Acknowledgments
The authors would like to thank the previous authors of this chapter, Owen L. Ala, MD and T. Shane Johnson, MD, for their contributions.
Physical Examination of the Wrist
When performing a physical examination of the wrist, it is helpful to approach the wrist in a systematic fashion based on anatomical regions. Knowledge of anatomical landmarks will help the practitioner develop a differential diagnosis, while several exam maneuvers can help in making the correct diagnosis. The importance of wrist palpation based on anatomical region cannot be understated. Imaging can then be used to aid in the diagnostic process. In this chapter, we will use an anatomical approach to the wrist to identify and better understand commonly seen wrist pathologies and their treatments.
Radial-Sided Wrist Pain
The bones, tendons, and ligaments that lie on the radial side of the wrist can all be sources of pain for the patient. The first extensor compartment contains the extensor pollicis brevis (EPB) and abductor pollicis longus (APL) tendons and can be a source of significant pain, known as De Quervain’s tenosynovitis. These tendons can be identified making up the lateral/radial border of the anatomical snuffbox at the level of the radial styloid. Pain with palpation over this compartment or a Finkelstein’s test – clasping the thumb in the palm with ulnar deviation of the wrist – will confirm the diagnosis. Pain elicited with this maneuver more proximal in the forearm can be the result of intersection syndrome. Palpating just distal to the radial styloid in the anatomical snuffbox can indicate scaphoid pathology. The distal pole of the scaphoid can be palpated at the base of the thenar eminence. This can be confirmed by ranging the wrist from radial to ulnar deviation and feeling the scaphoid flex and extend. Palpating just distal to the anatomical snuffbox can elicit tenderness in the scaphoid–trapezium–trapezoid (STT) or trapezium–metacarpal (carpometacarpal, CMC) joint of the thumb. Pain in this region is commonly seen in CMC arthritis.
Central Wrist Pain
Just ulnar to the anatomical snuffbox on the dorsal side of the wrist is the “soft spot,” which is the region of the scapholunate (SL) ligament. The Watson shift test can be performed to assess the integrity of the SL ligament; this is performed by moving the wrist from ulnar hyperextension to radial flexion while pressing on the scaphoid tubercle volarly and assessing for a “clunk.” The contralateral wrist should always be assessed for comparison. Pain in the dorsal midline of the wrist can also be the result of Kienböck’s disease. The distal radioulnar joint (DRUJ) is assessed more proximally. A radioulnar ballottement test can be used to test for congruency of the DRUJ.
Ulnar-Sided Wrist Pain
The ulnar-sided wrist exam should start by palpating the ulnar head and ulnar styloid. Pain with palpation of the ulnar soft spot or fovea can be an indication of triangular fibrocartilage complex (TFCC) pathology. Injury to the TFCC can be confirmed with magnetic resonance imaging (MRI) or arthroscopy. Pain elicited by pushing the distal ulna in a radial direction like a “piano key” is a sign of DRUJ instability or injury. Immediately dorsal to the ulnar soft spot lies the extensor carpi ulnaris (ECU) tendon. Pain and snapping while performing ulnar flexion and supination of the wrist indicates ECU tendinitis and subluxation respectively. Pain on the volar side of the ulnar wrist is likely due to injury to the pisiform or hook of the hamate. This can be seen from both arthritic and traumatic pathologies.
Kienböck’s Disease
Pest first described avascular necrosis of the lunate based on cadaveric dissections although the disease gets its name from radiologist Robert Kienböck. Despite Kienböck’s disease being described over 100 years ago, the etiology and natural history are not clearly understood and it is thought to result from a vascular etiology. Kienböck’s disease presents in patients as wrist pain, usually located centrally over the dorsum of the wrist, and is rarely bilateral. Pain is usually present with wrist motion, but as the disease progresses, pain at rest is frequent. This diagnosis is commonly seen in men 20–40 years of age. It was originally thought that trauma to the lunate likely results in Kienböck’s disease, although long‐term follow-up studies have shown that lunate fractures do not reliably develop avascular necrosis, even with an ulnar negative variance. Ulnar negative variance has been associated with Kienböck’s disease, prompting many to point to this anatomical feature as a possible etiology. Hulten’s classic study in 1928 showed an association between ulnar negative variance and Kienböck’s disease but more recent studies do not demonstrate a clear correlation.
Clinical Evaluation
The clinical evaluation of the patient with Kienböck’s disease starts with a thorough physical exam of the wrist. However, the exam findings for Kienböck’s disease are usually nonspecific. Dorsal wrist pain, often with motion or activity, is the main physical exam finding. Radiographic evaluation is followed by the physical exam. Kienböck’s disease can be diagnosed by correlating the physical exam with radiographs, however imaging using MRI is often necessary for definitive diagnosis. Radiographs are evaluated for sclerosis of the lunate and negative ulnar variance.
Staging and Treatment
Radiographs and MRI are used to stage Kienböck’s disease. The staging system as presented by Lichtman and associates is the most popular and helps guide treatment and determine prognosis. The treatment of Kienböck’s disease is based on the staging and will be discussed together. Treatments of Kienböck’s disease are evolving with advances in microsurgery and 3D printing technology.
Stage I
Symptoms of stage I Kienböck’s disease are usually nonspecific and mimic wrist sprains or synovitis. Radiographs are usually normal in stage I Kienböck’s and represent precollapse and early appearance of the disease. MRI will demonstrate low signal on T1- and T2-weighted images in the entirety of the lunate. If the low signal is localized to a specific area of the lunate on T1-weighted images then other more focal diagnoses are more likely. If T2-weighted images show increased signal, this can be a sign of revascularization and MRI can be used to monitor resolution of disease. Stage I disease is best treated nonoperatively with a splint. Most people advocate for at least 3 months of splint use. If symptoms do not improve a radial shortening osteotomy in the setting of negative ulnar variance would then be considered.
Stage II
Radiographs become diagnostic or at least suggestive of Kienböck’s disease during stage II. One typically finds increased sclerosis of the lunate with possible fracture lines or early collapse. MRI continues to be diagnostic with the entire lunate showing low T1 and T2 signal intensity. Lunate height is maintained and symptoms usually mimic chronic wrist synovitis. Treatment usually involves a trial of splinting, although at this stage a radial shortening osteotomy is more aggressively pursued to prevent advancement of the disease and collapse of the lunate.
Stage IIIA
Stages II and IIIA are usually discussed together because the treatment options are the same. Stage IIIA Kienböck’s disease demonstrates collapse of the lunate; however, there is maintenance of carpal height and no signs of arthritis. The anterior–posterior distance of the lunate is usually increased on the lateral radiograph. Sclerosis is present and MRI findings of low signal intensity on T1- and T2-weighted images remain consistent. The treatment of stage II and stage IIIA disease should start with initiating supportive care and immobilization. A surgeon should not rush to surgery during this stage of treatment and a course of nonoperative treatment should be initiated. The severity of the patient’s symptoms should dictate whether to proceed with surgical treatment or continue with nonoperative management. The surgical treatment options differ based on ulnar variance.
Stage II and IIIA with Ulnar Negative Variance
When ulnar variance is negative, it is thought that increased pressure from the lunate fossa of the distal radius is the cause of the symptoms. Unloading and decreasing the pressure on the lunate through a radial shortening osteotomy is usually the treatment of choice. One study demonstrated 93% of patients had diminished pain after a radial shortening osteotomy. Ulnar lengthening osteotomy has been described for stage II and IIIA disease with ulnar negative variance but this is generally considered a more difficult procedure with higher complications. Ulnar lengthening requires bone grafting and healing at two osteotomy sites, both the proximal and distal ends.
Stage II and IIIA with Ulnar Positive or Neutral Variance
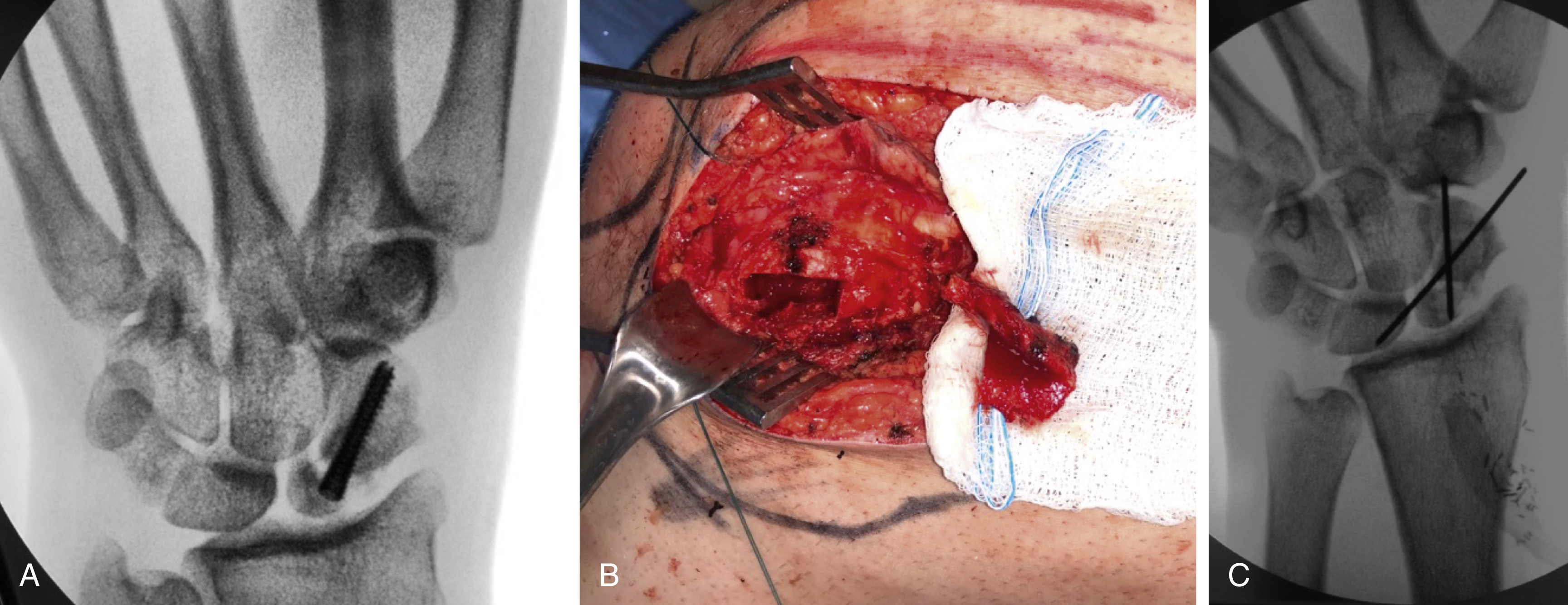
Revascularization procedures are most appropriate in stage II and IIIA with ulnar positive or neutral variance. A number of revascularization procedures have been described, including vascularized pedicle transfer from the distal radius, vascularized pisiform transfer, vascularized bone graft from the metacarpal, and free vascularized grafts from the iliac crest, medial femoral condyle or medial femoral trochlea. The most popular vascularized pedicle transfer is from the distal radius and is the 4,5 extensor compartment artery (ECA) ( Fig. 58.1 ). In more advanced disease, coronal fractures of the lunate can be fixated in preparation for a vascularized osteochondral graft. These procedures may be accompanied by joint leveling procedures such as capitate shortening, radius osteotomy or external fixation.
Stage IIIB
Stage IIIB is characterized by lunate collapse, rotation of the scaphoid, and loss of carpal height. Revascularization procedures are usually not considered after lunate collapse and loss of carpal height because of the destruction of normal anatomical architecture. There is some controversy about the best way to treat stage IIIB disease and some authors have had good results with limited wrist arthrodesis, including scaphocapitate arthrodesis and scaphotrapezium–trapezoid arthrodesis. Although controversial, other authors have had good results with radial shortening osteotomies. Lunate excision with interposition arthroplasty or proximal row carpectomy are additional salvage procedures that have been described. A recent case report has been published describing the implantation of a 3D-printed lunate prosthesis with near normal recovery of wrist motion at 12 months postoperatively. Another very promising new treatment for stage II and III Kienböck’s is the free medial femoral trochlea flap. In a report by Bürger et al, 13 of 16 patients went on to have complete pain relief with similar range of motion as before surgery. Grip strength was 85% of the contralateral hand. ,
Stage IV
Stage IV is characterized by collapse of the lunate and degenerative changes in the radiocarpal and midcarpal joints. Joint leveling or revascularization procedures are not performed once degenerative changes appear. Many patients can be managed nonoperatively with supportive care such as splints, antiinflammatory medications, and activity modification. In patients who have failed nonoperative management, salvage procedures can be considered such as limited or total wrist arthrodesis or proximal row carpectomy. For a proximal row carpectomy to reliably provide pain relief, the proximal capitate and lunate fossa must be free from degenerative changes since they will make up the new radiocarpal articulation.
De Quervain’s Tenosynovitis
De Quervain’s tenosynovitis causes pain on the radial side of the wrist, centered over the first extensor compartment. Active radial deviation of the wrist or passive ulnar deviation causes severe pain over the tendons of the first dorsal compartment. This presentation is commonly seen with new mothers or more recently with frequent cell phone texting users. The first dorsal compartment tendon sheath can be up to five times its normal size due to fibrous tissue deposition and may feel like a mass over the radial styloid. De Quervain’s tenosynovitis is a myxoid degeneration of the first dorsal compartment. Although nonsteroidal antiinflammatory medications are a first-line treatment, it is not an acute inflammatory process. Often the tendon sheath can be easily palpated due to its enlarged size. There is known to be significant anatomical variation of the first extensor compartment that is associated with increased risk for developing tenosynovitis, including multiple slips of APL. A septum within the first compartment has also been found in 50% of individuals in cadaver studies.
Intersection syndrome should be ruled out during the diagnosis of De Quervain’s tenosynovitis. Intersection syndrome causes pain in an area about 4 cm proximal to the radial styloid and there are subtle differences. It is caused when pain, swelling, and sometimes crepitus occurs because the outcropping muscle bellies of the APL and EPB intersect with the long wrist extensors on the dorsum of the forearm. This should also not be confused with a neuropathy of the radial sensory nerve as it exits the deeper tissues between brachioradialis and extensor carpi radialis longus. This is known as Wartenberg’s syndrome.
Diagnosis of de Quervain’s tenosynovitis is usually done with a patient history and physical exam. Imaging studies are usually not required. Pain over the first dorsal compartment on the radial aspect of the wrist, which is made worse with palpation, is the first physical exam finding. A thickened tendon sheath and pain with active radial deviation are also common symptoms. Finkelstein’s maneuver is the classic physical exam maneuver used to aid in the diagnosis, although this can also be positive in intersection syndrome. Ultrasound can also be used to visualize the tendons and inflammation present within the tendon sheath, and can aid in steroid injection directly into the sheath, but is rarely used.
Initiation of nonsurgical treatments such as watchful waiting, splinting, nonsteroidal antiinflammatory medication, and steroid injections into the first extensor compartment should be considered. Multiple studies have shown successful treatment with each of these nonsurgical techniques. The authors recommend nonsurgical treatment be initiated upon diagnosis. We recommend a removable thumb spica splint, nonsteroidal antiinflammatory medications, and steroid injection into the compartment. Cases that are refractory to nonsurgical management should be considered for surgical release.
Surgical release of the first extensor compartment is very successful and produces reliable, long-term pain relief. Open surgery through a small horizontal incision is the authors’ preferred method of treatment. Care is taken to make incision through the sheath as dorsal as possible to decrease the risk of volar tendon subluxation postoperatively. Branches of the radial sensory nerve are in or near the operative site and should be carefully identified and/or avoided. These branches lie deep to the venous structures of the forearm. If a branch of the radial sensory nerve is cut during surgery, a painful neuroma can result. When performing the surgery, it is necessary to thoroughly explore the first extensor compartment due to significant anatomical variation, including presence of a septum and multiple slips of APL.
Scaphoid Fractures
The scaphoid is most commonly injured by a fall on an outstretched hand, resulting in radial-sided wrist pain and, classically, snuffbox tenderness. Scaphoid fractures represent 60% of all carpal bone fractures. They can occur as isolated trauma or in association with other bony or ligamentous injuries of the distal radius and carpal bones. The triquetrum is the second most commonly fractured carpal bone.
The distally based blood supply of the scaphoid makes fractures notoriously difficult to heal. The result is a high incidence of delayed union, nonunion, and avascular necrosis. Eighty percent of the scaphoid is covered in articular cartilage, which limits its blood supply entry points. The majority of the blood supply to the scaphoid comes from the dorsal scaphoid branches of the radial artery, with a minority of the blood supply from the volar scaphoid branches. These vessels enter the scaphoid relatively distally, resulting in purely intraosseous perfusion of the proximal pole. The tenuous blood supply of the proximal pole explains the reported avascular necrosis that occurs in 13%–50% of scaphoid fractures.
Early diagnosis and treatment are important since scaphoid fractures that are left untreated have a higher incidence of nonunion, malunion, and avascular necrosis. Scaphoid nonunion is difficult to treat and if left untreated will progress to carpal collapse and degenerative arthritis, specifically, scaphoid nonunion advanced collapse (SNAC wrist).
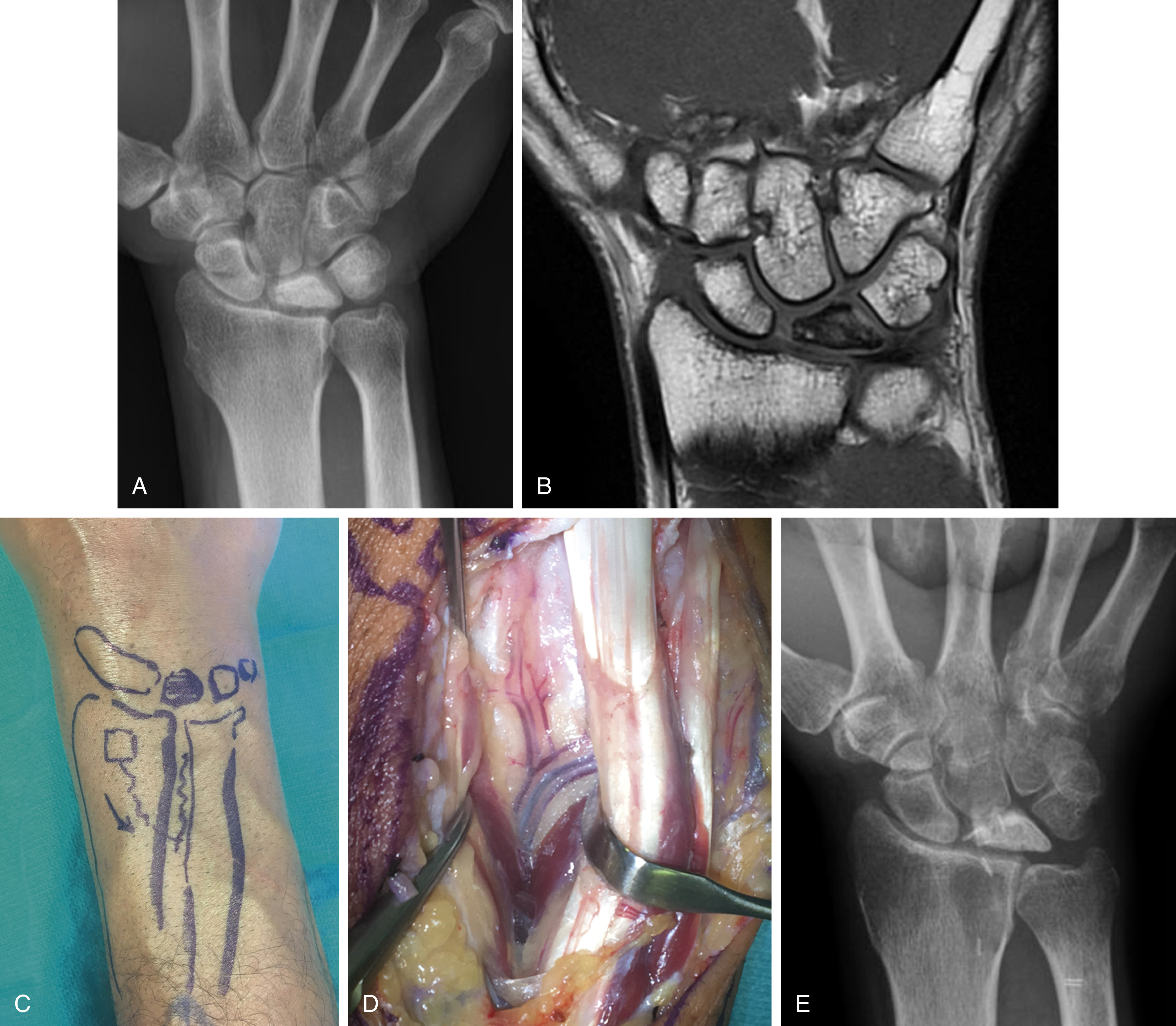
Diagnosis begins with a good history and physical exam as most patients report a recent history of trauma to the hand and wrist. Patients typically have pain on the radial side of the wrist, specifically in the anatomical snuffbox, decreased range of wrist motion, decreased grip strength, and pain with thumb motion. Adequate radiographs, including posteroanterior (PA), lateral, pronated oblique, supinated oblique, and a PA with ulnar deviation (scaphoid view), are essential. However, X-rays often show no evidence of fracture. It is thought that initial radiography misses 15%–20% of scaphoid fractures. When there are signs of scaphoid fracture on history and physical exam but no radiographic evidence, the traditional algorithm would be to cast the patient in a short arm thumb spica cast and repeat the X-rays in 2 weeks. After 2 weeks, there is usually sufficient resorption at the fracture site to visualize a scaphoid fracture on radiographs. Advanced imaging studies, such as MRI and computed tomography (CT) scans, can help in the diagnosis and in surgical planning. MRI is the preferred test if the diagnosis of a scaphoid fracture is in question. The sensitivity and specificity of diagnosing a fracture is 90%–100% with MRI. CT scans show bone structure better than MRI and are therefore better in determining displacement of a fracture or in visualizing bony healing. However, a CT scan should be used with caution in diagnosing an occult scaphoid fracture because there is a high rate of overdiagnosis with CT (false-positives), most likely due to normal vascular foramina appearing as a fracture. Therefore, a CT scan is better used in surgical decision-making to determine if a fracture seen on radiographs is displaced or not and for operative planning.
Cast immobilization for 8–12 weeks can successfully treat most nondisplaced or minimally displaced scaphoid fractures. There is no consensus on whether to use a short arm or long arm cast, although data from a small randomized trial suggests an initial period of long arm casting can decrease time to union and decrease the nonunion rate. A long arm cast theoretically prohibits wrist rotation and shear stresses on the scaphoid. If treating a scaphoid fracture nonoperatively, the authors recommend using a long arm cast for the first 4–6 weeks and then changing to a short arm cast for the duration of the treatment. The disadvantages to cast treatment are increased time to healing, frequent office visits, increased need for radiographs, potential for skin breakdown, and stiffness. There is high-level data that suggests immobilization of the thumb is not necessary and may in fact be detrimental to healing.
Some authors have advocated immediate percutaneous internal fixation of minimally displaced and nondisplaced scaphoid fractures. The reasons behind the trend to immediate percutaneous fixation of these fractures are a decreased time to healing, faster return to sports or work, and a favorable economic analysis in terms of social life and return to work. , There appear to be similar functional outcomes at 2 years of cast versus percutaneous treatment of nondisplaced or minimally displaced scaphoid fractures.
Internal fixation is indicated in scaphoid fractures with displacement of more than 1 mm on any radiographic view. Scaphoid fractures with displacement have a much higher probability of progressing to a nonunion with cast immobilization compared with internal fixation. The headless compression screw is the most accepted technique for fixation of scaphoid fractures. It has the advantage of being recessed below the articular surface of the scaphoid. The scaphoid is a curved bone, shaped much like a cashew, although there can be significant variability. The shape of the scaphoid makes screw fixation challenging; for the best results, the screw should be placed down the central axis of the scaphoid in all dimensions. Breaching of the scaphoid with the screw during fixation should be avoided since it will lead to early degenerative arthritis. Healing rates of 95% have been reported with adequate screw fixation.
Scaphoid Nonunions
Scaphoid nonunions develop in about 5% of scaphoid fractures and are usually the result of inadequate immobilization, displacement, or delayed diagnosis. Scaphoid nonunions are difficult because of the tenuous blood supply of the proximal pole and resultant avascular necrosis, the tendency for the scaphoid to collapse into a “humpback deformity,” and the systematic progression to a SNAC wrist.
Successful treatment of scaphoid nonunions without collapse, using open and percutaneous techniques, has been described. Percutaneous fixation of scaphoid nonunions without collapse or displacement has been described with a cannulated compression screw. Even with resorption of bone at the fracture site, the union rates of percutaneous screw fixation is high. The Matti–Russe technique has traditionally been the treatment of choice for scaphoid nonunions without collapse. This technique uses corticocancellous pieces of bone taken from the iliac crest and placed into the scaphoid after the fibrous tissue from the nonunion has been removed. Modifications of the Matti–Russe technique have taken the graft from the distal radius instead of the iliac crest or placed a corticocancellous graft with either screw or Kirschner wire (K‐wire) fixation. The Matti–Russe technique has been reported to provide union in up to 90% of patients. The majority of patients who did not achieve union were reported to have avascular necrosis of the proximal pole, and this technique is not generally recommended for patients with avascular necrosis.
Treatment of scaphoid nonunions with collapse (“humpback deformity”) is more difficult since this requires a need for reduction of the deformity, replacement of the lost bone, and stable fixation. Volar wedge fixation with a tricorticocancellous bone graft taken from the iliac crest has been a successful way to correct a humpback deformity and replace lost bone. This technique is usually augmented with screw fixation down the central axis of the scaphoid. Recently, there have been published reports of treating scaphoid nonunions with humpback deformity without structural bone graft and only placing cancellous bone graft. The reduction is performed open through a volar approach and assisted with K-wires, while a screw fixation provides a stable construct for healing. Bone graft is then harvested from the distal radius or iliac crest and packed around the screw to replace the bone lost from the collapse. The good results reported with these techniques have not been duplicated when the proximal pole has gone onto avascular necrosis.
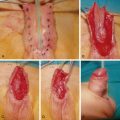
Treatment of scaphoid nonunions with avascular necrosis of the proximal pole have been most successfully treated with vascularized bone grafts. The most popular vascularized bone graft is the 1,2 intercompartmental supraretinacular artery (1,2 ICSRA) bone graft taken from the dorsal aspect of the distal radius. The vascular anatomy of the 1,2 ICSRA is consistent, but very small. Recent reports have shown about a 75% union rate with use of the 1,2 ICSRA vascularized graft. A capsular-based vascularized bone graft has been described by Sotereanos et al and based off the artery of the base of the fourth dorsal compartment. This technique has been praised for its simplicity. However, both these techniques provide only a small amount of bone for reconstructing a humpback deformity and the vascular pedicles are tenuous. Recently, free vascularized bone grafts taken from the medial femoral condyle (MFC) or trochlea (MFT) and based off either the longitudinal (MFC) or transverse (MFT) branches of the descending geniculate artery has been described. The free vascularized medial femoral condyle flap can be taken without articular cartilage or with articular cartilage as a medial femoral trochlea flap utilizing the transverse branch of the descending geniculate artery, with or without a chimeric skin paddle. The vascular pedicle of these flaps is sufficiently robust and is usually anastomosed end-to-side into the radial artery in the anatomical snuffbox or volar wrist. The donor sites of each of these flaps appear to be well tolerated, with patients returning to normal lower extremity function ( Fig. 58.2 ). In the setting of proximal pole avascular necrosis, we prefer to core out the proximal pole bone, preserving the cartilaginous cap and the scapholunate ligament, and place the bone graft as a dowel into the scaphoid.