Pemphigus vulgaris (PV) is an autoimmune blistering disorder with a complex etiology involving an interplay of genetic as well as environmental factors, most of which remain unknown. Despite the identification of several human leukocyte antigen (HLA) alleles as risk factors for disease, no other non-HLA genes have clearly been implicated in disease susceptibility. Newer candidate gene and whole-genome approaches are needed to illuminate the full palate of genetic risk elements in PV. Based on this information, genetic-based tools can be expected to provide a scientific rationale for future clinical decision-making by physicians and facilitate an era of personalized medicine.
Pemphigus is 1 of 4 major groups of autoimmune blistering diseases (AIBD) characterized by autoantibodies against intercellular junctions in the skin. Pemphigus vulgaris (PV) is representative of the pemphigus group. It is a prototypical organ-specific human autoimmune disease that presents clinically with flaccid blister formation on the skin and mucous membranes. Histologically, there is an intraepidermal split resulting from acantholysis of suprabasilar keratinocytes. Autoantibodies against desmoglein 3 (Dsg3) in humans have been primarily implicated in lesional disorders, and can passively transfer the disease in mice. More than 50% of patients with PV also have circulating autoantibodies against desmoglein 1 (Dsg1), and their presence may be required for the development of nonmucosal lesions. More recently, nondesmoglein targets are also being uncovered in PV. Nevertheless, the etiopathogenesis of PV, particularly the role of genetic factors in the generation of autoantibodies that are relevant to disease development and clinical presentation, remains poorly understood.
Numerous studies provide support for the importance of genetic factors in PV. As for many autoimmune conditions, several lines of evidence make a compelling argument for a complex, polygenic basis for disease involving multiple, but an unknown number of genetic loci, as well as nongenetic factors. Genetic and nongenetic/environmental elements are believed to contribute to the dysregulation of normal immune tolerance that ultimately leads to the development of autoimmunity in the skin. At present, the full complement of susceptibility loci remain unidentified, and it is not possible to identify individuals at risk.
This article reviews the evidence for the genetic basis of disease from studies on (1) population analyses, (2) autoimmune comorbidity, and (3) familial aggregation. It reviews the data implicating specific (1) human leukocyte antigen (HLA) and (2) non-HLA genes in the development of PV. It also outlines current and emerging strategies intended to identify disease susceptibility loci including (1) candidate gene and (2) whole-genome approaches. The role of genetics in terms of biologic and clinical relevance to our understanding and management of disease is also discussed.
Genetic basis of disease: what is the evidence?
Population Studies
The overall prevalence and incidence of PV is low, but it varies significantly in various ethnic populations. As discussed by Gupta and colleagues elsewhere in this issue, the incidence of PV to is reported to range between 0.076 per 100,000 person years and 5 per 100,000 person years. This wide range of incidence across worldwide geographic locations supports the hypothesis that genetic factors linked to specific racial and ethnic populations affect disease incidence. However, population-associated environmental factors such as those related to microbial variance and social-psychological and economic variables cannot be ruled out and are likely to be intertwined with relevant genetic factors. In general, PV incidence seems lower in more northern locations. A north-south gradient has previously been observed in type 1 diabetes mellitus and multiple sclerosis and has been suggested to be related to the relative dominance of Th1-type versus Th2-type pathogens in particular geographic locations. It remains to be determined how specific genetic and environmental factors influence the development of PV across the globe.
Similarly, the literature and our own studies indicate a female predominance in PV, as is often reported in autoimmune conditions. The female/male ratio ranges from 1.1:1 to 2.25:1 (see the article by Gupta and colleagues elsewhere in this issue). This female bias can be interpreted as evidence for the role of genetics in disease. Nongenetic mechanisms may be involved, likely in concert with heritable elements, in influencing the expression of PV in women rather than men.
For several autoimmune conditions, twin and adoption studies have been able to provide strong evidence for genetic susceptibility. The comparison of concordance rates among monozygotic versus dizygotic individuals allows a focus on the relative contribution of genetic risk to disease. However, because large-scale population-based assessments are required and the prevalence of PV is low, twin and/or adoption studies are not available.
Autoimmune Comorbidity
It has long been observed that patients with 1 autoimmune condition have an increased likelihood to develop 1 or more other autoimmune diseases. Work from others, as well as our group, indicates that this is the case in PV. In a survey of 171 patients with PV, we found that nearly 21% of patients report having a comorbid autoimmune condition (please refer to Fig. 1 in the article by Gupta and colleagues elsewhere in this issue). Thyroid disease was by far the most common PV-associated condition, occurring in nearly half of those reporting a concomitant autoimmune disorder. Autoimmunity affecting the thyroid has been similarly linked with several autoimmune diseases. This may be, at least in part, because of the higher incidence of thyroid disease compared with other autoimmune conditions. Nevertheless, these data suggest that PV and autoimmune thyroid disease may share at least some pathogenetic factors. Recently, a common-cause theory for the development of autoimmunity based on shared disease genes and polymorphisms has emerged. Data from our laboratory on overlapping gene expression changes within several autoimmune skin diseases, such as alopecia areata, psoriasis, and vitiligo, and with other nonskin autoimmune conditions, lend experimental support to this hypothesis. The subset of PV-associated susceptibility loci that are disease specific and those that are related to an overall autoimmune diathesis remain to be determined.
Familial Aggregation
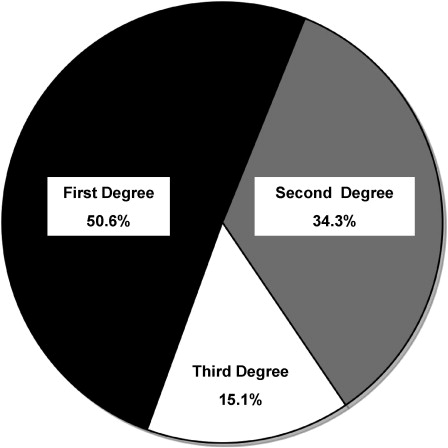
Additional support for the genetic basis of disease comes from the increased incidence of autoimmunity in blood relatives of autoimmune patients. In our survey of 171 patients with PV, we found that 48% reported having 1 or more relatives with autoimmune disease (please refer to Fig. 2 in the article by Gupta and colleagues elsewhere in this issue). Type 1 diabetes was reported most commonly, followed by autoimmune thyroid disease. PV itself was reported less commonly, occurring in the relatives of 2.9% of patients. Although familial cases of PV have been cited in the literature, they are rare.
Multiple PV cases within families are most likely to occur in first-degree relatives. There is a significantly higher incidence of all autoimmune diseases in first-degree relatives of patients with PV compared with second-degree and third-degree relatives ( Fig. 1 ). These findings, in which autoimmunity correlates more strongly with an increased genetic commonality (first>second>third degree), strongly support the genetic basis of PV. However, the influence of environmental factors cannot be ruled out.