20 Ear Reconstruction
Summary
Ear deformities are common in the pediatric population. Disorders include: constricted ears, microtia, accessory cartilage, lobule anomalies, prominent ears, cryptotia, Stahl deformity, keloids, and traumatic defects. The reconstructive procedure and timing depends on the type of deformity.
20.1 Introduction
Several types of ear anomalies affect the pediatric population. These are reconstructed using different otoplasty strategies. Construction of an absent ear (microtia) is presented in a separate chapter. The primary morbidity of an ear anomaly is psychosocial. Children can develop lowered self-esteem if peers notice their malformation. Ear anomalies are not painful and reconstruction does affect hearing; consequently, insurance companies may not cover the procedure. Correction of some deformities (e.g., cryptotia) can facilitate the use of eyeglasses. Ear anomalies may be broadly divided into three categories: (1) excess structures, (2) deficient structures, and (3) structures present but malformed.
In contrast to most other types of congenital anomalies managed by pediatric plastic surgeons, ear malformations can cause increased anxiety for families because (1) they often have to pay for the procedure and (2) they are subjecting their child to general anesthesia for a “cosmetic” problem. Consequently, surgeons treating ear anomalies should be well trained on the subject and ensure that patients and families have reasonable expectations (similar to rhinoplasty). For example, I emphasize that the goal of the operation is to improve the child’s appearance, but that I “guarantee” there will be some residual asymmetry between the ears.
20.2 Diagnosis
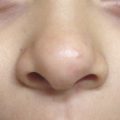
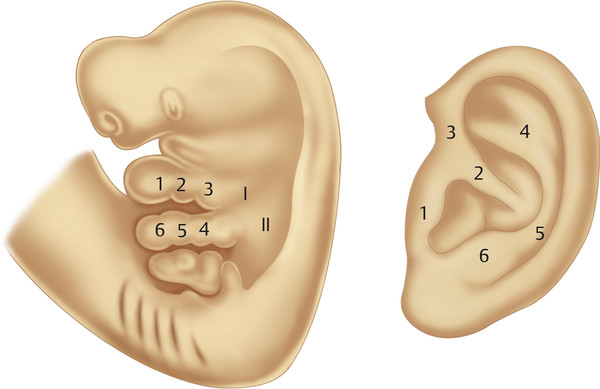
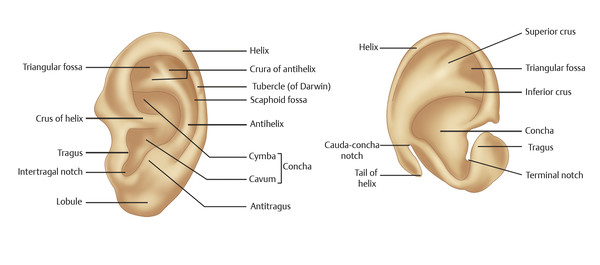
Diagnosis of an ear anomaly is made by physical examination (Fig. 20‑1, Fig. 20‑2). The surgeon should be able to identify the abnormality and list reconstructive options. A framework for the operative approach usually can be made by determining if structures need to be removed, added, or rearranged. Ear anomalies can be better understood by knowing the embryology of ear development. The first mandibular arch gives three anterior hillocks: (1) tragus, (2) root of helix, and (3) superior helix. The second hyoid arch results in three posterior hillocks: (1) antihelix, (2) antitragus, and (3) lobule. The external auditory meatus is formed by the first branchial groove (Fig. 20‑3).
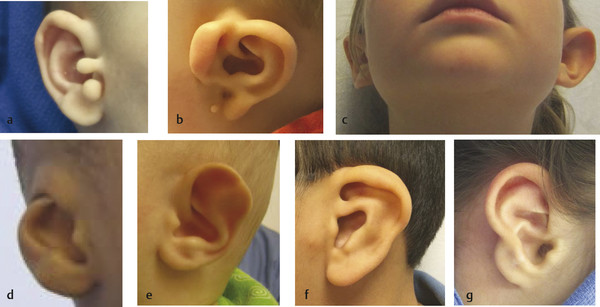
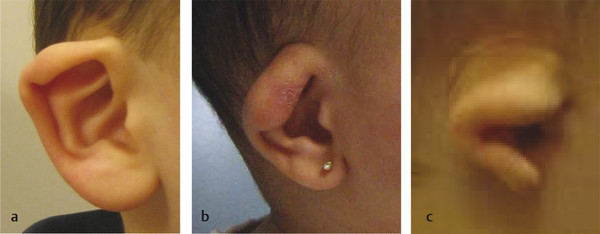
The surgeon must understand normal ear anatomy and can use a contralateral normal ear, if present, as a guide (Fig. 20‑4). The axis of the ear is angled approximately 20 degrees more posterior than a vertical line. The top of the helix is at the level of the eyebrow and the lobule is at the base of the columella. The height of the ear is 6.5 (±1) cm and its width is 4.0 (± 0.5 cm). It is located 6 to 7 cm posterior to the lateral canthus. Although several measurements for normal protrusion from the mastoid have been described, I use 15 mm (top helix), 20 mm (midhelix), and 22 mm (lobule).
Imaging is rarely necessary to manage an ear deformity. If a branchial cleft cyst or sinus is suspected, however, I will obtain a magnetic resonance imaging to confirm the diagnosis and determine the extent of the tract. Occasionally, a sinus can be deep and approximate the facial nerve; this information is helpful prior to operative intervention.
20.3 Nonoperative Treatment
A window exists up to 3 months of age when the ear can be molded. Because one-third of anomalies improve during the first 2 weeks of age (particularly a prominent helical root), molding should not be initiated until after this time. Ideally, molding is started between 2 and 4 weeks of age when maternal estrogens in the child facilitate cartilage manipulation. The ear can be molded using wire and tape or a commercially available system (Fig. 20‑5). Both methods are equally effective; the commercial system is easier to use, but more expensive.
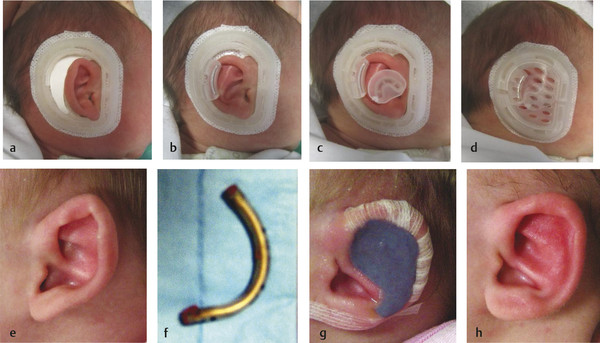
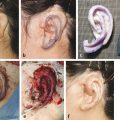
Unfortunately, patients often are referred after the window to mold their ear has passed. I will not attempt to mold the ear after 3 months of age because of the poor response. The ear deformity that is most amenable to molding is a prominent ear (Fig. 20‑6). The antihelical fold is straightforward to create and the concha is able to be set back. Another anomaly that is able to be molded includes a mild constricted ear with overhanging helical cartilage; the cartilage can be “lifted” into better alignment. A Stahl deformity also can be improved by flattening the abnormal crus. Children commonly are referred for ear anomalies that cannot be helped with molding (e.g., severe constricted ear, cryptotia).
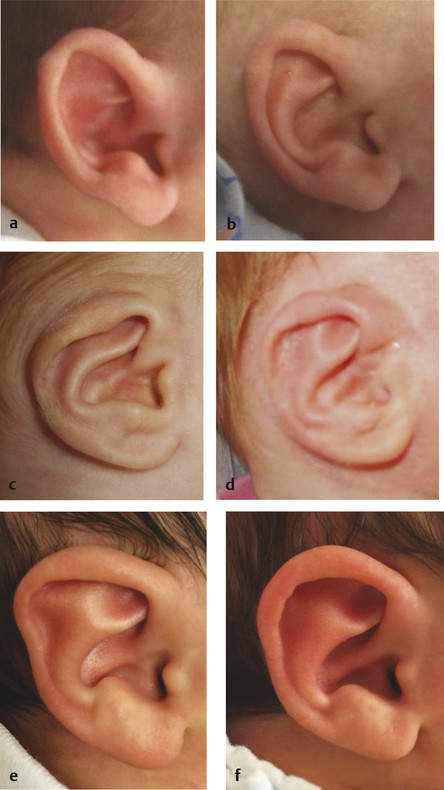
20.4 Operative Treatment
20.4.1 Indications
The primary indication for correction of an ear anomaly is to improve the patient’s self-esteem. Cryptotia and moderate-severe constricted ears can make it difficult to wear eyeglasses. Timing of intervention falls into three categories: (1) 6 to 12 months, (2) 3 to 4 years, (3) late childhood or adolescence. Simple excisions of excess structures can be performed during infancy under local anesthesia. Parents with preauricular cartilage remnants are usually eager to remove them as soon as possible. Most are small and able to be excised under local anesthesia in the office. I prefer that he or she is at least 6 months of age, when their physiology approximates that of an adult, before doing an elective procedure. After 12 months of age, however, it is too difficult to restrain an awake child and I will perform the procedure in the operating room with sedation or wait until the child is old enough to be able to cooperate in the clinic. If a child has an ear deformity that cannot be corrected simply using local anesthesia during infancy, I prefer they be at least 3 years of age before operative intervention under general anesthesia.
Because long-term memory and self-esteem begin to form at approximately 4 years of age, many parents decide to correct deficient or malformed structures when the child is 3 years of age. At this time, the ear has achieved 85% of its size and the risk of scar limiting enlargement of the ear is reduced (ear growth is complete by approximately 6 years of age). Although studies have shown that operating on ears before 3 years of age does not inhibit growth, I prefer to wait until at least 3 years of age because (1) the ear is larger, which facilitates the procedure, and (2) it is not urgent to intervene before this time because memory and self-esteem have not yet formed.
If a child has a severe deformity that he or she will definitely want improved, then it is best to do the procedure prior to 4 years of age, before memory and self-esteem form. Children at this age tolerate the procedure more favorably than older patients who are more anxious about the operation. If an individual has a mild deformity that he or she may not want to have improved, then it is prudent to wait until they are older to determine if they become bothered by it.
Some families choose to wait to correct an ear anomaly until the child is old enough to participate in the decision to have the procedure. Children between 5 and 8 years of age typically are bothered by a deformity, but fear of the operation outweighs their desire to improve their appearance. After 8 years of age, the child’s interest in correcting the anomaly begins to outweigh the anxiety about the operation. Girls typically present later than boys because they are better able to camouflage their deformity with hair. Boys often will grow their hair long to hide their ear.
Except for small excisional procedures in infancy, I prefer to correct other ear anomalies under general anesthesia. Even in adolescents who may tolerate procedures under local anesthesia, they typically have a better experience if they are asleep. General anesthesia also facilitates the procedure for the surgeon and increases the likelihood of achieving the most favorable outcome.
During any ear procedure, I ensure that betadine has been placed into the external auditory meatus. I do not administer a perioperative dose of antibiotic unless I am (1) doing an extensive cartilage dissection or (2) placing permanent intracartilaginous sutures. Although these procedures are clean cases, the benefit of reducing the risk of infection, chondritis, and destruction of the ear after a “cosmetic” operation outweighs the potential disadvantages of the antibiotic. I do not prescribe postoperative antibiotics.
The ears are particularly tolerant of asymmetry because of their lateral position over the temporal bones. The full appearance of the ears cannot be appreciated on frontal view. To compare the ears, an individual must look at one ear first and then change positions to see the contralateral side. Consequently, it is most important to have the ears as symmetrical as possible on frontal view. Because most ear anomalies cause a “cosmetic problem,” patients and families can be focused on minor asymmetries. It is important that families understand that slight asymmetries between the ears are common in the general population and that there will be residual asymmetry following the procedure.
20.5 Types of Abnormalities (Listed in Order of Frequency)
20.5.1 Accessory Cartilage
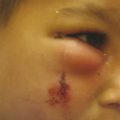
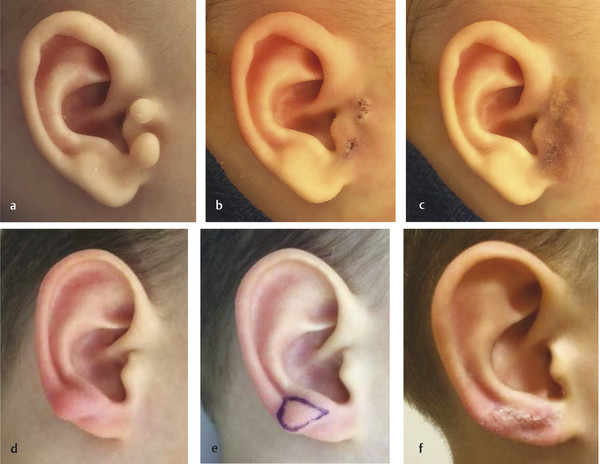
Accessory cartilage in the preauricular area is the most common ear anomaly (Fig. 20‑7). I do not advocate placing a suture around the area to cause necrosis because (1) there will be a residual bump that has a high likelihood of needing a surgical procedure to improve and (2) restraining the child to perform a definitive excision is not much more involved than tying a suture around the lesion. I prefer to remove the accessory cartilage between 6 and 12 months of age under local anesthesia in the office to obviate general anesthesia. Patients who present after 12 months of age are managed with sedation in the operating room because it is too difficult to restrain the child in the clinic. If an infant has a large lesion with a wide cartilage base that cannot be easily excised and closed, the procedure is performed in the operating room. Only dissolving 6–0 or 7–0 chromic sutures are placed followed by cyanoacrylate glue and steri-strips.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree