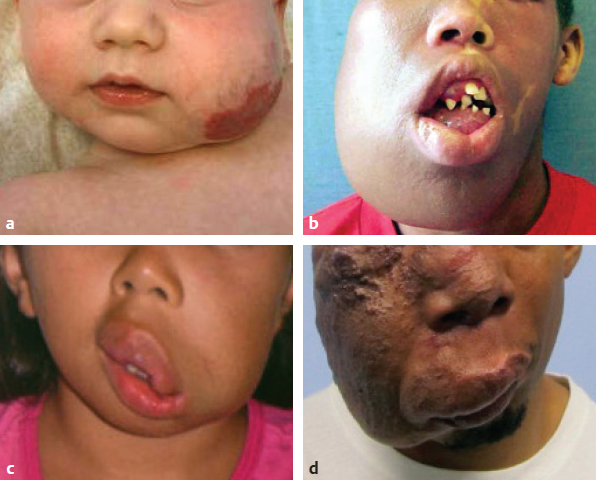
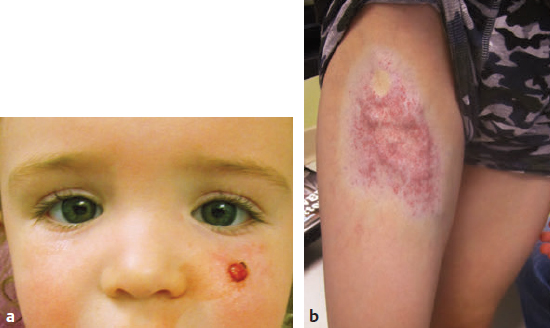
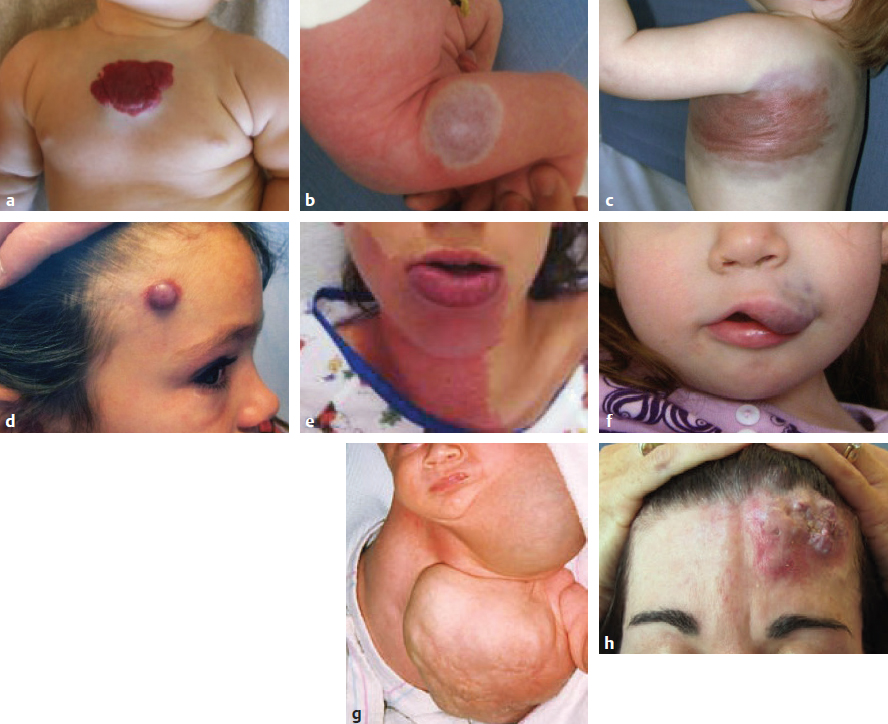
CHAPTER Vascular anomalies are disorders of the endothelium that usually present during childhood. Lesions affect all parts of the vasculature: capillaries, veins, arteries, or lymphatics. Although vascular anomalies are benign, they can cause local destruction and systemic morbidity. The field is difficult because (1) different lesions look similar and (2) terminology is confusing (Fig. 45.1). The most important principle in managing these conditions is to use correct terminology and confirm the diagnosis before intervention (Tables 45.1 and 45.2). Fig. 45.1 Vascular anomalies can look similar. These four patients each have an enlarged cheek caused by a different type of vascular anomaly. It is critical to accurately diagnose the patient, because each of the lesions has unique morbidity, prognosis, and treatment. (a) Infantile hemangioma. (b) Capillary malformation. (c) Lymphatic malformation. (d) Arteriovenous malformation. Summary Box The Most Common Problems Associated with Vascular Anomalies • Error in diagnosis • Overaggressive treatment • Managing complex patients outside of an interdisciplinary vascular anomalies center • Failure to recognize potential comorbidities associated with the vascular anomaly The most common cause of complications in the field of vascular anomalies is misdiagnosis leading to incorrect treatment (Figs. 45.2 and 45.3). Seventy percent of papers published in 2009 used the term hemangioma erroneously to describe another type of vascular anomaly.1 This is despite peer review and authors who are more likely to be familiar with this field. In practice, the diagnostic error rate likely is even higher. In our Vascular Anomalies Center, 47% of patients have an incorrect referral diagnosis; malformations are more likely to be mislabeled (54%) compared with tumors (30%).2 Patients with misdiagnosed lesions are at risk for receiving incorrect management (21% in the literature).2 For example, if a patient with a venous malformation is diagnosed with a cavernous hemangioma (this term has been abandoned), then the individual is more likely to be treated with propranolol or corticosteroid. These drugs are used to treat infantile hemangioma and have no efficacy for venous malformation. Fig. 45.2 Misdiagnosis of a vascular anomaly. (a–c) A female infant referred with a “hemangioma” of the upper lip. The patient has a venous malformation. It was present at birth, enlarged slowly, and did not regress. (a) Early infancy. (b) At 1 year of age. (c) At 3 years of age. (d–f) Infantile hemangioma. Rapid growth during infancy was followed by involution. (d) Early infancy. (e) At 1 year of age. (f) At 3 years of age. Fig. 45.3 Patients erroneously diagnosed with a “hemangioma.” (a) A young child with a pyogenic granuloma of the cheek. (b) An adolescent with a congenital hemangioma of the lower extremity. One method to reduce diagnostic errors is to manage patients in an interdisciplinary vascular anomalies center with providers who focus on these diseases. Treatment errors resulting from misdiagnosis also can be minimized by using the biologic classification of vascular anomalies and avoiding outdated terms for these lesions.3,4 With this framework 90% of vascular anomalies can be identified by history and physical examination; 10% require radiographic tests to confirm their diagnosis. Ultrasound usually is the first imaging modality used to diagnose a vascular anomaly. Ultrasound can determine whether the lesion is fast flow or slow flow and does not require sedation in children. If the diagnosis is unclear after ultrasound, the area is evaluated using magnetic resonance imaging (MRI). Rarely, a vascular anomaly cannot be diagnosed using imaging studies (less than 1%) and a biopsy is necessary to identify the lesion. Although numerous types of vascular anomalies exist, if a physician can differentiate between the 8 major lesions, he or she will be able to successfully manage at least 95% of patients5 (Fig. 45.4, Table 45.3). Fig. 45.4 Major types of vascular anomalies (a) Infantile hemangioma. (b) Congenital hemangioma. (c) Kaposiform hemangioendothelioma. (d) Pyogenic granuloma. (e) Capillary malformation. (f) Venous malformation. (g) Lymphatic malformation. (h) Arteriovenous malformation. Table 45.3 Differentiation of vascular anomalies
45
Vascular Anomalies
Avoiding Unfavorable Results and Complications in Vascular Anomalies
Error in Diagnosis
Main vascular tumor types | Characteristic |
Infantile hemangioma | Has a unique growth phase: lesions enlarge rapidly over the first few months of life and then regress in early childhood |
Congenital hemangioma | Is fully-grown at birth and has a peripheral halo |
Kaposiform hemangioendothelioma | Is typically large, is present at birth, and causes severe thrombocytopenia, bruising, and bleeding |
Pyogenic granuloma | Small, red lesion that presents in childhood and bleeds |
Vascular malformation types (present at birth) | Characteristic |
Capillary malformation | Becomes purple and thickens over time |
Venous malformation | Is bluish, enlarges with dependent positioning, and causes pain from localized thrombosis |
Lymphatic malformation | Most commonly affects the neck and axilla and can leak lymphatic fluid |
Arteriovenous malformation | Exhibits fast flow on handheld Doppler examination |
Overaggressive Treatment
After diagnostic errors the next most common cause of complications in the field of vascular anomalies is overaggressive treatment. Vascular anomalies are benign, and intervention is not mandatory. Patients can be managed with active nonintervention. The primary morbidity of vascular anomalies is psychosocial, because lesions cause a deformity. Intervention generally should be reserved for symptomatic vascular anomalies causing a deformity, obstruction, bleeding, infection, and/or pain. Generally, asymp tomatic lesions are observed unless they are at significant risk for problems. Management of a vascular anomaly should not leave a worse deformity than the appearance of the lesion. If a vascular anomaly is located on the face and is likely to cause psychosocial morbidity, I usually wait to intervene until the child is between 3 and 4 years of age, because long-term memory and self-esteem begin to form at this time.
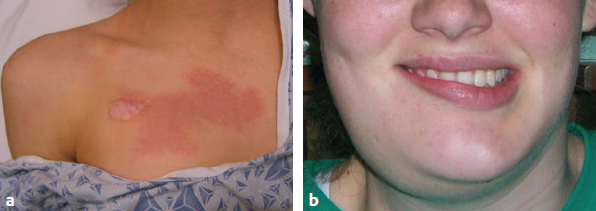
In addition to treating asymptomatic vascular anomalies, another form of overaggressive treatment is resection of a lesion that can be managed with a “less-invasive” intervention (Fig. 45.5). For example, a problematic proliferating infantile hemangioma should be treated with intralesional corticosteroid or oral pharmacotherapy (e.g., prednisolone, propranolol) rather than resection.6 Typically, first-line intervention for a problematic vascular malformation is not resection. Before excision several interventions should be considered: pulsed-dye laser (capillary malformation), sclerotherapy (lymphatic malformation, venous malformation), and embolization (arteriovenous malformation).7–10
Vascular anomalies are not malignant and do not require radical resection with negative margins. The goal of intervention is to improve the patient’s symptoms with the least amount of morbidity. Lesions have a high recurrence rate, and often the goal of therapy is to control rather than cure the deformity. If a patient has a diffuse arteriovenous malformation of the face, for example, localized resections are carried out rather than radical resection and free-tissue reconstruction, which would leave a worse deformity than the lesion. Aggressive dissections to remove as much tissue as possible generally should not be performed, because the risk of nerve injury or other problems is increased. When I improve diffuse lesions of the face I perform subtotal resections without facial nerve dissections or parotidectomies.
Managing Complex Patients Outside a Vascular Anomalies Center
Some types of vascular anomalies (e.g., infantile hemangioma, capillary malformation, pyogenic granuloma) can be managed by a single specialist focused on vascular anomalies. However, most vascular malformations (venous malformation, lymphatic malformation, arteriovenous malformation, overgrowth syndromes) require interdisciplinary care in a vascular anomalies center.2 Patients with complicated vascular malformations are more likely to receive incorrect treatment and experience complications if they are managed outside of a vascular anomalies center. Individuals with diffuse vascular malformations require the care of multiple providers (e.g., plastic surgeons, orthopedic surgeons, pediatric surgeons, interventional radiologists, neurosurgeons, and hematologists). Many major academic medical centers have vascular anomalies programs, and it is best for complicated patients to be managed in these centers.
Failure to Recognize Comorbidities
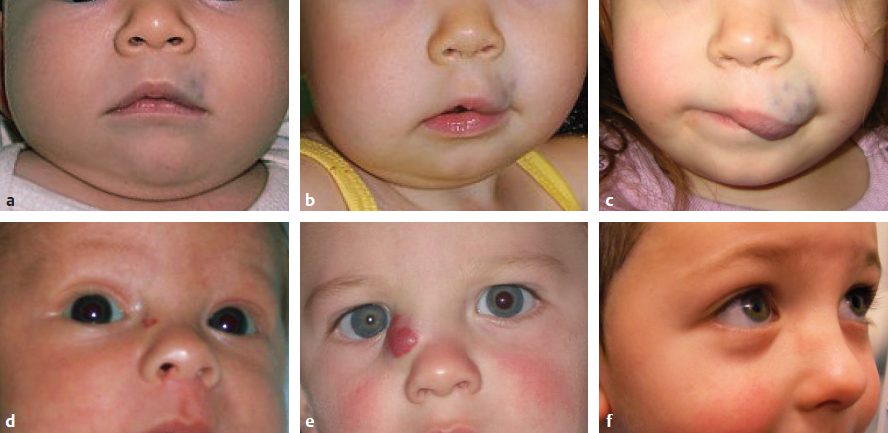
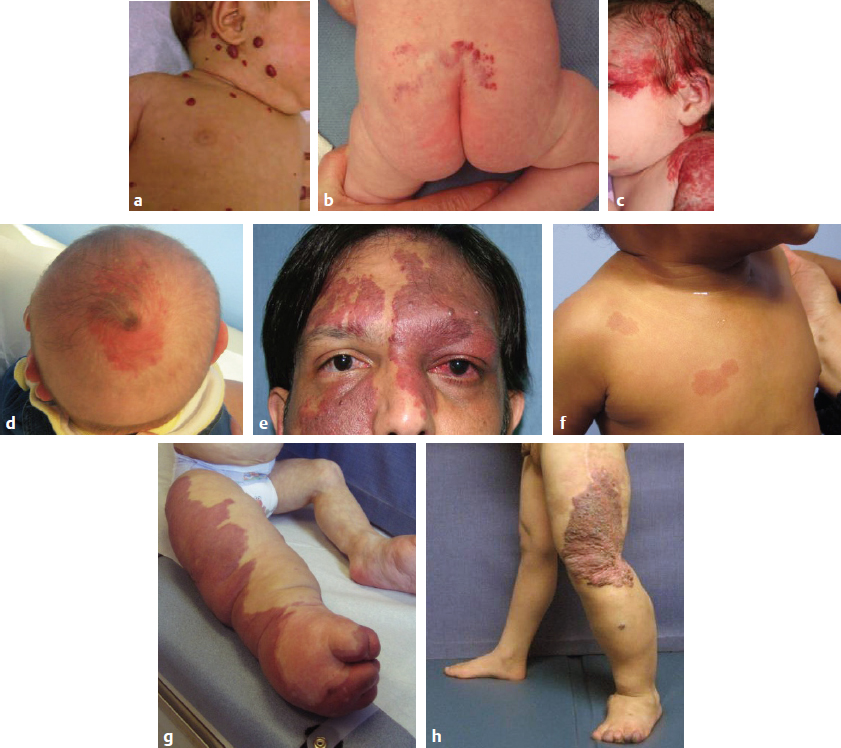
Several types of vascular anomalies are associated with underlying structural abnormalities (Fig. 45.6; see also Tables 45.1 and 45.3). If an association is not recognized, then the patient may experience a preventable complication. Patients with five or more infantile hemangiomas have a 16% risk of hepatic hemangiomas; liver ultrasound screening should be considered.11 PHACES association affects 2.3% of patients with infantile hemangioma and consists of a plaquelike lesion in a regional distribution of the face with at least one of the following anomalies12:
Fig. 45.6 Examples of vascular anomalies that have associated morbidities outside of the area of the lesion. (a) Multiple cutaneous infantile hemangiomas (five or more). (b) Diffuse reticular hemangioma of the lumbosacral area. (c) PHACE association. (d) Heterotopic neural nodule. (e) Sturge-Weber syndrome. (f) Capillary malformation–arteriovenous malformation. (g) CLOVES syndrome. (h) Klippel-Trénaunay syndrome.
PHACES
Posterior fossa brain malformation
Hemangioma
Arterial cerebrovascular anomalies
Coarctation of the aorta and cardiac defects
Eye/Endocrine abnormalities
Sternal clefting or Supraumbilical raphe
Cerebrovascular anomalies are the most common associated finding (72%). Because 8% of these children have a stroke in infancy, patients should have an MRI to evaluate the brain and cerebral vasculature. Infants are referred for ophthalmologic, endocrine, and cardiac evaluation to rule out these associated anomalies and LUMBAR association (Lower body infantile hemangioma, Urogenital anomalies, Myelopathy, Bony deformities, Anorectal malformations, Renal anomalies).13 The hemangioma is flat, reticular, and diffusely involves the midline lumbosacral area.
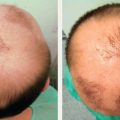
Capillary malformations can be associated with underlying anomalies. A lesion involving the parietal or occipital scalp with alopecia and a ring of long hair (“hair collar sign”) may be a heterotopic neural nodule.5 These lesions have a 50% risk of extending intracranially and contain heterotopic leptomeningeal tissue. MRI is obtained to determine whether a connection to the dura exists and, if present, the patient is referred to a neurosurgeon. Children with a capillary malformation in the first trigeminal nerve distribution are at risk for Sturge-Weber syndrome.5 Individuals undergo MRI to identify leptomeningeal vascular anomalies that can cause seizures. They are referred for ophthalmology evaluation to rule out ocular abnormalities (glaucoma, choroidal vascular anomalies). A midline capillary malformation of the posterior trunk may be associated with an underlying spinal arteriovenous malformation.14 Patients likely have CLOVES syndrome (Congenital, Lipomatosis, Overgrowth, Vascular malformations, Epidermal nevi, and Scoliosis/Skeletal/Spinal anomalies) or capillary malformation–arteriovenous malformation (CM-AVM).15 Children with CM-AVM also are at risk for intracranial arteriovenous malformations and may undergo MRI screening.
Vascular malformation overgrowth conditions should be recognized, because they may have significant associated morbidity. Individuals with CLOVES syndrome are at risk for Wilms tumor and undergo renal ultrasound screening. Patients with Klippel-Trenaunay syndrome (capillary-lymphatic-venous malformation of a lower extremity with overgrowth) may have thrombophlebitis and pulmonary embolism from a large, embryonic vein.16 Patients undergo MRI evaluation and removal of this vein.
Managing Unfavorable Results and Complications in Vascular Anomalies
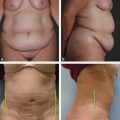
Resection of vascular anomalies have a higher complication rate compared to the removal of other benign lesions. Vascular anomalies cause local ischemia and have increased blood flow. Consequently, the risk of postoperative infection, hematoma, and wound dehiscence is elevated (Fig. 45.7). Surgeons should have a low threshold to use perioperative antibiotics, drains, and strategies to control blood loss.
After subtotal or complete resection of a vascular anomaly, it is generally best to reconstruct the area by advancing skin flaps or using regional flaps. Skin grafts have a higher risk of failure if they are placed on a recipient site that contains disease (Fig. 45.8). Grafts placed over an anomaly are difficult to vascularize because the underlying wound bed is ischemic.
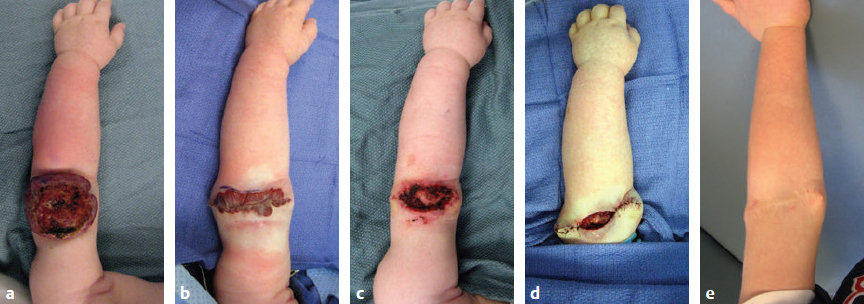
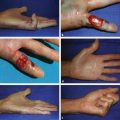
Less-invasive interventions than resection for vascular anomalies are also at risk for complications. A common consult from interventional radiology colleagues is to manage wounds. Sclerotherapy for venous and lymphatic malformations has a 5% risk of cutaneous ulceration (Fig. 45.9). Embolization of arteriovenous malformation can cause wounds. Carbon dioxide laser for bleeding lymphatic vesicles has a high risk for full-thickness skin loss. Wounds are allowed to heal secondarily; aggressive intervention to close the wound generally should be avoided. If the scar is unfavorable and causes a deformity, it can be revised at a later time.