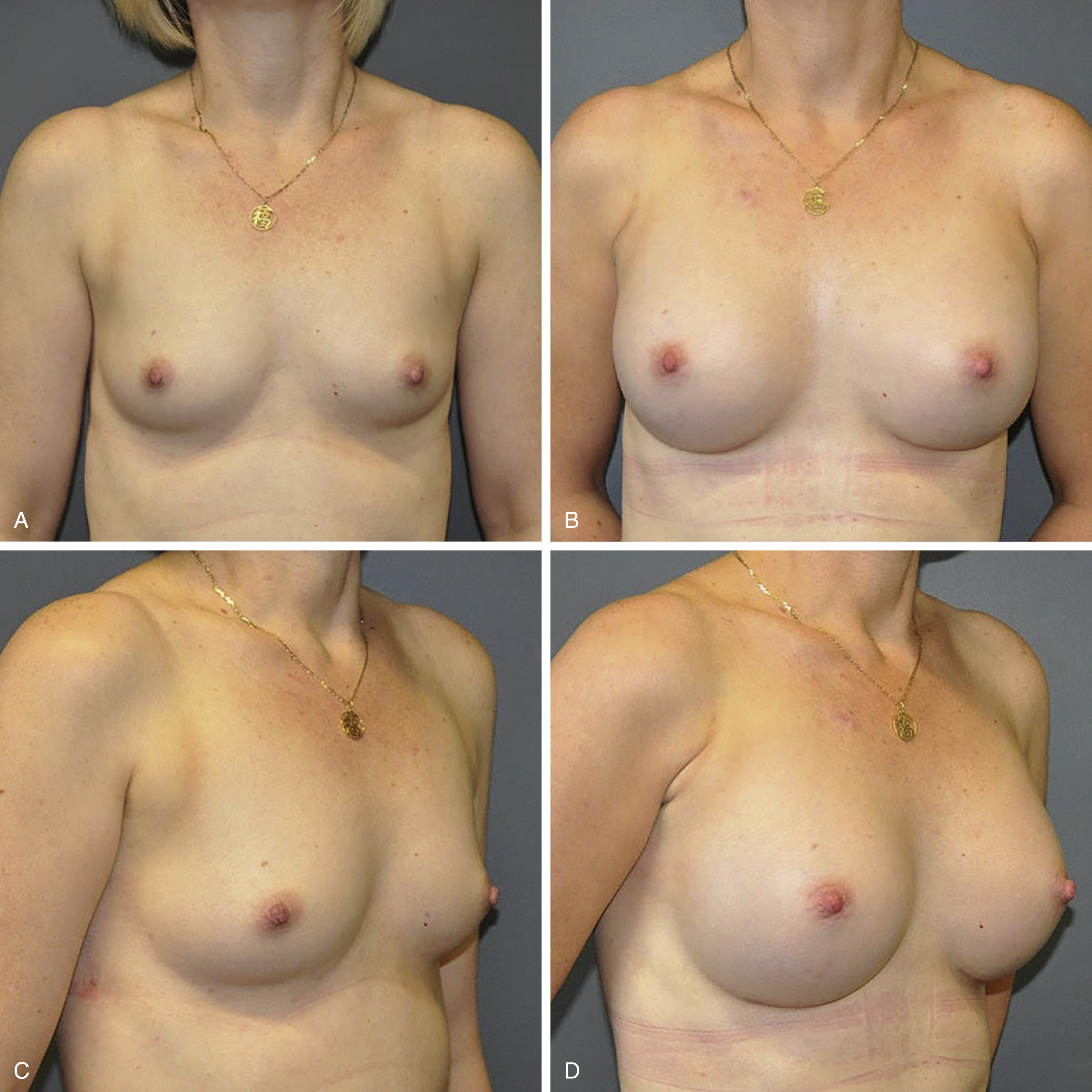
![]() For video accompanying this chapter see ExpertConsult.com . See inside cover for access details.
For video accompanying this chapter see ExpertConsult.com . See inside cover for access details.
Introduction
Breast augmentation is the most popular esthetic surgery procedure. In the United States 450,000 implants are placed for reconstructive surgery purposes and 5,083,717 for breast augmentation annually. Given its history, there is a vast amount of literature devoted to differing opinions, philosophies, and controversy regarding its safety. Many injectable and implantable materials have also been used over the years, including shellac, beeswax, epoxy resin, petroleum jelly, Teflon, polyurethane and polyvinyl alcohol. In recent times, controversy again has surrounded silicone implants, with an increased association with breast implant-associated anaplastic large cell lymphoma (BIA-ALCL). The popularity of this procedure would behoove all plastic surgeons to remain abreast of the science and the clinical literature in this rapidly evolving field.
Implant Evolution
Silicone (dimethylpolysiloxane) was first developed during World War II as part of aircraft engineering. Its soft, inert nature made it a perfect prosthetic substrate for medicine. Silicone breast implants can be divided into five generations based on shell thickness, texture, and implant fill ( Table 35.1 ). Current implants are divided by gel fill into saline or silicone. The silicone fill is further subdivided into stable and soft, based on level of molecular cross-linkage. By shell, they are divided into textured and smooth, all made of silicone polymers, and finally, by shape, into round and anatomical.
| Implant Generation | Production Period | Shell Characteristics | Silicone Gel Characteristics | Shape |
|---|---|---|---|---|
| First | 1962–1970 | Thick shell (average 0.25 mm), Dacron patch | Thick viscous | Anatomical |
| Second | 1970–1982 | Thin shells (average 0.13 mm), no patch, smooth surface | Less viscous softer gel | Round |
| Third | 1982–1992 | Thick low bleed shells, smooth surface | More viscous than second generation | Round |
| Fourth | 1993–present | Thick, strong, low bleed shell, smooth and textured surfaces | More viscous cohesive gel | Round and anatomical |
| Fifth | 1993–present | Thick, strong, low bleed shell, smooth and textured surfaces | Cohesive and form stable gel | Round and large array of anatomical |
First-generation implants manufactured by Dow Corning in 1962 were composed of viscous silicone gel containing a thick teardrop shell. In the 1970s, high capsular contracture rates led to an evolutionary step towards a second generation of softer implants; these were smooth, round, seamless, and contained less viscous silicone. Although reducing capsular contracture rates, they were associated with “gel bleed” from small molecular weight particles of silicone diffusing across the thin silicone elastomer shells, which has also been implicated in capsular contractures. This led to third-generation implants which had a strengthened silicone shell, two layers of high performance elastomer with a fluorosilicone barrier between layers. The fourth generation of textured implants were made in part to emulate polyurethane foam’s natural pores. Foam delamination resulted in a noncontractile capsule, yielding immobile implants that had a soft feel. They were, however, withdrawn in 1990 after animal studies showed the carcinogenic nature of the polyurethane byproducts. Kirkpatrick et al implanted 1266 polyurethane implants in 490 animals (3 implants each) which caused the formation of 340 tumors over a 2-year period. The study concentration, however, was 3000 times that used in humans.
Biocell, developed by McGhan Medical (owned by Allergan), is composed of an irregular open pore textured surface with an average density of 3.1 pores/mm 2 and an average pore size of 289 μm. These pores, created by a lost salt technique, are developed through an adhesive effect. This is similar to polyurethane except for delamination. This shell characteristic has been used for both saline- as well as silicone-filled implants, resulting in immobile yet soft feel implants and lower contracture rates. Biocell was withdrawn from the European markets in December 2018, followed by the Canadian and Australian regulators, the latter banning a large number of textured implants and tissue expanders. In July 2019 Allergan issued a worldwide recall of their textured implants and breast expanders after the US Food and Drug Administration (FDA) notified the company of the increased risk associated with their textured implants. Eighty percent of the reported 573 cases were Allergan textured implants. Of these, there have been 33 deaths. Given the rarity of BIA-ALCL the FDA have not recommended removal from women with implants currently in situ. The FDA conducted their own analysis and concluded that the use of Biocell textured implants from Allergan was associated with a sixfold risk of BIA-ALCL compared with textured implants from other companies. It is clearly evident that research into the appropriate implant materials is an evolving field and will require greater understanding of biocompatibility.
Siltex, developed by Mentor (owned by Johnson & Johnson), has a textured surface characterized by a pattern of nodules ranging in height of from 65 μm to 150 μm and in width from 60 μm to 275 μm. These do not adhere to the surrounding tissues, meaning they are mobile in the pocket resulting in lower contracture rates (saline- or silicone-filled). Fifth-generation silicone implants are textured with form stable silicone achieved by increasing the cross linkage of the silicone gel, which assists in maintaining their shape ( Table 35.1 ).
Saline implants were first reported by the French in 1965, aiming for a less invasive and smaller scar surgery. The main disadvantage of these devices is their deflation rate and feel. The deflation rates in the initial offerings were as high as 75% at 3 years. The shells were thickened by a room temperature vulcanized process which reduced their deflation rates. However, they contained surface wrinkles and a round-ball appearance when overfilled. The current accepted rate of deflation is approximately 1–4% for the first 10 years.
Indications
Glandular hypomastia may occur as a developmental or involutional process and affects a significant number of women. Developmental hypomastia is seen as primary mammary hypoplasia, or as a sequela of thoracic hypoplasia (Poland’s syndrome). Involutional hypomastia may develop in the postpartum setting and be exacerbated by breastfeeding or significant weight loss. Inadequate breast volume may lead to a negative self-body image, feelings of inadequacy, and low self-esteem. Such disturbances may adversely affect a patient’s interpersonal relationships, sexual fulfillment, and quality of life.
Initial consultation for augmentation should begin with open-ended questions about the patient’s goals and expectations. Patients today have often invested time researching the internet, Facebook forums, chat rooms and learning from friends who have had the procedure. The surgeon should be able to form an impression of the patient. Any concerns about the patient’s level of understanding, unrealistic expectations, or self-esteem issues should be explored prior to proceeding with surgery. A medical history and physical examination is essential for assessment of risk factors and candidacy for breast augmentation.
The ideal size and shape of the female breast is subjective and relates to both personal preference and cultural norms. However, most surgeons agree there are certain shared characteristics representing the esthetic ideal. These include a profile with a sloping or full upper pole and a gently curved lower pole with the nipple–areola complex at the point of maximal projection. The breast structure itself contains breast parenchyma, skin, and subcutaneous fat. The resulting form of the breast after augmentation will be determined by the dynamic interaction of the breast implant, parenchyma, and soft tissue envelope.
Assessment and Implant Selection
Physical examination begins with observation of signs of chest wall deformity or spinal curvature. It is imperative to document and draw attention to any asymmetry of breast size, nipple position, or inframammary fold (IMF) position. Palpation of all quadrants of breasts and axillae is required to rule out masses or suspicious lymph nodes. The surgeon should carefully assess the quantity and compliance of the parenchyma and soft tissue envelope. The soft tissue pinch test involves gathering the superior pole of the breast between the examiner’s thumb and index finger, measuring the thickness of the intervening tissue. A pinch test result of <2 cm indicates a need for subpectoral placement of the implant. It is also important to characterize the amount, quality and distribution of the breast parenchyma as it may be necessary to reshape or redistribute tissues to achieve the desired shape of the breast mound. The elasticity of the skin should also be characterized by observing its resistance to deflection and noting any signs of skin redundancy or stretch marks. Most manufacturers have developed a preoperative planning system to facilitate patient assessment and implant selection. Standard discussion centers around information about implant shapes (anatomical vs. round) or projection levels. The patient’s perception of the desired look is simulated utilizing different size and style implants and sizers or with computer simulation. Interestingly, Hidalgo and Weinstein’s level 1 study did not demonstrate a statistically significant difference between anatomical or round implants.
Operative Techniques
Preoperative Markings
Preoperative markings are a useful reference point during the procedure and are made with the patient in the upright position. The surgeon should mark the chest midline in the frontal view from the suprasternal notch to the xiphoid process, the existing IMFs, and the likely position of the new IMFs as the proposed limits of the dissection. The senior surgeon’s approach is dynamic, using the intrinsic mobility, movement and excursion of the patient’s tissues, as well as implant measurements and dimensions. The central sternum is used as a static reference point to the more laterally placed breast and nipple areolar complex (NAC) during standard arm excursion. In the seated position, a line from the center of the nipple is marked with the arms in both the resting position and with the hands behind the head to the sternal point. The lower pole of breast skin and glandular elasticity is assessed by simulating expansion with an implant. This allows the surgeon to assess how much skin may be recruited from below the native IMF and optimal placement of the incision. The dimensions of the implant in relation to the breast mound footprint is used as another measure for establishing the new IMF position. The ideal natural appearance of the NAC is at mid-height of an anatomical form stable implant. The NAC is elevated with augmentation. Based on these markings the mid-height of the implant is determined on the chest wall. This translates to approximately 1.0 cm for small breasts, 1.0–1.5 cm for moderately sized breasts, and 1.5–2.0 cm for larger breasts.
Access Incisions
Inframammary
This is the most common and versatile access incision. It allows for clear dissection and subsequent visualization of the implant pocket. The technique may reduce the rate of capsular contracture through clear visualization and “dry” technique. This allows good hemostasis and reduced risk of bacterial contamination through the incision and operative field. Its direct access to the glandular–muscular interface and overlap allows manipulation for the best symmetrical and esthetically pleasing outcome. The drawback of this incision is the relative visibility of the scar. The incision should be placed in the predicted location of the new IMF, which has been determined and marked preoperatively. An incision length of up to 5.0–7.5 cm (depending on shape and size of the implant) is required, especially for anatomical implants. The incision should be designed lateral to the breast midline, this will place the resulting scar in the deepest portion of the new IMF.
Periareolar
The principle advantage of this incision is the resulting scar, which is usually considered well camouflaged and inconspicuous. The periareolar approach allows easy adjustment of the IMF and direct access to the parenchyma for scoring and release when the lower pole of the breast is constricted. Whether this is a transparenchymal approach, or periparenchymal via dissection to the inferior pole, the incision allows for access and creation of subglandular, and submuscular pockets. In addition to the higher risk of contamination and capsular contracture rates, it is associated with reduced sensitivity of the NAC as well as increased rates of periareolar pain. It is also more likely to interfere with lactation and breastfeeding.
In pigmented skin the risk of keloid and/or depigmentation remains high. It is also exceedingly difficult to place many cohesive or anatomically shaped implants through this incision. Despite the evidence, this access is used by many surgeons who appreciate the “direct” access, “remote” control of the IMF and “invisible” incision. In the opinion of the authors, the basic science and clinical series evidence suggests there is no justification for using the periareolar incision.
Transaxillary
This was first developed in the 1970s as an open technique, before being adapted to a minimally invasive approach. Although this technique theoretically offers access and creation of all three implant pockets, the traditional open technique involved blunt dissection of the inferior origins of the pectoralis major muscle, for creation of the submuscular pocket. The open blunt technique led to a series of suboptimal esthetic outcomes, due in part to uneven coverage of the implant and its malposition owing to a lack of controlled dissection of the vascular inferomedial aspect of pectoralis major. In this space lie the major intercostal vessels, which increases the risk of hematoma. As saline implants became popular, especially in the United States, proponents of this technique increased. Introduction of endoscopic assistance allowed the insertion of saline implants through a 2–3 cm incision, enabling precision view, thus becoming a new standard. Currently, the technique is performed with a slightly longer incision of 4–5 cm. Although textured implants can be placed via the incision, anatomical-shaped implants cannot. The IMF is lowered 1–2 cm depending on size and asymmetry. Proponents of this technique support its safety, small incision hidden in the axilla, remote access, and better control of the IMF. Despite evidence suggesting this incision increases capsular contractures, the authors believe that this technique is more anatomically and surgically sound than the periareolar technique, but less versatile than the inframammary incision. However, the purported control over the IMF is offset by the fact that there is little or no possibility for a nuanced manipulation of the gland–muscle interface or position at the IMF. This is imperative in the view of the authors, as it gives a more direct control of the NAC to IMF distance as well as the shape of the lower pole. However, Tebbetts reports excellent equivalent outcomes with dual plane and transaxillary approaches at long-term follow-up.
Transumbilical
This technique was developed in the 1990s, and the first large series reported by Dowden argued for its use based on its versatility (access to both subpectoral, as well as subglandular pockets), minimal scarring with no implant tension on the wound, reduced loss of nipple sensation, elimination of dead space, and good control of the implant pocket. This technique has limited application and is only suited for inflatable saline implants. It involves blunt dissection over the rectus sheath toward the breast. Creation of the subglandular or subpectoral pockets is achieved by avulsion of the tissues, including the inferior pectoralis major (for the subpectoral pocket). Although good results have been reported by Dowden and other surgeons, the authors feel this is not a surgically sound technique. It has poor visibility of the operative field. Its safety, based on basic surgical principles, cannot be recommended in comparison to the other incisions.
Pocket Creation
Subglandular
The original implant pockets were dissected in a subglandular plane. Where the soft tissue envelope is considered thick enough, many surgeons continue to use the subglandular pocket for implant placement.
Submuscular
This approach was first developed to reduce the risk of capsular contractures, the theory and clinical supportive evidence being that increased movement over the implants disrupt capsular contracture formation. However, these are associated with implant animation superiorly on pectoralis major contraction independent of the overlying breast tissues.
Dual Plane
This approach is developed in either a complete or partial submuscular pocket. The lower edge of the pectoralis major is dissected off the chest wall and lower sternum. For a total submuscular pocket it may be necessary to also dissect serratus anterior and the external oblique from the chest well. The issues of animation of the implants with contraction, the implants riding too high, and the wide cleavage are well-known drawbacks of the submuscular plane. By definition, with this technique, it can sometimes be difficult to place the NAC on the most projected part of the breast tissue.
Tebbetts described the dual-plane technique as a means of addressing the various shortcomings of each technique and allowing a nuanced approach to implant placement and pocket dissection. He described dissection of tissue in both submuscular and subglandular planes. The lower edge of the pectoralis major is dissected, followed by dissection of the breast tissue from the underlying pectoralis major. He described three different levels of dissection of the breast tissue–muscle interface and redraping of the pectoralis major muscle on the implant as the projected NAC level ( Fig. 35.1 ):
- •
Type I: No dissection, the inferior border of the muscle covers the implant and entire NAC level.
- •
Type II: The inferior border of the muscle drapes over the implant to the level of the NAC.
- •
Type III: The inferior border of the muscle retracts to the superior margin of the NAC.
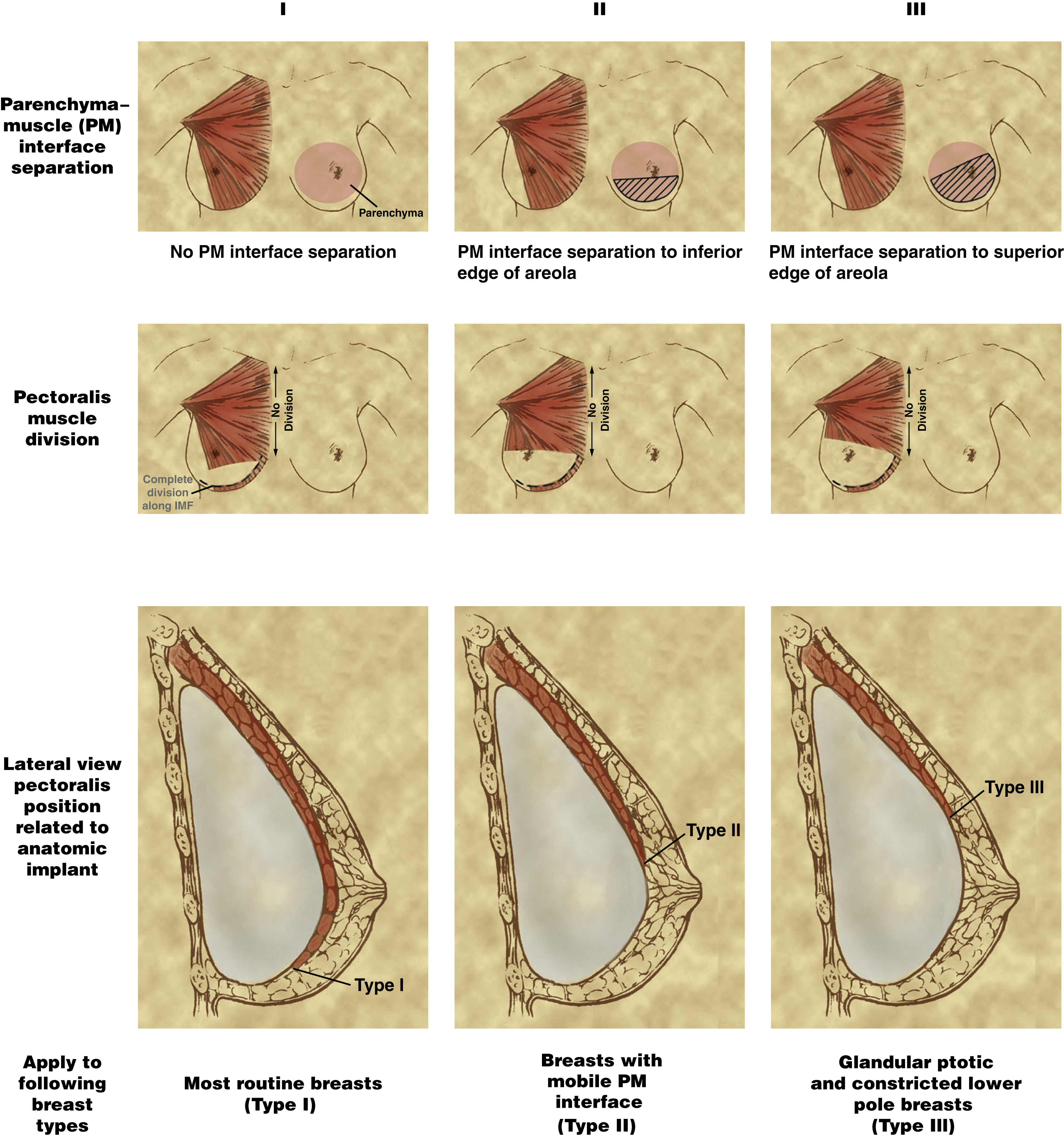
Utilization of dual-plane technique provides the most flexible and nuanced approach for implant positioning and placement. It mostly alleviates implant animation problems due to muscle contraction by allowing good soft tissue coverage of the upper pole. Dual-plane technique also facilitates expansion of the lower pole and eases adjustment of the IMF should it be necessary. In addition, this technique has been shown to reduce capsular contracture rates.
Subfascial
Subfascial plane is a more recent technique. Here the implant is placed under the pectoralis major fascia which results in very low or no capsular contracture. The authors have little experience with this technique, however the senior author has extensive experience with pectoralis major in reconstructive settings and we contend that the pectoralis major fascia is rather thin and would not alleviate the issues of implant animation or optimal NAC position on the breast mound.
Sterile Technique, Betadine, and Adams’ Solution
Authors’ Preference
The general principles of surgery need to be observed even more stringently with the use of prosthetic material. Once informed consent has been obtained by the surgeon, decisions relate to access incision and pocket creation, use of sterile technique, no touch approach, and perioperative antibiotics.
The patient is then placed in the supine position, centered on the operating table with the pelvis directly over the flexion point of the bed. The arms are tucked in with adequate ulnar nerve protection. This way the patient may be placed in the upright seated position as needed during the procedure. Sterile preparation and draping of the anterior chest must provide visualization of the patient’s shoulders as an important anatomical reference point. Triple antibiotic solution irrigation is used for all implant cases regardless of the incision type.
The authors use the IMF incision approach in all procedures. The senior author in most cases creates a dual-plane II–III pocket with silicone implants. The pocket is precisely dissected to snugly accommodate the implant. After creation of the subglandular plane, the lateral border of the pectoralis major muscle is identified. The muscle edge can be lifted by forceps to allow easy entry into the submusculofascial plane. This plane is readily identified by the loose areolar connective tissue and ease of dissection. An extended electrocautery instrument is used to complete the dissection. The inferior origin of the pectoralis major is released from lateral to medial at the level of the IMF. Division of the pectoralis continues medially to the sternal border. The internal mammary vessels in the subpectoral dissection pocket are visualized and cauterized as the dissection is extended medially maintaining a “dry” dissection field. Partial deep division may selectively be carried out 1–3 cm above the xiphoid, depending on implant shape and size. Lateral dissection is avoided in order to let the implant push the pocket out itself and to reduce the risk of injury to the lateral neurovascular bundles. The nerves can be stretched to accommodate the implant but should be preserved to minimize postoperative sensory changes. When the pectoralis major muscle is elevated, care must be taken to leave the pectoralis minor down on the chest wall. This will minimize bleeding and allow correct placement of the implant. Once adequate hemostasis is obtained and pocket dimensions are finalized, the pocket is irrigated with betadine first and then antibiotic-containing solution, and the implants are carefully placed by a minimal-touch technique and change of glove. Repeated use of implant “sizers” is avoided to reduce unnecessary pocket manipulations and possible contamination. A multilayer closure is performed with absorbable suture. It is important to close off the implant pocket with a separate layer of suture before closing the skin prior to application of skin glue. No drains are placed, the NAC is covered with film and chloramphenicol antibiotic ointment ( Figs. 35.2 and 35.3 ).