue curve) versus volume augmentation (yellow curve) compared with the grafted amount (x axis). The most difficult question is to know when to stop grafting, since the last-drop effect dictates that one cannot graft more than what the recipient tissue bed can tolerate. This point is called the “point beyond max capacity” (black arrow), where the volume augmented and graft survival are optimized compared with the amount grafted. This is the point at which the more grafts, the worse the results will be.
Therefore percentage graft survival, the most commonly used yardstick of success in fat grafting, is only a reflection of the tendency to overgraft. Since the ultimate goal is tissue augmentation, a more relevant measure of success should be the percentage augmentation of that recipient area, regardless of whatever amount of fat preparation is grafted. The upper limit of percentage augmentation is determined by the amount of Rs present in the recipient and not by the amount of added Gs. Grafting beyond the point where all available Rs interact with Gs is counterproductive. Of course, the craftsmanship of the surgeon in ensuring that Gs are delivered in optimal-sized packages that do not coalesce and that every G is inserted in close proximity to a recipient R remains a critical step.
Because fat grafting is done to add volume to a mastectomy defect, the final percentage volume augmentation obviously should be one of the most relevant measures of success. Assuming we are using the very best graft tissue, augmentation is still limited by (1) the stoichiometry, or the ratio of graft to recipient, (2) the craftsmanship of graft delivery, and (3) the recipient site’s capacity to accommodate the graft. We reviewed the literature and performed some calculations to derive this often unreported number; this revealed that augmentation is limited to 40% in the best-documented reports and in nonirradiated recipient tissues. 19 It is only after preexpansion of the tissue of the recipient site (and thus increasing the Rs and compliance) that percentage augmentation reaches the 80% to 90% range. 7 , 8 , 13 – 17
Thus in practice, achieving success of large-volume fat grafting is a complex, multifactorial process. Focusing on a single factor or a single step in the series of events leading to graft survival is a common but erroneous oversimplification. The ultimate survival of the grafts depends on on a number of individual factors and steps that are chain-linked in series. Regardless of the strength of the other links, it is the weakest link that seals the outcome. The following analogy of a farmer optimizing his crop nicely illustrates this multistep process. The four Ss of a successful crop are:
The “seeds”: These must be gently harvested—viable, fine fat tissue droplets.
The “soil”: The recipient site, which needs to be large enough, well ploughed, and fertilized to accept the large number of “seeds” required for the necessary crop.
The “sowing”: The fat grafting technique must skillfully distribute the “seeds,” one at a time, over the entire field to maximize their individual chance of taking root.
The “support”: After grafting, the area must be immobilized to protect the “seedlings” from stamping, drying, and shaking as they engraft.
Most of the scientific literature focuses on the graft, the method of harvesting and preparation, the selection of better seeds, such as stem cells, and the addition of growth-promoting factors. However, even the very best seeds with the best additives will not yield much if they are crammed in a tiny and rocky plot. The success of large graft amounts is most often limited by the fact they are used to enlarge small hostile (irradiated) recipient sites. Injected fat is not a tissue expander; grafted seeds/fat droplets are not capable of significantly stretching the recipient site and also surviving. They will do better when they are comfortably seeded in an already stretched recipient site. Furthermore, the sowing technique is unfortunately the least studied and the most variable operator factor. Even the best seeds in an ideal recipient field will not yield much if they are dumped all together in a clump. The ideal seeding technique should disperse a fine mist of fat droplets instead of a “caulking gun” stream. Skillful dispensing is a critical factor that requires time and craftsmanship to gain mastery.
Microvascular surgeons know that free flaps can fail despite a perfect anastomosis, because success and failure depend on many factors besides the anastomosis. Similarly, perfect grafting materials, such as adipose-derived stem cells, do not guarantee success, because there may be other bottleneck rate-limiting factors that can lead to a poor result. An appropriate analogy in this case is that of a moped and a Ferrari competing in heavy traffic—adding horsepower is fruitless when traffic is the rate-limiting factor.
For practical purposes, the farmer analogy still stands; all four Ss need to be optimized, and struggling to improve a non-rate-limiting factor is futile, because the remaining bottleneck will still limit the outcome.
This brief overview is still incomplete, since we still do not fully understand the intricate biology of fat grafting and the multitude of additional cytokines and factors involved in the sequence of events leading to successful fat graft survival. But again, farmers knew what it takes to feed our civilization for centuries, long before they understood the complex biologic processes that led them to reap successful crops. Further investigations will allow us to achieve ever-better results in breast reconstruction after lumpectomy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree