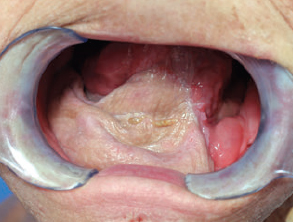
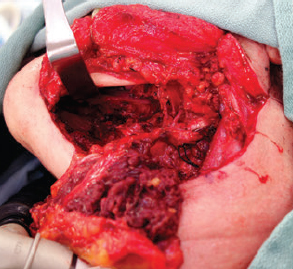
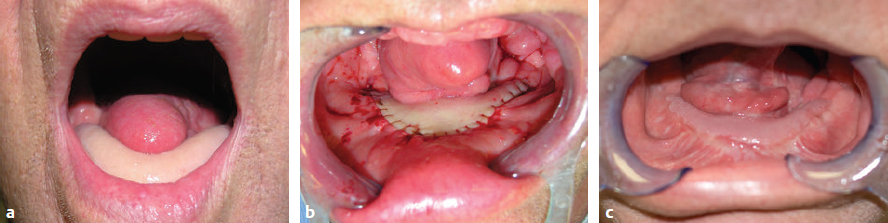
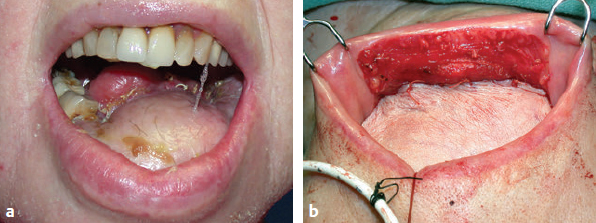
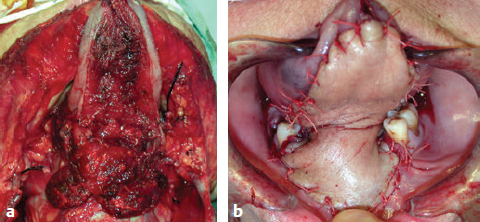
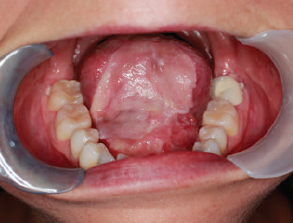
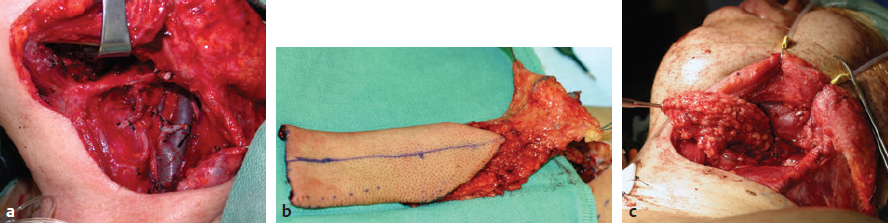
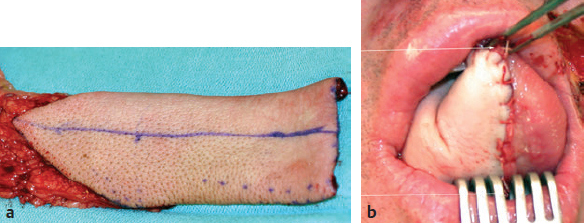
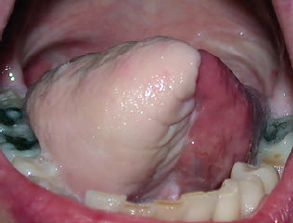
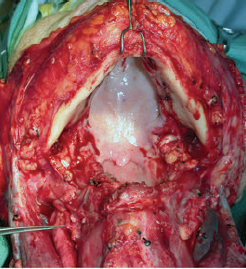
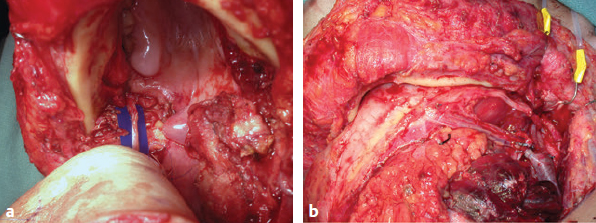
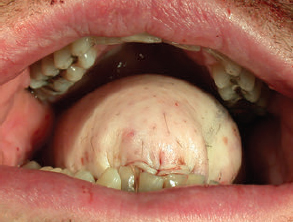
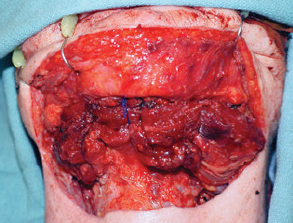
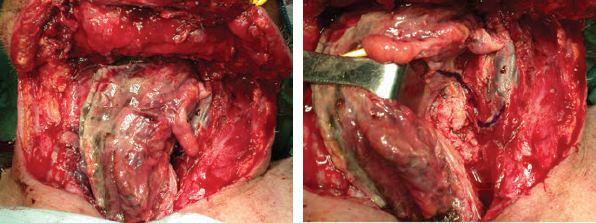
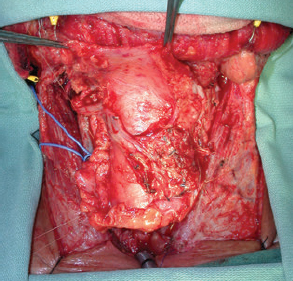
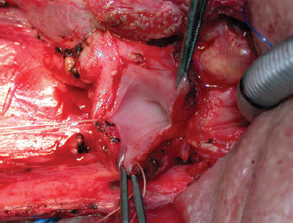
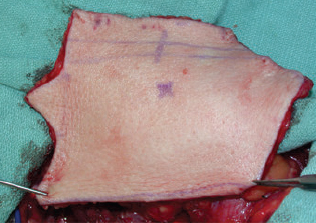
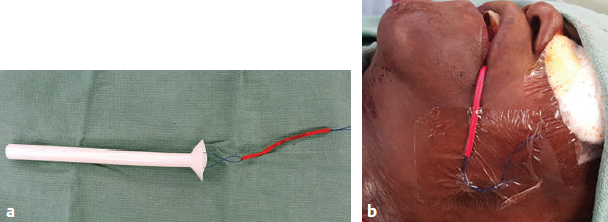
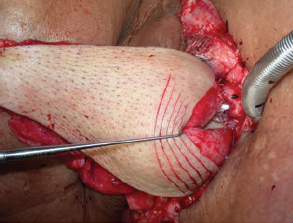
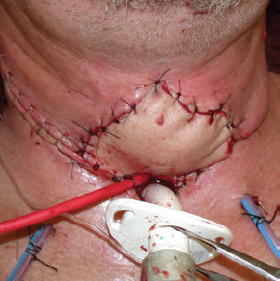
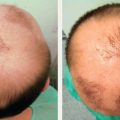
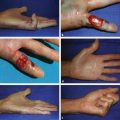
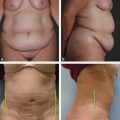
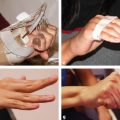
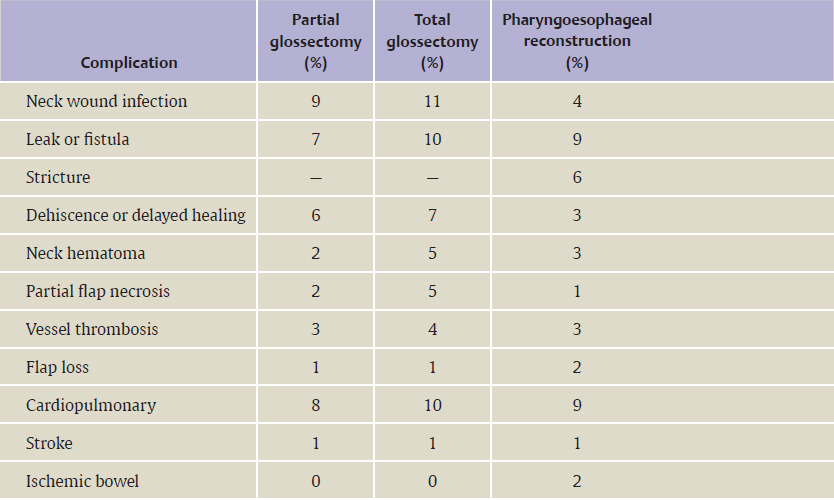
CHAPTER The oral cavity consists of the following structures: the lips, buccal mucosa, mandibular and maxillary alveolar ridge, retromolar trigone, hard palate, floor of the mouth, and anterior (oral or mobile) tongue. The pharynx is anatomically divided into the oropharynx, nasopharynx, and hypopharynx. The oropharynx is bounded by the nasopharynx superiorly, the oral cavity anteriorly, and the hypopharynx and larynx inferiorly. The main structures in the oropharynx are the base of tongue, tonsillar pillars, lateral and posterior oropharyngeal walls, and soft palate. The hypopharynx extends from the level of the hyoid bone to the lower border of the cricoid cartilage, which is congruous with the cervical esophagus below. This is a critical area that is responsible for airway protection and swallowing and speech functions. Common malignancies in the oral and pharyngeal regions include those of the lip, buccal area, floor of mouth, tongue, tonsils, hypopharynx, and larynx. The treatment of these lesions often results in surgical defects that can significantly affect the appearance and function of these vital structures. Providing basic tissue coverage for these defects without considering the functional and aesthetic outcomes is no longer acceptable in modern-day practice. Unfavorable outcomes are common in these complex reconstructions. Proper reconstruction of such defects may prevent life-threatening complications, restore essential functions such as swallowing and speech, and improve patients’ quality of life and self-image. Major head and neck surgery and free flap reconstruction pose significant perioperative risks for patients. Medical complications, not surgical complications, have been proven responsible for slow recovery, prolonged hospital stay, and poor outcomes.1 A thorough medical evaluation and consultation to the medical services before surgery are often required. Many head and neck cancer patients have a long history of tobacco and alcohol abuse. Pulmonary function should be evaluated in those with chronic obstructive pulmonary disease, and cardiovascular workup should be performed as indicated. Preoperative optimization of heart and lung functions is essential to reduce perioperative mobility and mortality. A history of peripheral vascular disease and carotid artery disease has a great impact on the flap selection, recipient vessel selection, and risks for perioperative cerebrovascular accidents. Carotid artery diseases can also be identified on head and neck CT images, which may demonstrate calcification and stenosis. Vascular specialists should be consulted when significant peripheral vascular diseases and carotid stenosis are present. Diabetes is also prevalent in this patient population and increases the risk of surgical site infection. Proper glucose control is essential. Summary Box Complications After Glossectomy and Pharyngoesophageal Reconstruction Choice of flaps is extremely important for oral and pharyngeal reconstruction. Specific defects require several considerations: • Specific flaps with appropriate thickness • Pedicle length • Ability to carry both skin and muscle • Possibility of two skin islands • Skin color match • Donor site morbidity The thickness of the flap affects not only postoperative function, but aesthetic outcomes as well. The paucity of recipient vessels may demand a flap with a long vascular pedicle to obviate vein grafting in an already-complex setting. Therefore, depending on the defects, surgical goals, and availability of recipient vessels, the choice of flap should be carefully evaluated in the preoperative planning session. The patient and family should be made fully aware of potential functional deficits, including the possibility of long-term tube feeding and tracheostomy dependency. Common donor sites such as the forearm, anterolateral thigh (ALT), abdomen, and anterior chest should be evaluated for skin quality and thickness, history of surgery or trauma, and vascular disease. Hand dominance should also be documented. A bracelet with the words “NO IV” is placed on the potential donor site arm, typically the one with the nondominant hand. Defects in the floor of mouth may vary from a small mucosal defect to a large defect involving the musculature with exposed bone. The latter often require postoperative radiotherapy, which significantly influences the methods of reconstruction. Isolated floor-of-mouth defects that do not include the suprahyoid musculature may be skin grafted or reconstructed with a facial artery musculomucosal (FAMM) flap.2,3 However, ligating and dividing the facial artery is common practice during a neck dissection. Therefore reconstruction using the FAMM flap may not be possible for patients who have had a neck dissection. When the floor-of-mouth defect extends to the upper neck, with resection of part of the suprahyoid musculature and exposure of the mandible, more extensive flap reconstruction is indicated. In high-risk patients, the pectoralis major flap is a viable option to fill the dead space in the floor of the mouth and upper neck. In young and more functional patients, however, a free flap is preferred to minimize neck deformity and maximize function. Fig. 51.1 A forearm flap can provide a thin and pliable skin for floor-of-mouth reconstruction that allows full mobility of the tongue. Fig. 51.2 When there is a large soft tissue defect in the floor of mouth, an anterolateral thigh flap with a segment of vastus lateralis muscle can provide reliable coverage and dead space filling. Fig. 51.3 (a) A bulky flap in the floor of mouth can interfere with tongue and lip function. (b) Flap thinning is possible with suction-assisted lipectomy and direct excision at the end of debulking. (c) One year later. The common problems after this type of reconstruction may include a bulky flap that limits tongue mobility and function or inadequate filling of dead space causing infection, fistula, and subsequent bone exposure that may delay adjuvant therapy and cause osteoradionecrosis. The goals are to effectively obliterate the dead space and protect the mandible from postoperative radiotherapy without unwanted bulk that may impede tongue mobility and oral function. A radial forearm flap or ulnar artery perforator (UAP) flap is ideal because they provide a thin and supple lining (Fig. 51.1). A thin ALT flap can also be used to reconstruct the floor of the mouth, with a segment of the vastus lateralis muscle used to fill the dead space (Fig. 51.2). This flap is particularly useful for large defects involving the entire suprahyoid musculature, especially after previous radiation. A shallow floor-of-mouth defect should not be covered with a bulky flap, because it may restrict tongue mobility and obliterate the lingual sulcus, causing drooling (Fig. 51.3). When the resection involves the mandibular gingiva and labial mucosa up to the vermilion border, a common mistake is to suture the flap all the way to the vermilion. This will push the lip downward and completely obliterate the labial sulcus, causing drooling, oral incompetence, and inability to eat and drink. Therefore the flap edge should be sutured to the labial tissue at the base of the labial sulcus. The remaining labial mucosal defect can be left to remucosalize or can be skin grafted. The remucosalization process takes approximately 1 week. The same principle applies to mandibular reconstruction with a fibular osteocutaneous flap when the labial mucosa defect extends to the vermilion (Fig. 51.4). Floor-of-mouth defects commonly involve the ventral tongue. A common practice is to close the tongue to itself by folding the lateral edges of the tongue ventrally and to reconstruct the floor of the mouth with a flap. This technique creates a small “snake tongue” (see Fig. 51.3a) that often causes impairment of speech and swallowing functions. A better approach is to resurface the ventral tongue and the floor of the mouth simultaneously with a thin forearm flap (Fig. 51.5) to preserve the bulk and mobility of the tongue. The tongue is a highly functional organ. A common problem after partial tongue reconstruction is the tethering of the remaining tongue, limiting tongue mobility and function. The most important goal for partial or hemiglossectomy reconstruction is, therefore, to preserve the remaining tongue function. Small defects of up to one quarter or even one third of the tongue, with minimal resection of the floor of the mouth and no communication to the neck, may be closed primarily or with a skin graft (Fig. 51.6). Resection of the floor of the mouth combined with a neck dissection creates a connection between the oral cavity and the neck along the lingual cortex of the mandible. These defects are best reconstructed with a flap to eliminate the dead space, minimizing the risk of infection and fistula formation. The remaining tongue has the ability to move intelligibly as long as it is not tethered. A bulky flap will push the remaining tongue to the side and obstruct its mobility. Therefore a thin and pliable flap should be chosen. The radial forearm flap or UAP flap is usually the first choice. Some adipose tissue in the upper forearm may be included to obliterate the submandibular dead space (Fig. 51.7). A thin ALT flap with a strip of the vastus lateralis muscle can be a good option as well. Fig. 51.4 (a) When resection of labial mucosa approaches the vermilion border, suturing the flap to the mucosal margin may obliterate the labial sulcus and push the lower lip down, causing drooling. (b) A better approach is to inset the flap to the bottom of the labial sulcus and leave the raw surface on the lip to remucosalize. This will preserve the labial sulcus and prevent drooling. Fig. 51.5 (a) After resection of the ventral tongue, closure of the remaining mobile tongue to itself may result in a “snake tongue,” with significant volume loss. (b) An alternative approach to reconstructing the ventral tongue defect is to use a thin forearm flap, part of which is also used to reconstruct the floor of the mouth. To avoid tethering of the remaining tongue, careful flap insetting is important. The length of the ventral tongue, defined as the distance from the tip of tongue to its junction with the floor of mouth and gingiva, is approximately 5 to 6 cm. The distal end of the flap is oriented toward the tip of the remaining tongue. The distal flap width, usually 6 cm, matches the ventral tongue length (Fig. 51.8). Thus one corner of the distal flap is sutured to the tip of tongue, and the other corner to the gingiva. The rest of the flap is sutured to the dorsal tongue medially and gingiva laterally. Such insetting re-creates a nice ventral sulcus and lateral gutter (Fig. 51.9). When there is no gingiva left to attach the flap to, it can be sewn to the teeth by tying the suture knots around the teeth. A single layer of interrupted 3–0 Vicryl sutures usually suffices, although some surgeons prefer horizontal mattress sutures. It is important, however, not to tie the horizontal mattress sutures too tight, which may lead to flap–mucosa edge necrosis and fistula formation. Fig. 51.6 For partial tongue defects without bone exposure or communication to the neck, full-thickness skin grafts can provide excellent coverage with preservation of tongue mobility. Fig. 51.7 (a) Partial glossectomy with neck dissection often leaves a submandibular or upper neck dead space. A conventional radial forearm flap may not have extra tissue to obliterate the dead space. (b,c) A portion of the adipofascial tissue from the upper forearm based on a proximal perforator can be included in the radial forearm for this purpose. Fig. 51.8 For hemiglossectomy reconstruction, the goal is to preserve the remaining tongue mobility without tethering. Adequate ventral sulcus and lateral gutter should be re-created with a 6 to 7 cm wide flap. The distal width of the radial forearm flap is used to re-create the ventral sulcus of the mobile tongue to prevent tethering. Fig. 51.9 With proper flap insetting, the ventral sulcus and lateral gutter of the tongue can be re-created to achieve tongue mobility. Sensory reinnervation can improve swallowing function and patient satisfaction. This can be achieved by coapting the sensory nerve of the flap to the divided lingual nerve. Sensory recovery of the flap with this technique has been well documented. The radial forearm flap, UAP flap, ALT flap, and lateral arm flap can all potentially provide sensory reinnervation.4–9 Unlike hemiglossectomy, a total or subtotal glossectomy leaves no meaningful tongue muscle. Functional outcomes are poor, and aspiration can be a serious complication necessitating a total laryngectomy in some patients. Common problems after total and subtotal glossectomy reconstruction include inadequate bulk resulting in a “funnel tongue,” inadequate vascularized tissue filling resulting in infection and fistula formation, and risks of aspiration related to resection of all floor-of-mouth musculature (Fig. 51.10). The goal of reconstruction is to create enough bulk for the neotongue. Tissue bulk is important for reconstructing large defects of the tongue for two reasons. First, it is needed to help the neotongue touch the palate to produce better speech and push food toward the hypopharynx. Second, the tissue bulk diverts saliva and food to the lateral gutters during swallowing to minimize aspiration.10 With these goals, a bulky flap should be chosen. Some muscle is also needed to obliterate the dead space in the floor of mouth and upper neck. This makes the ALT flap the ideal choice. It also has the advantages of providing sensory and motor reinnervation (Fig. 51.11). The rectus abdominis myocutaneous (RAM) is another good option. Obese men often have more adipose tissue in the abdomen than in the thighs. The opposite may be true for women. It has been suggested by some surgeons to remove the thick cutaneous portion of the flap and use the muscle only. Muscle flaps with or without skin grafts are, however, prone to atrophy and will lose significant bulk, especially after radiotherapy, resulting in a funnel shape that allows food to pour into the hypopharynx and larynx, causing aspiration. A forearm flap is usually too thin for total or subtotal glossectomy defects. Fig. 51.10 A total glossectomy with resection of all floor-of-mouth musculature leaves no tongue muscle to function and increases the risk of aspiration because of the drop of the larynx. Fig. 51.11 Both sensory and motor reinnervation is possible with the anterolateral thigh flap with a segment of the vastus lateralis muscle for total glossectomy reconstruction. (a) The lingua nerve is used for sensory reinnervation. (b) The hypoglossal nerve is used for motor reinnervation. During flap insetting, a dome of neotongue is created so that it can touch the palate easily (Fig. 51.12). Flap insetting can be difficult in a narrow jaw, particularly when no mandibular gingiva is left. It is helpful to inset the flap before revascularization while the flap is still “empty.” In addition to a funnel tongue, drop of the larynx can cause aspiration, which often occurs during a total glossectomy because the suprahyoid musculature is removed. The risk can be reduced with laryngeal suspension. Laryngeal suspension is often performed with circumhyoid sutures (0–0 Prolene) placed through drill holes in the mentum on both sides of the midline.11 The distance between the hyoid bone and the mentum is approximately 4.5 to 5 cm (Fig. 51.13). Care should be taken to avoid compression of the vascular pedicle by these suspension sutures. Fig. 51.12 Volume is necessary for total glossectomy reconstruction to reduce the risk of aspiration and improve swallowing and speech functions. Functional muscle transfer has been successfully used to restore upper extremity functions and for facial reanimation. However, it is rarely used in tongue reconstruction and produces uncertain results. My experience and that of others12,13 have shown minimal muscle movements after a functional muscle flap such as a gracilis flap or ALT flap with the vastus lateralis muscle by coapting the motor nerve of the flap to the hypoglossal nerve. My current approach is to use the ALT flap with vastus lateralis muscle with both sensory and motor reinnervation, because these additional procedures are easy to perform and add little extra time. Without postoperative radiotherapy, reinnervation of the muscle may prevent muscle atrophy. Postoperative radiotherapy, however, may destroy the motor end plates before reinnervation occurs. As with partial glossectomy reconstruction, sensory reinnervation may improve swallowing functions and patient satisfaction.8 Fig. 51.13 After a total glossectomy, static laryngeal elevation can be achieved with circumhyoid sutures to suspend the larynx to the mandible. The difficulties of postlaryngopharyngectomy reconstruction are mainly the results of previous radiotherapy and neck surgery. This increases the risk for delayed wound healing, fistula formation, infection, carotid artery rupture, lack of recipient vessels, a combined pharyngoesophageal and neck skin defect, and tracheoesophageal puncture (TEP) complications. Functional outcomes such as speech and swallowing can be disappointing as well. In addition to being nonvocal after a total laryngopharyngectomy, postreconstruction complications such as pharyngocutaneous fistulas and anastomotic strictures may render patients dependent on tube feeding. Radiotherapy has become the primary treatment for early stages of squamous cell carcinoma in the larynx and hypopharynx. Therefore the majority of laryngopharyngectomy cases are recurrent cancers that have failed radiotherapy and, in some cases, previous neck dissection. These defects are among the most difficult to repair in head and neck reconstruction. The reconstructive goals are to provide alimentary tract continuity, protect great vessels, maximize TEP speech and swallowing functions, reduce the risk of fistula formation, and minimize neck deformity and donor site morbidity. The choice of flaps for pharyngoesophageal reconstruction is important. The flap has a great impact on defect coverage, restoration of speech and swallowing functions, simultaneous neck resurfacing and tracheostoma reconstruction, and donor site morbidity. Commonly used flaps include the ALT flap, jejunal flap, radial forearm flap, and pectoralis major myocutaneous flap.14–20 The pectoralis flap gained popularity in the early 1980s but is used less now because of advances in free flap reconstruction. In general, enteric flaps heal fast but tracheoesophageal (TE) speech is poor and donor site morbidity is higher. Conversely, fasciocutaneous flaps heal slower but TE speech is superior and donor site morbidity is minor.21,22 ALT flaps have largely replaced jejunal and radial forearm flaps in the last decade. If the ALT flap is unavailable because of a lack of perforators or is deemed too bulky, a radial forearm flap or ulnar artery perforator flap is the next choice. In otherwise healthy patients with a circumferential defect, the jejunal flap is another good choice. Contraindications of the jejunal flap include significant comorbid diseases, obesity, and a history of abdominal or gastrointestinal surgeries. The pectoralis major flap can be used for partial defects in male patients, for salvage after failed free flap reconstruction, or in very high-risk patients who cannot tolerate a free flap surgery. The advantage of ALT flaps over forearm flaps is that an ample amount of fascia, the vastus lateralis muscle, and a large second skin paddle can be included for better coverage of the soft tissue defect in the neck, thus potentially reducing the risk of fistula formation. The flap is usually partially inset before revascularization. Recipient vessel choices include the branches of the external carotid artery and facial vein. In a previously radiated and operated neck, using the transverse cervical vessels may avoid difficult and risky dissection around the carotid artery.23,24 Before closure of the neck incision, the neck is slightly flexed with the head in a neutral position. This repositioning may cause kinking or twisting of the vascular pedicle. Therefore the vascular pedicle should be carefully inspected again before closing the neck incision. The second skin paddle is then turned outward to resurface the neck, to reconstruct the tracheal defect, or for flap monitoring. To monitor a buried flap without an external skin island, I prefer externalizing a small skin island based on a second cutaneous perforator or externalizing a small amount of the vastus lateralis muscle based on a muscular branch.25 This technique is highly reliable, because three-dimensional assessment of the entire skin or muscle is possible. This second skin island or muscle is then brought out through the neck incision and covered with petroleum jelly–impregnated gauze to prevent desiccation. A 2–0 silk tie may be placed around the perforator at the skin level so that the pedicle can be easily tied when it is time to remove the monitor segment. The monitor is removed 5 to 7 days later, before the patient is discharged from the hospital. Alternatively, a handheld Doppler device can be used to monitor the perforator or the main vascular pedicle through the neck skin. The Flow Coupler (Synovis) provides an additional method of flap monitoring. With the jejunal flap, a 3 to 4 cm long monitor segment is created from the remaining jejunum based on branches of the pedicle vessels. Both ends of the bowel segment are left open to avoid distention. At the time of neck closure, the tracheostoma is sutured to the surrounding neck and anterior chest skin, and a No. 8 Shiley tracheostomy tube is placed in the tracheostoma and secured to the chest skin. Fig. 51.14 During flap insetting for circumferential pharyngoesophageal reconstruction, positioning the perforators of the anterolateral thigh flap posteriorly may cause perforator compression against the vertebrae and flap failure. Fig. 51.15 The longitudinal seam of the tubed anterolateral thigh flap is positioned more posteriorly with the perforators in the anterior location to avoid compression and kinking. When the ALT flap is tubed to reconstruct a circumferential defect, the perforators need to be carefully positioned. If they are positioned posteriorly against the prevertebral fascia, the swelling of the flap can easily compress the perforators against the hard surface of the vertebrae, causing flap failure (Fig. 51.14). During flap insetting, the proximal anastomosis between the flap and the base of tongue is usually performed first. The longitudinal seam is positioned posterolaterally at the 4 or 8 o’clock position for easy suturing of the T junction. It can be positioned at 6 o’clock if the flap is tubed first. Anastomosis starts with two corner sutures at the 3 and 9 o’clock positions. The posterior wall is then completed, followed by the anterior wall. The tubed flap is slightly twisted so that the longitudinal seam at the distal anastomosis is facing posteriorly at the 6 o’clock position. Such positioning of the tubed flap places the perforator vessels anteriorly between the 11 and 1 o’clock positions to avoid compression of the perforators against the prevertebral fascia. The entire vascular pedicle can be placed anteriorly across the flap to avoid sharp kinking (Fig. 51.15). Anastomotic strictures can result from a circular scar formation, inadequate diameter, a thick flap, postoperative radiation, or fistula. To obtain a sufficient lumen, a flap width of 9.4 cm is required to achieve a 3 cm diameter (3π) for the tubed neopharynx or esophagus.19 For partial defects, the width of the flap is calculated by subtracting the width of the remaining pharyngeal mucosa from 9.4 cm. The flap can be thinned as needed by direct excision of subcutaneous fat at the periphery of the flap. Ample subcutaneous tissue should be left around the cutaneous perforators, and 2 to 3 mm of subcutaneous tissue is left on the dermis to protect the subdermal plexus. The anterior wall of the cervical esophagus is split longitudinally for approximately 1.5 cm to spatulate the distal anastomosis (Fig. 51.16). This is important to enlarge the distal anastomosis and minimize the risk of ring stricture. On the flap side, a triangular lip is created and inserted into the longitudinal split of the esophagus to complete the spatulation (Fig. 51.17). A Montgomery salivary bypass tube with a diameter of 14 mm can be placed inside the tubed flap during circumferential reconstruction in some patients. This technique may further reduce stricture and fistula formation. The indications for the use of the Montgomery tube include a thick flap, difficult flap insetting, very low location of the transected cervical esophageal end, small cervical esophagus, or poor tissue quality because of radiation injury. The tube is inserted through the mouth before the longitudinal seam of the flap is completed. The proximal flange of the bypass tube is placed above the proximal anastomosis, and the distal end is placed in the esophagus below the distal anastomosis. A 1–0 Prolene suture is attached to the flange of the tube, brought out through the mouth, and taped to the cheek to prevent distal migration and facilitate easy removal of the tube. A 10 Fr red rubber catheter is placed over the Prolene suture to protect the oral commissure (Fig. 51.18). Fig. 51.16 To spatulate the distal anastomosis to minimize ring strictures, the anterior cervical esophagus is incised longitudinally for approximately 1.5 cm. Fig. 51.17 A triangular lip of flap is created to be inset to the longitudinal opening of the cervical esophagus to complete the spatulation. Fig. 51.18 For some patients, a Montgomery salivary bypass tube can be placed in the reconstructed esophagus to minimize the risks of leakage and stenosis. (a) A Prolene suture is placed on the flange of the tube and taped to the cheek. (b) A small red rubber catheter is placed over the Prolene suture to prevent the suture cutting through the oral commissure. If a feeding tube has not yet been placed, a Dobbhoff feeding tube is placed through the nose and flap or inside the Montgomery salivary bypass tube, and through the cervical esophagus to the stomach. The Montgomery tube is usually removed 4 to 6 weeks after reconstruction. This can be done in the clinic without sedation by pulling out the previously placed Prolene suture. In my experience, pharyngocutaneous fistulas occur in 6% of patients.19,20 Fistula rates are similar in partial and circumferential reconstructions. Pharyngocutaneous fistulas occur more commonly at the distal anastomosis than at the proximal anastomosis, most likely related to the thin, less well-vascularized cervical esophagus compared with the base of the tongue. In fact, proximal fistulas are rare with the ALT flap unless there is a dehiscence from poor tissue quality. It has been speculated that the longitudinal seam of a tubed fasciocutaneous flap or two longitudinal suture lines in a partial defect might have contributed to a higher incidence of fistula formation with the radial forearm flap. However, in my experience, the fistula rate with the ALT flap is no higher than with the jejunal flap, and no fistulas have occurred through the longitudinal suture lines. Fistulas usually develop between 1 and 4 weeks postoperatively and manifest as leakage of saliva or liquids or, in some patients, as a neck infection. Therefore any neck infection or abscess that occurs after a pharyngoesophageal reconstruction should raise suspicion for anastomotic leakage. Risk factors for fistula formation include improper suturing techniques, poor tissue quality at the anastomosis site, previous radiotherapy, lack of well-vascularized tissue protection of the anastomosis, and turbulent postoperative course. At the time of surgery, any questionable tissue in the proximal pharynx and cervical esophagus should be trimmed until well-vascularized tissue is seen. Although a watertight seal using a horizontal mattress suturing technique is advocated by some, I believe that such tight horizontal mattress suturing may actually cause necrosis of the mucosal edges and thus increase the risk for fistula. I prefer single-layer, simple interrupted 3–0 Vicryl sutures for flap insetting, taking relatively big bites, inverting the skin and mucosal edges into the lumen, spacing each suture 5 to 7 mm apart, and avoiding tight knots. To further protect the suture lines, a second layer of fascial closure is recommended. Fig. 51.19 A novice mistake is to deepithelialize a strip of skin and externalize a portion of the flap for neck coverage. This results in a high fistula rate. Once a leakage occurred in this patient, the de-epithelialized portion reepithelialized quickly because of the lack of contact inhibition, resulting in a nonhealing fistula. During ALT flap harvesting, a wider amount of fascia than skin is included in the flap. When making the anterior incision, the fascial incision is made 1 to 2 cm more medially than the skin incision. Likewise, an additional 1 to 2 cm of fascia is recruited posteriorly. After completion of flap insetting, the extra fascia of the flap is wrapped around the flap to reinforce the suture lines and cover the anastomoses. Placement of the Montgomery salivary bypass tube inside the flap may also reduce the risk of fistula in high-risk patients by protecting the anastomosis from constantly soaking in saliva. If neck resurfacing is needed but only one cutaneous perforator remains, an attempt might be made to externalize a portion of the distal flap by deepithelializing a strip of skin around the distal anastomosis (Fig. 51.19). This, however, results in a high fistula rate.19 Even with a minor leakage, the deepithelialized portion will reepithelialize, causing a nonhealing fistula. The timing for resuming an oral diet after surgery may also have an effect on fistula formation. The radial forearm flap has a reputation of having high fistula rates and having delayed fistulas that occur 4 to 5 weeks after surgery.14,15 A fasciocutaneous flap does not heal as quickly as a jejunal flap once it is immersed in saliva 24 hours a day. This may explain the high incidence of delayed fistula formation. In fact, the anastomosis may experience secondary healing, which could take 4 to 6 weeks. If an oral diet is started early, for instance 7 days after surgery, as with the jejunal flap, fistulas will likely develop with the unhealed fasciocutaneous flap. After fasciocutaneous flap reconstruction, I routinely delay oral intake for 2 weeks in nonirradiated patients and for 4 to 6 weeks in patients who have undergone radiotherapy. Before oral intake, a modified barium swallowing study is performed. If no leaks or fistulas are identified, a liquid diet is started and then advanced to a regular diet as tolerated. In the presence of a leak, oral intake is delayed, and the modified barium swallowing study is repeated in two weeks. Most leaks heal within 2 weeks. In a previously irradiated patient, the tracheostoma is at risk for necrosis and dehiscence. This could be the result of an ischemic tracheostoma itself or ischemic neck and chest skin or tight closure. The blood supply to the trachea arises laterally. Extensive lateral dissection on both sides to skeletonize the trachea should be avoided. When the tracheal stoma is short, excessive undermining of the previously irradiated anterior chest skin to reach a low tracheostoma under tension can easily cause skin necrosis, because it is compressed over the clavicular heads. An alternative approach to mature the anterior wall of the tracheostoma is to use a pedicle internal mammary artery perforator (IMAP) flap.26 Questionable neck skin around the tracheostoma should be débrided, and well-vascularized skin from the flap should be used instead. Full-thickness skin grafting over the vastus lateralis muscle to mature the posterior tracheostoma and anterior neck can provide very stable coverage. Another common problem with the tracheostoma is partial obstruction related to a bulky second skin paddle for external coverage. The skin paddle should be thinned as much as possible at the time of reconstruction. Alternatively, muscle and skin grafting can provide excellent results. When a Shiley tube is placed in the tracheostoma with a bulky skin paddle, the flange of the Shiley tube can cause compression and necrosis of the skin around the tracheostoma. To avoid this, the superior flange of the Shiley tube is trimmed and a longer tube should be used (Fig. 51.20). The flap can also be thinned using suction-assisted lipectomy several months after surgery; this method works extremely well. For tracheoesophageal voice rehabilitation, patients commonly wear a hands-free stoma button. This button needs to sit inside the tracheostoma securely. Ideally, the tracheostoma should have a circumferential flange to secure the voice button without the bowstring of the neighboring sternocleidomastoid (SCM) muscles. This can be achieved by dividing the medial edges of the SCM before neck closure and maturing the tracheostoma using a semivertical mattress suturing technique to create a flange (Fig. 51.21). A frozen or woody neck, defined as severe fibrosis in the neck without tissue planes, is the result of neck surgery (such as neck dissection) combined with radiotherapy.24 Long-standing pharyngocutaneous fistulas usually result in the worst cases of frozen neck. There are three major problems associated with managing a frozen neck during reconstruction: 1. Risk of carotid artery rupture 2. Lack of recipient vessels 3. Concomitant neck skin or tracheal defects
51
Oral and Pharyngeal Reconstruction
Avoiding Unfavorable Results and Complications in Oral and Pharyngeal Reconstruction
Preoperative Planning
Medical Evaluation
Flap Choices
Surgical Goals and Intraoperative Considerations for Specific Defects
Floor-of-Mouth Defects
Partial or Hemiglossectomy Defects
Total or Subtotal Glossectomy Defects
Funnel Tongue
Aspiration
Motor Reinnervation
Postlaryngopharyngectomy Defects
Flap Choices for Pharyngoesophageal Reconstruction
Flap Insetting
Perforator Compression or Kinking
Stricture
Pharyngocutaneous Fistula Formation
Tracheostoma Problems
Managing a Frozen Neck
Plastic Surgery Key
Fastest Plastic Surgery & Dermatology Insight Engine