Vulvodynia is a multifactorial chronic pain disorder that is distressing to the patient and exigent to the physician. Although the condition is common, it remains little understood, so patients remain undiagnosed and untreated or undertreated for many years. Although multiple therapies exist in the treatment of vulvodynia, few randomized controlled clinical trials have been performed. Thus, treatment should be individualized and tailored to a patient’s diagnosis, symptoms, and psychosexual functioning. Patient education is also important and is facilitated by patient brochures providing assurance that vulvodynia is a real disease.
Vulvodynia is defined by the International Society for Study of Vulvovaginal Disease (ISSVD) as “vulvar discomfort, most often described as burning pain without relevant visible findings or a specific, clinically identifiable, neurologic disorder.” Patients with vulvodynia often describe it as chronic vulvar burning, stinging, irritation, rawness, and, rarely, pruritis. It may be felt only during sexual intercourse, experienced continually, or triggered by nonsexual activities such as walking. Although believed in the past to be an uncommon condition, vulvodynia is a major contributing cause for patient referral. Data from a population-based study funded by the National Institutes of Health found that 15.7% of women reported lower genital tract discomfort persisting 3 months or longer. Recent population-based studies show estimates as high as 28%, with 1 study showing that 39% of women who suffer from chronic vulvar pain fail to seek treatment. Frequency of vulvodynia is underestimated partially because of the belief of the medical community that this problem is psychological and thus is not in their realm, and also because affected women are reluctant to discuss their symptoms because of fear of neglect. Vulval pain has been highlighted as a highly prevalent condition that is associated with substantial disability. Although vulvodynia is a multifactorial pain syndrome in which psychological, social, and sexual function interact, it is a diagnosis of exclusion, in which treatable causes such as dermatoses, infection, neoplasia, and neurologic disorder must be ruled out; patients must be properly classified and also appropriate education and psychological support/counseling must be administered. As vulvodynia receives increased attention by both the medical profession and the media, more women are seeking care, information, and guidance.
Causes
Infection and Vulvodynia
The causes of vulvodynia are unknown; however, several hypothesis have been proposed to identify causative factors. One of the most consistently reported clinical findings associated with the onset of vulvodynia is a history of frequent yeast infections. A chronic subclinical yeast infection was believed to play a role in the development of symptoms, but the use of antifungal medications has been shown to be inadequate for patients with undocumented yeast infection. It is not clear whether the culprit is the yeast itself, the treatments undertaken that can sensitize the tissue, an underlying sensitivity present in the tissue, or simply the most common diagnosis made for unexplained symptoms. A recent study suggested that diverse urogenital infections such as yeast infection, urinary tract infection, trichomonas, and human papilloma virus (HPV) may precede the onset of vulvodynia, with multiple assaults significantly compounding risk. However, this has not been a consistent finding within other studies, and prospective studies documenting urogenital infections in association with vulvodynia are warranted.
Although HPV was initially reported as a frequent cause of vulvodynia, testing for HPV has shown that this virus is absent in most women with vulvar pain. One recent study observed that the low rate of observed infection in women with vulvodynia, and the diversity of HPV types detected in the patient population studied, suggest incidental virus carriage rather than direct cause and effect.
Genetic Factors
Gerber and colleagues conducted studies on genetic predisposition and the onset of vulvodynia. They found that more affected women were homozygous for allele 2 in the interleukin-1β receptor antagonist and for allele 2 interleukin-1β gene than nonaffected women. Each of these alleles has been associated with prolonged inflammatory response. Susceptibility to vulvodynia might be influenced by carriage of this polymorphism. They concluded that these findings strongly imply that women with vulvodynia may be at increased risk for a proinflammatory immune response to be triggered by a variety of stimuli and may have difficulty in terminating an inflammatory event that involves interleukin-1β production. A similar deficit in interleukin-1receptor antagonist production has been shown to contribute to chronic inflammation in individuals with inflammatory bowel disease. The investigators also stated that some women have a genetic predisposition to develop a chronic inflammatory response after an inciting event, such as a yeast infection. The prolonged inflammation could trigger other events such as increased sensitivity in both genital and nongenital areas of the body. It has been reported that affected women have more somatic pain disorders and show increases in sensitivity to nongenital touch, pain, and temperature.
Vulvodynia as a Neuropathic Disorder
Vulvodynia has features that are characteristic of other chronic neuropathic pain conditions. These features include the persistent and burning quality of the pain, the allodynia and hyperpathia, the absence of physical findings on examination, lack of associated pathologic condition of the tissues, and strong association with depression, and are all reminiscent of other neuropathic syndromes such as regional pain syndrome (formerly reflex sympathetic dystrophy), and pudendal neuralgia. The transition from the nociceptive to neuropathic pain is key in vulvodynia because it underlies the shift from a pain disorder in which sexual intercourse elicits pain to a pain disorder that is progressive and ongoing even with the avoidance of any further intercourse. The role of neuropathic pain in vulvodynia is supported by a documented response to agents used to treat neuropathic pain. Immunohistochemistry has shown altered density of nerve endings such as the vanilloid receptor VR1 (TRPV1), which is expressed by nociceptors, and is triggered by capsaicin, noxious heat, protons, and chemicals produced during inflammation; as well as increased number of intraepithelial free nerve endings, calcitonin-related gene peptide (peptide found in nerve fibers), lowered tactile and pain thresholds, nociceptor sensitization, and overall peripheral nerve hyperplasia. Several studies report successful treatment of localized vulvodynia with botulin toxin A. Moreover, increased blood flow and erythema in the posterior vestibular mucosa have been shown in vulvodynia via laser Doppler perfusion imaging of the superficial blood flow in the vestibular mucosa. Researchers postulated that such observation is the result of both neovascularization and angiogenesis along with the release of neuropeptides from C fibers in the skin, which produces an axon reflex causing vasodilatation and increased blood flow.
Pelvic Floor Abnormalities and Vulvodynia
Most women with vulvodynia exhibit pelvic floor abnormalities. Pelvic floor performance is significantly lower in affected patients in terms of contractile and resting ability and stability and efficiency of contraction. Pelvic floor abnormality may serve as causative or aggravating factors in the development of vulvodynia. Rehabilitation of pelvic floor muscles via surface electromyography has been successful in reducing pain and increasing sexual interest, pleasure, and activity.
Hormonal Influence
Hormones have a role in the genesis and continuance of many pain syndromes. Some clinic-based studies support an association between hormonal contraception and vulvodynia. The effect of oral contraceptives (OCs) on vulvar epithelium is largely unknown, however they may “alter the vaginal epithelium by promoting loss of a cyclic pattern, low karyopyknotic index and the appearance of navicular cells with marked curling and folding.” The presence of estrogen from OCs and the increased number of parabasal cells (a marker of atrophy), is unexpected. According to a recent study, women taking OCs have lower mechanical pain thresholds in the vestibular region compared with controls. Other studies suggest that there is only a modest risk of OC use and vulvodynia, and that risk is confined to women whose exposure occurred before the age of 18 years. It is often difficult to assess whether OCs truly influence the risk of vulvodynia because OC preparations are variable.
However, hormone supplementation influences sensory discrimination and pain sensitivity. Estrogen is known to affect inflammatory neuropeptides involved in chronic pain, in which the lack of estrogen is associated with increased density of sympathetic, parasympathetic, and sensory nerve fibers in the vulva, whereas acute or chronic estrogen administration may result in a decrease in the total and sympathetic fiber numbers. Moreover, estrogen regulates uterine sympathetic nerve remodeling through actions on myometrium, ganglion, and intermediary pituitary factors. Vaginal dysfunction during menopause is generally assumed to occur because of diminished estrogen-mediated trophic support of vaginal target cells. In addition, studies show that increased sympathetic innervation may change vasoconstriction and promote vaginal dryness, whereas sensory axon proliferation may contribute to symptoms of pain, burning, and itching associated with menopause and vulvodynia. Estrogen can therefore both lower the pain threshold and be a potent mediator of peripheral nerve remodeling.
Vulvodynia is a multifactorial disease. Although most clinicians find that depression and anxiety are often associated with the disease and sometimes exacerbate it, studies do not support a psychosexual dysfunction as a primary cause of vulvodynia. Data show that vulvodynia results in a significant psychosocial effect and, compared with dermatologic disorders conventionally regarded as affecting well-being, patients with vulvodynia experienced a more severe effect on quality of life. However, some experienced vulvologists are certain that psychosexual dysfunction is the major causal factor. Also, conditions such as interstitial cystitis, headaches, fibromyalgia, and irritable bowel syndrome are overrepresented in women with vulvodynia, with depression compounding, and at times worsening, the condition. Although clinical management should begin with careful examination to rule out skin disease and infection, overall psychosocial effects should also be addressed during treatment.
Diagnosis
Getting the terminology
Assessment of a woman with possible vulvodynia includes both history and physical findings. A good assessment is essential to differentiate between vulvar pain caused by an objective abnormality, and vulvodynia, in which pain is unassociated with abnormal clinical findings; to differentiate between the different subsets of vulvodynia (localized, generalized, provoked, unprovoked, or overlapping) according to ISSVD terminology ( Box 1 ). A systematic assessment is essential in both early diagnosis and appropriate management of vulvodynia. Previous descriptions of vulvodynia have grouped patients according to whether pain is provoked by coitus (vulvar vestibulitis syndrome) or generalized and neuropathic pain (dysesthetic vulvodynia). Recent terminology debates have questioned whether the term vulvodynia should be replaced by dysesthesia, and the term vestibulitis avoided because there is no inflammation in vulvodynia. Definitions of pain provocation, quality, duration, and distribution vary. Terminology developed by the ISSVD recommends standardization of the definition of vulvar pain and the classification of vulvodynia into subtypes of generalized versus localized and provoked versus unprovoked. Generalized specifies involvement of the whole vulva, and localized specifies involvement of a portion of vulva, such as vestibule (vestivulodynia), clitoris (clitorodynia), hemivulva (hemivulvodynia). Unprovoked implies discomfort that occurs spontaneously, without a trigger, provoked asserts that the discomfort is caused by physical contact, such as intercourse, clothing pressure, tampon use, cotton-tipped pressure or fingertip pressure. The ISSVD has recommended elimination of the term dysesthesia because it is an “unnecessary modification of previous terminology.”
A) Vulvar Pain Related to a Specific Disorder
1) Infectious (eg, candidiasis, herpes, etc)
2) Inflammatory (eg, lichen planus, immunobullous disorders, etc)
3) Neoplastic (eg, Paget’s disease, squamous cell carcinoma, etc)
4) Neurologic (eg, herpes neuralgia, spinal nerve compression, etc)
B) Vulvodynia
1) Generalized
a) Provoked (sexual, nonsexual, or both)
b) Unprovoked
c) Mixed (provoked and unprovoked)
2) Localized (vestibulodynia, clitorodynia, hemivulvodynia, etc)
a) Provoked (sexual, nonsexual, or both)
b) Unprovoked
c) Mixed (provoked and unprovoked)
Data from Moyal-Barracco M, Lynch PJ. 2003 ISSVD terminology and classification of vulvodynia: a historical perspective. J Reprod Med 2004;49:772–7.
Moreover, vulvodynia is a diagnosis of exclusion; it occurs in the absence of clinically identifiable findings such as active or chronic infection of the vulva, inflammation, neoplasia, spasmodic pelvic musculature, trauma, or a neurologic disorder as evident in peripheral neuropathy, pudendal neuralgia, herpes neuralgia, spinal nerve compression, and so forth. These can often be ruled out on examination. Sphincter dysfunction, weakness in the lower limbs, sensory changes such as anesthesia of the affected area, allodynia, and symmetric sensation of bilateral limbs should be assessed.
Examination
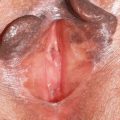
A complete genitourinary physical examination is recommended in assessing a patient with vulvodynia. The examination should include visual inspection of the external genitalia and labia for erythema, erosions, crusting, pallor, dryness and ulceration, and hypopigmentation, and it should include a speculum. A pediatric-sized speculum, and single-digit internal examination to evaluate pelvic floor muscle strength and tenderness is advised for patient comfort. A wet mount is often helpful in evaluation of vaginal secretions for yeast, pH, and white blood cells.
Candida is usually the first working diagnosis that needs to be ruled out. Itching is a predominant symptom in Candida albicans infection with secondary pain and rawness. Some organisms, such as Candida glabrata , Candida parapsilosis , Candida krusei , and Saccharomyces cerevisae , can cause symptoms similar to vulvodynia such as burning, soreness, rawness, and irritation. These organisms are difficult to detect and treat, and they are often not responsive to initial antifungal therapy. A fungal culture is imperative in diagnosis, because it is easy to miss these organisms on microscopic analysis. In contrast with C albicans , the organisms listed earlier do not produce pseudohyphae or hyphae in the vagina, but form small, budding yeast. Isolation of a fungal organism does not ensure that the cause of the symptoms has been identified, but the treatment could help in case the symptoms are related. On physical examination, creamy white curds are visible on the vaginal walls, and there is vulvar erythema, rarely with pustules.
A routine vaginal culture is indicated for a patient with vulvar pain, because some patients exhibit a heavy growth of group B Streptococcus . Although group B Streptococcus is usually an asymptomatic colonizer of vagina, many clinicians believe that it occasionally produces vulvar burning or irritation. These patients may benefit from penicillin administration.
Skin diseases of the vulva or vagina can also cause pain. Such conditions as lichen planus are a common cause of vulvar pain. Approximately 50% of women with cutaneous lichen planus have genital involvement. Erosive lichen planus is a distinct subtype of the disease, which manifests as vestibular, introitus, and vaginal erosions, resulting in inflammatory vaginal discharge even in the absence of vulvar involvement. There is also contact bleeding and marked erythema of vaginal mucosa. Erosive lichen planus is a scarring disease, therefore loss of the normal architecture of the vulva is common, and, in severe cases, results in obliteration of the vaginal canal. Inflammatory vaginitis may occasionally be caused by other erosive skin diseases, such as cicatricial pemphigoid, pemphigus vulgaris, bullous pemphigoid, erythema multiforme, and fixed drug eruption. More common noninfectious causes of vaginal inflammation include atrophic vaginitis and a clinical syndrome seen in premenopausal women consisting of diffuse vaginal erythema and vaginal secretions that are microscopically purulent and exhibit increase in immature epithelial cells. Sobel named this syndrome desquamative inflammatory vaginitis. Lichen sclerosis is another chronic inflammatory disease with a predilection for the anogenital region. It often presents with pruritis as the most frequent symptom, and clinical signs such as pallor, atrophy, fissures, and foci of hyperkeratosis. Dyspareunia is a common complaint.
Vulvar dermatitis may also present with symptoms of dyspareunia; however, the main symptom is again pruritis. It has traditionally been classified into endogenous dermatitis, such as seborrheic dermatitis, atopic dermatitis, and lichen simplex chronicus, and exogenous dermatitis from irritants and allergic contact dermatitis. However, there is often an overlap. Lichenification, excoriations, and, at times, fissures are evident on examination. Biopsy of a specific skin finding is necessary. Recent study reported that 61% of women who presented with chronic vulvar pain had identifiable disease on biopsy. The remaining 39 % had nonspecific findings. The investigators recommended performing biopsies in patients who present with vulvodynia symptoms even in the absence of skin or mucosal changes. This recommendation has been debated in the literature. However, biopsy of a specific skin finding is often best to avoid false positives, and evaluation by a trained dermatopathologist is essential.
Evaluation of Psychosocial Effects
Vulvodynia often significantly affects a woman’s psychological health. Reports of psychosocial stress are common in the literature and include depression, altered body image, impaired social relationships, altered sexual function, and difficulty in physical activities and daily activities of life. An overall decrease in quality of life is seen in women with vulvodynia.
As with other areas, conflicting studies exist regarding the psychosocial effect of vulvar discomfort. The only consistent psychological effect in women with vulvodynia was difficulty with sexual functioning. Although studies find that affected women’s physiologic sexual arousal is not impaired, because of fear of sexual intimacy from previous experiences with pain with intercourse, patients become fearful and thus sexual arousal is decreased. It is often necessary to perform a psychosexual assessment or to send patient and partner to be properly evaluated.
Treatment
The management of vulvodynia includes nonspecific supportive measures ( Box 2 ) as well as specific therapies directed toward the treatment of neuropathic pain, pelvic floor muscle dysfunction, and the psychosexual factors and sequelae ( Box 3 ).
Validate symptoms, be supportive
Treat any objective abnormalities
Topical estrogens (estradiol vaginal cream [Estrace] can be used intravaginally or topically) (conjugated equine estrogen [Premarin])
Discontinue irritants (eg, excessive washing, irritating lubricants, tight clothing, douching, nonessential medications, sanitary pads, hair dryers)
Apply lubrication during sexual activity (eg, vegetable oil, Astroglide)
Apply lylocaine 2% jelly or 5% ointment for pain 20 minutes before sexual activity
Apply cold compresses (eg, crushed ice, frozen peas, gel pack)
Address and manage depression
Offer education (including written material) for both patient and partner
Refer patient for membership in National Vulvodynia Association
Refer both patient and partner for sex therapy and counseling to help cope with symptoms
Treat abnormal visible conditions such as infections, dermatoses, and both malignant and premalignant conditions
Vulvar care measures; avoidance of irritants
Topical medications
Lidocaine 5% jelly at introitus at bedtime
Nitroglycerine
Amitriptyline 2%, baclofen 2% (±ketofen 2%)
Capsaicin
Oral medications:
Antidepressant class
Tricyclic medications (≤150 mg/d)
Venlafaxine extended release (150 mg/d)
Duloxetine (60 mg twice a day)
Anticonvulsant class
Gabapentin (≤3600 mg/d)
Pregabalin (≤300 twice a day)
Injections
Triamcinolone 10 mg/mL, 0.2–0.4 mL into trigger point
Botulinum toxin A injections
Intralesional interferon (IFN)-α (no longer used)
Pelvic floor physical therapy
Pelvic floor surface electromyography and biofeedback
Low-oxalate diet with calcium citrate supplementation (controversial)
Cognitive-behavioral therapy (CBT), sexual counseling
Surgery (for vestibulodynia only) localized excision/vestibulectomy/perineoplasty
Education
Patients who have vulvodynia often endure multiple therapeutic modalities. First, it is essential to properly diagnose and identify the pain pattern of vulvodynia. It is important to fully educate the patient and the partner and to fully explain both the condition and treatment options. Always introduce the concept early in the treatment process and warn that initial treatment is a trial of therapy. Validation of the patient’s symptoms is invaluable in treatment. Many women are convinced that their symptoms result from a yeast infection or are fearful that their symptoms signify a serious underlying medical illness or future infertility. Patients need reassurance regarding these concerns and that their symptoms are not caused by a sexually transmitted or life-threatening disease. The self-management program that the Robert Wood Johnson Medical School-University of Medicine and Dentistry of New Jersey used during a vulvodynia clinical trial introduces empowerment through individual self-management. It consists of 3 components, including a psychoeducation component that involves understanding of exacerbating and alleviating factors, mental preparation and generalized awareness of their condition, and the ability to control factors affecting the condition. Learning to manage factors was empowering to patients. The second component involved physically training the pelvic floor through understanding the physiology of pelvic pain and learning exercises to decrease the painful sensations. The third component of self-management is sexual preparation of both patient and partner, which consists of learning other forms of sexual pleasure. The study found these techniques to be highly effective because the woman empowers herself through taking control of the condition and her response to the condition.
There are almost no scientific data on the efficacy of therapies for vulvodynia. Clinical trials are primarily limited to small, open series of patients, and placebo-controlled studies are too small to yield useful data. However, because vulvodynia has gained recognition as a common and treatable entity, more studies are ongoing. A broad range of possible management strategies exists, but the trial and error approach is necessary to find the most effective treatment of a patient. The concern is that few of the treatment strategies have been confirmed in randomized controlled trials. However, therapies should not be disregarded because of lack of randomized controlled clinical trials, because this is a complicated and difficult-to-treat condition.
Vulvar Care and Topical Preparations
A variety of general nonspecific measures are available to increase the comfort level of women with vulvodynia (see Box 2 ). All potential irritants should be eliminated, including the frequent application of medications, particularly creams that contain alcohols and other irritating substances. Excessive washing of the vulvar region by patients is common, and many commercial lubricants (eg, K-Y lubricating jelly [Ortho McNeil, Raritan, NJ, USA]) may cause irritation. Astroglide (BioFilm, Vista, CA, USA) and vegetable oil are good alternatives. Xylocaine (AstraZeneca, Wilmington, DE, USA) 2% jelly (does not burn on application) and 5% ointment (brief burning sensation on application but is more potent) can help relieve the symptoms of burning in many women and, when applied liberally 20 minutes before sexual activity, may facilitate intercourse. Zolnoun and colleagues trialed 5% lidocaine ointment in 61 patients with vulvodynia, and a significant increase in patients’ ability to have intercourse was noted (76% of women reported ability to have intercourse, compared with 36% before treatment). In this study, patients applied the ointment on the cotton ball and placed it in the vestibule overnight. Patients continued to apply the preparation for 7 weeks, although some applied it for a longer period of time. Danielsson and colleagues compared application of topical lidocaine gel with biofeedback in 46 women and found improved sexual function in both groups at 12 months. Lidocaine application does have side effects, so it is important to instruct patients that transient penile numbness may occur for sexual partners and that a remote chance of lidocaine toxicity exists. A condom may decrease such side effects. Application of topical anesthetics may result in significant increase in the degree of comfort during intercourse.
The application of cold compresses or ice to the vulva may help relieve symptoms. Rinsing and patting dry the vulva after urination may be helpful. Use of hair dryers should be discouraged. Benzocaine is the anesthetic in Vagicaine (Clay-Park Laboratories Inc, Bronx, NY, USA) and Vagisil (Combe Inc, White Plains, NY, USA), but this may cause allergic contact dermatitis and should be avoided. Diphenhydramine (Benadryl; Warner Wellcome, Morris Plains, NJ, USA), present in many topical anesthetics and anti-itch preparations, is also is a common sensitizer that should be avoided.
The topical immune response modifier imiquimod (Aldara, 3M Pharmaceuticals) has been suggested as a potential therapy because of its stimulation of the cellular immune system and induction of cytokines such as IFN-α. However, this medication is a potential irritant and no clinical studies on its use in vulvodynia have been published. Topical and oral corticosteroids are not useful for vulvar pain, except in the case of accompanying inflammatory skin disease such as lichen planus.
Some clinicians anecdotally describe improvement in premenopausal and non–estrogen-deficient women by avoidance of all painful stimuli, including intercourse, for 1 to 2 months, along with the application of estrogen creams to the affected area. A study by Eva and colleagues showed decreased estrogen receptor expression in women with vulvar dysfunction; thus introduction of estrogen, both vaginally and topically, may improve both vulvar and vaginal atrophy and associated pain.
Steinberg and colleagues retrospectively evaluated the effects of capsaicin 0.025% applied daily for 12 weeks with preventative application of lidocaine. Although results showed a significant improvement of vulvar pain, the investigators did not indicate a specific number of patients in whom the therapy was effective. Moreover, given the prior application of lidocaine, it is impossible to isolate the effect of capsaicin. Murina and colleagues also studied the application of capsaicin cream preceded by an application of lidocaine, but did so with a prospective design, a higher dosage, and a longer treatment duration. Results indicated that 59% of participants reported an improvement of their vestibular pain, but the symptoms recurred 2 weeks after capsaicin discontinuation. Despite the preventive application of lidocaine, all participants indicated an intense burning sensation after capsaicin use. Care should be taken with the application of capsaicin because of its potential as a strong irritant. Topical nitroglycerin has been reported to improve symptoms associated with vulvodynia, but headache was a significant side effect of treatment.
Oral Medications
Pharmacologic therapies, both oral and topical, are a mainstay of vulvodynia management. Specific first-line therapy for most patients with either form of vulvodynia is standard therapy for neuropathic pain. A survey of 167 providers who treat vulvar pain was conducted in 2005. The most commonly used treatment of vulvodynia was tricyclic antidepressants (TCAs). There was no difference in the use of physical therapy, estrogens, injected or topical steroids, IFN, or laser therapy to treat generalized and localized vulvodynia. Respondents were more likely to use TCAs, gabapentin, and psychiatric care, and less likely to use local anesthesia and vestibulectomy.
Antidepressants
Although most antidepressants do not confer specific relief from neuropathic pain, several are useful in this regard: tricyclic medications, venlafaxine, and duloxetine. Tricyclic medications, particularly amitriptyline (Elavil, AstraZeneca) and desipramine (Norpramin, Aventis Pharmaceuticals, Bridgewater, NJ, USA), improve pain substantially in most patients who can tolerate doses of 100 to 150 mg. Patients must be counseled that, although these medications are known primarily for their antidepressant effects, they are also being used for their beneficial effects on neuropathic pain. Many information sheets provided by pharmacies currently list these medications as commonly indicated for pain (although the pain indications are not approved by the US Food and Drug Administration). Patients who believe they are receiving the medication to treat depression are likely to feel deceived and therefore may not take their medication. With amitriptyline, patients should be started on half of a 10-mg tablet and the dose should be gradually increased to minimize potential adverse reactions until the total dose is 150 mg or until symptoms are controlled, whichever occurs first. Other side effects include constipation, weight gain, urinary retention, tachycardia, blurred vision, and confusion. Serious side effects include seizures, stroke, infarctions, agranulocytosis, and thrombocytopenia. If unacceptable drowsiness or fatigue occurs, the dose can be decreased slightly and the patient given a week to acclimate before trying again to increase the dose. If the patient continues to experience drowsiness, desipramine, which is less sedating, can be substituted using the same dosing schedule (ie, target dose of 125–150 mg/d). However, desipramine is more likely to produce anxiety and tremulousness compared with amitriptyline, so, occasionally, patients might benefit from combination therapy with these 2 drugs to minimize these side effects while maintaining the beneficial effects. Other side effects of tricyclic medications include dryness of the mouth and eyes, constipation, increased appetite, and, rarely, urinary retention. Reed and colleagues showed that of 83 women taking a TCA at the first follow-up, 49 improved by more than 50%, compared with 30 of 79 not taking a TCA at follow-up. They concluded that women prescribed TCAs in general were more likely to have pain improvement compared with those women not taking these medications.
Other antidepressants have been used for pain control. These selective serotonin and norepinephrine reuptake inhibitors, such as venlafaxine and duloxetine, have been used for women with vulvodynia. These medications are often used when a patient does not respond readily to the common tricyclics, and are often used in conjunction with anticonvulsants. Venlafaxine is started at 37.5 mg daily in the morning with an increase to 75 mg daily after 1 to 2 weeks. Medication can be increased gradually to 150 mg daily. When stopping the medication, it is important to wean slowly. Duloxetine is begun at 20 to 30 mg each day and titrated to as much as 60 mg twice a day. Because most selective serotonin reuptake inhibitors can theoretically inhibit the metabolism of tricyclics, monitoring tricyclic levels in these patients is prudent.
Anticonvulsants
For women who cannot tolerate adequate doses of tricyclic medications or who fail to improve, gabapentin (Neurontin, Parke-Davis, Morris Plains, NJ, USA) may be considered. This medication is effective in diabetic neuropathy and postherpetic neuralgia at doses of 3600 mg/d or less. Anecdotally, many clinicians have reported beneficial effects of gabapentin in treating vulvodynia. Overall, this anticonvulsant is better tolerated and than tricyclic medications. However, it should be administered in divided doses 3 to 4 times a day, it is more expensive than amitriptyline, and it also has several side effects, including drowsiness, fatigue, dizziness, and ataxia. Serious reactions include leucopenia. Similar to tricyclic medications, this drug can be administered at low doses initially and gradually increased. Gabapentin comes in 100-, 300-, 400-, 600-, and 800-mg tablets. It is started at 300 mg by mouth daily for 3 days, then 300 mg by mouth twice daily for 3 days, and then 300 mg by mouth 3 times daily. It can be increased gradually to 3600 mg. Do not exceed 1200 mg in a dose and, for elderly patients, do not exceed 2700 mg/d. If there is a partial response to tricyclics, they may be continued at lower doses, such as 10 to 20 mg, in combination with gabapentin, and the combination of the 2 drugs may be better than either alone.
A similar medication, pregabalin (Lyrica), can be used, titrating to as much as 150 mg twice a day. Side effects are similar to those of gabapentin. Carbamazepine (Tegretol; Novartis Corporation Pharmaceuticals) and Topiramate (Topamax, Ortho Pharmaceutical Corporation, Raritan, NJ, USA) have also been used to treat vulvodynia.
Some women remain refractory to most commonly used oral therapy. Recent study evaluated the efficacy of a central nervous system agent, lamotrigine, which is an anticonvulsant with demonstrated benefits in both mood and pain syndromes. Lamotrigine (Lamictal, GlaxoSmithKline Pharmaceuticals, Durham, NC, USA) is an anticonvulsant and mood stabilizing agent that works by stabilizing the slow, inactivated conformation for the type IIA neuronal sodium channels, which prevents ongoing firing of action potentials in conditions of sustained neuronal depolarization. A study of 31 patients who completed the trial showed a clinically significant response to treatment with lamotrigine as shown by decreased pain scores and improved mood and anxiety symptoms. Subjects from the vulvodynia group had especially robust reductions on all measures of pain at both the 8- and 12-week visits.
Pelvic Floor Physical Therapy and Biofeedback
Although it was once a second-line treatment of vulvodynia, pelvic floor therapy has become a major therapy in the treatment of vulvodynia. Physical therapy has been shown to be efficacious in the treatment of vulvodynia and, in this author’s opinion, is essential in successful management of vulvodynia. Pelvic floor physical therapy is widely available and effective. It involves the assessment of the patient history and pelvic musculature, joints, and muscle tension. The function of related structures, such as bowel and bladder, is assessed as well. Most therapists use a weekly session focused on exercise for the pelvic girdle and floor, soft tissue mobilization, and joint manipulation.
Physical therapy has the advantage of treating the associated abnormalities that may worsen the symptoms of vulvodynia, such as joint pain, fibromyalgia, and interstitial cystitis. A study performed by Hartman and colleagues sought to identify current practice trends of physical therapists in the United States. treating women with localized, provoked vulvodynia. Assessment modalities used by more than 70% included detailed history; assessment of posture, tension in the pelvic floor, pelvic girdle, associated pelvic structures, and bowel/bladder function; strength testing of abdominals and lower extremities; and voiding diaries. Nearly 70% used exercise for the pelvic girdle and pelvic floor; soft tissue mobilization/myofascial release of the pelvic girdle, pelvic floor, and associated structures; joint mobilization/manipulation; bowel/bladder retraining and help with contact irritants, dietary changes; and sexual function. An evaluation by a physical therapist may identify and help alleviate dysfunctional aspects of the musculoskeletal system, such as the obturator internus and coccygeus muscles, and the sacrospinous and sacrotuberous ligaments. Other areas that can be targeted by physical therapy include fascial attachment and tissue tension levels of the bladder and urethra, uterine mobility, and sacrococcygeal mobility and positioning. Physical therapy treatment techniques include both internal and external soft tissue mobilization and myofascial release.
A recent retrospective study reported the response rate of 24 patients with either vulvar vestibulitis syndrome or dysesthetic vulvodynia who were treated with pelvic floor rehabilitation by the Glazer method and concomitant physical therapy. Patients with vulvodynia are more likely than asymptomatic women to exhibit increased resting pelvic floor muscle tension with fasciculation, but overall weakness. This profile probably predates symptoms and may predispose these women to the development of vulvodynia. These abnormalities are too subtle to be identified during physical examination, but they can be identified by surface electromyography (Glazer method). Biofeedback training helps patients learn exercises to strengthen weakened pelvic floor muscles and to relax these muscles, with a resultant reduction in pain. The Glazer method uses a small vaginal probe (about the size of a tampon) with electrical sensors connected to a computer. If pelvic floor abnormalities are identified by surface electromyography, then retraining the pelvic floor muscles with twice-daily exercises can be extremely beneficial. A home training device can be attached to the vaginal probe so the patient can monitor the effectiveness of her exercise regimen as a biofeedback procedure. The exercises must be performed regularly, and improvement is generally observed after several months. After 8 to 12 months, the exercises can be discontinued and patients generally retain the improvement in symptoms, although the profile of high resting tension of pelvic floor muscles, fasciculation, and weakness usually returns. A retrospective study in Italy assessed a total of 145 women diagnosed with vulvodynia who were treated with weekly biofeedback and transcutaneous electroanalgesia (TENS), in association with functional electrical stimulation and home therapy with stretching exercises for the pelvic floor. An improvement of vulvar pain was seen in 75.8% of subjects. The study concluded that pelvic floor relaxation with biofeedback and electroanalgesia is safe and effective in improving vulvar pain and dyspareunia in women with vulvodynia. Another study in Italy assessed 40 women with vulvodynia who underwent transcutaneous electrical nerve stimulation. Twice-weekly active TENS or sham treatments were delivered through a vaginal probe via a calibrated dual channel YSY-EST device. Women of both groups underwent 20 treatment sessions. The study concluded that marked improvement was shown in most women compared with placebo. Moreover, recent literature on vulvodynia also mentions a novel therapeutic approach to treatment of the disease with noninvasive cortical stimulation. This may be effective in very resistant cases.
Intralesional Injections
Clinically, botulinum toxin A blocks the cholinergic innervation of the target tissue. Recently, it has been shown to be effective in women with vulvodynia. Several studies have concluded that conditions and symptoms that are caused by pelvic floor spasms, daily pelvic pain, and dyspareunia are most likely to be improved by botulinum toxin A. Limited data regarding use for provoked vestibulodynia indicate an improvement in pain scores. A recent study in Korea examined 7 women with pain on genitalia that could not be controlled with conventional pain managements. Between 20 and 40 U of botulinum toxin A were used in each injection. Injection sites were the vestibule, levator ani muscle, or the perineal body. Repeat injections were administered every 2 weeks if the patient’s symptoms had not fully subsided. In all patients, pain disappeared with botulinum toxin A injections. Five patients needed to be injected twice; the other 2 patients needed only 1 injection. The study did not observe complications related to botulinum toxin A injections, such as pain, hemorrhage, infection, or muscle paralysis, but potential problems with botulinum toxin A injection include toxin reactions, urinary and fecal incontinence, urinary retention, and secondary treatment failure caused by antibody production. In a case study by Romito and colleagues, the 2 participants reported complete pain relief within 2 to 7 days after the injections, lasting for 5 to 6 months. Dykstra and Presthus conducted a study with 12 patients who reported a significant reduction of their pain for 8 to 14 weeks after the injection, with a higher dosage leading to a longer-lasting effect. Nevertheless, only 25% of patients reported a significant improvement of their quality of life on follow-up. The optimum dose and injection technique of botulinum toxin has not been determined, which probably explains the different conclusions of these studies.
IFN-α has been reported to be beneficial, primarily when injected locally. It was initially advocated in vulvar vestibulitis, because there was an association with HPV and vulvar pain. Recent studies have disputed that notion. However, a report of a focal depression in natural killer lymph cell activity in patients with vulvodynia supports the use of IFN. The most common regimen consists of IFN-α 1 million units injected 3 times per week for 4 weeks circumferentially at the periphery of the vestibule. In this procedure, the vestibule is divided into 12 areas as in a clock face; for example, a first injection could be given at the 6:00 position, the second injection at the 7:00 position, and so on until the entire periphery has been injected once. Patients may experience flulike symptoms such as fever, malaise, and myalgias; pretreatment with acetaminophen or ibuprofen may minimize these symptoms. In addition, patients may experience significant injection-site pain, which may be relieved by pretreatment of 20 to 30 minutes with a topical anesthetic. Improvement 1 year after IFN-α therapy is variable.
Although topical steroids do not help patients with vulvodynia, trigger point injections are useful, especially when a patient occasionally reports pain that is localized in origin. When one of these trigger points is identified, 0.2 to 0.3 mL of 3 mg/mL triamcinolone acetonide injected into the affected area may substantially improve pain within 1 to 2 weeks. An additional injection 4 to 6 months later occasionally resolves the pain permanently. Some investigators combine triamcinolone acetonide 0.1% injections with bupivacaine, with injections into a specific area or as a pudendal block. Segal and colleagues examined the effects of subcutaneous injections of betamethasone plus lidocaine administered at 1-week intervals in the vestibule. At follow-up, the participant indicated a complete relief of her pain and improvement in sexual intercourse. The efficacy of methylprednisolone injections plus lidocaine were studied prospectively by Murina and colleagues. Results revealed that 32% of participants had a complete remission of their pain symptoms, and 36% of the patients showed improvement.
Surgical Therapy
Another treatment option for patients with vulvar vestibulitis syndrome is surgical excision of the vestibule. It is commonly the last treatment option for patients with vestibulodynia because most experts believe that surgery should be reserved for long-standing cases of severe vulvar pain and after all other managements have yielded unsatisfactory results.
Three approaches are often used in the surgical treatment of vulvodynia, including a local excision (clinical identification and removal of extreme painful areas), total vestibulectomy (skin, mucous membrane, hymen, and adjacent tissue are removed, along with vestibular glands and transaction of Bartholin ducts), and perineoplasty (tissues of perineum removed ending just above the anal orifice), in which denervation of the vestibule (vestibuloplasty) has been shown to be ineffective. Many surgeons remove all areas of the vestibule, including areas that do not exhibit pain, because vestibulectomy failures result in recurrences in remaining vestibule tissue. About 85% of patients experience a cure or remarkable improvement in their symptoms after surgery. However, dehiscence, recurrence of symptoms, or worsening of pain occasionally occurs after vestibulectomy. Although vestibulectomy was once the treatment of choice for vulvar vestibulitis syndrome, the scarcity of experienced surgeons, the discomfort of the procedure, the cost, and the success of less-aggressive therapies have relegated this procedure to second- or third-line therapy.
Laser Therapy
Early reports of some forms of laser ablation described improvement in vulvodynia. However, worsening pain and recurrence are common, and laser treatment is now contraindicated in women with vulvar pain. Laser ablation of the vulvar epithelium is an alternative to the vestibulectomy, but laser therapy for vulvodynia remains a controversial issue. Reid and colleagues advocated the use of flashlamp-excited dye laser to selectively photocoagulate symptomatic subepithelial blood vessels in 168 women and the removal of painful Bartholin glands in 52 women not responsive or not suited to flashlamp-excited dye laser photothermolysis. The study showed statistically significant clinical improvement. Results of laser therapy for vulvodynia compare similarly with vestibulectomy. Complete response occurs in 62% versus improvement in 92%. Ketoprofen-neodymium:yyttrium-aluminum-garnet (KTP-Nd:YAG) laser treatment has been used in the treatment of vulvodynia, with a study examining patient response after 2 years. Sixty-eight percent reported less pain with sexual intercourse and 29% reported no change. The KTP-Nd:YAG laser and pulsed-dye laser offer the benefit of being absorbed by vasculature and thus encouraging collagen remodeling. Because angiogenesis and increased nerve density are characteristics of vulvodynia, the laser is used to disrupt this phenomenon and to advance collagen remodeling without changing the innate structure. CO 2 laser therapy for vulvodynia was not recommended in a study of 3 cases of vulvodynia after CO 2 laser treatment of condylomata acuminata or bowenoid papulosis of the female genital mucosa. Laser treatment was associated with a delay in healing and chronic pain.
Dietary Modifications
The theory that high urinary and tissue oxalate levels cause pain in some patients has led to the use of a low-oxalate diet and mealtime calcium citrate supplementation. The addition of oral calcium citrate (Citracal), 2 tablets (200 mg and 950 mg) orally 3 times a day, is used to neutralize oxalates in the urine. One theory is that oxalate may irritate the vestibulum and may be a contributing cause to vulvodynia pain over a long period. Other studies have indicated that patients with vulvodynia do not have increased oxalate levels, and that there is no correlation between oxalate levels and symptom improvement, so most vulvologists do not find dietary modification useful for vulvodynia.
Counseling
Vulvodynia is often devastating, affecting a patient’s personal relationships and quality of life. Depression is a common symptom and should be addressed. Sargeant and O’Callaghan reported that women with vulvar pain reported significantly worse mental health-related quality of live than women without vulvar pain. In their study, illness perceptions played an important role in the women’s mental health-related quality of life. The clinician should make it clear that counseling and antidepressant therapy are recommended not to treat their pain, but because they can minimize the depression and disruption of personal relationships. A referral to couples counseling is ideal and can help the patient and her partner cope with vulvodynia. Patients need to understand that referral to therapy does not meant that a patient’s condition is imagined; therapy is just another tool in the treatment of vulvodynia. Sex therapy, couples counseling, and psychotherapy can be important in proper management of patient. A therapist with expertise in sex therapy is preferred to help the couple to discover alternative types of painless sexual activity.
Behavioral interventions for chronic pain emphasize a self-management approach, deemed more effective than more conventional medical and rehabilitation therapies. CBT for vulvodynia can help to decrease pain, reduce fear and anxiety associated with pain, and reestablish satisfying sexual functioning. Kuile and colleagues evaluated the efficacy of therapy in a group format; the investigators reported that participants had a significant reduction of vulvar pain, in addition to a significant improvement of sexual satisfaction and perceived pain control. Masheb and colleagues tested the efficacy of CBT and supportive psychotherapy (SPT) in women with vulvodynia. Participants had statistically significant decreases in pain severity, with 42% achieving clinical improvement; participants in CBT reported greater treatment improvement and satisfaction than participants in SPT. Bergeron and colleagues conducted the only randomized study that examined CBT. More specifically, participants were randomized either to CBT, biofeedback, or vestibulectomy. The participants of the 3 treatment groups reported noteworthy improvements of their pain. The average pain reduction was 47% to 70% for vestibulectomy, 19% to 35% for biofeedback, and 21% to 38% for CBT. Although vestibulectomy was shown to be appreciably more successful than the 2 other treatments in terms of pain decline, the 3 treatments resulted in equal levels of improvement. Moreover, the CBT group presented a significantly lower dropout rate than the vestibulectomy group, and participants were more content with their treatment than those who took part in biofeedback.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree