Patients with recessive dystrophic epidermolysis bullosa develop numerous life-threatening skin cancers. The reasons for this remain unclear. Parallels exist with other scarring skin conditions, such as Marjolin ulcer. We summarize observational and experimental data and discuss proposed theories for the development of such aggressive skin cancers. A context-driven situation seems to be emerging, but more focused research is required to elucidate the pathogenesis of epidermolysis bullosa–associated squamous cell carcinoma.
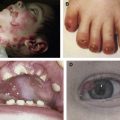
It has been apparent since 1974 that patients suffering from recessive dystrophic epidermolysis bullosa (RDEB) are at increased risk of developing life-threatening skin cancers. Comprehensive analysis of epidermolysis bullosa patients in the continental United States over a 20-year period has now translated this risk into figures pertaining to incidence, mortality, and morbidity. The data are shocking and clear: The cumulative risk of developing squamous cell carcinoma (SCC) and subsequent death in patients with generalized severe RDEB (gs-RDEB) at age 55 is greater than 90% and 78%, respectively. Such frequency of SCC development in a genetic disease is only exceeded by patients suffering from xeroderma pigmentosum, where mutations in DNA repair machinery result in dramatic hypersensitivity to sunlight and a greater than 2000-fold increase in skin cancers. The comparison with xeroderma pigmentosum is interesting, though. Although an increase in incidence is seen, with a frequency of 50% for all nonmelanoma skin cancer in patients under 10 years, metastatic disease is comparable to spontaneous SCC: Metastasis in xeroderma pigmentosum is reported at 4% compared to 2% to 4% for the general population. In xeroderma pigmentosum, internal malignancies are also increased (20-fold increase in brain tumors for instance), which is not the case in RDEB. Therefore, clear differences exist in the pathogenesis of SCC between xeroderma pigmentosum and RDEB.
A number of studies have tried to explore the reasons for such an unrivalled high incidence of aggressive SCC in RDEB patients. This review summarizes what is currently known about the pathogenesis of RDEB-associated SCC and speculates as to what the true causes of this phenomenon may be.
Dystrophic epidermolysis bullosa
Dystrophic epidermolysis bullosa (DEB) is caused exclusively by mutations in COL7A1, the gene that encodes type VII collagen. The generally accepted dogma is that patients with the most severe form of the disease, gs-RDEB, harbor premature termination codon mutations in COL7A1 . This means there is less type VII collagen present in the patients’ skin, with decreased or absent anchoring fibril formation, resulting in more severe disease. However, there are exceptions: In patients with gs-RDEB, 9.4% of identified mutated alleles were missense substitutions and 2.4% of this series of patients harbored missense substitutions on both alleles.
Cancer in epidermolysis bullosa
The first comprehensive review of the literature brought together a number of case reports and summarized that over two thirds of RDEB patients eventually die from SCC. Since this publication, additional cases have been generally reported on a case-by-case basis, although in some instances up to 10 patients have been described. Reports have also identified patients suffering from dominant DEB (DDEB) and junctional epidermolysis bullosa (JEB) who have developed SCC, although few details of metastasis or death were observed. In the largest cohort of epidermolysis bullosa patients studied to date, Fine and colleagues demonstrated solid statistics for RDEB and that SCC does occur with an increased incidence in JEB patients but not in DDEB patients. Some reports suggest that DDEB patients develop aggressive SCC. However, this evidence does not represent a comprehensive cohort study. One report of malignant melanoma in a child with RDEB can be found in the literature and three cases of melanoma were reported by Fine and colleagues in 2009, suggesting possible increased risk in childhood (2.5% in RDEB by age 12 compared to 1.35%–2.7% lifetime risk in the general population). Other tumors were reported by Fine and colleagues but not in a frequency significant enough to suggest any difference in the RDEB population; clearly cutaneous SCC is the principle cancer.
Cancer in epidermolysis bullosa
The first comprehensive review of the literature brought together a number of case reports and summarized that over two thirds of RDEB patients eventually die from SCC. Since this publication, additional cases have been generally reported on a case-by-case basis, although in some instances up to 10 patients have been described. Reports have also identified patients suffering from dominant DEB (DDEB) and junctional epidermolysis bullosa (JEB) who have developed SCC, although few details of metastasis or death were observed. In the largest cohort of epidermolysis bullosa patients studied to date, Fine and colleagues demonstrated solid statistics for RDEB and that SCC does occur with an increased incidence in JEB patients but not in DDEB patients. Some reports suggest that DDEB patients develop aggressive SCC. However, this evidence does not represent a comprehensive cohort study. One report of malignant melanoma in a child with RDEB can be found in the literature and three cases of melanoma were reported by Fine and colleagues in 2009, suggesting possible increased risk in childhood (2.5% in RDEB by age 12 compared to 1.35%–2.7% lifetime risk in the general population). Other tumors were reported by Fine and colleagues but not in a frequency significant enough to suggest any difference in the RDEB population; clearly cutaneous SCC is the principle cancer.
Open questions in recessive dystrophic epidermolysis bullosa
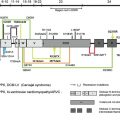
A number of unanswered questions remain concerning the pathology of RDEB-associated SCC: Is it the SCC keratinocyte itself that is tumorigenic? Is COL7A1 a tumor suppressor? Or is it the microenvironment of RDEB that predisposes to SCC alone? Other key questions need to be answered: What is the mutagen in RDEB? Why do other blistering or proliferative skin diseases not show such high incidence of SCC? And why is SCC the major cancer to affect this patient group? No single experiment or investigation has provided clear-cut answers to these questions and the data can be conflicting and controversial.
Clinical-pathological studies
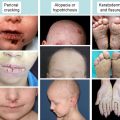
Clinically, the risk of developing SCC appears to parallel the severity and extent of ulceration and scarring of the skin, which in turn correlates with loss of type VII collagen expression and decreased or absent anchoring fibrils. However, no data are available and no detailed analysis has been carried out of the COL7A1 mutation spectrum in a large cohort of RDEB patients who have gone on to develop SCC. Such clinical, genetic, and immunohistology data could potentially shed light into the matter. Reported RDEB SCCs generally occur over joints and on the distal extremities in areas of chronic wounding and scarring, paralleling what is seen in cases of Marjolin ulcer and other genetic skin diseases predisposing to scar formation. Marjolin ulcer (reviewed by Ogawa and colleagues ) and SCC arising in burn scars (reviewed by Kowal-Vern and Criswell ) do recur and are typically more aggressive than UV-induced SCC: A mortality rate of 21% is seen in all burn-scar cancers. Burn-scar malignancies also include melanoma, but the frequency, like in RDEB, is much less than SCC. SCCs have been observed in various other chronic skin disorders, such as lupus vulgaris, discoid lupus erythematosus, necrobiosis lipoidica, and chronic radiodermatitis (reviewed by Kaplan ). Clearly however, there is a shorter latency period in RDEB (tumors developing in second decade of life as opposed to 30–50 years in Marjolin ulcer) and increased mortality is also seen in RDEB. One might speculate that this shorter latency could be due to the extent and persistence of chronic wounds and scarring in RDEB. Certainly this clinical parallel exists and has yet to be fully explored.
Other genetic skin diseases have been associated with cutaneous SCC. Anecdotal reports have linked nonbullous congenital ichthyosiform erythroderma and Darier disease, with aggressive SCC, as well as with Huriez and KID (keratitis, ichtyosis, deafness) syndromes. Up to 10% of Kindler syndrome patients, another condition associated with chronic scarring, suffer from SCC and reports of recurrence and metastatic SCC exist, but the numbers are too small for statistical analysis and incidence in these reported cases are much lower than that for RDEB.
Goldberg and colleagues postulated that repetitive tissue stress leads to tumor promotion and that this was likely to be relevant for RDEB. In support of this theory, Smoller and colleagues observed growth-activated keratinocytes in biopsies from RDEB scarred skin based on the expression of protein markers similarly regulated in psoriatic skin, such as intense suprabasal expression of K16, involucrin, and filaggrin. Six RDEB patients and four patients with epidermolysis bullosa simplex (EBS) were studied. RDEB biopsies from clinically normal, unwounded skin did not show a growth-activated pattern of antibody immunoreactivity. The investigators speculated as to whether this growth activation could be a reason for SCC development.
Such a study in RDEB patients asks: Are observed phenomena a result of the phenotype rather than a factor predisposing to SCC? The answer has not been determined; many factors related to wound healing, tissue stress, and remodeling have been shown to promote tumorigenicity in other experimental systems. To follow up on a single line of enquiry would certainly require increased numbers, more controls, and good experimental data. Demonstrating a similar scenario in other non–tumor bearing skin diseases would suggest that indeed these observations are a result and not a cause. The same logic can be applied to studies investigating the presence of natural killer cells, fibroblast growth factor (FGF), and metalloproteinase (MMP) proteins in patients with RDEB. The activity of natural killer cells, which play a key role in immune response to certain viruses and cancers, was analyzed in a cohort of 11 RDEB, 6 DDEB, 7 JEB, and 10 EBS patients, as well as in 20 normal and 12 malabsorption control subjects. Reduced activity was seen in RDEB, DDEB, and JEB patients, and in malabsorption controls. As junctional and dystrophic EB patients are frequently malnourished this could well explain the observation. Nevertheless, a reduction in circulating immune cells may well be an important compounding factor in SCC development.
Are increased levels of urine FGF–2 observed in RDEB patients a result of repetitive tissue injury or the distinct pathology of RDEB generated by mutations in COL7A1 ? Indeed, elevated FGF–2 was seen in 51% of 39 RDEB patients but also in 21% of 33 unaffected family members and 13% of 30 EBS patients. No elevated levels were seen in 12 JEB patients ruling out a response to blister formation but the identification of variation in the unaffected family members and EBS patients suggest the absence of a direct link to SCC formation. Similarly in wound healing, matrix MMPs are expressed during keratinocyte, fibroblast, and inflammatory cell migration and during connective tissue remodeling. The increase in MMP in some RDEB patients should come as no surprise given the extent of tissue remodeling that occurs. However, a polymorphism increasing the activity of the MMP1 promoter has been recently shown to modify the severity of disease in a French RDEB cohort. Whether patients developing SCC have higher levels of MMP1 expression has yet to be determined. One study has identified the expression of MMP in RDEB-associated SCC. The investigators looked at expression of MMP7, MMP13, and MMP9 using immunohistochemistry in archival samples (25 RDEB SCC, 61 UV SCC, 29 Bowens disease). MMP7 and MMP13 were present in the majority of SCCs studied and MMP9 was present in infiltrating stromal cells in all SCCs studied (RDEB and non-RDEB). The report suggests there was an increase in intensity of MMP7 staining in RDEB SCC, particularly within the invasive edge of tumors, yet distribution and incidence remained comparable to UV-induced SCC.
Expression of MUC1 (mucin-1), a cell surface protein associated with breast cancer, has been observed in 25 of 25 RDEB SCCs, 5 of 5 JEB SCCs, and 52 of 55 UV-induced SCCs, suggesting that this glycoprotein is a marker of SCC similarly expressed in epidermolysis bullosa and non–epidermolysis bullosa cancers.
In the only study of its kind to date, gene expression analysis was carried out in three RDEB patients presenting with SCC. RNA from tumor and peritumoral skin was compared with three spontaneous SCC patients. For the most part, an identical pattern of gene expression was observed except for the lack of IGFBP-3 (insulin growth factor-binding protein 3, a mediator of apoptosis) expression in RDEB SCC tumors, findings which were corroborated with protein expression in 7 of 11 RDEB SCCs compared with 1 of 21 non-RDEB SCCs. The gene expression platform used for this experiment was a nylon array spotted with around 4000 skin-related genes. This made it possible to relate the lack of discernible differences to the number of genes present. Another problem that will be experienced by such in vivo expression profiling experiments is the heterogeneity seen in SCC tumors from both RDEB and non-RDEB patients: The degree of differentiation, the degree of infiltrating cells, and the size of invading tumor keratinocyte islands will add numerous confounding factors to the expression profiles of tumor cells at the time of RNA isolation, which could well mask true differences between RDEB and non-RDEB SCC.
COL7A1 , type VII collagen, and cancer
Since the identification of type VII collagen and development of monoclonal antibodies, a number of studies have looked for expression in a wide range of tumors. The resulting data have been variable and in some cases contrasting. Initially, a study of a wide range of tumors concluded that, in general, invasive and metastatic tumors do not express extensively type VII collagen. However, exceptions to this rule exist in bladder cancer, squamous carcinomas of the lung, tumors of the head and neck region, female genital tract tumors, and in some adenocarcinomas of the breast.
In a separate study of head and neck SCC, Wetzels and colleagues noted that type VII collagen was localized to the basement membrane in all 42 SCCs studied and was also present in the cytoplasm of 36% of tumors. This finding was later confirmed by an independent group. Type VII collagen expression was reported in well-differentiated or moderately differentiated colon cancer and one study concluded that this expression was transiently involved in the progression from dysplastic epithelia to colon cancer. Conflicting data were later published identifying no type VII collagen expression in colon cancer. Type VII collagen expression was observed in neoplastic nervous system tissues and cell lines and in thyroid tumors. In prostate cancer, early stromal invasion is associated with the loss of a number of hemidesmosome-associated adhesive elements, such as laminin 5, type VII collagen, and α6 and β4 integrins. Invasive tumor cells then go on to produce basement membrane–like matrices and express related integrin receptors. In addition, anticancer drugs shown to be effective against prostate cancer down-regulate COL7A1 expression in vitro. Decreased type VII collagen has been observed in cutaneous SCC and dissolution of type VII collagen was associated with more invasive melanoma cell lines, which parallels in vivo observations. More recently, a study of esophageal SCC identified a direct correlation with type VII collagen and increased aggressiveness in tumors. Studying 109 patients, 35% presented with intracellular type VII collagen expression and patients with negative type VII collagen had an increased 5-year survival rate. This year, Kita and colleagues have confirmed these findings using messenger RNA analysis, demonstrating an increased 5-year survival rate in patients with low levels of COL7A1 (22 patients) versus those with high levels of COL7A1 messenger RNA (44 patients ). A similar study examining the messenger RNA expression of COL7A1 in colorectal cancer (n = 33), adenoma (n = 29), and normal tissue from the same patient and healthy volunteers (n = 20) noted that COL7A1 was up-regulated in both adenoma and cancer and concluded that this was an early event.
This histological and messenger RNA expression data are very difficult to distil into any conclusive statement as to the role of type VII collagen in cancer. It seems apparent that certain cancers will up- or down-regulate type VII collagen in a seemingly opposing manner. The observations in prostate cancer are interesting in that initial loss of type VII collagen leads to dysplasia while up-regulation leads to metastasis. The lack of prostate cancer in RDEB patients and the obvious metastasis of RDEB SCC would suggest this mechanism is not relevant to RDEB SCC. One explanation from all the in vivo expression data could be that there is no definitive relationship in non-RDEB cancers, and type VII collagen expression is inconsequential. To complicate matters, COL7A1 has recently been identified as a gene with a high rate of methylation silencing in breast cancer cell lines. In this study, COL7A1 was identified as being methylated in 10 of 11 and mutated in 1 of 11 breast cancer lines. Analysis of publicly available expression array data sets identified decreasing COL7A1 expression to correlate with increasing tumor grade in breast cancer. Interestingly, no COL7A1 methylation (or mutation) was identified in colon cancer lines, even though the colon cancer lines had an overall increase in genomic methylation.
Intracellular expression of type VII collagen is reported in RDEB SCC and in wounded RDEB skin, suggesting that either intracellular expression is associated with cancerous and precancerous lesions or is a transient response to wound healing. Again, whether this is truly a mechanism or a consequence remains an open question.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree