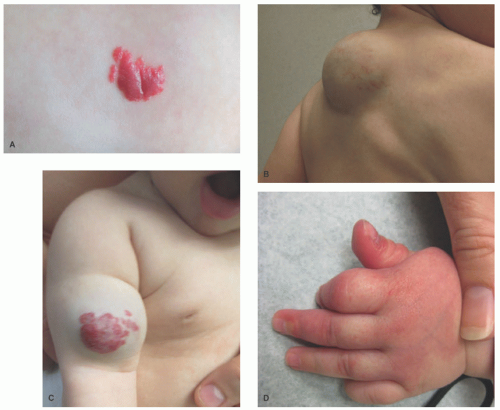
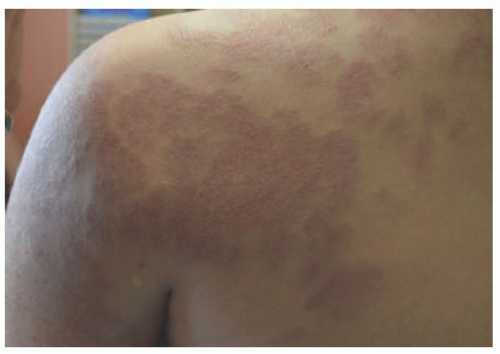
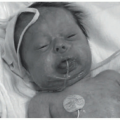
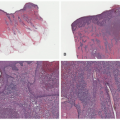
area of pallor or telangiectasia; initially, they may resemble a bruise. Hemangiomas may occur anywhere on the body but may have a predilection for the head and neck. Hemangiomas grow rapidly over the first 5 to 7 weeks of life, ultimately growth stabilizes, and IH involute or regress slowly over the course of years.4 There are several clinical subtypes, including superficial, deep, mixed, and IH with minimal or arrested growth. All have a typical clinical appearance in the skin. Superficial IH are well-demarcated bright red vascular plaques. Deep IH are bluish-hued subcutaneous nodules. Mixed IH demonstrate both a red superficial component and a deeper underlying bluish tumor. IH with minimal or arrested growth tend to favor the lower body and are often flat or telangiectatic in their appearance, with proliferation occurring only in <20% of the lesion, most often at the periphery (Figure 25-1A-D clinical subtypes). IH may be solitary or multifocal in their presentation. When they appear to occupy a region or territory of the body, for example large facial hemangiomas on the frontotemporal area, they may be associated with underlying structural anomalies as seen in Posterior fossa anomalies, large facial Hemangioma, Arterial anomalies, Cardiac anomalies, Eye anomalies (PHACE).5
TABLE 25-1. Classification of vascular anomalies: basic overview2 (ISSVA, 2014) | ||||||
|---|---|---|---|---|---|---|
| ||||||
TABLE 25-2. Vascular tumors (ISSVA, 2014) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
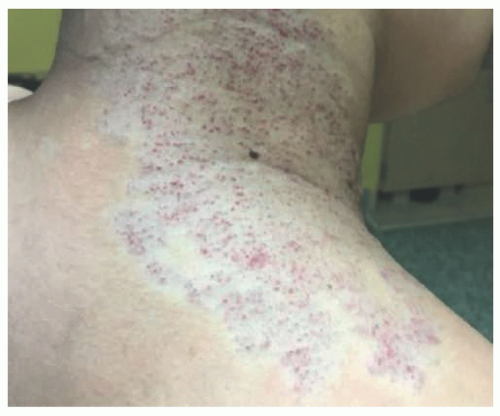
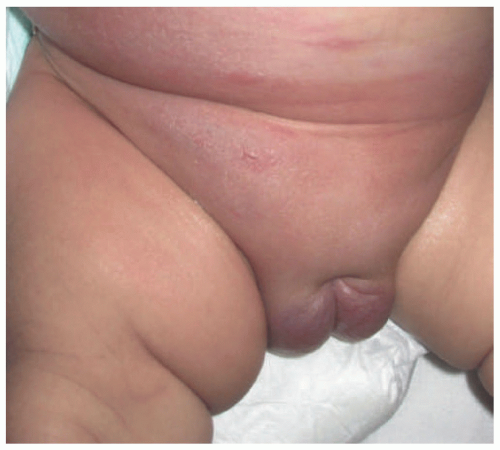
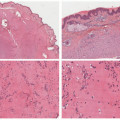
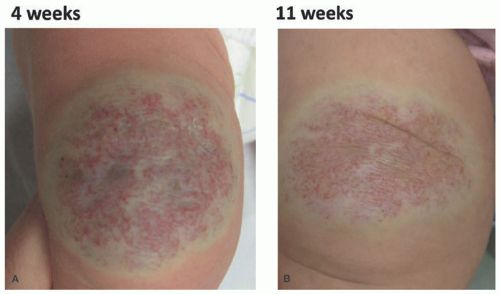 FIGURE 25-3. Rapidly involuting congenital hemangioma of the left leg (A) at 4 weeks (B) at 11 weeks. |
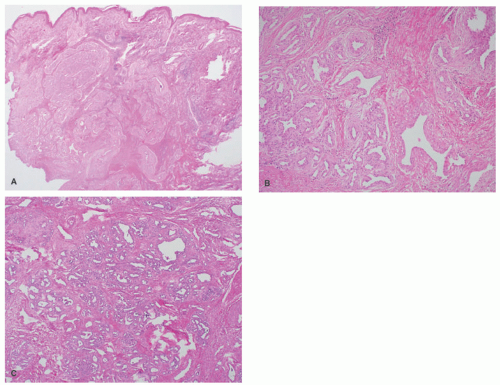
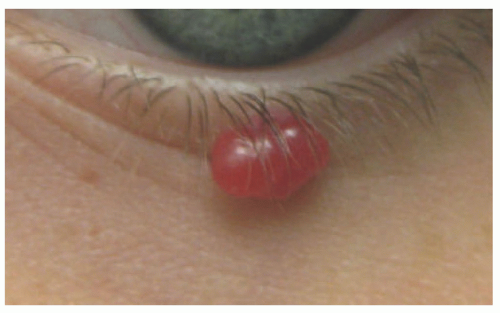
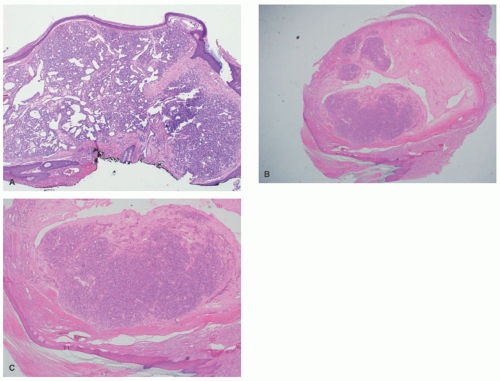
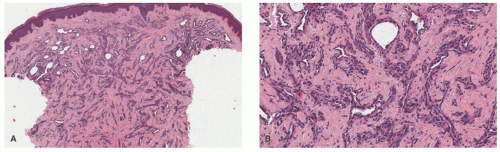
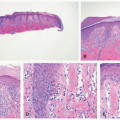
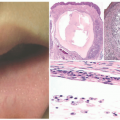
demonstrates increased fibrosis and diminished vascular lobules. Deep dermal or subcutaneous presentations of PG are not uncommon and may be intravascular. Intravascular PG presents within the lumen of a vein or artery. Intravenous PG was originally described by Cooper et al. in 1979,18 as an intraluminal polyp comprised by lobules of capillaries lined by flattened or rounded endothelial cells (Figure 25-7B). A surrounding fibrous stroma is also usually observed.19,20
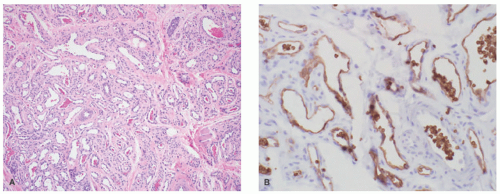
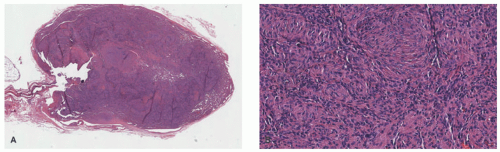
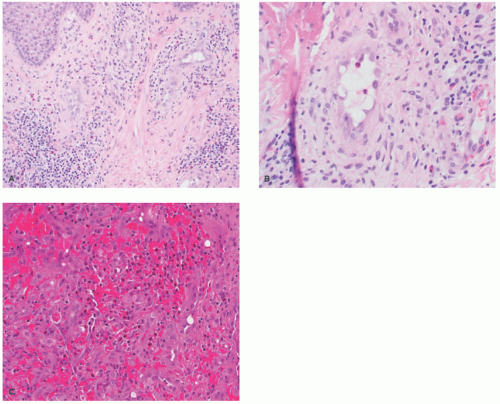
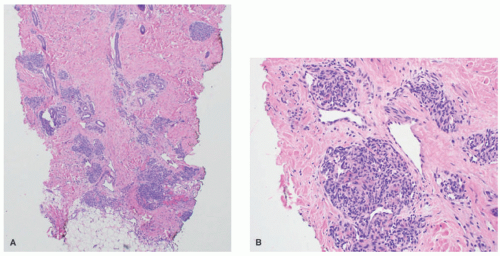
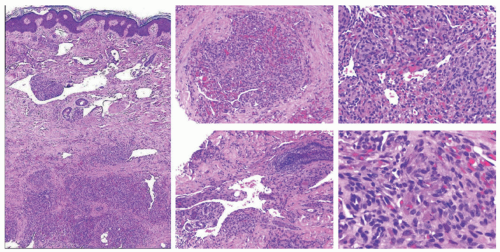
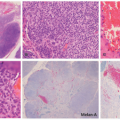
lymphoid follicles) and a lesser vascular element. More cellular EH types may mimic malignant tumors such as epithelioid hemangioendothelioma (EHE) or even epithelioid angiosarcoma (AS) (Figure 25-9C). Immunohistochemistry and molecular studies such as fluorescent in situ hybridization (FISH) can be helpful in ruling in these malignant tumors, because 90% of EHE demonstrate a characteristic WWTR1-CAMTA1 fusion, which results from a t(1;3)(p36;q25) translocation,29 and many ASs demonstrate MYC amplification on FISH and/or MYC-positivity on immunohistochemistry.30
histopathologic features and thus it is felt that they may exist together on a spectrum. On one end, TAs are considered benign and tend to be smaller in size, whereas KHE is locally aggressive and potentially life threatening. Both may be associated with a consumptive coagulopathy known as Kasabach Merritt phenomenon (KMP), which is the main source of mortality in the setting of KHE. This phenomenon does not occur in the more common entity of IH. KMP occurs because of platelet trapping within the aberrant vessels of these two unique tumors. In extensive, infiltrative lesions, more commonly with KHE, this leads to profound thrombocytopenia, hypofibrinogenemia, elevated D-dimer, and prolonged prothrombin time and activated partial thromboplastin time. Infants with KMP are at risk for life-threatening bleeding/hemorrhage.36
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree