28 Local Perforator Flaps in Oncoplastic Breast-Conserving Surgery: The Nottingham Experience
Fasciocutaneous chest wall perforator flaps such as the lateral intercostal artery perforator (LICAP), the lateral thoracic artery perforator (LTAP) flap, and the thoracodorsal artery perforator (TDAP) flap have been described for reconstruction of partial breast defects in breast-conserving surgery/breast-conserving therapy (BCS/BCT) for breast cancer and also have a role in postmastectomy reconstruction. 1 – 4 In this chapter we present our approach to using these flaps in BCT and present surgical and patient-reported outcomes.
Local perforator flaps harvested from the chest wall adjacent to the breast have inherent advantages. The lax lateral chest wall subcutaneous tissue acts as an ideal filler to replace volume in breast defects. In addition, the resultant scarring from harvesting tissue lateral or inferior to the breast can usually be hidden in the bra line and full function of the latissimus dorsi (LD) muscle is preserved, keeping donor-site morbidity to a minimum.
Harvested without associated muscle bulk, they have homogeneous flap thickness. They may also be raised on a solitary vascular pedicle and can safely be oriented 90 degrees to the axis of the vascular pedicle, maximizing use of the frequently available axillary skin fold for good volume and skin transfer if required. Similarly, they can quickly be raised on a small mesentery of tissue including perhaps two or three perforators and turned over into the lateral or lower regions of the breast. These characteristics, and their close proximity to the breast, enable these flaps to be used for a wide variety of indications in primary cancer excisions and breast reconstructive surgery.
Nottingham Experience
The experience presented in this chapter relates to all local perforator flap surgeries performed for BCT at the Nottingham Breast Institute (Nottingham, UK) from 2007 to 2015. Patients were closely followed after surgery, and outcomes and complications were identified prospectively. Routine cancer surveillance was performed annually by means of clinical examination and mammography.
Patient satisfaction was assessed at 1 year after surgery by a self-rated questionnaire consisting of two parts; the first was the validated Hopwood Body Image Score 5 that assessed the patient’s affective, behavioral, and cognitive response to treatment; the second part consisted of questions formulated to elicit feedback specifically on local perforator flap surgery.
Indications for Local Perforator Flaps in Breast-Conserving Surgery
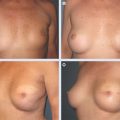
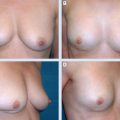
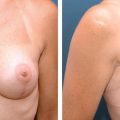
Local perforator flaps were primarily performed in the same procedure with tumor resection, the volume of which would otherwise have resulted in a poor cosmetic outcome.
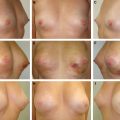
Typically, women in our series had nonptotic breasts, a degree of lateral chest wall laxity with adequate subcutaneous volume, and a tumor situated in the lateral third or lower pole of the breast. Such cases would usually be thought of as ideal for a local perforator flap, and this group of women represented our most common indication for this surgery. However, in women with breasts of similar morphology and a suitable donor site, a local perforator flap would be considered a defect in any location if the scar resulting from flap harvest was acceptable.
The key selection criteria were the degree of lateral chest wall laxity and adequate subcutaneous tissue volume. In most cases flaps would be replacing more than 20% of breast volume, but the range is estimated to be 5% to 50%, reflecting the fact that flap size varied greatly, depending on clinical need and the volume of available donor tissue.
As our experience grew with the LICAP and LTAP flaps, the indications for a TDAP flap in BCT became much less common and more specific.
The specific elements of TDAP flap design and dissection are described separately.
Principles of LICAP and LTAP Flap Design
By using the appropriate chest wall perforator located around the periphery of the breast, a flap can be raised to permit reach into any breast defect location in any woman who is a suitable candidate.
Characterizing the position of the perforating vessels used for these flaps was initially based on clinical experience, using preoperative handheld Doppler (8 MHz) mapping and intraoperative observation. A detailed study using anatomic dissections and a clinical series of Doppler observations assisted in confirming and better defining the anatomic basis of the flaps most commonly used. Other good anatomic descriptions of chest wall perforator locations exist, but these have been mainly defined in terms of their proximity to the anatomic landmark of the anterior border of the latissimus dorsi muscle or axillary fold. The aim of our studies was to define the location of the perforators in terms of their proximity to the breast. In so doing, we identified consistent perforator locations, which reinforced our clinical experience that local perforator flaps could be raised based on perforators lying very close to the breast periphery, giving them easy and potentially long reach within the breast. In addition, identification of these perforator locations allowed scar placement to be within the bra line in most cases.
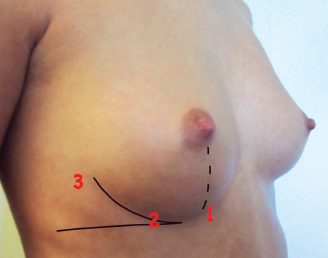
The vascular basis of the vast majority of local perforator flaps used in our series is illustrated. Although considerable anatomic variation exists in the precise location of individual perforators, the figure shows a simplification of where perforators are initially looked for preoperatively. The consistency of these sites is such that it is exceptional for perforators not to be found in close proximity to these landmarks. The most commonly used and consistent LICAP perforators are indicated: located close to the point where the breast meridian crosses the IMF (1); located in the angle between the lateral breast curve and a horizontal line level with the IMF (2); and located just lateral to the lateral breast curve (3).
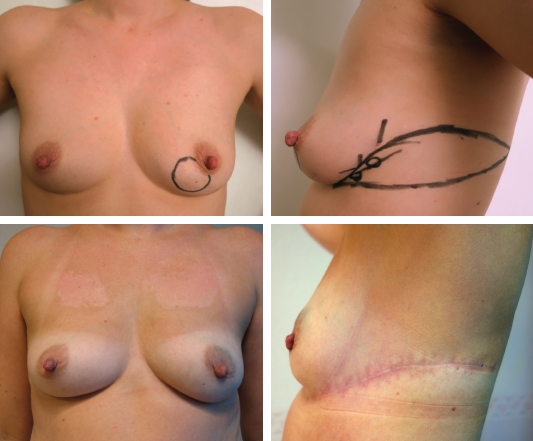
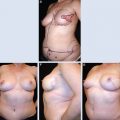
The flap is planned to include one or more perforators near to the breast then varied according to the distribution of skin laxity, and the volume and reach of tissue required. Flap design followed a typical pattern in most cases, the guiding principles of which are as follows; the inferior border of the flap is based on a lateral upwardly curving extension of the inframammary fold (IMF). The superior border is then mapped according to skin laxity, perforator location, and natural skin tension lines. Potential tension on wound closure is avoided and the flap incorporates existing skin laxity so that the resulting closure does not displace the breast laterally. The flap design typically forms a leaf pattern, as shown for a typical LICAP case.
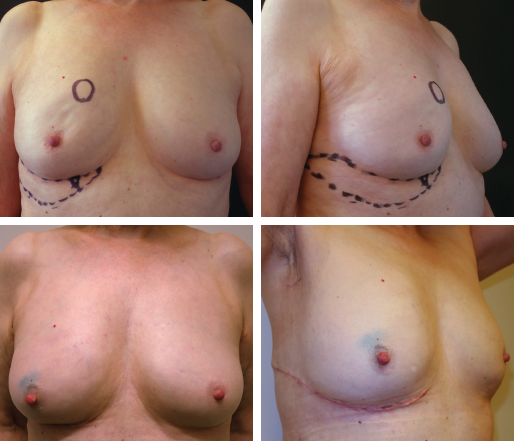
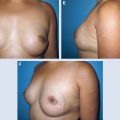
For women with skin laxity below the IMF, a crescent-shaped flap is used that may be extended laterally, if required. A flap based on an inframammary perforator may be used for a wide excision of a cancer in the upper inner quadrant.
LICAP and LTAP Flap Dissection
General anesthesia is administered, and the patient is positioned supine with an inflatable 3 L pressure bag below the ipsilateral hemithorax to tilt the torso. This provides good access to the lateral chest wall and breast, and deflation of the pressure bag after flap harvest and closure of the wound posteriorly allows insetting with the patient supine, or sitting up if necessary. Local anesthetic with epinephrine is infiltrated around the planned wide local excision and is used as a means of hydrodissection of the subcutaneous tissue overlying the cancer. It is also infiltrated around the flap, carefully avoiding the marked LICAP perforators and the potential LTAP territory.
For most breast defect locations, the LICAP flap is based on a perforator located at the junction of the IMF and lateral breast fold. In most cases, an additional perforator located above this could also be included and a mesentery of tissue between the two is preserved. In addition to the LICAP perforators, we found intraoperatively that a high percentage of LICAP flaps could be either converted into or supplemented by LTAP vessels.
In many cases, an LTAP vessel had been suspected preoperatively on Doppler during planning of the flap, but in an equal number it was sought and found intraoperatively, and the course and size of the vessel was such that it became the serendipitous source of flap perfusion. In some cases, anatomic variants of the chest wall vessels, such as a long subcutaneous branch of the thoracodorsal vessels, the serratus branch of the thoracodorsal vessels, or a subcutaneous branch of a vessel accompanying the long thoracic nerve was used. In many such cases, preserving both LTAP (or a variant vessel) and LICAP vessels did not compromise the mobility of the flap, whereas in others the flap was rationalized to the largest and most clinically well-located source. In such situations this was usually the LTAP vessels because of their larger caliber and the surgeon’s ability to convert the flap into one with a longer pedicle, enabling greater reach and more flexibility of orientation and means of transposition.
For breast defects in the lower pole or medial half of the breast, perforators along the IMF are used, creating the anterior and medial intercostal artery perforator flaps (AICAP or MICAP). The most constant of these is located at the point at which the breast meridian crosses the IMF or just lateral or medial to this point. This allows a flap to be designed along the IMF, extending for the required distance along the bra line, and to be transposed into a defect, which could even lie in the upper inner quadrant. Similarly, a skin-bearing flap can be used to replace the nipple when a central excision is indicated.
Because of the rich diversity of options available to vascularize a chest wall flap, the learning curve for this operation includes developing a sense of when one ideal source of vascularization has been identified so that others can be sacrificed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree