Chapter 6
The Lower Limb
Leg ulcers
- Most common chronic wounds in developed countries.
- Affect 1–3% of the British population.
Aetiology
- Leg ulceration may be caused by ‘VATIMAN’:
- Venous disease
- Arterial disease
- Trauma
- Insect bites
- Trophic ulcers
- Self-inflicted injuries
- Burns and frostbite
- Radiation
- Insect bites
- Infection
- Bacterial
- Fungal
- Mycobacterial
- Syphilis
- Bacterial
- Metabolic disorders
- Diabetes
- Necrobiosis lipoidica diabeticorum
- Pyoderma gangrenosum
- Porphyria
- Gout
- Calciphylaxis
- Diabetes
- Autoimmune diseases
- Vasculitis
- Systemic lupus erythematosus (SLE)
- Systemic sclerosis (scleroderma)
- Rheumatoid arthritis
- Polyarteritis nodosa
- Systemic lupus erythematosus (SLE)
- Vasculitis
- Neoplasia
- Squamous cell carcinoma (SCC)—Marjolin ulcer
- Basal cell carcinoma (BCC)
- Melanoma
- Kaposi sarcoma
- Lymphoma.
- Squamous cell carcinoma (SCC)—Marjolin ulcer
- Venous disease
The leg ulcer patient
Medical history
- Time and mechanism by which the ulcer started
- Previous treatment
- Ambulatory status
- Type of footwear
- Symptoms suggestive of a cause, e.g. claudication, rest pain
- Relevant comorbidities, e.g. diabetes, smoking.
Examination
- The ulcer itself
- Examined for features suggestive of aetiology:
- Venous ulcers—classically in the gaiter area, sloping edges.
- Arterial ulcers—usually on toes, feet or ankles, ‘punched out’ edges.
- Features of malignancy.
- Venous ulcers—classically in the gaiter area, sloping edges.
- Examined for features suggestive of aetiology:
- State of the circulation
- Temperature, capillary refill.
- Peripheral pulses.
- Varicose veins, oedema, venous eczema, hyperpigmentation—all suggest underlying venous disease.
- Temperature, capillary refill.
- Sensation
- ‘Glove and stocking’ sensory loss suggests peripheral neuropathy.
Investigation
- Microbiology wound swab
- Ankle brachial pressure index (ABPI)
- Vascular studies
- Radiology
- X-rays, CT and bone scans assess bony involvement.
- Biopsy of long-standing ulcers to exclude malignancy
- Onward referral to a rheumatologist for vasculitis or connective tissue disorder.
Venous ulcer disease
- An area of epidermal discontinuity that persists for four weeks or more, occurring as a result of venous hypertension and insufficiency of the calf muscle pump.
- Accounts for 80% of lower limb ulceration.
- Venous hypertension may be caused by:
- Reflux of venous blood due to valvular incompetence
- Venous obstruction
- Insufficiency of the calf muscle pump.
- May affect the superficial system (long and short saphenous veins), deep system or interconnecting perforators.
- Valvular incompetence occurs due to thrombophlebitis, previous thrombosis or trauma.
- Pathogenesis is not fully understood; mooted mechanisms include:
- Venous hypertension causes protein-rich exudate to leak into subcutaneous tissue.
- Forms a pericapillary fibrin cuff, causing local tissue hypoxia.
- Venous hypertension causes extravasation of erythrocytes.
- Triggers an inflammatory response, with deposition of haemosiderin within macrophages.
- Transforming growth factor (TGF)-β may mediate dermal fibrosis, lipodermatosclerosis and eventual ulceration.
- Lipodermatosclerosis is characterised by:
- Scarring
- Fibrotic, hyperpigmented skin
- ‘Inverted champagne bottle’ appearance.
- Scarring
- Triggers an inflammatory response, with deposition of haemosiderin within macrophages.
- Accounts for 80% of lower limb ulceration.
Management
Non-operative
- Bed rest and leg elevation
- Impractical for most patients; ulceration tends to recur with ambulation.
- Compression
- Gold standard treatment; used continuously until healing occurs.
- Mean time to healing is 5 months.
- Ulcer recurrence rate 30% in 5 years.
- Mean time to healing is 5 months.
- Four-layer bandaging enables the shortest time to healing.
- The four layers are applied from toes to knee and consist of:
- Orthopaedic wool
- Crêpe bandage
- Elastic bandage
- Cohesive retaining layer.
- Orthopaedic wool
- The four layers are applied from toes to knee and consist of:
- Compression stockings are easier to use; there is limited evidence of their superiority over bandaging.
- Stockings are classified as:
- Class 1 (light): 14–17 mmHg
- Class 2 (medium): 18–24 mmHg
- Class 3 (strong): 25–35 mmHg.
- Class 1 (light): 14–17 mmHg
- Stockings are classified as:
- Gold standard treatment; used continuously until healing occurs.
- Local treatments
- Pentoxifylline (unlicensed indication).
Operative
- Venous ulceration is not usually managed surgically.
- Indications for surgery may include:
- Intractable pain
- Failure of non-operative treatment
- Surgically treatable venous disease.
- Intractable pain
- Surgery addresses both the underlying venous disease and the ulcer.
Treatment of venous disease
- Ablation of superficial and perforating veins.
- Achieved by open surgery or endovenous laser ablation.
- Subfascial endoscopic perforating vein surgery (SEPS).
- Reconstruction of the deep venous system.
Treatment of the venous ulcer
- Debridement and skin grafting.
- High recurrence rate if underlying venous pathology is not corrected.
- Excision with flap reconstruction.
- In exceptional circumstances, pedicled or free flaps are used after ulcer excision.
Arterial ulcer disease
- Results from reduced blood supply to the lower limb.
- Most common cause is atherosclerosis, risk factors for which include:
- Age
- Family history
- Smoking
- Diabetes
- Hypertension
- Hyperlipidaemia
- Obesity.
- Age
- Generalised changes in the limb:
- Dusky erythema
- Lower surface temperature
- Lack of hair growth
- Thin, brittle, atrophic skin
- Thickened or missing toenails
- Absent peripheral pulses.
- Dusky erythema
- Ulceration may develop after seemingly trivial trauma.
- Patients should be taught how to care for their skin.
Management
- Combination of operative and non-operative methods.
- Debridement or negative pressure wound therapy may enlarge the area of ischaemia.
Non-operative management
- Control of modifiable risk factors, e.g. diabetes, smoking
- Graded exercise regime
- Foot care
- Infection prevention
- Cilostazol or pentoxifylline.
Operative management
- Indications for invasive treatment:
- Non-healing ulceration
- Gangrene
- Rest pain
- Progression of disabling claudication.
- Non-healing ulceration
- Aim to improve blood flow into the affected limb.
- This accelerates healing time.
- Achieved by reconstructive vascular surgery or angioplasty.
- This accelerates healing time.
Lower limb trauma
Epidemiology
- Annual incidence of open lower limb fractures is approximately 5 per 100,000 population.
- Analysis of open tibial fractures treated by the Edinburgh Orthopaedic Trauma Unit (1988–1990) revealed:
- 21% Gustilo type I injuries.
- 19% Gustilo type II.
- 60% Gustilo type III.
- Of type III fractures: 27% type IIIA, 60% type IIIB, 8% type IIIC.
- 21% Gustilo type I injuries.
- Average age 42 years.
- Most common mechanisms of injury:
- Fall from height
- Motor vehicle collision
- Interpersonal violence.
- Fall from height
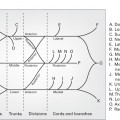
Classification of open fractures
- Numerous classifications have been described:
- Gustilo and Anderson (1976)
- Hannover Fracture Scale-97 (HFS-97) (1982)
- Byrd and Spicer (1985)
- Predictive salvage index (PSI) (1987)
- Mangled Extremity Severity Score (MESS) (1990)
- The AO soft tissue grading system (1991)
- Limb salvage index (LSI) (1991)
- Nerve injury, ischemia, soft tissue, skeletal, shock and age of patient (NISSSA) (1994)
- Ganga hospital score (2006)
- Bastion classification (2012).
- Byrd and Spicer (1985)
- Gustilo and Anderson (1976)
Gustilo and Anderson
- Widely used; relatively simple to apply.
- Based on a retrospective study of long bone fractures.
- Open fractures are best classified after wound excision.
- Poor inter-observer reliability.
- No account is taken of fracture characteristics or nerve/muscle injury.
- Grade IIIB is therefore a diverse group.
- Open fractures were originally classified into three categories:
Type I
- Open fracture with a wound <1 cm long and clean.
Type II
- Open fracture with a laceration >1 cm long without extensive soft tissue damage, flaps or avulsions.
Type III
- Either open segmental fracture, open fracture with extensive soft tissue damage or traumatic amputation.
- This key point is often overlooked: all high energy pattern injuries are Type III.
- Special categories in Type III:
- Gunshot injuries
- Any open fracture caused by a farm injury
- Any open fracture with accompanying vascular injury requiring repair.
- Gunshot injuries
- In 1984, Gustilo et al. subclassified type III injuries:
Type IIIA
- Adequate soft tissue coverage of a fractured bone despite extensive soft tissue laceration or flaps.
- High energy trauma irrespective of the size of the wound.
Type IIIB
- Extensive soft tissue injury with periosteal stripping and bony exposure.
- This is usually associated with massive contamination.
Type IIIC
- Open fracture associated with arterial injury requiring repair.
MESS
- Designed to predict whether a lower limb is salvageable.
- Total score ≤6 is consistent with a salvageable limb, using modern multidisciplinary surgical reconstruction techniques.
- May aid decision making when primary amputation is considered.
- Does not replace experienced clinical judgement.
A. Skeletal/soft tissue injury
- Low energy (stab, simple fracture, civilian gunshot wound): 1
- Medium energy (open or multiple fractures, dislocation): 2
- High energy (close-range shotgun, military gunshot wound, crush injury): 3
- Very high energy (above + gross contamination, soft tissue avulsion): 4
B. Limb ischaemia (double the score for ischaemia >6 h)
- Pulse reduced or absent but perfusion normal: 1
- Pulseless, paraesthesias, diminished capillary refill: 2
- Cool, paralysed, insensate, numb: 3
C. Shock
- Systolic blood pressure always >90 mmHg: 0
- Hypotensive transiently: 1
- Persistent hypotension: 2
D. Age (years)
- <30: 0
- 30–50 years of age: 1
- >50 years of age: 2.
NISSSA
- Aims to address MESS’s shortcomings by including nerve injury, and more detailed information about muscle and soft tissue injuries.
- Applies only to open tibial fractures.
- More sensitive and specific than MESS.
AO system
- The most comprehensive soft tissue classification.
- However, it is very complex; usually reserved for research or coding.
Bastion classification
- Named after Camp Bastion, the main British military base in Afghanistan.
- Classifies lower limb injuries caused by improvised explosive devices.
Initial management of lower limb trauma
- As with all trauma, initial assessment is an ATLS-style primary survey.
- The purpose of the primary survey is to identify and treat all immediately life-threatening problems.
- The <C>ABC approach, adopted by the military, may be appropriate.
- This attends to <C>, catastrophic haemorrhage, early on in the algorithm.
- A tourniquet may be required to stop bleeding.
- The purpose of the primary survey is to identify and treat all immediately life-threatening problems.
History
- Mechanism of injury is a guide to the level of energy transferred.
- Factors suggestive of high energy injury:
- High speed motor vehicle accidents, particularly pedestrian.
- Falls from significant height
- Missile wounds
- Any injury involving crushing.
- High speed motor vehicle accidents, particularly pedestrian.
Examination
- Establish the neurovascular status:
- Capillary refill time.
- Dorsalis pedis and posterior tibial arterial pulse.
- Active movement of toes and ankle (common peroneal and tibial nerves).
- Sensation on the sole of foot (tibial nerve) and first webspace (deep peroneal nerve).
- Re-examine neurovascular status regularly, particularly after fracture manipulation.
- Capillary refill time.
- Factors suggestive of high energy injury:
- Transverse or segmental fracture pattern with comminution
- Associated injuries
- Large soft tissue defect
- Closed degloving, where skin is intact but perforating vessels are divided.
- Tyre prints.
- Transverse or segmental fracture pattern with comminution
Treatment
- Current UK Standards recommend this initial treatment:
- Analgesia.
- Restore limb alignment; splint using plaster of Paris backslab.
- Remove gross contamination.
- Photograph the wound.
- Cover the wound with sterile, saline-moistened gauze covered with semi-occlusive film dressing.
- Repeat neurovascular examination.
- Intravenous antibiotics—co-amoxiclav or cefuroxime 8 hourly:
- Continue until definitive soft tissue cover or 72 hours, whichever is sooner.
- Additional ‘single shot’ gentamicin 1.5 mg/kg at the time of debridement.
- Additional ‘single shot’ gentamicin and either teicoplanin or vancomycin at the time of skeletal stabilisation and definitive soft tissue closure.
- Penicillin allergy: give clindamycin instead of coamoxiclav/cefuroxime.
- Continue until definitive soft tissue cover or 72 hours, whichever is sooner.
- Tetanus prophylaxis if required.
- X-ray—two views, including the joints above and below the fracture.
- Analgesia.
Surgical management
- Summarised as:
- Debridement
- Skeletal stabilisation
- Soft tissue cover.
- Debridement
Timing
- The traditional ‘6-hour rule’ of open fracture surgery was based on the doubling time of Staphylococcus.
- This is the time it takes one bacterium to reach an infective load of 105 organisms.
- However, many studies fail to demonstrate a relationship between the timing of debridement and outcome.
- UK Standards recommend primary surgery within 24 hours of injury by senior plastic and orthopaedic surgeons.
- Exceptions mandating immediate surgery:
- Gross contamination
- Compartment syndrome
- Devascularised limb
- Another injury requiring immediate surgery.
- Gross contamination
Primary surgery
- A major aim of primary surgery is to convert a contaminated traumatic wound into a clean surgical wound.
- This is achieved by three ‘Es’:
- Extend
- Explore
- Excise.
- Extend
- Use of a thigh tourniquet is preferred by some surgeons.
Wound extension
- Extend along lines of election for fasciotomy to assess the entire zone of trauma.
- Should not injure perforators that supply local fasciocutaneous flaps.
Degloving injuries
- Degloving is avulsion of skin and subcutaneous tissue from underlying muscle or bone.
- Degloving is classified by Arnež:
- Pattern 1—Limited degloving with abrasion/avulsion
- Tissue loss due to abrasion/avulsion and limited degloving of remaining skin.
- Typically occurs around bony prominences; may expose bone or joint.
- Tissue loss due to abrasion/avulsion and limited degloving of remaining skin.
- Pattern 2—Non-circumferential degloving
- Most skin remains as a flap or undermined area, usually just superficial to muscle fascia.
- Pattern 3—Circumferential single-plane degloving
- More extensive than pattern 2; skin does not usually survive.
- Pattern 4—Circumferential multiplane degloving
- Similar to pattern 3, but with additional breach of muscle fascia.
- Degloving can run between muscles and between muscle and periosteum.
- Indicative of high energy transfer.
- Pattern 1—Limited degloving with abrasion/avulsion
- The margins of excision following degloving can be difficult to determine.
- Findings suggestive of non-viable skin:
- Fixed staining and thrombosis of subcutaneous veins.
- Circumferential degloving.
- Poor perfusion, demonstrated by intravenous fluorescein and a Wood’s lamp.
- Newer technologies using indocyanine green (ICG) fluorescence have been reported.
- Fixed staining and thrombosis of subcutaneous veins.
Muscle
- Muscle viability is assessed by four Cs:
- Colour (pink)
- Contraction
- Consistency (dead muscle tears easily in the jaws of forceps)
- Capacity to bleed.
- Colour (pink)
Bone
- Deliver the ends of the fracture out of the wound.
- This allows complete assessment and debridement of the wound and bone.
- The deep posterior compartment can be inspected this way.
- This allows complete assessment and debridement of the wound and bone.
- Loose bone fragments that fail the ‘tug test’ are removed.
- Large articular fragments that can be fixed with absolute stability are preserved.
- Bone viability is determined by its capacity to bleed:
- Punctate bleeding from exposed cortical surfaces (paprika sign).
- Extent of periosteal stripping and muscle/fascia connections.
- Punctate bleeding from exposed cortical surfaces (paprika sign).
- Following excision, irrigate the wound with large volumes of warm saline.
- High pressure pulse lavage is not recommended
- Associated with deep bacterial inoculation and tissue damage.
- High pressure pulse lavage is not recommended
- Second look within 24–48 hours is occasionally indicated; multiple serial debridements are associated with worse outcomes.
Fracture stabilisation
- Provisional stabilisation is achieved with a spanning external fixator.
- Indicated when definitive stabilisation and soft tissue cover cannot be achieved at primary surgery.
- Pins are inserted through ‘safe corridors’ to avoid neurovascular structures.
- The construct should allow access to the wound for soft tissue cover.
- If conversion to definitive internal fixation is planned, UK Standards recommend this occurs within 72 hours of primary surgery.
- Indicated when definitive stabilisation and soft tissue cover cannot be achieved at primary surgery.
- Definitive stabilisation is usually achieved by internal fixation (intramedullary nail, plate, screws).
- Internal fixation should not be placed if immediate soft tissue cover cannot be achieved.
- Orthopaedic implants within open wounds are associated with higher infection rate.
- Internal fixation should not be placed if immediate soft tissue cover cannot be achieved.
Vascular injury
- Limb devascularisation requires emergency surgical exploration.
- Muscle suffers irreversible ischaemic damage in 3–4 hours.
- The maximum acceptable delay is 6 hours of warm ischaemia.
- Diagnosed by absent dorsalis pedis or posterior tibial pulse on palpation or Doppler.
- Capillary refill in the toes can be misleading.
- Preoperative angiography unnecessarily prolongs ischaemia time.
- The level of vascular injury is predicted by the fracture or dislocation.
- Management of vascular injuries:
- Direct exploration of the suspected site of injury.
- Immediate revascularisation by temporary shunts, e.g. Javid, Pruitt-Inahara or Sundt.
- Venous injury at, or proximal to, the popliteal vein is also shunted.
- Risk of cardiovascular events as toxic metabolites enter the systemic circulation.
- Renoprotective measures may prevent complications of myoglobinuria.
- Venous injury at, or proximal to, the popliteal vein is also shunted.
- Once circulation is restored, reasses the limb.
- If salvageable, stabilise the skeleton with an external fixator.
- Replace shunts with reversed vein grafts.
- Fasciotomy is required following revascularisation—muscle tends to swell post-operatively.
- If not done, compartment pressure monitoring should be performed repeatedly.
- Direct exploration of the suspected site of injury.
Wound cover
- The decision to proceed with definitive soft tissue cover at primary surgery depends on:
- Immediate definitive bony stabilisation possible?
- Is the fracture configuration fully understood?
- May require further imaging with CT.
- Is the fracture configuration fully understood?
- Plastic surgeon available?
- What flaps are available?
- Zone of injury well defined?
- Recipient vessels available?
- May require CT angiography.
- Patient stable enough to withstand long surgery?
- Immediate definitive bony stabilisation possible?
Temporary
- Topical negative pressure dressing
- Should not be used instead of definitive vascularised soft tissue cover.
- Use for >7 days associated with increased risk of deep infection.
- Should not be used instead of definitive vascularised soft tissue cover.
- Antibiotic bead pouch
- Supplies higher local concentrations of antibiotics than systemic administration.
- Polymethylmethacrylate (PMMA) cement is impregnated with an antibiotic—usually gentamicin or tobramycin.
- PMMA beads are placed in the wound, then covered with a semi-occlusive film dressing.
- Antibiotic elutes from the cement; can remain effective for 21 days.
- Polymethylmethacrylate (PMMA) cement is impregnated with an antibiotic—usually gentamicin or tobramycin.
- Supplies higher local concentrations of antibiotics than systemic administration.
Definitive
- Definitive cover requires vascularised soft tissue.
- UK Standards state this be done by senior specialist teams on a semi-elective basis within 7 days of injury.
- Evidence favours early closure to avoid infection.
- Delay >7 days increases likelihood of friable/fibrotic recipient vessels.
- Evidence favours early closure to avoid infection.
Local or regional fasciocutaneous and muscle flaps
- Reserved for low energy injuries with limited zone of trauma.
- Fasciocutaneous flaps are typically raised on septocutaneous or myocutaneous vessels from peroneal or posterior tibial arteries.
- Can be located with hand-held Doppler to aid flap planning.
- The pedicled medial sural artery perforator flap and reverse flow sural neurocutaneous flap can cover defects over the proximal and distal tibia, respectively.
- Anterolateral thigh flap can be pedicled distally for proximal tibial wounds.
- Common pedicled muscle flaps for lower leg coverage:
- Medial gastrocnemius for knee and proximal third.
- Medial hemisoleus for middle third.
- Bipedicled tibialis anterior for middle third.
- Medial gastrocnemius for knee and proximal third.
Free tissue transfer
- Mainstay of treatment for high energy open tibial fractures.
- Ongoing debate about fasciocutaneous versus muscle flap superiority.
- There is little robust, high quality evidence to support use of one flap over another.
- Proponents of fasciocutaneous flaps say:
- Aesthetically superior.
- Easier to re-elevate for secondary bony reconstruction.
- May be sensate.
- Minimal donor site morbidity.
- Aesthetically superior.
- Proponents of muscle flaps say:
- Conform better to cavities.
- Act as ‘muscle macrophages’, reducing likelihood of infection.
- Experimental data demonstrates improved bone healing.
- Muscle thins over time to provide a good aesthetic result.
- Conform better to cavities.
- Recipient vessels in the lower leg:
- Posterior tibial artery
- Approached medially between flexor digitorum longus and soleus.
- The neurovascular bundle is between soleus and tibialis posterior.
- Also approachable posteriorly, known as the ‘Godina split’:
- Posterior mid-calf incision deepened between the heads of gastrocnemius and through soleus.
- Approached medially between flexor digitorum longus and soleus.
- Anterior tibial artery
- Approached just lateral to the subcutaneous border of the tibia.
- The neurovascular bundle is on the interosseous membrane, between tibialis anterior and long toe extensors.
- Approached just lateral to the subcutaneous border of the tibia.
- Posterior tibial artery
- More proximal recipients include popliteal and superficial femoral vessels.
- Often requires interposition vein grafts.
- Long and short saphenous veins provide additional drainage options if venae comitantes are unsuitable.
- A ‘single vessel leg’ can support a free flap by anastomosing end-to-side.
- Reconstruction of the other injured vessels with vein grafts is considered.
Management of segmental bone defects
Primary bone shortening
- Can be done for segmental defects <5 cm.
- Acute shortening >5 cm may cause circumferential full thickness necrosis of a doughnut-shaped block of adjacent soft tissue.
- May also kink vessels, resulting in distal ischaemia.
Temporary placement of a spacer
- Antibiotic-impregnated PMMA spacers bridge bony defects and maintain limb length.
- They are subsequently removed and the defect reconstructed by other means.
- The Masquelet technique relies on the ‘induced membrane’ that forms around the cement spacer to revascularise morsellised cancellous bone graft, placed at a second stage.
Bone grafting
- Delayed cancellous bone grafting is usually performed 6 weeks after injury, when soft tissues have healed.
Primary bone shortening and subsequent lengthening
- Bone lengthening is accomplished by a circular frame.
- Examples: Ilizarov, Taylor Spatial Frame™.
- Lengthening at the site of fracture is bone distraction.
- Lengthening by corticotomy at a remote site is bone transport.
- The bone is lengthened by turning special screws on the frame.
- Lengthening usually proceeds at 1 mm/day.
- After lengthening, the frame is left in situ for a period of consolidation.
Reconstruction with vascularised bone
- Common sources include the free fibula and deep circumflex iliac artery (DCIA) flap.
- Ribs are usually too thin.
- Up to 18 months or more is required for bone strengthening and hypertrophy.
- During this period, full weight-bearing risks fracture.
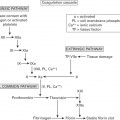
Acute compartment syndrome
- Compartment syndrome is elevation of interstitial pressure in a closed osseofascial compartment that results in microvascular compromise.
- It is a surgical emergency.
- Missed diagnosis causes irreversible neuromuscular ischaemia and significant long-term morbidity.
- Compartment syndrome can be caused by:
- Fractures, both closed and open
- Never assume open fractures concomitantly decompress compartments.
- Soft tissue trauma
- Arterial injury
- Prolonged limb compression in an obtunded patient
- Burns.
- Anterior and deep posterior compartments of the lower leg are most affected.
- These are also the most frequently missed during fasciotomy.
- It is a surgical emergency.
Pathophysiology
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree