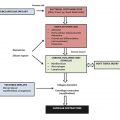
13 Management of Acute Complications of Aesthetic Breast Surgery Part I—Local and Systemic Complications
Summary
This book chapter provides a thorough discussion on the incidence, diagnosis, and management of the most commonly encountered acute complications following aesthetic breast surgery. Local complications discussed include hematoma, seroma, surgical site infection, delayed wound healing, fat necrosis, nipple areolar complex necrosis, and Mondor’s disease (superficial thrombophlebitis of the thoracoepigastric veins). Systemic complications reviewed include venous thromboembolism and pneumothorax.
13.1 Introduction
Aesthetic breast surgery has seen a tremendous growth over the last few decades and currently represents one of the most commonly performed cosmetic surgical interventions in North America. According to the American Society for Aesthetic Plastic Surgery (ASAPS) statistical reports, the annual number of breast cosmetic surgical procedures has increased significantly over the years with procedures performed in 2018 exceeding 670,000. 1 In 5 years, the annual number of breast augmentations increased by 15.2%, mastopexy by 55.9%, reduction mammaplasty in women by 27.6%, and male breast reduction for correction of gynecomastia by 47.9%. 1
Unfortunately, not all breast surgical procedures result in a favorable outcome. When complications occur, they can be dealt with in the office setting, but more severe cases may require emergency room visits, hospital admissions, or even reoperations. More importantly, whether minor or major complications, they can result in a potentially devastating cosmetic outcome and pose significant financial burden on the patient and the surgeon. The cosmetic surgeon can be faced with numerous unfavorable situations following aesthetic breast surgery, and likewise there are many ways to treat these problems. As such, the focus of this chapter is to review the acute complications encountered following aesthetic surgical procedures and discuss their management.
13.2 Local Surgical Complications
13.2.1 Hematoma
Postoperative bleeding and hematoma represent one of the most common complications in aesthetic breast surgery. Several risk factors predisposing toward hematoma formation following these procedures have been previously identified, including age, medical comorbidities such as high blood pressure and malignancy, anticoagulant and antidepressant medications, operative and anesthetic techniques, as well as postoperative management. 2 , 3 , 4 , 5 As a result, postoperative hematoma rates vary. Although the incidence is less than 2% in the majority of the studies on aesthetic breast surgery, some series have reported rates as high as 8%. 5 , 6 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14
Postoperative hematomas typically develop within the first 24 hours after surgery. Less commonly, hematomas can present days, weeks, or even months after surgery and may be associated with minor injury or trauma to the breast, overactivity, or clotting disorders. Hematomas may present with a variety of symptoms and signs, some of which may be nonspecific. Suspicion should arise if there is increasing unilateral pain or tenderness, edema, extensive ecchymosis of the skin, or new volume discrepancy. Other signs include erythema or warmth, especially if complicated by an infectious process, and in some instances hemodynamic changes.
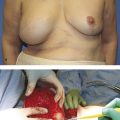
Small hematomas can be observed and usually resolve without intervention. However, larger hematomas should be taken back to the operating room for evacuation under sterile conditions. Reoperation should involve removal of the implant if present, evacuation of the clot, and irrigation of the breast pocket, followed by meticulous hemostasis and identification of any causative vessels if possible, reirrigation of the pocket, implant replacement, and incision closure. Any underlying coagulation abnormality must also be addressed and corrected. Prompt identification and management of these hematomas is crucial, because if left untreated they can result in a variety of negative long-term sequelae. In cases of delayed presentation with a liquefied hematoma, the hematoma may be aspirated by the physician or with imaging guidance. The compressive effect of a sizable fluid collection or clot can cause wound dehiscence, flap compromise, or even necrosis of the nipple areolar complex (NAC). These complications can further lead to exposure of an underlying implant and subsequent implant loss. Poor cosmesis and asymmetries may also become an issue. In addition, periprosthetic hematomas have been associated with increased risk of both subclinical infection and capsular contracture (Fig. 13‑1). 15 , 16 , 17 As our knowledge of capsular contracture risk factors grows, as well as its link to implant-associated anaplastic large cell lymphoma, the plastic surgeon must be aware how hematoma formation plays a role. It has also been shown that submuscular implant placement in breast augmentation has a higher hematoma incidence compared with the subglandular position. 10 If surgeons attempt to mitigate the risk of capsular contracture by choosing to place the implant in the submuscular plane, they must be more vigilant with hemostasis to prevent hematoma formation.
Hematoma risk can be generally minimized with careful pocket dissection using direct vision and, if needed, special lighted retractors, with special attention to hemostasis. Patients should be advised to abstain from certain medications and supplements that may predispose to bleeding for at least a week preoperatively, such as platelet inhibitors, anticoagulants, androgenic hormones, nonsteroidal anti-inflammatory drugs (NSAIDs), and vitamin E. The use of drains in the breast pocket is controversial, with no convincing evidence in the literature to support decreased hematoma recurrence rates. 18 Postoperative breast compression after breast augmentation is still practiced by some surgeons; however, in a randomized study of 130 patients it was not shown to reduce the frequency of postoperative bruising or hematoma formation. 19 In fact, 37.5% of the patients complained about the use of compression. Also, although supportive mild compression with a surgical bra or wrap is used for breast reductions and mastopexies, excessive compression should be avoided, as it can compromise the circulation of the breast flaps and the NAC.
13.2.2 Seroma
Seroma is not frequently observed following primary aesthetic breast procedures, but its reported incidence ranges from 0.2 to 5.5%. 8 , 12 , 13 It is more common for troublesome seromas to occur after revisionary implant surgery, especially after capsulectomy or extensive capsulotomy. 20 Likewise, the use of acellular dermal matrix, mostly in revisionary surgery, has been advocated to increase the incidence of seroma formation, which if not treated may in its turn lead to non-adherence of the acellular dermal matrix. Significant fluid accumulation within the breast implant pocket or breast cavity after mastopexy and reduction mammoplasty may cause breast enlargement and asymmetries, discomfort, pain or tenderness, or wound dehiscence with drainage of clear/yellowish fluid. Occasionally, an undrained seroma may cause ongoing cellulitis in the absence of infection, and has to be excluded.
When an early seroma is detected, percutaneous aspiration should be considered if it is palpable or ballotable. This can be done with ultrasound guidance. If an implant is present, extreme care must be taken not to puncture it during aspiration. A safe approach involves positioning the patient in the lateral decubitus position and performing the aspiration through the skin of the lateral aspect of the breast at the most dependent aspect of the fluid collection, while manually protecting the implant by displacing it medially. 20 A sample of the drained fluid should be sent to the microbiology laboratory for culture if an infection is suspected, as well as for histology in the case of a late seroma if anaplastic large cell lymphoma is suspected. At times, a short-term catheter may be left in place for continuous drainage. If repeated aspirations do not effectively resolve the seroma, open drainage in the operating room under sterile conditions may be required. If surgical exploration reveals a seroma capsule, then the surgeon should perform complete capsulectomy, implant removal (with possible placement of a new implant), and possible drain placement.
One should be aware that different surgical approaches might lead to different outcomes. For example, Hall-Findlay and Shestak suggested that seromas after breast reduction are less likely to resolve spontaneously with the inverted-T skin resection pattern than with the vertical pattern because the inframammary scar can block drainage. 21 Significant drainage may also result from increased movement of the implant, due either to an excessively large pocket or, more likely, to too much activity by the patient. The patient should be cautioned to reduce activity if seroma develops while a drain is still in place. It is important to remember that undrained seromas can have other untoward consequences such as implant rotation, malposition, and infection. To our knowledge, the current literature does not identify prognostic factors associated with conversion of simple seroma to infected seroma and possible implant loss. Furthermore, if a recurrent seroma is not regularly drained, it can sometimes form a hardened mass of scar tissue.
There is no strong evidence in the literature to support the placement of drains to reduce the occurrence of seromas after primary aesthetic breast procedures. In the United States, common practice is not to leave a drain in the breast pocket after breast augmentations or augmentation mastopexies. Drain placement after mastopexies and breast reductions is less standardized and comes down to surgeon’s preference. Other authors prefer to leave a drain after significant liposuction to the breast or lateral axillary roll has been performed, as this appears to contribute to seroma formation. 20 In the authors’ practice, they are more likely to leave a drain after revisionary implant surgery that involves substantial modification of the capsule, such as capsulectomy or extensive capsulotomy. Typically, the authors allow removal of the drain catheter when drainage is less than 20 mL over a 24-hour period for 2 consecutive days. The timing of drain removal is critical, but can become tricky if there is persistent drainage. If the drain is removed too early, there is a risk of fluid reaccumulation, but if it is left in place for a prolonged period of time, there is a risk of introducing infection into the breast pocket.
13.2.3 Surgical Site Infection
Surgical site infection (SSI) is also one of the most frequently encountered complications following aesthetic breast surgery. Published rates of SSI have been shown to be lowest in breast augmentation, ranging from 0.001% to 7%, and highest in breast reduction, ranging from 0.11% to 24.5%. 5 , 10 , 12 , 16 , 22 , 23 , 24 , 25 , 26 , 27 , 28 , 29 , 30 Although minor SSIs might be common, the incidence of major SSIs that require emergency room visit, admission to the hospital, or reoperation is very low and less than 1% in two of the largest studies analyzing the CosmetAssure database over different time intervals. 5 , 22 Hanemann et al reviewed 51,235 cosmetic breast surgery procedures between 2003 and 2009, and revealed a major SSI rate of 0.22%. 22 Similarly, Gupta et al found an overall major infection rate of 0.25% in over 73,608 aesthetic breast procedures. 5 This was broken down to an infection risk for breast augmentation of 0.19%, augmentation–mastopexy of 0.43%, mastopexy of 0.32%, and breast reduction of 0.55%. Another significant study by Fisher et al analyzed the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database for complications following reduction mammoplasty. 31 This study included 3,538 patients and found an overall postoperative complication rate of 5.1%, and specifically an SSI rate of 3.0% (superficial infection: 2.7%, deep infection: 0.2%, organ space infection: 0.1%). As advocated by prior studies, the impact of surgical technique may play an important role on infection rates including meticulous hemostasis and dissection, avoiding the use of drains, use of antibiotic/antimicrobial irrigations, careful handling of the implants, and avoidance of external exposure of the implant to the air and patient skin. 10 , 32 Several other factors play a role in influencing postoperative SSIs after cosmetic breast procedures, and it is not surprising that many of these have a synergistic effect. These include age, increased body mass index, smoking, diabetes, steroids and other immunosuppressive medications, pregnancy, vigorous exercise, prolonged operative times, and combining procedures such as augmentation with mastopexy. 5 , 29 , 30 , 33 , 34 , 35 , 36 , 37 , 38
The majority of SSIs manifest within the first month after surgery, but there is usually a lag of at least 5 days and up to 2 weeks postoperatively before the SSI appears. 39 Some infections, especially related to implants, can occur months or even years after the initial operation. 16 , 36 , 40 In rare occasions, SSIs manifest within the first 48 hours after surgery and are almost always due to Clostridium species or Streptococcus pyogenes. These infections can progress very rapidly if not addressed in a timely fashion and can have devastating consequences. For such necrotizing infections, serial aggressive surgical débridements may be required. Postoperative toxic shock syndrome caused by Staphylococcus aureus wound infection can also be observed within the first 48 hours after surgery. 41 In these cases, the incision appearance can be deceptively benign on examination, and diagnosis is based upon fulfillment of the following six criteria: fever, rash, desquamation, hypotension, multisystem organ involvement, and negative culture results and serologies. 39 , 42
Local signs of swelling, pain, erythema, and purulent drainage are usually reliable in diagnosing an SSI. However, in morbidly obese patients or those with deep, multilayer wounds, external signs of SSI may be delayed. 43 Other adjunct tests, such as laboratory or imaging studies (such as ultrasound), can assist with the diagnosis. Certainly, any fluid collections around a breast implant should be drained and cultured, whenever possible. The causative organisms may be different, depending on the type of surgical procedure, but are mostly bacteria. For example, the most frequently isolated bacteria following breast augmentations include S. aureus, S. epidermidis, streptococci A and B, Klebsiella pneumoniae, Bacillus, and Propionibacterium, whereas Corynebacterium species, Propionibacterium acnes, Pseudomonas aeruginosa, Escherichia coli, and Enterobacteriaceae are less common. 36 , 44 , 45 , 46 , 47 Sporadic cases of mycobacteria have been described and should be suspected for late infections with an indolent course when common techniques fail to isolate a cause. 14 , 46 , 48 , 49 , 50 Acid-fast stains and cultures should be obtained in all cases of subacute infection, even when routine culture reveals skin flora. 51 It is not unusual to observe some erythematous skin changes around or near the surgical incision during the first week after surgery without the other symptoms and signs of infection. Most of these changes resolve without any treatment and may be related to tape sensitivity or other local tissue insult not involving bacteria. In fact, previous data suggest that antibiotics begun immediately postoperatively or continued for long periods after the procedure do not prevent or cure this inflammation or infection. 52 , 53 , 54 , 55 Special attention should be paid among individuals who underwent implantation of acellular dermal matrix (ADM) during revisionary breast surgery, as erythema of the breast within the first weeks postoperatively could also reflect “red breast syndrome.” 56 , 57 This is self-limited and believed to be related to an immune response. Unlike an infection, it is not associated with significant leukocytosis or systemic signs of infection, and the erythema only overlies the ADM. 57
Management of breast infections in the setting of implants is slightly different compared to infections without implants, and it will thus be discussed separately. In patients without implants, simple cellulitis from a suspected infectious etiology can be managed with an oral course of antibiotics. If a deeper infection is suspected, typically the first and most important step in the management is incision and drainage. The incision should be opened, the infected material should be evacuated, the cavity should be thoroughly washed out, and appropriate dressing changes should be initiated to promote wound healing by secondary intention. Incision and drainage of superficial abscesses rarely cause bacteremia; hence prophylactic antibiotics are not necessary. 58 Making the incision too small may fail to control the infection. It is critical to open the incision adequately, not only to control the infection but also to diagnose and treat associated problems such as necrosis of the subcutaneous tissue and fascia that require debridement. However, there is a fine balance between effective debridement and unnecessary overexcision; this balance is particularly important in the aesthetic surgery population, where cosmetic outcomes are crucial to the success of the operation. The latest practice guidelines for the management of skin and soft tissue infections published by the Infectious Diseases Society of America (IDSA) states that adjunctive systemic antimicrobial therapy is not routinely indicated but, in conjunction with incision and drainage, may be beneficial for SSIs associated with a systemic response such as erythema and induration extending > 5 cm from the wound edge, temperature > 38.5°C, heart rate > 110 beats/min, or white blood cell count >12 000/µL. 43 The antibiotic choice is usually empiric but can be supported by Gram stain, culture of the wound contents, and the site of surgery.
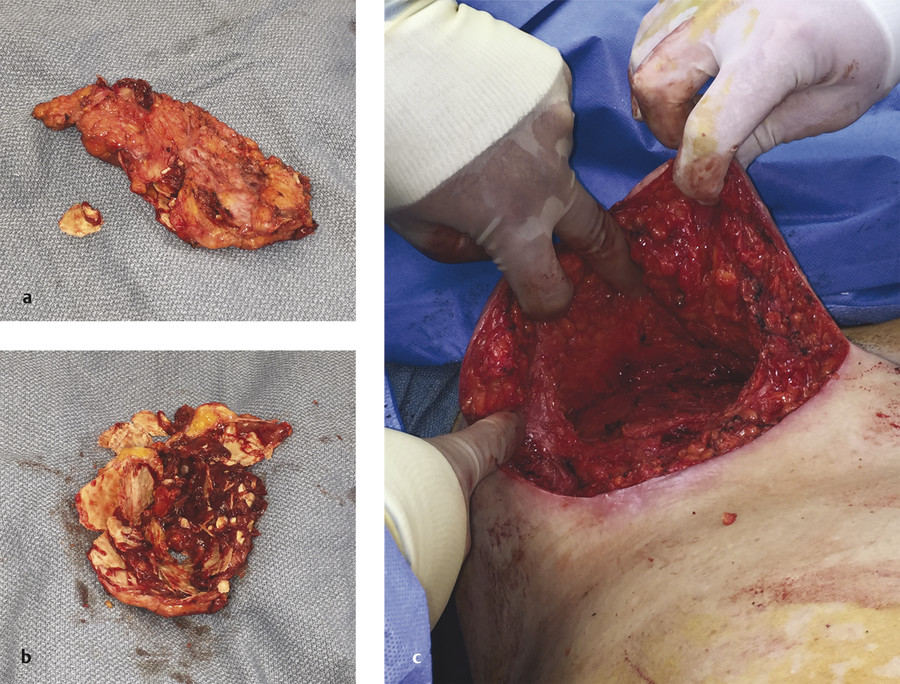
When implants are present, infections tend to occur late (months and occasionally years after surgery) and are often associated with limited clinical manifestation. 59 A high level of suspicion is thus required. Management remains challenging, and it is still not clearly defined given the absence of high-level evidence in the literature. In the past, immediate removal of infected prostheses was standard practice. However, nowadays it is not uncommon for the patient and the surgeon to be unwilling to accept the loss of the implant, with its associated physical and financial consequences, until they are convinced that an attempt at medical therapy has failed. As a result, plastic surgeons have explored other options in an attempt to salvage these devices, such as systemic antibiotics combined with either conservative wound drainage, antibiotic lavage, capsulotomy with implant exchange, and capsule curettage with implant exchange. 60 , 61 , 62 , 63 , 64 , 65 In 2004, Spear et al proposed treatment guidelines for breast implant salvage in the setting of infection with and without implant exposure (Fig. 13‑2). 62 Patients with mild infections can be started immediately on oral antibiotics, whereas patients with severe infections should be started on parenteral antibiotics. Those patients who respond completely require no further treatment. However, surgical intervention is recommended for persistent infections or threatened or actual exposure. In these cases, drainage of the fluid collection is indicated. This is followed by partial or total capsulectomy, debridement, pocket lavage, site change, implant exchange, and possible local tissue and/or flap coverage, which is chosen based on the surgeon’s experience and clinical judgment. Salvage attempts for implant infection and exposure depend on the degree of infection, response of the infection to initial antibiotic therapy, and availability of adequate soft tissue coverage. In addition, if aggressive salvage measures must be taken, both the patient and the surgeon should be prepared to go through this process together. In some occasions, implant salvage is contraindicated. Examples include identification of atypical pathogens on wound culture (such as nontuberculous mycobacteria), fungal infections, and persistence of systemic signs of sepsis despite appropriate antibiotic therapy. 51 , 63 In these cases, timing of implant replacement depends on the degree of infection and the causative organisms. 16 Some experts recommend waiting 3 to 6 months before reimplantation, but there is no good evidence to support this approach.
Over the years, a variety of perioperative prevention measures have been proposed to assist in reducing the incidence of SSIs. Although aesthetic breast procedures are not required to follow the Surgical Care Improvement Project (SCIP) guidelines, the prophylactic use of antibiotics by board-certified plastic surgeons, as shown by prior surveys, has steadily increased over the last few decades. 66 , 67 , 68 In 2015, the American Association of Plastic Surgeons selected experts to evaluate the evidence for efficacy and safety of antibiotic prophylaxis in plastic surgery procedures, and consensus recommendations were published. 69 Meta-analysis of randomized controlled trials has shown that routine antibiotic prophylaxis for patients undergoing clean cosmetic breast surgery results in a significant reduction in the risk of SSIs (2.5% versus 11.4%; odds ratio 0.16; 95% confidence interval 0.04–0.61; p = 0.01). Further subgroup analyses of patients undergoing breast reduction and breast augmentation with implants suggested that both groups benefit from antibiotic prophylaxis, but it was not possible to determine the best antibiotic and optimal dose from the available data. Irrigation of a surgical pocket at the time of implant placement is commonly performed by most plastic surgeons despite the lack of high-level evidence that this technique is actually effective in preventing SSIs. There are, however, a few retrospective and limited prospective cohort studies that demonstrated lower rates of SSIs following irrigation of the implant pocket with a combination of antibiotics (e.g., cefazolin, gentamicin, and bacitracin) and antiseptics (povidone-iodine). 70 , 71 , 72 Finally, several regimens have been tested for their decolonization efficacy to prevent methicillin-resistant Staphylococcus aureus (MRSA) infections, including chlorhexidine body washes, povidone-iodine body washes, mupirocin nasal ointment, and oral antibiotics. 73 , 74 Of those, prophylactic use of intranasal mupirocin was shown to reduce the rate of postoperative S. aureus infections significantly among carriers, 75 but it did not provide long-term MRSA control, and it did not prevent recolonization or subsequent infections. 76 , 77 Therefore, since the optimal decolonization regimen is unclear at this point in time, it should be reserved for patients who are colonized with MRSA or are known to have had a MRSA infection in the past.
13.2.4 Delayed Wound Healing
Delayed wound healing, although not frequently seen after mastopexy, it is the most common complication after breast reduction with reported incidences ranging from less than 1% to approximately 65%. 12 , 13 , 78 , 79 , 80 Perhaps one of the main reasons for this wide variation in the range of reported incidence is the different inclusion criteria for this complication among different studies. The majority of delayed wound healing complications are minor and can be managed in the outpatient setting with local wound care. However, a small proportion of these cases has extensive tissue involvement and may require reoperation to debride the devitalized tissue and create a more favorable environment for healing by secondary intention. If an unsightly scar develops, it can be revised at a later date. Another reason for the wide variation in the range of reported incidence of delayed wound healing and skin necrosis following reduction mammaplasty and mastopexy is the broad variety of techniques used. The Wise pattern of skin resection compromises the skin flaps to a greater degree than a vertical or periareolar reduction does. 81 Tension on the skin flaps at the time of the closure can lead to ischemia at the areas that are most remote from the blood supply; this includes the distal skin flaps at the central lower breast and at the point of convergence of the vertical and horizontal suture lines, commonly referred to as the inverted T point. 81 Even for the vertical reduction pattern, wound healing problems commonly occur where the NAC meets the vertical limb. This becomes more of a concern with augmentation mastopexy patients because of the risk of implant exposure. In these patients, a thorough assessment of the wound will determine imminent exposure of the implant, which may change the approach to the management and may prompt earlier implant removal and plan for delayed replacement, if at all. The incidence of these complications, as shown by several studies, is also affected by other factors such as age, obesity, cigarette smoking, and increased resection weights. 13 , 21 , 78 , 81 , 82 , 83
Wound healing problems and skin flap necrosis following breast reduction and mastectomy are vexing complications to both the patient and the surgeon; thus, minimizing their occurrence is in the best interest of both. Ways to achieve this include preservation of the subdermal plexus, gentle retraction of the skin flaps, and minimization of thermal damage. 84 This is especially important in all revisional or reoperative breast surgery. Inherently, when a mastopexy is done in conjunction with a revision breast augmentation, the increased pressure on the flaps by the implant can lead to higher rates of skin necrosis and delayed wound healing. Techniques that involve wide skin undermining should be avoided in these cases, especially if the original augmentation was in the subglandular plane. 20
Following breast augmentation, wound healing problems are rare. When wound dehiscence occurs in the early postoperative period, it is usually due to a technical error related to closure or an inappropriately selected heavy implant (Fig. 13‑3). In these cases, reoperation should be considered to salvage the implant by irrigating the pocket and reclosing the wound. During closure of the incision, particularly with the inframammary approach, the superficial fascial system of the breast should be identified and carefully approximated, not only to provide soft tissue coverage of the inferior aspect of the implant but also to prevent transmission of the weight of the implant directly to the incision. 20 If dehiscence occurs after the first postoperative week, an infectious process should be suspected. As previously discussed, management of these cases remains challenging. Implant removal is frequently required; however, in recent years several options to salvage these devices have been proposed by several groups. 60 , 61 , 62 , 63 , 64 , 65 , 85
13.2.5 Fat Necrosis
Fat necrosis is predominantly seen after reduction mammaplasty but can occasionally be seen after mastopexy. Although not very common, it can lead to anxiety for the patient and the surgeon. It results from vascular compromise of the adipose tissue of the breast parenchyma or from ischemic necrosis. For example, when large suture bites are used to approximate the breast parenchyma under tension, the perfusion of the adipose tissue can be disrupted, leading to fat necrosis. Likewise, the blood flow to the fatty component of the breast parenchyma may not be as robust at the distal end of the chosen pedicle, leading to necrosis.
In the vast majority of patients, fat necrosis presents as a breast lump. A firm, discrete, mobile breast mass can be found on physical examination. Occasionally, yellow drainage may be expressed from the incision in the early postoperative period without other signs of infection. Associated skin changes are not usually present. Examination can resemble other benign or even malignant breast pathology; therefore, the physician should take such findings seriously. Imaging studies, such as mammography and ultrasound, may be obtained to evaluate the lesion and establish a baseline for future follow-up. If any concerning features are noted on imaging, the lesion must be excised. Excision can also be done if it is bothersome to the patient. Resection can be accomplished through a pre-existing scar. Otherwise, such a mass can be observed for at least a year, since a fair number of these lesions will become softer or even resolve with time and massaging.
13.2.6 NAC Necrosis
Partial or total necrosis of the NAC represents one of the most feared complications following breast reduction and mastopexy, especially after revisionary cases. It is not only disappointing for patients but can also be discouraging for the surgeons. Its incidence ranges from 0 to 10%. 12 , 78 , 86 , 87 , 88 The spectrum of NAC ischemia spans from spontaneous, completely reversible nipple congestion to total loss of the nipple with extensive necrosis of subadjacent breast tissue. 89 Vascular compromise of the NAC can be caused by arterial insufficiency but is more commonly due to venous congestion. 88 Understanding the vascular anatomy of the breast is thus critical not only to preserving the arterial inflow but also to maintaining an adequate venous drainage system for the NAC. As demonstrated from previous studies, the most reliable blood supply to the NAC comes from the inferomedial aspect of the breast via the internal thoracic–anterior intercostal system, and less so from the superolateral aspect of the breast via the lateral thoracic artery and other minor branches. 88 The most reliable pattern of venous drainage, as shown with breast venograms, is located in the superomedial, medial, and inferior pedicles. 90
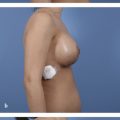
Arterial compromise of the NAC manifests with delayed capillary refill, dark discoloration, and pale nipple that is cool to touch (Fig. 13‑4). On the other hand, signs of venous congestion include excessively brisk capillary refill and dark, rapid bleeding on pinprick, as well as cyanotic and edematous nipple. Causes of venous congestion include inadequate preservation of venous drainage, long pedicles, kinking or compression of the pedicle, excessively tight skin closure, and hematoma. 89 In addition, other factors have been shown to increase the risk of NAC ischemia, such as obesity, cigarette smoking, diabetes, large-volume tissue removal, and long distance of transposition of the NAC (more than 15 cm). 91 Patients who have had breast augmentation previously and wish to undergo secondary mastopexy are also at increased risk of ischemic complications. 92 Breast implants can cause tissue stretching and thinning that primarily affect the inferior pole of the breast, which has to be taken into consideration when selecting a pedicle for the secondary mastopexy. For instance, choice of an inferior pedicle will be more prone to vascular compromise because of thinning of the soft tissue of the inferior pole. For this patient population, it is wise to select the least aggressive mastopexy that will achieve the desired result. It is also important to avoid insertion of excessively large implants, which can lead to undue pressure on the tissues and possible ischemic changes.
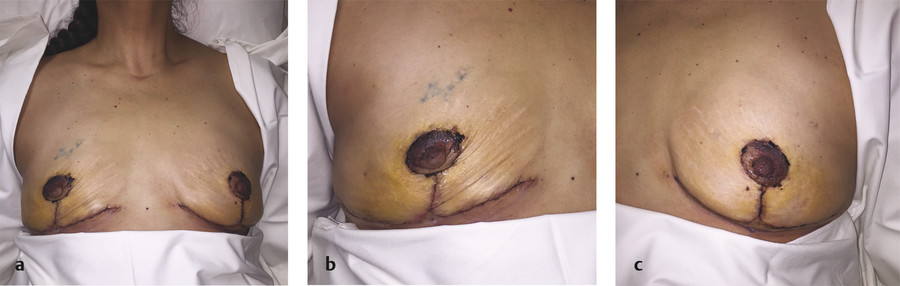
Meticulous preoperative planning is also critical for patients undergoing revision breast reduction or mastopexy. Previous patient records, including the previous surgeon’s operative report, should be obtained to determine which technique and pedicle were utilized. Special attention should be paid to the effect of the prior skin incisions on the blood supply of the nipple and the skin flaps. The safest approach during revision surgery is to reuse the pedicle that was used in the previous surgery. However, often surgeons do not have access to the previous operative report and do not have information regarding the previous pedicle used. In these cases, safe options include the use of bipedicled techniques and breast reduction by liposuction alone with skin-only mastopexy. 20 Techniques that involve wide undermining of skin flaps, especially in situations where the integrity of the vascular supply is unclear, should be avoided.
The viability of the nipple and areola should be evaluated frequently intraoperatively. Generally, the decision is made clinically, but if there is uncertainty, other techniques can be used. such as intravenous fluorescein and indocyanine green videofluorography. 93 , 94 The advantages of the latter method include repeated use during the same operation and simultaneous evaluation of both the arterial microcirculation and venous drainage. If it becomes obvious that the NAC is nonviable, it is advisable to convert it from a pedicle flap to a full-thickness graft. Fundamentally, the free nipple graft should be affixed to a healthy dermal bed with good blood supply, but not directly onto breast tissue or fat. Even though free nipple grafts have a higher incidence of projection loss and depigmentation, these consequences are preferable to frank necrosis. 20 If viability is questionable, either intraoperatively or immediately postoperatively, a few simple maneuvers can be attempted to improve the perfusion. Releasing the dermal and subdermal sutures around the periphery of the areola can improve the venous outflow significantly within minutes. 89 Application of vasodilating ointments to the area may also help. In our experience, Venelex ointment (balsam Peru and castor oil; Stratus Pharmaceuticals, Miami, FL), applied 5–6 times per day, offers promising results. Steroids such as methylprednisolone (Medrol Dosepak, Pfizer, New York, NY) have also been advocated to reduce local tissue edema and promote venous outflow. 89 Other methods that are not commonly used include leeches and hyperbaric oxygen therapy. 95 , 96 Although ischemia of the nipple and areola are a source of great angst for both the patient and the surgeon, it is not easy initially to gauge how much tissue will ultimately survive; thus, patience from both parties is required, and aggressive treatment should be avoided until the tissues demarcate.
Depending on the size of the necrotic area, one could either allow the nonviable tissue to slough or debride it. The remaining defect will then be managed with local wound care to promote healing by secondary intention.
NAC reconstruction should be considered, but it is advisable to delay the process for 3 to 6 months in order to allow for resolution of inflammation and improvement in local circulation as well as maturation and softening of the scar tissue. 89 Several reconstructive options are available. The choice is usually made based on the nature of the defect and the preference of the surgeon. For partial loss of the nipple or the areola, the contralateral NAC is the ideal donor site for a natural-appearing result. A full-thickness skin graft can be used for areola reconstruction and a composite nipple graft for nipple reconstruction, assuming there is enough tissue for sharing. If the damage is more extensive, a type of local flap is elevated and rotated or folded to create a nipple of desirable size and shape. Full-thickness skin graft for areolar reconstruction can also be obtained from other body regions, such as the upper inner thighs and labia minora. It is very common for the grafted areola to lose pigmentation over time, leaving behind a circular scar around the nipple. Intradermal tattooing is a simple method to restore that pigmentation. 97 , 98 More recently, three-dimensional tattooing has gained popularity. Prior authors have also advocated tattooing the intact contralateral NAC to provide the best possible color match between the two sides. 89
13.2.7 Mondor’s Disease
Mondor’s disease, or thrombophlebitis of the thoracoepigastric system of veins, is an unusual condition that can be seen after breast augmentation. It has also been reported following breast reduction. 99 The literature for aesthetic surgery is scarce, but it is probably more prevalent than is commonly thought. In the largest series reported to date, there were 13 cases of Mondor’s disease, involving 11 patients, in 2,052 breasts (0.63%), for an incidence of 1.07% in 1,026 cases of breast augmentation. 100 It is believed to occur more commonly after a transverse inframammary incision, which results in division of the vertically oriented superficial veins. As a result, retrograde blood flow is prevented because of the presence of valves in these superficial veins, causing venous stasis followed by thrombosis. Physicians should be familiar with several other etiologies linked with this diagnosis, including breast cancer, hypercoagulability disorders, connective tissue diseases, and extensive physical activity as well as external compression by tight-fitting dresses and tight bras. 101 , 102
The disease presents as a firm or painful cord on the thoracoabdominal wall and can be visible when the arms are abducted. 100 Typically, it appears within the first 6 weeks after surgery, but it can occur even months later. 100 , 102 It generally resolves after a short period of time since the thrombus becomes organized, and the veins eventually recanalize after a few weeks. Treatment includes reassurance and supportive care with warm compresses and NSAIDs. While anticoagulation has been proposed by some authors in the context of concomitant prothrombotic risk factors, there is no robust evidence to support its use at this point in time. 100 , 101
13.3 Systemic Complications
13.3.1 Venous Thromboembolism
Venous thromboembolism, defined as either deep venous thrombosis (DVT) or pulmonary embolus (PE), is an important patient safety issue among aesthetic surgery patients and undeniably represents one of the most feared complications in cosmetic surgery. In a small percentage of patients, PE can be rapidly fatal or lead to debilitating chronic cardiac dysfunction. 103 , 104 DVT can also damage the vein wall and the venous valves, predisposing patients to chronic venous reflux and the postthrombotic syndrome. 105 Although the incidence of venous thromboembolism in aesthetic breast surgery is reported to be less than 1%, this rate is believed to be underestimated. 5 , 13 , 80 , 87 , 104 , 106 , 107 , 108
As previously advocated by many, including the Joint Commission and the American College of Chest Physicians, venous thromboembolism prevention is the most effective strategy to minimize its associate morbidity and mortality. 109 , 110 , 111 , 112 , 113 Individualized risk stratification by using the patient’s personal characteristics (e.g., age, body mass index, hypercoagulable state) to conceptualize and quantify her venous thromboembolism risk has been proposed. As a result, several individualized risk stratification tools have been developed, but the 2005 Caprini score is currently the most widely used and well-validated among various surgical populations. 114 , 115 , 116 , 117 , 118 , 119 , 120 , 121 , 122 It contains 40 risk factors, some of which can be potentially modified before elective surgery, such as obesity, use of estrogen-based contraceptives, and history of prior major surgery within 1 month. Of note, the 2005 Caprini score has also been compared with the modified 2010 Caprini score and was found to provide superior risk stratification for adult plastic surgery patients. 123 Although the 2005 Caprini score has not previously been validated among the outpatient surgery population, theoretically there should be no reason why it would predict risk differently between these two populations. This is important for the aesthetic breast procedures, since they are typically performed in ambulatory surgery centers or outpatient surgical suites. Certainly, there are high-risk patients within this patient population who would benefit from individualized risk stratification to better conceptualize their venous thromboembolism risk and enable surgeons to propose risk modification and risk reduction strategies. Numerous other factors may affect the risk of venous thromboembolism. For instance, Kim et al examined more than 1.4 million cases from the ACS-NSQIP database and showed a clear relation between increased operative time and venous thromboembolism risk. 110 Likewise, other studies have suggested that general anesthesia, and particularly paralysis, may predispose patients to lower-extremity venous stasis by eliminating the positive action of the calf muscle pump. 124 , 125 , 126 , 127
The existing plastic surgery literature recommends the use of mechanical devices and chemoprophylaxis for high-risk patients, as stratified by the 2005 Caprini score. 128 , 129 A meta-analysis comparing sequential compression devices (SCDs) and elastic compression stockings has demonstrated superiority of the sequential compression stockings for venous thromboembolism risk reduction. 130 To further support this finding, SCDs are believed to affect two limbs of Virchow’s triad (stasis and hypercoagulability) by re-creating the calf muscle pump and stimulating the body’s endogenous fibrinolytic system, as opposed to elastic compression stockings, which affect only one limb of the triad (stasis) by shunting blood from the superficial to the deep system. 105 It is for these reasons that the latest American Association of Plastic Surgeons guidelines favor the use of SCDs for all patients. 128 The same guidelines did not support the use of chemoprophylaxis in the overall plastic surgery population. Instead, chemoprophylaxis was recommended based on risk stratification for perioperative venous thromboembolism using the 2005 Caprini score. Specifically, chemoprophylaxis should be considered for patients with Caprini score greater than 8. In addition, the American Society of Plastic Surgeons recommendations state that chemoprophylaxis should be strongly considered for patients with 2005 Caprini scores of 7 or greater. 131 Based on these data, it is felt that chemoprophylaxis should be provided for patients with a 2005 Caprini score 7 or greater. Unfortunately, there is no strong data with regard to the optimal duration of chemoprophylaxis, especially for outpatient surgeries. In certain occasions, such as in patients with a recognized hypercoagulable state not clearly characterized by the 2005 Caprini score, a preoperative hematology consultation may be extremely valuable to assist with hypercoagulability workups, as well as perioperative venous thromboembolism risk modification and chemoprophylaxis. 132 Although the apparent risk of bleeding is a concern for plastic surgeons, several studies have shown increased rates of reoperative hematomas to be less than 1% after use of chemoprophylaxis, with only a minority of studies demonstrating statistically significant differences. 128 , 133 , 134
Diagnosis of DVT is an ongoing clinical challenge. Accurate and timely diagnosis in patients with suspected DVT is critical because, if left untreated, thrombi can break off and travel into the pulmonary circulation, resulting in detrimental PE, whereas anticoagulation in the absence of thrombosis is medically inappropriate. In fact, previous studies have shown that more than 40% of patients with DVT have asymptomatic PE on imaging, and the majority of patients with a fatal PE have a detectable DVT on autopsy, mostly asymptomatic before death. 135 , 136 , 137 DVT classically presents with leg pain and edema. The pain can be worse with dorsiflexion of the foot (Homans’s sign). Occasionally, warmth and erythema of the skin over the area of thrombosis are noted. PE presentation varies from nonspecific fatigue to gradually progressive dyspnea to sudden onset of pleuritic chest pain, cough, hemoptysis, hypoxia, or even cardiopulmonary collapse. Unfortunately, the majority of patients with a pulmonary embolus do not have a typical presentation, but rather nagging symptoms, which makes diagnosis more challenging. This is why a high index of suspicion is required, particularly when a patient has predisposing risk factors for the condition. Routine laboratory tests are not very useful diagnostically but may provide clues as to the underlying cause and may also influence treatment decisions if a thrombus is detected. D-dimer has been consistently demonstrated to have high sensitivity but poor specificity in postoperative patients with acute DVT. 137 , 138 , 139 , 140 , 141 Although it is an adjunct test for ruling out DVT, especially in those with a low to moderate suspicion of thrombosis, it can be elevated in many other conditions, which makes it a nonspecific test. Compression ultrasonography with Doppler is currently the imaging modality of choice for diagnosis of DVT given its high sensitivity and specificity. 142 , 143 Contrast-enhanced computed tomographic (CT) venography and magnetic resonance (MR) venography are rarely used unless there is uncertainty about iliac vein or inferior vena cava thrombosis after ultrasonography. These diagnostic tests are considered less well validated, have associated complications of radiation and contrast exposure, and are costlier. 144 For PE, the diagnostic study of choice is CT pulmonary angiography. Ventilation-perfusion (V/Q) lung scan is reserved for patients in whom CT is unavailable or contraindicated (e.g., history of moderate or severe contrast allergy, high risk of contrast nephropathy, difficulty lying flat). Other imaging modalities that are less commonly used include MR pulmonary angiogram and catheter-based pulmonary angiography.
Treatment of venous thromboembolism should be individualized, and numerous factors have to be taken into consideration. The primary objectives are to prevent propagation of the thrombus and recurrence, to reduce morbidity, and to minimize the risk of complications. For many decades, the mainstay of treatment has been anticoagulation. However, especially in surgical patients, the risk of bleeding from anticoagulation should be carefully balanced against the risk of recurrent thrombosis. Placement of an inferior vena cava filter, pharmacologic thrombolysis, and catheter-assisted or surgical thromboembolectomy may also be considered in some circumstances.
13.3.2 Pneumothorax
Pneumothorax is a rare complication after aesthetic breast surgery with an incidence of less than 1%, but it appears to be more common than it is generally thought following augmentation mammaplasty. 78 , 145 In most cases, it is difficult to determine the exact etiology, but various mechanisms have been proposed. They can be related to iatrogenic injury by the surgeon or the anesthesiologist, or even pre-existing pulmonary pathology. Examples include accidental needle penetration of the pleura during local anesthetic infiltration, direct damage or thermal damage to the pleura during dissection with electrocautery, rupture of a pre-existing bleb, raised air pressure within the surgical pocket during implant insertion, high-pressure ventilation or intubation maneuvers, a rush of air created while changing oxygen tanks during surgery, and a plugged exit valve. 145 , 146
A pneumothorax can present immediately, hours, or even days after surgery. Presentation may range from completely asymptomatic to cardiopulmonary collapse. Typically, symptoms include pleuritic chest pain (with possible radiation to the ipsilateral shoulder and back), shortness of breath, diaphoresis, and anxiety. There have been reports of bilateral tension pneumothoraxes after breast augmentation, which represent a medical emergency. 147 Patients will present with severe dyspnea, cyanosis, tachypnea, tachycardia, and hypoxia. Pneumothorax can lead to decreased cardiac output as a result of compromised venous return due to compression of the vena cava and right atrium, which may potentially be life-threatening. Physical examination may reveal asymmetric lung expansion and distant or absent breath sounds on the affected side.
If the intercostal fascia and pleura are traumatized during the pocket dissection and the small defect is noted intraoperatively, this should be communicated with the anesthesiologist and managed immediately. The defect should be closed using a purse-string or interrupted suture over a catheter, if possible. The catheter is withdrawn during deep inspiration delivered by positive pressure ventilation or with a Valsalva maneuver if performed under intravenous sedation and local anesthesia. 145 If the parietal pleura and intercostal fascia cannot be reapproximated, other methods can be utilized to close the defect. These techniques would also be used in conjunction with the placement and removal of a catheter as just described. These other techniques include using a patch of breast implant capsule from another part of the posterior capsule, a de-epithelialized skin graft, or a biologic matrix patch, and covering with fibrin glue or similar material to create a seal. A postoperative chest radiograph is always obtained to ensure that there is no residual pneumothorax. If the patient is discharged home, the family or caregiver are instructed to observe for symptoms and signs of pneumothorax and have a low threshold to call or return to the emergency department.
Postoperatively, when pneumothorax is suspected, chest radiography will not only allow for confirmation of the diagnosis, but it will also act as a baseline study, determine the extent of the pneumothorax and potential causes, and assist with the treatment plan. Occasionally, a CT scan is needed to diagnose an occult pneumothorax. Tension pneumothorax is primarily a clinical diagnosis based on patient’s presentation, and obtaining imaging may delay treatment, resulting in a detrimental outcome. In these cases, needle decompression should be performed, followed by tube thoracostomy. In general, the decision to observe or to treat with an immediate intervention (such as aspiration, small-bore catheter, or chest tube placement) should be guided by a risk stratification that takes into account the patient’s presentation and the likelihood of spontaneous resolution and recurrence. 148
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree