Abstract
Phototherapy represents the use of ultraviolet (UV) radiation in the treatment of skin diseases. Currently, phototherapy encompasses irradiations with broadband UVB (290–320 nm), narrowband UVB (311–313 nm), 308 nm excimer laser, UVA1 (340–400 nm), UVA (320–400 nm) with psoralens (PUVA) or alone, and extracorporeal photochemotherapy (photopheresis). Therapeutic success depends upon proper selection of the appropriate phototherapy modality for a given disease. Proper dosimetry is required to avoid acute side effects such as a sunburn reaction, blistering, and burning sensations. Despite the recent development of new drugs such as systemic immunomodulators (“biologics”), phototherapy is still a mainstay for the treatment of a variety of inflammatory dermatoses. The potential risk of skin carcinogenesis represents the major long-term side effect of phototherapy; with the exception of PUVA, the magnitude of this risk is not yet clearly delineated.
Keywords
phototherapy, broadband UVB, narrowband UVB, BB-UVB, NB-UVB, UVA, UVA1, PUVA, psoralens, 308 nm excimer laser, extracorporeal photopheresis, photocarcinogenesis, dosimetry
- ■
Phototherapy represents the use of ultraviolet (UV) radiation for the treatment of skin diseases
- ■
Currently, phototherapy encompasses irradiation with broadband UVB (290–320 nm), narrowband UVB (311–313 nm), 308 nm excimer laser, UVA1 (340–400 nm), UVA (320–400 nm) plus psoralens (PUVA) or alone, and extracorporeal photochemotherapy (photopheresis)
- ■
Therapeutic success depends upon proper selection of the phototherapy modality for a given disease
- ■
Proper dosimetry is required to avoid acute side effects such as a sunburn reaction, blistering, and burning sensations
- ■
The risk of skin carcinogenesis represents the major long-term side effect of phototherapy; with the exception of PUVA, the magnitude of this risk is not yet clearly delineated
Introduction
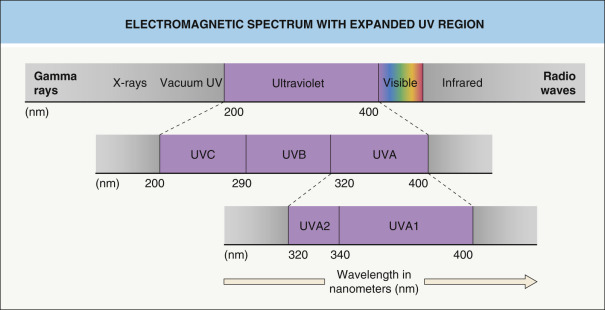
Over the past half-century, phototherapy has greatly influenced treatment concepts in dermatology. Studies focusing on the effects of electromagnetic radiation ( Fig. 134.1 ) on the skin have induced fruitful collaborations between basic scientists and clinicians.
Although ultraviolet (UV) radiation had been used for decades in the management of common skin diseases such as psoriasis and atopic dermatitis, the introduction of PUVA in the mid-1970s sparked a whole new series of discoveries, including high-intensity UV sources and selective spectra in the UVB and UVA range, e.g. narrowband UVB (311–313 nm) and UVA1 (340–400 nm).
Phototherapy With UVB
Historical Aspects
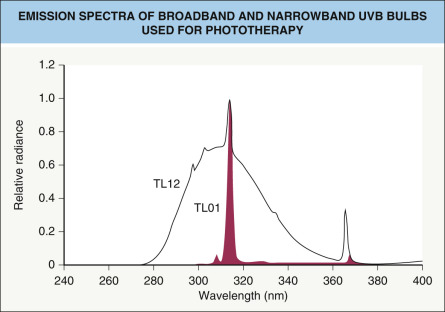
To date, the only dermatologist to be awarded a Nobel Prize in medicine was Dr Niels Finsen in 1903. The award recognized his demonstration of the beneficial effect of UV light on lupus vulgaris, and he eventually became known as the “father of photomedicine”. The combination of topical crude coal tar followed by UV irradiation for the treatment of psoriasis was introduced by Goeckerman in 1925 and became a standard therapy for psoriasis for half a century, particularly in the US. In the 1970s, it was observed that broadband (BB) UVB radiation alone, if given in doses that produce a slight erythema, could clear the milder forms of psoriasis, particularly the seborrheic and guttate types. A major advance was the development of fluorescent bulbs that emitted narrowband (NB) UVB radiation at 311–313 nm in the mid-1980s. This narrow spectrum is superior in clearing psoriasis and thus represents the most effective and most frequently used UVB phototherapy for psoriasis; it is also beneficial for a variety of other dermatoses that were previously treated with PUVA (see below).
Principles and Mechanisms
UVB phototherapy refers to the use of artificial UVB radiation without the addition of exogenous photosensitizers. The radiation is absorbed by endogenous chromophores, and photochemical reactions involving these UV-absorbing biomolecules mediate a variety of biologic effects, ultimately leading to the therapeutic effects. The most important chromophore for UVB is nuclear DNA. Absorption of UV by nucleotides causes the formation of DNA photoproducts, primarily pyrimidine dimers (see Ch. 86 ) .
UVB exposure reduces DNA synthesis and thus is used to suppress the accelerated DNA synthesis found in psoriatic epidermal cells. UVB also induces the expression of the tumor suppressor gene TP53 , and this can lead to either cell cycle arrest (allowing time for DNA repair) or apoptosis of keratinocytes (“sunburn cells”) if the DNA damage is too severe to be repaired. Through these mechanisms, p53 prevents photocarcinogenesis.
In addition to its effect on the cell cycle, UV induces the release of prostaglandins and cytokines. Interleukin (IL)-6 and IL-1, for example, seem to play important roles in producing the systemic symptoms of UV phototoxicity (sunburn) and immune suppression, respectively . These responses may, however, prove to be equally important for therapeutic effectiveness.
There is also increasing evidence that UV radiation can affect, in addition to DNA, extranuclear molecular targets located in the cytoplasm and cell membrane. These targets include cell surface receptors, kinases, phosphatases, and transcription factors. It was shown that nuclear and cytoplasmic/membrane effects are not mutually exclusive – rather they independently contribute to the multiple biologic effects of UVB.
Many of the therapeutic effects of UVB may be due to its immunosuppressive properties which have been linked to the formation of pyrimidine dimers. UV radiation suppresses contact allergy, delayed-type hypersensitivity, and immune surveillance against UV-induced non-melanoma skin cancers in mice. Of note, Langerhans cells are very sensitive to UVB, which alters their antigen-presenting function. Keratinocytes also secrete soluble mediators such as IL-1 and IL-6, prostaglandin E 2 and TNF-α in response to UVB, which by themselves can alter the immune response. Therapeutic UVB suppresses the type 1 (proinflammatory) axis as defined by IL-12, interferon-γ and IL-8, and can selectively reduce proinflammatory cytokine production by individual T cells .
The interplay of the various photobiological pathways is far from being completely understood. In psoriasis, both epidermal keratinocytes and cutaneous lymphocytes may be targeted by UVB. Immune suppression, alteration of cytokine expression, and cell cycle arrest may all contribute to the suppression of disease activity in psoriatic plaques . In addition, a subset of T cells, Th17 cells, which seem to play a central role in the immunopathogenesis of psoriasis, are also down-regulated by UVB . More recently, evidence was presented that keratinocyte apoptosis may be a key mechanism in the clearance of psoriatic plaques .
Action Spectrum and Radiation Sources
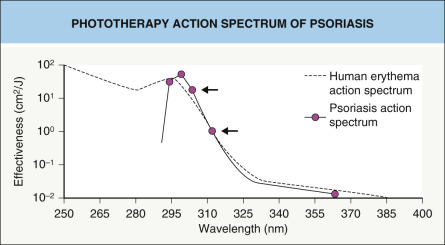
Parrish and Jaenicke demonstrated that for antipsoriatic activity, 304 and 313 nm were the optimally effective wavelengths ( Fig. 134.2 ), even at suberythemogenic doses. Erythemogenic UVA doses are also therapeutically effective, but 1000 times higher fluences are needed (as compared with UVB) which practically speaking is not feasible. The addition of UVA also does not enhance the therapeutic efficacy of UVB in psoriasis (unlike atopic dermatitis). The Philips TL01 fluorescent lamp that emits NB-UVB (311–313 nm) was introduced to optimally meet the requirements for antipsoriatic activity ( Fig. 134.3 ).
Devices that contain UV light-emitting bulbs are available in a wide range of sizes and designs including enclosed whole-body cabinets, portable flat panels, and hand-held units. When reliable patients live far distances from physician-supervised phototherapy centers or for other reasons are unable to schedule multiple treatments per week, UVB units designed for home use can be utilized. Practical guidelines for home phototherapy have been published .
Exposure to natural sunlight is called heliotherapy. Natural sunlight contains sufficient UVB radiation such that it is sometimes used to treat photoresponsive dermatoses. However, due to variables that affect UVB irradiance, including time of day, cloud cover, season, altitude and latitude, appropriate exposure times cannot be standardized. As a result, there is a greater probability of sunburn and an inadequate therapeutic response.
Treatment Protocols
Before starting phototherapy, determination of the patient’s UV sensitivity via phototesting is recommended, since skin typing by history alone does not always reflect the actual sensitivity of a particular individual. However, phototesting is not mandatory for the experienced therapist and, for practical reasons, is often not done. Testing is performed by exposing four to six small template areas (e.g. 1 cm diameter circles or squares) of usually non-sun-exposed skin (lower back, buttocks) to an incremental series of UVB doses. In successive test sites, the incremental increases can be fixed (e.g. 10 or 20 mJ/cm 2 ) or proportional (e.g. 40% higher). An example is given in Table 134.1 . Note that the actual doses delivered as specified in fluence units (mJ/cm 2 ) strongly depend on the type of UVB unit; for example, NB-UVB is much less erythemogenic than BB-UVB and consequently higher numerical doses are required.
The minimal erythema dose (MED) is defined as the lowest dose that causes a minimally perceptible erythema reaction at 24 hours after irradiation. Sunbathing or exposure to tanning lamps should be avoided before phototesting. It is crucial to document the type of lamp used for the MED determination, since the values obtained with broadband and narrowband sources differ by up to ten-fold (see Table 134.1 ).
Despite continued discussion as to whether visual assessment of the MED is the optimal method for establishing the initial UVB dose, it is an easily performed procedure that does not require any specialized equipment. An initial therapeutic UVB dose equal to 70% of the MED is typically recommended. Subsequent treatments are given two to five times weekly. Since UVB erythema peaks within 24 hours after exposure, doses may be increased with each successive treatment. However, if treatments are given five times weekly, the doses should be increased every other treatment. The rate of increase depends on treatment frequency and the response to the preceding UVB exposure. The objective of the dose increments is to achieve a minimally perceptible erythema as a clinical indicator of optimal dosimetry. For example, with thrice-weekly exposures, doses are increased by a maximum of 40% if no erythema appears, and by a maximum of 20% upon development of slight erythema. If mild erythema persists, the dose should be maintained. With daily exposures, these rates are no more than 30%, 15% and 0%, respectively. If more intensive or painful erythema develops, irradiations are stopped until the symptoms disappear. Treatment is then continued until complete remission is achieved or no further improvement can be obtained with continued phototherapy ( Fig. 134.4 ).
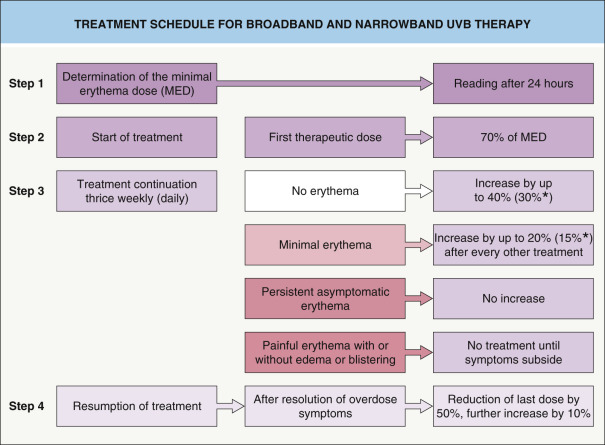
Whether maintenance treatment leads to a more prolonged remission time is still a matter of debate, since minimal precise data are available. In cutaneous T-cell lymphoma (CTCL), most therapists perform maintenance treatment for several months to a year. For psoriasis, some centers use a 2-month maintenance phase with twice-weekly exposures for 1 month and once-weekly exposures for another month. The last effective UVB dose is given throughout the maintenance phase. If relapses occur during the maintenance phase, treatment frequency and UVB dose are again increased until clearing. Of note, there was some evidence in a randomized study that maintenance NB-UVB may lead to longer remission times in plaque-type psoriasis .
Excellent guidelines for the use of NB-UVB have been prepared by the British Photodermatology Group. They present evidence-based guidance for treatment of patients with a variety of dermatoses and photodermatoses, with identification of the strength of evidence available at the time of preparation .
Phototherapy for Psoriasis
Guttate and seborrheic (minimally elevated) forms of psoriasis respond most favorably and rapidly to BB-UVB, while chronic, plaque-type psoriasis is more resistant. In terms of clearing efficiency and duration of remission, BB-UVB is inferior to both NB-UVB and PUVA. As a result, in many countries, NB-UVB has largely replaced conventional BB-UVB phototherapy. Based on several publications, NB-UVB phototherapy appears to be somewhat less effective than PUVA.
In addition to the treatment protocols mentioned above, other regimens may be used. Some schedules utilize skin type-dependent starting doses and fixed increments regardless of skin reaction. Sometimes, the patient’s extremities (in particular the lower extremities which tend to respond more slowly) can be exposed to higher doses than the trunk.
Adjunctive topical agents and combination therapies are often used to improve efficacy and reduce the cumulative UVB dose, with the ultimate aim of minimizing the risk of long-term side effects. Narrowband UVB has been successfully combined with therapies such as anthralin and vitamin D analogues. The use of bland emollients before UVB treatment is laborious and time-consuming, and in our experience does not dramatically increase treatment efficacy. The concurrent use of topical corticosteroids may reduce remission times and thus is discouraged by some dermatologists.
Systemic drugs such as retinoids increase efficacy, particularly in patients with chronic plaque-type psoriasis . In addition, retinoids may reduce the carcinogenic potential of UVB phototherapy. There have been open-label studies and small comparison studies regarding the efficacy of combining UVB phototherapy and biologic agents . To date, there have been no large-scale studies, and long-term data, including remission times, are unavailable.
Phototherapy for Cutaneous T-Cell Lymphoma, in Particular Mycosis Fungoides
For early stages (IA, IB, and IIA) of mycosis fungoides (MF), conventional treatment strategies include topical corticosteroids, UV radiation, and topical cytotoxic agents such as mechlorethamine (nitrogen mustard). More advanced stages are treated with various combinations that include total body electron beam radiation therapy (see Ch. 139 ), X-irradiation, systemic retinoids, PUVA, systemic chemotherapy, and immunomodulators (e.g. brentuximab, IFN), but none of these regimens has been demonstrated to induce permanent remission.
Lesions of MF frequently occur in non-sun-exposed areas of the body, and patients with early stage MF often benefit from exposure to natural sunlight. Response to phototherapy is related to the type (i.e. patch stage MF does better than the plaque type) but not the extent of skin involvement. In a more recent study, prolonged maintenance therapy (up to 30 months) resulted in a relapse-free period of 26 ± 10 months .
The proposed mechanisms for UVB phototherapy of MF include impairment of epidermal Langerhans cell function and alterations in cytokine production and adhesion molecule expression by keratinocytes . Moreover, NB-UVB can induce apoptosis of T lymphocytes, which may more specifically contribute to the beneficial effect of this light source .
UVB phototherapy is clearly less efficient than PUVA, and the duration of treatment in the clearing and maintenance phases is longer and thus requires higher patient compliance. Whether patients would benefit from adding UVA, as is sometimes recommended, remains questionable. Likewise, NB-UVB and BB-UVB therapy have not been compared in MF. It is possible that combining a systemic retinoid (e.g. bexarotene) with NB-UVB may increase efficacy. Lastly, a pilot study documented the efficacy of medium- to high-dose UVA1 therapy (see below) in patients with stage IA and IB MF. However, there is no evidence that UVA1 is superior to PUVA and no long-term follow-up is available.
Phototherapy for Vitiligo
Many patients observe follicular repigmentation in areas of vitiligo following sun exposure. Because of seasonal and weather-dependent variations in sunlight intensity in moderate climates, exposure to natural sunlight is often not a practical option for inducing repigmentation. In the past, PUVA was most frequently used for the treatment of vitiligo, but exposure to artificial UVB irradiation, in particular NB-UVB, can also provide an acceptable therapeutic effect, if given for a sufficiently long period. Although the action spectrum for phototherapy of vitiligo is not known, NB-UVB has clearly become quite popular in recent years. In comparison studies of NB-UVB with PUVA, NB-UVB was as effective as PUVA but had fewer side effects ( Fig. 134.5 ). Also, in a randomized double-blind trial of non-segmental vitiligo, NB-UVB therapy was superior to oral PUVA therapy and the color match of repigmented skin was considered to be better in the patients treated with NB-UVB . UVA alone is of limited benefit.
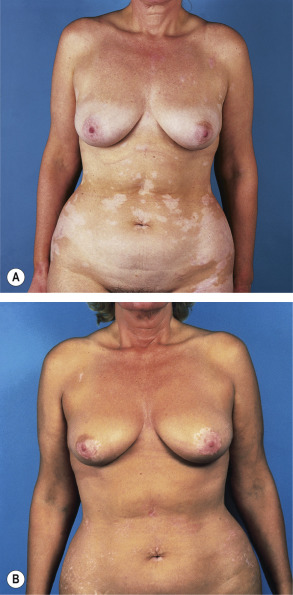
Since intense erythema may induce the Koebner phenomenon and worsening of the disease, it is necessary to stay within a dose range that induces minimally perceptible erythema. Therefore, initial assessment of the MED in a vitiliginous area that is normally not sun-exposed (e.g. buttocks, lower back, abdomen) is recommended. The UVB doses have to be increased more carefully than in other disorders, because of the increased photosensitivity of depigmented skin.
The initial exposure is 70% of the MED in lesional skin, and subsequent doses are chosen according to the response in vitiligo areas, i.e. the goal is to induce a barely perceptible erythema that has a light pink color. The presence or absence of faint erythema is the only useful parameter for determining dosage increments. The dose should not be increased more than once weekly by 5–20% of the preceding dose. Despite the lack of pigment, vitiliginous skin does develop photoadaptation, probably as a result of epidermal hyperplasia and thickening of the stratum corneum. Usually, a maximum dose will be reached during the first couple of months which is then used throughout the entire treatment period. Most commonly, two to three treatments per week are given. Although few controlled studies are available for any particular phototherapy regimen to be preferred over another, consensus recommendations are available .
Recently, the combination of an afamelanotide implant plus NB-UVB phototherapy resulted in clinically apparent, statistically significant superior and faster repigmentation compared with NB-UVB monotherapy. Afamelanotide is an analogue of α-melanocyte stimulating hormone that can induce diffuse hyperpigmentation of the skin, especially in sun-exposed sites. Of note, the response was more noticeable in patients with skin phototypes IV to VI .
Phototherapy for Atopic Dermatitis
Broadband UVB (BB-UVB) phototherapy
Based on the empirical experience that sun exposure was beneficial for patients with atopic dermatitis, BB-UVB has been used to treat atopic dermatitis since the late 1970s.
UVA/UVB phototherapy
More recent studies suggest that a combination of UVA plus UVB irradiation (UVA/UVB therapy) is superior to conventional BB-UVB, conventional UVA, or low-dose UVA1 therapy in the management of chronic, moderate atopic dermatitis. A paired-comparison study demonstrated statistically significant differences in favor of UVA/UVB therapy, compared with BB-UVB therapy .
Narrowband UVB (NB-UVB) phototherapy
Earlier studies showed that NB-UVB irradiation not only improved the total clinical score, but also substantially reduced the need for potent topical corticosteroids. These beneficial effects were still present in the majority of patients 6 months after cessation of phototherapy. Narrowband UVB therapy has also been used successfully in conjunction with UVA1 therapy. However, due to the lack of baseline data on the duration and severity of atopic dermatitis, a final conclusion on preference of phototherapy in the acute or chronic phases cannot be drawn from available studies .
Phototherapy for Photodermatoses
In the majority of patients, polymorphous light eruption (PMLE) gradually abates with continual outdoor light exposure. For patients prone to PMLE, NB-UVB phototherapy can be used prophylactically to photoharden the skin (see Ch. 87 ). Two to three treatments per week for a total of 15 sessions are typically administered prior to an anticipated increase in exposure to sunlight (e.g. springtime). Despite using reduced UVB doses (as compared to those for psoriasis), this phototherapy regimen can induce a transient photoeruption; the latter usually responds to topical corticosteroids and smaller incremental increases in subsequent UVB doses. The precise mechanisms by which phototherapy induces adaptation to sunlight are not known, but may include hyperpigmentation, thickening of the stratum corneum, and modulation of cutaneous immune functions.
Narrowband UVB phototherapy was recently shown to be capable of inducing photohardening in patients with erythropoietic protoporphyria .
Phototherapy for Pityriasis Lichenoides and Lymphomatoid Papulosis
Both pityriasis lichenoides acuta and chronica can have a prolonged course and be rather resistant to therapy. As sunlight may lead to some improvement, UVB phototherapy has also been associated with some success. However, PUVA seems to be more effective (see below), especially in the acute form of the disease (perhaps because of the deeper extension of the dermal inflammatory infiltrate). Although some authors have recommended PUVA as the treatment of choice for the acute form , PUVA may be reserved for cases of pityriasis lichenoides chronica that are UVB-resistant.
Phototherapy for Seborrheic Dermatitis
Seborrheic dermatitis generally improves during the summer and “sunny” vacations. Accordingly, UV radiation also seems to have a beneficial effect on this condition. However, flares of dermatitis may occasionally occur. In an open prospective study, NB-UVB was found to be an effective treatment in severe cases .
Phototherapy for Pruritus
Both narrowband and broadband UVB therapy can be beneficial in various forms of pruritus, particularly those associated with diabetes and hepatic disorders or those that are idiopathic. Remissions in hepatic disease, however, are relatively short-lived. In a case series, NB-UVB led to improvement of uremic pruritus .
Side Effects of Phototherapy With UVB
Short-term side effects include erythema ( Fig. 134.6 ), xerosis accompanied by pruritus, occasional blistering, and an increased frequency of recurrent herpes simplex viral infections. Painful erythema resulting from overexposure is treated with topical corticosteroids. Systemic nonsteroidal anti-inflammatory drugs and corticosteroids can prove useful in severe cases if administered early.
Long-term side effects include photoaging and carcinogenesis. Although UVB is a known carcinogen, its carcinogenic potential seems to be much lower than that of PUVA. In a British study involving 3867 patients who had been treated with NB-UVB, no significant association between NB-UVB treatment and basal cell carcinoma (BCC), squamous cell carcinoma (SCC), or melanoma was found . There was a small increase in BCCs amongst those patients who had also been previously treated with PUVA. To determine the true carcinogenic risk of NB-UVB phototherapy, additional longitudinal studies are essential.
Phototherapy With the 308 nm Excimer Laser or Lamp
With the 308 nm excimer, laser monochromatic UVB radiation can be delivered to specific sites of affected skin at higher fluences than are typically given with whole-body exposure. Clinical use of this laser for plaque-type psoriasis was first reported in 1997 and it is FDA-approved for this indication. Possible advantages are the lower number of treatments that may be necessary to induce clearing as well as the ability to selectively target affected skin with a reduced cumulative dose, thus perhaps reducing the long-term risk of carcinogenicity . However, widespread psoriasis generally cannot be treated with this modality because the spot size is limited to just a few square centimeters.
Clearly, the excimer laser is most suitable for patients with stubborn plaques unresponsive to other treatments and/or those with difficult-to-treat localized areas such as the palms, soles, knees, and elbows. Whether it really represents a therapeutic advancement still remains to be determined by larger and longer-term studies, particularly in view of the high costs of laser treatments. However, both non-laser excimer lamps and hand-held targeted UV devices are less expensive and appear to be similarly effective .
Based upon case series, targeted phototherapy with the excimer laser or excimer lamp may also serve as a treatment modality for the management of stable vitiligo as well as a variety of chronic inflammatory localized dermatoses such as granuloma annulare, lichen planus, lichen simplex chronicus, and alopecia areata .
Phototherapy With UVA1
The UVA spectrum (320–400 nm) has been arbitrarily subdivided into two parts: UVA1 (340–400 nm) and UVA2 (320–340 nm) (see Fig. 134.1 ). The main reason for this subdivision was the observation that UVA2 resembled UVB in its ability to induce erythema, immunomodulation, and photocarcinogenesis. Because of its longer wavelength, UVA1 radiation penetrates more deeply into the skin than UVA2 (see Ch. 86 ), and thus it affects not only epidermal structures, but also mid and deep dermal components, especially blood vessels. Since the skin is a large organ, exposure of circulating immune cells to UVA1 irradiation may have significant systemic ramifications. In particular, its ability to induce T-lymphocyte apoptosis is likely to be relevant in the treatment of atopic dermatitis and possibly of MF. UVA1 irradiation may also reduce the number of Langerhans cells and mast cells in the dermis in atopic dermatitis and cutaneous mastocytosis. In addition, it has been shown that increased collagenase expression in treated lesions of localized scleroderma accompanies improvement with UVA1 irradiation. Perhaps the efficacy of UVA1 in localized scleroderma, and in other sclerosing conditions, is in part due to this action.
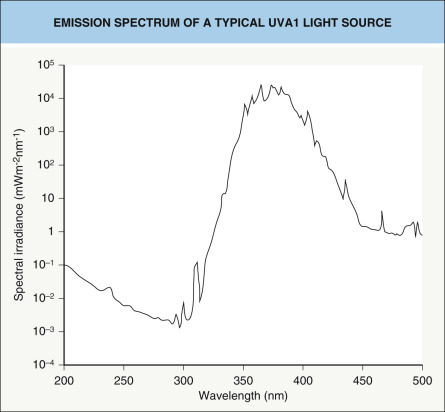
UVA1 phototherapy has been investigated as a safer alternative to PUVA for treating chronic conditions. Its major disadvantages are the higher fluences (e.g. 30–130 J/cm 2 per session) and therefore exposure times that are required as well as the higher costs for the lamps. To date, most of the UVA1 studies have been performed in Europe, where efficient sources of UVA1 radiation are more widely available ( Fig. 134.7 ).
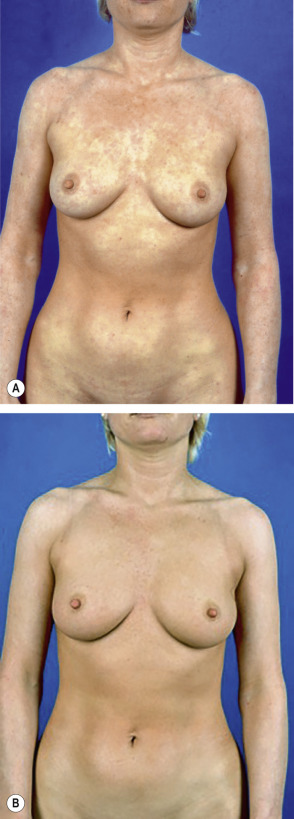
UVA1 can have a therapeutic effect on atopic dermatitis ( Fig. 134.8 ). It seems to be more effective than UVA/UVB therapy, at least for severe exacerbations of atopic dermatitis. If used as a monotherapy, medium and high daily doses are better than low doses. When UVA1 and NB-UVB were compared for the treatment of atopic dermatitis, they seemed to be equally effective . However, as noted previously, there is currently no consensus as to the preferred type of UV phototherapy for atopic dermatitis .
UVA1 therapy has been reported to be beneficial in several other dermatoses , in particular sclerosing dermatoses , acute GVHD, and chronic sclerodermoid GVHD . UVA1 has also been used to treat urticaria pigmentosa and MF . However, its efficacy, especially for the latter indication, requires confirmation in larger patient series.
Side Effects of Phototherapy With UVA1
The adverse effects of UVA1 are less severe than those of UVB and UVA2, although the high fluences usually induce marked tanning, especially in more darkly pigmented individuals. Until more is known about UVA1 therapy, its use (especially with high doses such as 130 J/cm 2 ) should be limited to treating diseases with periods of severe, acute exacerbations, and, in general, one treatment cycle should not exceed 15–20 successively administered exposures and should not be repeated more than once a year. A European prospective longitudinal study has been initiated to monitor patients treated with UVA1 phototherapy for the development of skin cancer and photoaging. An increased expression of TP53 and a slight increase in Bcl-2 protein expression in keratinocytes were found in human skin after UVA1 irradiation (even with suberythemogenic doses), suggesting that UVA1 may cause DNA damage . In addition, UVA1 induced pro-mutagenic cyclobutane pyrimidine dimers and oxidative DNA damage (at both the genomic and nucleotide resolution level) in normal human skin fibroblasts . Of note, in the years since the widespread use of UVA1 phototherapy in Europe began in 1992, no serious negative side effects in humans have been reported.
Photochemotherapy With Psoralens (PUVA)
Psoralen photochemotherapy (PUVA) combines the use of psoralens (P) and long-wave UV radiation (UVA). This combination results in a therapeutically beneficial phototoxic effect, which is not produced by either of the components alone. Psoralens can be administered orally or applied topically in the form of solutions, creams, or baths with subsequent UVA exposure.
Historical Aspects
Topical exposure to extracts, seeds, or parts of plants (e.g. Ammi majus , Psoralea corylifolia ) that contain natural psoralens, followed by exposure to sunlight, was used as a remedy for vitiligo for thousands of years in ancient Egypt and India . In 1974, it was shown that the oral ingestion of 8-methoxypsoralen (8-MOP) and subsequent exposure to a new, high-intensity, artificial UVA radiation source was highly effective for the treatment of psoriasis . This was followed by recognition of its therapeutic effect for multiple skin disorders ( Table 134.2 ). Over the past decade, the use of PUVA has decreased significantly, in part due to the increased risk of developing skin cancers, especially with prolonged use in fair-skinned individuals, and the effectiveness of NB-UVB. Even though PUVA often represents a second-line form of phototherapy, it is essential that dermatologists become familiar with treatment regimens, potential side effects, and indications.
| PUVA-RESPONSIVE DISEASES | |
|---|---|
| Therapy of disease | Prevention of disease |
| Psoriasis | Polymorphic light eruption † |
| Palmoplantar pustulosis | Solar urticaria † |
| Atopic dermatitis | Chronic actinic dermatitis * , † |
| Mycosis fungoides (stages IA, IB, IIA) | Hydroa vacciniforme * , † |
| Vitiligo | Erythropoietic protoporphyria * , † |
| Generalized lichen planus | |
| Urticaria pigmentosa | |
| Cutaneous GVHD | |
| Generalized granuloma annulare | |
| Prurigo nodularis | |
| Pityriasis lichenoides (acute and chronic) * | |
| Lymphomatoid papulosis * | |
| Pityriasis rubra pilaris * , † | |
| Pigmented purpuric dermatoses * | |
| Langerhans cell histiocytosis * | |
| Localized scleroderma * | |
* Experience is limited to a small number of patients.
Psoralens
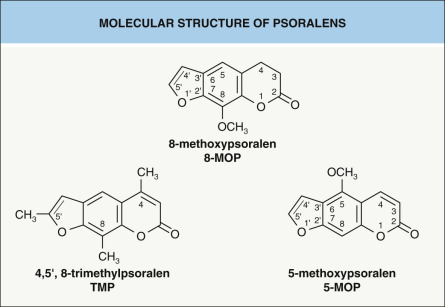
Psoralens are naturally occurring linear furocoumarins that are found in a large number of plants, and there are several synthetic psoralen compounds. For oral and topical (bath, cream) PUVA, mainly 8-MOP (methoxsalen) is used; it is of plant origin but also available as a synthetic drug. The synthetic compound, 4,5′,8-trimethylpsoralen (TMP, trioxsalen), is less phototoxic than 8-MOP after oral administration, but more phototoxic when delivered via bath-water. TMP is used primarily in Scandinavia for bath PUVA. 5-Methoxypsoralen (5-MOP, bergapten) is also therapeutically effective when given orally; it is less erythemogenic and is not associated with gastrointestinal intolerance ( Fig. 134.9 ). In some European countries, it was routinely used for PUVA, but it is no longer commercially available. Oral preparations of 8-MOP contain crystals, micronized crystals, or solubilized psoralens in a gel matrix. The liquid preparation induces earlier, higher, and more reproducible peak plasma levels than the crystalline preparations.
The steps between the ingestion of a psoralen and its arrival in the skin include disintegration and dissolution of the drug, absorption, first-pass effect, blood transport, and tissue distribution. The absorption rate of a psoralen from the gut depends on the physicochemical properties of the molecule, the rate of dissolution, the galenic characteristics of the preparation, and the fat content of concomitantly ingested food. Peak serum levels are usually reliably and predictably achieved by liquid preparations, whereas wide time variability occurs with crystalline formulations. Before reaching the skin via the circulation, psoralens are metabolized during their passage through the liver. Plasma levels of 8-MOP administered orally at different doses show a strong non-linearity, indicating a saturable first-pass effect. Thus, small differences in the ingested doses and absorption rates of psoralens lead to large differences in plasma levels. As a practical consequence, small amounts of the drug are almost completely metabolized by the liver during the first pass and therefore may be therapeutically inactive.
8-MOP serum levels show a wide range of inter-individual differences. Even on different occasions in the same patient, serum levels may differ; however, the levels are usually sufficiently constant to provide for relatively reproducible therapeutic results. This unpredictable pharmacokinetic behavior is probably due to inter- and intra-individual variations in intestinal absorption, first-pass effect, blood distribution in the body, and metabolism and elimination of the drug.
For a given patient, serum levels of 8-MOP correspond fairly well with skin reactivity, with the peak of skin phototoxicity coinciding with peak serum levels. A correlation between 8-MOP serum levels and epidermal concentrations does exist. Whether there is a significant correlation between maximum psoralen blood concentrations and the minimal phototoxicity dose (MPD) remains equivocal.
The pharmacokinetics of topical 8-MOP depend on the mode of application. 8-MOP administered topically as a 0.15% emulsion or solution has been shown to result in plasma levels comparable to those obtained by oral delivery, when large areas of the body are treated. In contrast, due to the much lower concentrations that are typically used, plasma levels following bath-PUVA treatment of almost the entire body surface are very low. Bath-water-delivered psoralens are readily absorbed into the skin, but promptly eliminated without cutaneous accumulation .
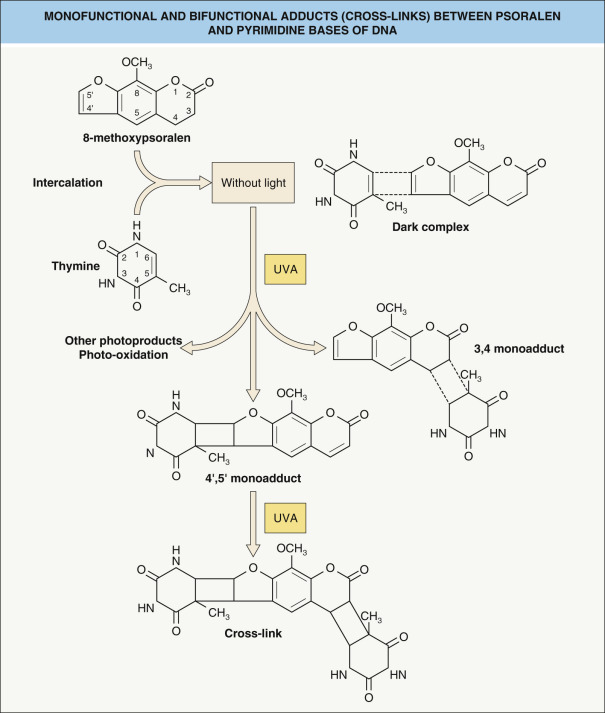
Psoralen photochemistry
Psoralens react with DNA in three steps. First, in the absence of UV radiation, the psoralen intercalates into the DNA double strand. Via a photochemical reaction, absorption of photons in the UVA range results in the formation of a 3,4- or 4′,5′-cyclobutane monoadduct with pyrimidine bases of native DNA. The 4′,5′ monoadducts can absorb a second photon and this reaction leads to the formation of an interstrand cross-link of the double helix with the 5,6 double bond of the pyrimidine base of the opposite strand ( Fig. 134.10 ).