1 The History of the Use of Perforator Flaps for Microsurgical Breast Reconstruction
Sarah E. Appleton and Steven F. Morris
The quest to perfect postmastectomy breast reconstruction has been accelerating for the past few decades. Results have steadily improved, and expectations have continued to mount as surgical techniques for autologous breast reconstruction have evolved. Although plastic surgeons have been performing surgery to improve breast aesthetics for centuries, postmastectomy breast reconstruction did not become widely accepted until the mid-1900s. Today, breast reconstruction is an essential part of the overall treatment plan and recovery of the patient with breast cancer. The psychological benefits of postmastectomy breast reconstruction are improved overall satisfaction, body image, self-esteem, and quality of life, as well as decreased anxiety and depression.1,2 Thus, breast reconstruction is popular among patients. In the United States, support for breast reconstruction was written into law in 1998 when President Bill Clinton signed the federal Breast Cancer Reconstruction Law as part of the Women’s Health and Cancer Rights Act that requires group health plans and individual insurance policies to cover the costs of reconstructive breast surgery.3,4
Plastic surgeons now have a large variety of alloplastic and autologous breast reconstruction options to perform breast reconstruction.5 Improved understanding of flap physiology and anatomy has yielded an increasing number of flap options. Advances in the field of microsurgery have provided surgeons with the ability to reconstruct a breast mound using autologous tissue that both looks and feels like the native breast with minimal donor-site morbidity. Cosmetic results of the reconstructed breast have steadily improved, which has led to increasing demand among breast cancer patients for breast reconstruction postmastectomy and postlumpectomy.6 The skill required to balance patient expectations and technical challenges and to produce an excellent cosmetic outcome has proven breast reconstruction to be a rewarding field within plastic surgery.
Cancer and Mastectomy
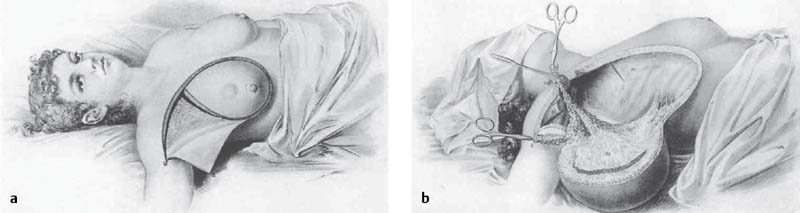
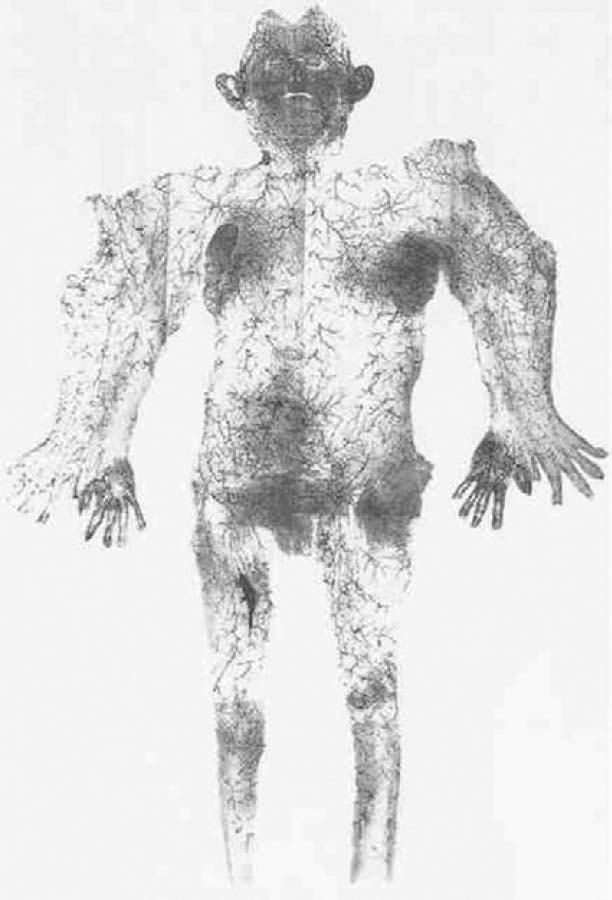
For over a century, surgeons have used autologous tissue to reconstruct the breast. Breast reconstruction after mastectomy, however, did not become popularized until the mid-20th century in part due to the widely publicized work of the famous American surgeon, Dr. William Stewart Halsted. Halsted, who was known for his radical mastectomy technique ( Fig. 1.1 ), was concerned that closure of the chest “might conceal tumour recurrence and increase the chance of tumour dissemination.”7 Therefore, it was common practice to allow mastectomy wounds to heal by secondary intention or to close primarily under high tension, resulting in significant morbidities including chest wall deformities and restricted shoulder movement.8 This erroneous belief led to a highly disfiguring cosmetic result and devastating psychological effects.1
The early 20th century women’s right movement began to question radical breast surgeries, and this prompted the development of breast conservation techniques.9 In 1948, Patey and Dyson10 introduced the modified radical mastectomy, which has since replaced Halsted’s mastectomy as the standard of care. Over the past 30 years, breast cancer surgery has steadily become less invasive.11 Breast conservation surgery techniques such as the lumpectomy, the skin-sparing mastectomy, and the nipple-sparing mastectomy have been shown in prospective randomized controlled trials to be safe, reliable, and effective options for the treatment of breast cancer in selected patients.12,13 These breast conservation methods treat the breast cancer effectively and yet preserve the skin, which improves the overall aesthetic result of breast reconstruction. When combined with immediate breast reconstruction, a skin-sparing mastectomy can reduce operating room costs, preserve the breast pocket, limit the anesthetic risk, and improve the overall aesthetic results.11,14,15
Early Flap Surgery
The word flap is derived from the Dutch “ flappe,” defined as something “to hang broadly and loosely, fastened only by one side.”9 Skin flaps have been used since ancient times to reconstruct defects throughout the body. According to translations by Bhishagratna, the earliest evidence of a pedicled forehead flap for nasal reconstruction comes from Sushruta in 700 A.D., who used a skin graft from the cheek to complete his reconstruction.16,17 In 1440 A.D., the Kanghiara family from India also described the forehead flap for nasal reconstruction.18 Joseph Constantine Carpue in England later popularized the same ancient Indian forehead flap for nasal reconstruction, which was widely adopted by surgeons in Germany, France, and Italy in the early 19th century.19 In 1597, Gaspare Tagliacozzi classically described a successful distally based tubed upper arm flap for nasal reconstruction.19,20
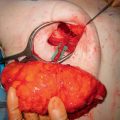
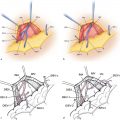
French surgeon Aristide Verneuil21 was the first to describe the use of autologous tissue to reconstruct the breast in 1887, when he transferred breast tissue on a pedicle to reconstruct the contralateral breast. However, it was the German surgeon Vincent Czerny22 who is credited with the first successful postmastectomy breast reconstruction in 1895, when he transferred a large lipoma from the lumbar region to fill a mastectomy defect. Shortly thereafter in 1896, the first documented muscle flap was performed by Iginio Tansini,23,24 when he described the successful application of the latissimus dorsi myocutaneous flap for postmastectomy breast reconstruction25( Fig. 1.2 ).
For the first half of the 20th century, randomly designed tubed flaps dominated all forms of reconstruction, including breast reconstruction. Over the next half-century, multiple attempts at breast reconstruction using local tissue transfers in the form of tubed pedicle flaps harvested from the contralateral breast, abdomen, or extended thoracoabdominal region were attempted. Ombredanne26 described the tubed axilloabdominal flap in 1924.
World War II stimulated a period of great innovation in the field of plastic and reconstructive surgery. Battlefield injuries, in particular difficult ballistic wounds and facial burns, proved to be significant reconstructive challenges for surgeons at the time. Innovative local flaps were developed and widely used to reconstruct these difficult defects. Conceptually, these flaps were all of a random pattern because they lacked any significant recognized orientation to its vascular blood supply.
Tubed Flaps
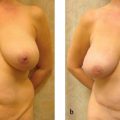
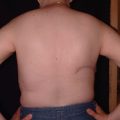
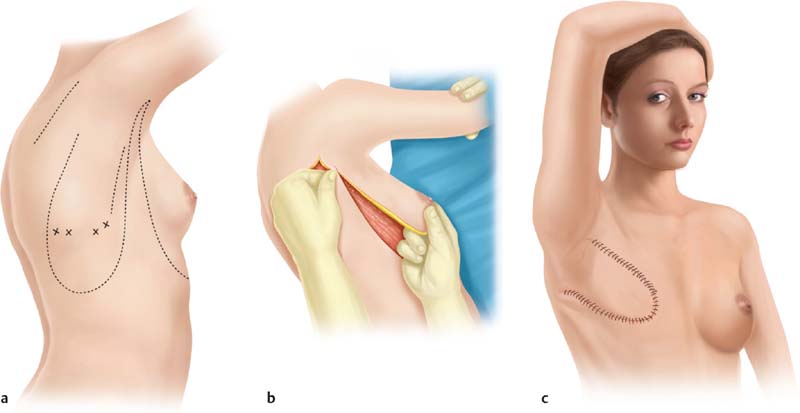
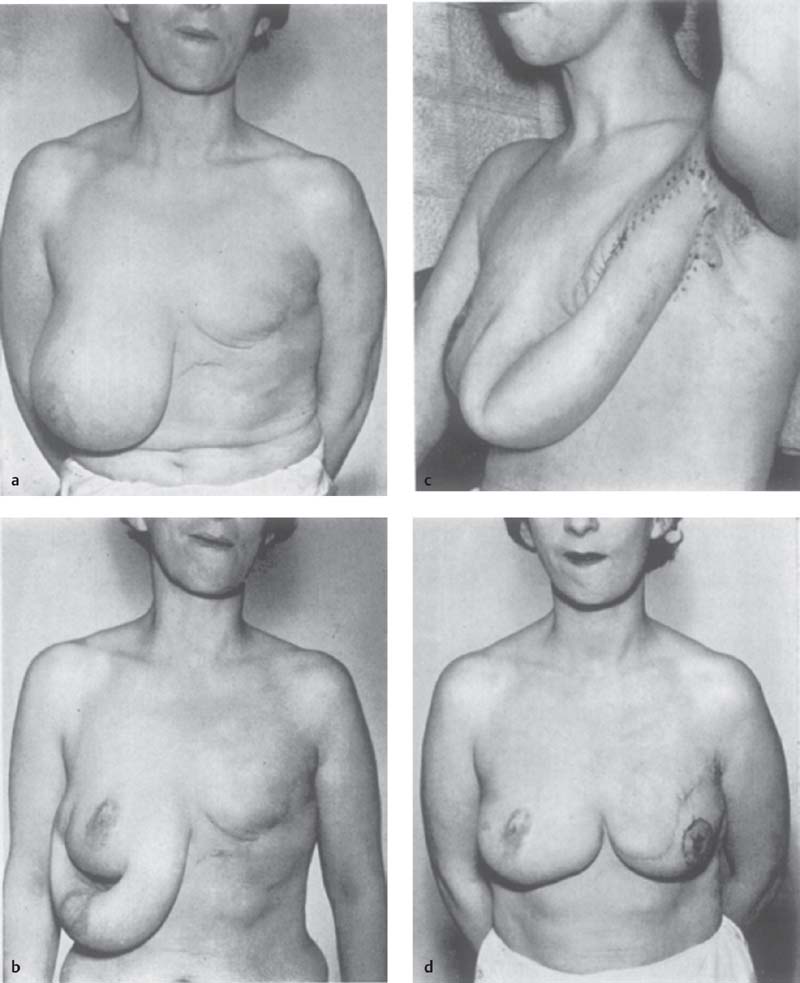
In 1942, Sir Harold Gillies27 successfully marched a tubed pedicle flap from the lower half of the contralateral breast, abdomen, and chest along the torso in multiple stages to re-create a breast mound. In 1950, Yannilos28 successfully performed a composite tubed pedicled contralateral breast flap to reconstruct the breast. In 1956, Holdsworth29 described his four-stage tube flap technique for breast reconstruction with a fairly good aesthetic result ( Fig. 1.3 ). Tubed flaps were eventually abandoned due to poor design and donor-site morbidity, and were replaced by flaps based on known blood supply ( Fig. 1.4 ).
Axial Pattern Skin Flaps
In 1918, Esser30 made the astute observation that skin overlying a vascular pedicle could be dispensed with as long as the feeding and draining vessels remained intact. In 1969, Milton31 confirmed Esser’s work. These observations are the earliest evidence of the axial pattern flap in the literature. McGregor and Jackson’s32 groin flap was the one of the first known axial pattern flaps. In 1973, McGregor and Morgan33 described differences between random and axial pattern flaps. Axial pattern flaps are characterized by the vascular pedicle situated along the flap long axis. The earliest axial pattern flaps described in the literature were the hypogastric flap,34 the deltopectoral flap,35 and the groin flap.32
Musculocutaneous Flaps
Shortly after Tansini’s24 ground-breaking work on musculocutaneous flaps, Morax36 described in 1908 the use of a platysma musculocutaneous flap for head and neck reconstruction. In the 1970s, McGraw,37 Mathes and Nahai,38,39 and others revisited and popularized musculocutaneous flaps recognizing their reliable vascularity from the large source vessel supplying the muscle. The increased activity and focus on muscle flaps led to the Mathes and Nahai38 classification of muscle and musculocutaneous flaps in 1981,38 which helped introduce these flaps to all reconstructive surgeons, including those performing breast reconstruction. Musculocutaneous flaps were readily adopted because a large skin flap could be reliably transferred based on a large source vessel supplying the muscle. The pedicled transverse rectus abdominis musculocutane- ous (TRAM) flap revolutionized breast reconstructive surgery and was widely accepted worldwide.40 However, musculocutaneous flaps were often bulky and often needlessly sacrificed the donor muscle, which was not required for the reconstruction.
Fasciocutaneous Flaps
Although musculocutaneous flaps have been in use since the early 20th century, septocutaneous perforators and their importance in flap design were not recognized until later. Septocutaneous perforators are vessels that follow the intermuscular septum and travel along the fascia to eventually supply the skin. Ponten41 first introduced the concept of fasciocutaneous flaps in 1981 with his work on lower leg reconstruction. Haertsch42 and Barclay and colleagues43 further developed the vascular basis of the fasciocutaneous flap. The major advantage of fasciocutaneous flaps over random skin flaps is the inclusion of fascia, which may contain a source vessel and its cutaneous branches. Cormack and Lamberty44 described in even greater detail the fascial structures throughout the body, and classified four main types of fasciocutaneous flaps.
Perforator Flaps
The advances in our knowledge of the anatomic characteristics of the musculocutaneous perforators eventually led to the evolution of the musculocutaneous perforator flap. Strictly defined, a perforator flap is a skin flap based on a musculocutaneous perforator, which is dissected through the muscle, preserving muscle function. A major advancement in this field was the discovery that the inclusion of a source vessel did not require the harvest of an entire muscle, and perforators could be dissected through the muscle. Perforator flaps require the incision of the desired skin paddle for transfer, followed by the identification of the perforating vessels that supply the skin and dissection of the perforators down to source artery. Practically, a perforator flap may be defined as a vascularized tissue transfer nourished by an adequate cutaneous perforator, which may be a musculocutaneous, septocutaneous, or direct perforator. Perforator flaps have enabled surgeons to tailor flaps to specific defects and minimize donor-site morbidity. For breast reconstruction in particular, perforator flaps have been widely adopted due to the close similarity between the skin and adipose tissue of a perforator flap and the native breast tissue lost after a lumpectomy or mastectomy. In particular, the following flaps have been used extensively for breast reconstruction: deep inferior epigastric perforator (DIEP), superior gluteal artery perforator (SGAP), superficial inferior epigastric artery (SIEA), thoracodorsal artery perforator (TDAP), and profunda artery perforator (PAP).
Microsurgery
Perhaps the most significant advancement in reconstructive surgery has been the development and refinement of microvascular techniques in the late 1960s and early 1970s. The introduction of the operating microscope for small-vessel anastomosis revolutionized microsurgery and opened the door to the era of free tissue transfer.45–47 In 1960, vascular surgeon Jules Jacobson48 performed the first microsurgical anastomosis of a 1.4-mm vessel under surgical magnification. Harry Buncke,49 the “father of microsurgery,” performed the first successful microvascular replantation of a rabbit’s ear in 1964 with vessels measuring 1 mm in size. In 1969, McLean and Buncke50 performed the first successful microvascular transplant using omentum to fill a large scalp defect. Taylor and Daniel51–53 performed the first successful skin free flap transfer of an iliofemoral island flap to reconstruct a lower limb defect in 1973. These refinements in microsurgical instrumentation and technique laid the groundwork for the field of microsurgery and free flap reconstruction.
After Taylor and Daniel’s52,53 1973 report of a successful free microvascular skin flap, the landscape of breast reconstruction was forever changed. In 1975, Toyomi Fujino54,55 performed the first successful microvascular reconstruction of a congenital breast aplasia defect with a “dermalfat-muscle” free flap harvested from the buttocks and anastomosed to the thoracoacromial artery. Hans Holmström56 performed the first abdominal free flap called the “abdominoplasty flap” based on the deep inferior epigastric artery, including the rectus abdominus muscle in the flap. Other free flaps proposed for breast reconstruction over the years include the deep circumflex iliac artery (groin) flap (Rubens flap),57,58 lateral thigh (tensor fascia lata) flaps,59 anterior thigh flap,60 superior and inferior gluteal musculocutaneous flaps,61–63 gracilis flaps,64 triceps flaps,65 and others.
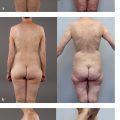
In 1987, Taylor66 published his landmark paper describing the angiosome theory of the vascular anatomy of the skin and integument, which he later elaborated on67,68 ( Fig. 1.4 ). This work has provided a roadmap to the vasculature of the integument for the reconstructive surgeon. The angiosome theory was greatly influenced by Manchot’s69 and Salmon’s70 earlier descriptions of human vascular anatomy and vascular territories. Taylor’s angiosome concept depicts the body as a mosaic of 40 threedimensional (3D) composite units of tissue supplied by named source arteries. An angiosome can further be broken down into smaller territories that are linked by reduced-caliber choke anastomotic vessels. The angiosome concept and understanding of the overall vascular anatomy of the skin has led to a more sophisticated surgical planning of free tissue transfers.65–67
Imaging
Taylor and colleagues71 used a handheld Doppler probe to outline the perforating vessels intraoperatively for flap planning. Preoperative imaging with Doppler flowmetry or Doppler sonography and color duplex imaging are commonly used for perforator flap planning.72,73 Masia et al74 introduced the preoperative computed tomography (CT) angiography for perforator mapping. Since that time, advances in the field of vascular imaging have led to the use of multidetector row CT, 3D CT, as well as magnetic resonance imaging (MRI) for preoperative perforator imaging and flap planning.75–79 Preoperative imaging can significantly reduce operative time. These new techniques have improved spatial resolution images, enabling the detection of stenotic or diseased peripheral vessels.80 Recently, several laser-assisted fluorescence angiography systems have been developed. For instance, the SPY laserassisted fluorescence angiography system (Novadaq, Bonita Springs, FL) has been employed for intraoperative real-time flap perfusion mapping. This technology uses indocyanine green fluorescence angiography and enables the reconstructive surgeon to confirm intraoperatively whether a perforator will provide adequate flap perfusion. This technology is able to predict postoperative mastectomy flap necrosis with great accuracy.81,82
Evolution of Breast Reconstruction
Breast reconstruction has a history that spans nearly 130 years, beginning with the late 19th-century work discussed earlier in the chapter. Over the next half-century, multiple attempts at breast reconstruction using local tissue transfers in the form of tubed pedicle flaps harvested from the contralateral breast, abdomen, or extended thoracoabdominal region were attempted. Tubed flaps were largely abandoned due to poor design, donor-site morbidity, and poor cancer survival rates. With an improved understanding of abdominal wall anatomy and advances in the abdominoplasty technique, efforts to use the abdomen as a donor site subsequently dominated the field.
Autologous flap reconstruction of the breast has used the abdomen as a donor site since the mid-20th century, when Gillies (discussed earlier) and D.R. Millard, Jr.83 transposed a racquet-shaped abdominal flap to the mastectomy defect. In 1977, Stephen Mathes and John Bostwick84 described the rectus abdominis musculocutaneous flap to reconstruct abdominal wall defects and later for use in breast reconstruction. In 1979, Robbins85,86 first reported the use of the rectus abdominis musculocutaneous flap for breast reconstruction. In 1977, Drever87 first described the vertically orientated epigastric island flap now commonly known as the vertical rectus abdominis musculocutaneous (VRAM) flap for use in breast reconstruction. By the end of the 1970s, the rectus abdominis musculocutaneous flap became the workhorse of autologous breast reconstruction in many centers.
In 1982, Carl Hartrampf88 first described and performed the pedicled TRAM flap, an abdominal island flap composed of a transverse ellipse of skin and subcutaneous tissue isolated on its superior epigastric vascular pedicle ( Fig. 1.5 ). The pedicled TRAM flap has the advantages of being a well-vascularized flap, with an adequate large arc of rotation and volume to reconstruct an aesthetic breast mound without the need for microsurgical anastomosis. Furthermore, the donor-site scar was cosmetically hidden in the lower abdomen.
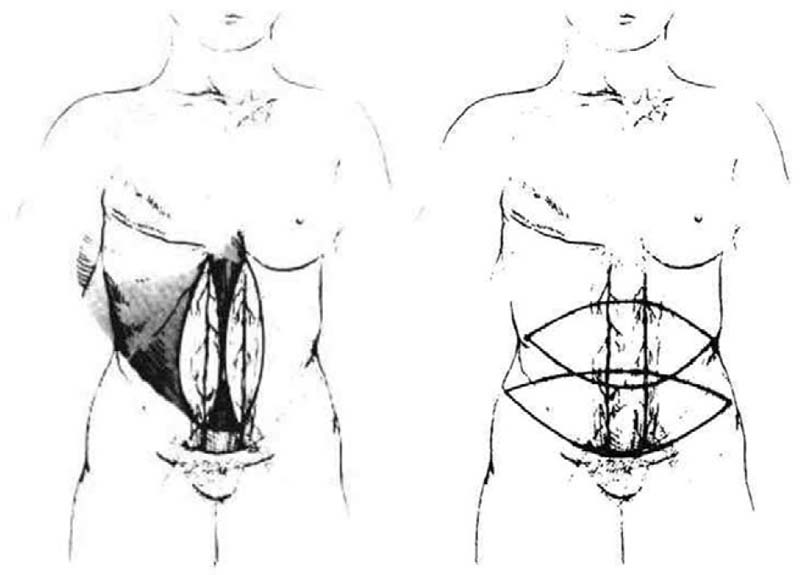
However, it was quickly recognized that the pedicled TRAM was associated with significant donor-site morbidity in the form of abdominal wall hernias and weakness. Abdominal wall fascial defects were commonly repaired by synthetic mesh and were associated with significant longterm complications.89 The field gradually focused on the generation of a muscle-sparing technique for the elevation of the rectus abdominis flap. In 1989, Grotting90,91 started to use the free microvascular TRAM (free TRAM) flap for primary breast reconstruction, with good results. The advantages of the free TRAM over the pedicled TRAM include the use of the dominant inferior epigastric pedicle (which is larger in diameter), the avoidance of a bulge in the epigastrium, reduced partial flap necrosis, and reduced donor-site morbidity. Both free and pedicled TRAM flaps remain widely used for breast reconstruction today, despite the risk of donor-site morbidity. The concept of musclesparing TRAM flaps evolved as surgeons reduced the amount of rectus abdominis muscle and fascia harvested with the flap to lessen the insult to the abdominal wall, while preserving the all-important periumbilical perforators.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree