Introduction
This chapter is our vision for the way forward from gaps to goals . We first summarize the major gaps that exist in order to provide optimal care for HS patients. We then outline the journey to address these gaps and the programs and initiatives required to do this ( Fig. 35.1 ).
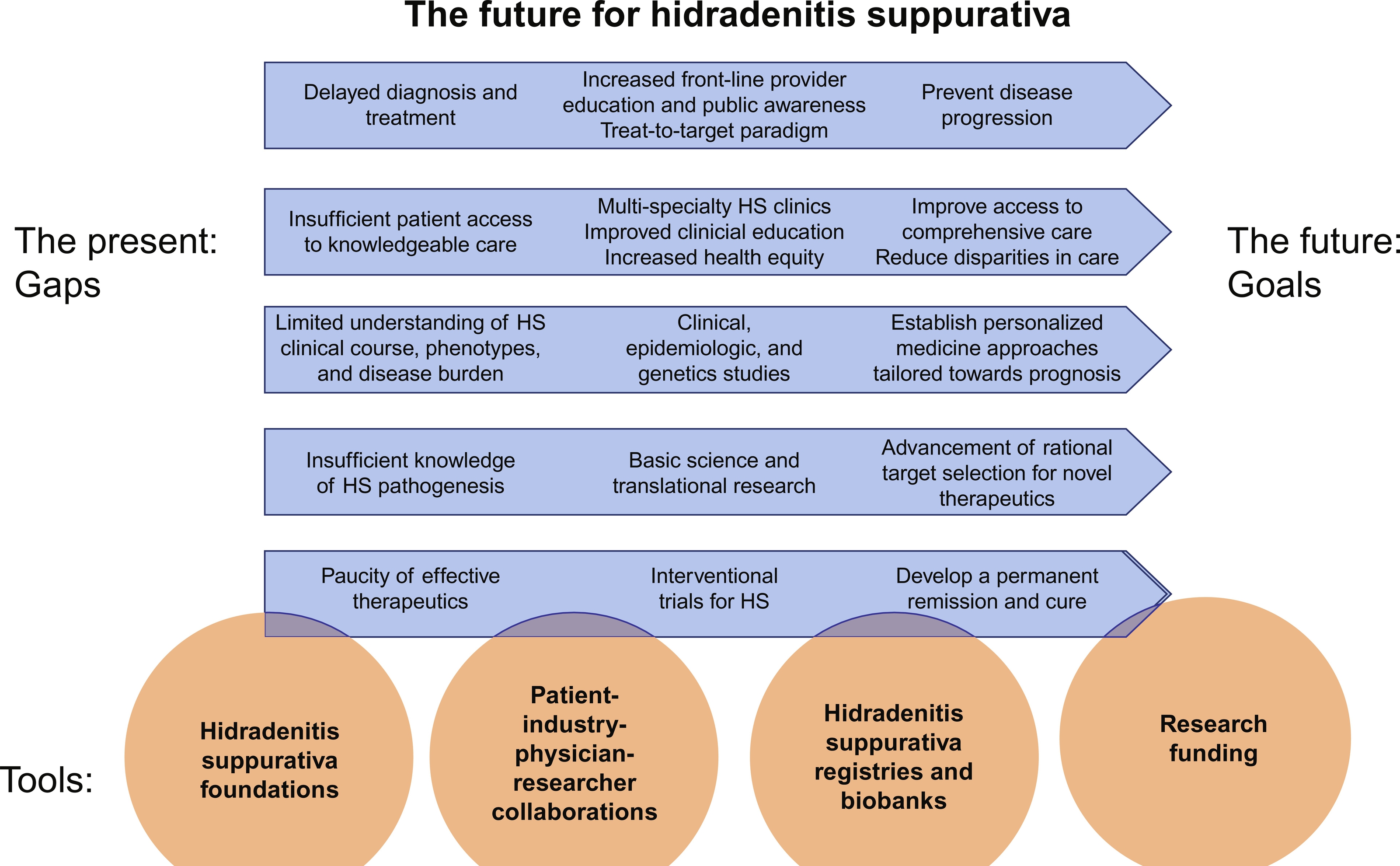
Major Gaps to Address Now for Optimal Care of HS Patients in the Future
- 1.
Delayed diagnosis and treatment may lead to uninhibited disease progression
It is unacceptable that there is a prolonged delay for most patients to be diagnosed with HS, on average, about a decade. It is critical that patients are diagnosed promptly so they may be treated much earlier in their disease course. As inflammation progresses, it causes irreversible tissue destruction and scarring in the region of disease activity. Given the relapsing and remitting nature of HS, a dynamic treatment plan is often needed to care for HS both during flares and periods of remission. Treatment goals are to improve quality of life, prevent permanent disfigurement, and reduce the need for visits to acute care settings. Another major gap is that it has not yet been definitively demonstrated that preventing HS from advancing will avert progression and scar formation. Early and proactive treatment has been shown in other destructive inflammatory diseases, including rheumatoid arthritis, to limit disease progression via the treat-to-target paradigm wherein treatment modalities are aggressively and proactively managed to maximize long-term health-related quality of life.
- 2.
Suboptimal access to care impedes HS management
Patients deserve timely access to knowledgeable care and effective treatment that leads to disease remission. HS patients describe insufficient access to healthcare providers who are educated about HS, as well as geographical restrictions to HS providers. In terms of treatment limitations, there is only one FDA-approved treatment for HS; adalimumab (Humira, AbbVie) based on the PIONEER I and II studies. Patients in some states face challenges accessing this therapy. There are also issues with access to off-label and procedural therapies, as well as financial difficulties in obtaining wound care supplies. Disparities in access to care have been suggested in this disease that predominates in women and Blacks, and it is already documented that Blacks with HS experience significant healthcare disparities.
- 3.
Limited understanding of HS clinical course, clinical phenotypes and disease burden prevents personalized medicine approaches
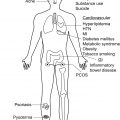
The ultimate goal of providing personalized treatment requires comprehensive knowledge of clinical course, clinical phenotypes, and disease burden. These features may hold important clues to predict disease prognosis and treatment response. Patients progressing along a given clinical course, as well as patients with different phenotypes, may respond to specific treatment approaches more positively than others. In addition, it is now appreciated that there are significant systemic co-morbidities associated with HS. Understanding these cardiometabolic, rheumatologic, oncologic, and psychological co-morbidities may also help tailor treatment for patients with HS.
- 4.
Insufficient understanding of HS pathogenesis inhibits therapeutic development
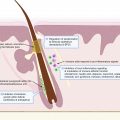
The unique HS pathogenic signature is still not known; this has hindered the development of novel targeted therapies. Accelerated basic and translational research is imperative to rationally target dysregulated pathways and guide therapeutic development.
- 5.
Paucity of novel therapeutics currently limits finding a permanent remission or a cure
There is a paucity of high-quality evidence for HS treatments. To address this deficit, carefully designed randomized controlled trials (RCTs) of novel targeted therapies are needed. While developing these HS clinical trial programs, special considerations include patient selection and recruitment, development of outcome measures, the role of placebo or active treatment comparators, and conduct of concomitant translational research for pathogenesis and biomarkers during the RCTs.
The Journey From Gaps to Goals
- 1.
Clinician, patient, and public education
To improve time to diagnosis and treatment, education of clinicians, patients, and the general public is paramount. Increasing awareness of HS diagnostic criteria across medical specialties will reduce under-diagnosis, misdiagnosis, and diagnostic delay. Education programs for front-line providers who first encounter patients with HS symptoms and signs could reduce the time to diagnosis and dermatology referral. Another major goal is to educate clinicians as treatment paradigms are developed to induce early disease remission and prevent progression, such as treat-to-target approaches. In addition, increased awareness of the clinical signs and symptoms of HS in the general public could decrease the stigma associated with HS. Physicians can work with patient organizations to improve patient education.
Given the high disease burden and healthcare costs associated with HS, efforts should be made to improve both pharmacologic and nonpharmacologic treatment plans. An individualized written home-management action plan has shown efficacy in other chronic inflammatory conditions, such as asthma and atopic dermatitis. Patients are provided with written instructions on flare recognition and corresponding step-up/step-down treatment at home. This is highly applicable to HS, another chronic disease with intermittent flares. Providing HS patients with proper disease education and a written treatment action plan may empower them to manage acute HS flares at home as opposed to seeking care in an acute care setting.
- 2.
Multi-specialty/interdisciplinary HS clinics
A key component of optimizing current HS management is developing specialized HS treatment centers and multi-specialty HS clinics anchored by the dermatologist, as discussed in Chapter 32 . Multi-specialty care can take different forms, including a multidisciplinary clinic with available specialists in one clinic, or referral to a network of specialists interested in and knowledgeable about HS. This comprehensive approach is modeled on the delivery of cancer care. First, those who provide direct care of HS lesions are essential, such as dermatologists, surgeons, nurses, and wound care specialists. Second, access to healthcare providers who care for those with comorbidities, as discussed in Chapter 8 , is also key. This includes assessment and treatment by pain specialists, psychologists, psychiatrists, rheumatologists, endocrinologists, cardiologists, primary care physicians, as well as counselors, dietitians, or nutritionists. Patient-led HS support groups established within a specialized HS treatment center can also provide a valuable resource for HS patients and their families, as discussed in Chapter 33 .
Caring for patients in the context of a multi-disciplinary setting can also improve our understanding of the most effective ways to integrate therapeutic approaches for HS management, including conventional medications, physical procedures such as surgery and lasers, and complementary and alternative medicine (CAM). An interdisciplinary approach can also facilitate the timely management of acute flares of HS. Developing multi-disciplinary HS clinical care in areas of higher HS prevalence can also help to reduce health disparities.
- 3.
Clinical, epidemiology, and genetics studies
Personalized medicine requires knowledge of patient and disease characteristics that guide treatment selection, which can be gained by long-term studies of clinical course, and large-scale epidemiology and genetics studies of HS patients. Earlier chapters have outlined our current knowledge of these areas, but there is still much to be learned. Studies on the role of imaging modalities to facilitate diagnosis, lesion assessment, and treatment planning are also needed. In addition, skin and serum biomarkers may also be useful to guide treatment choice and develop predictors of response, and further investigation in this area of research is warranted.
- 4.
Basic science and translational research
The current understanding of HS pathogenesis is described in earlier chapters. To expand and accelerate this knowledge, increased basic science and translational research are needed, such as in the fields of genetics, immunology, and the human microbiome. Increased funding for HS research is critical for these efforts.
- 5.
Clinical trials for HS
Chapter 34 outlined the current pipeline of treatments for HS. To reach our ultimate goal of permanent remission and a cure for HS, greater numbers of clinical trials in all disease stages are needed. Guidance for optimizing clinical trials in HS can be developed in conjunction with physicians, industry partners, regulatory bodies as well as patients. Some key considerations include the design of RCTs and pragmatic clinical studies, choice of comparator arms, dosage regimens, development and implementation of outcomes measures, biomarker studies within clinical trials, and patient recruitment.
Programs & Initiatives to Get Us There
Hidradenitis Suppurativa Foundations
Foundations have a very important role to develop and coordinate many activities to meet the presented goals. There are currently four international foundations for HS, listed as follows:
- •
United States Hidradenitis Suppurativa Foundation, Inc. (HSF)
- •
Canadian Hidradenitis Suppurativa Foundation (CHSF)
- •
European Hidradenitis Suppurativa Foundation (EHSF)
- •
Asia-Pacific Hidradenitis Suppurativa Foundation (APHiS)
Examples of activities that foundations have conducted and promoted include:
- •
Foundations play a key role in helping to identify healthcare providers who care for patients with HS and provide this information to the public (such as on the HSF website).
- •
There is an important role in clinician education for foundations. This can be through the development of treatment guidelines and providing recommendations for important aspects of HS care including screening of disease co-morbidities and pain management.
- •
Foundations can host scientific conferences. The EHSF has hosted nine scientific meetings, and the HSF and CHSF have jointly hosted the Symposium on Hidradenitis Suppurativa Advances (SHSA) for the past four years. Future education efforts should include healthcare providers from other specialties that are involved in HS care.
- •
They also play an important role in educating HS patients, friends and families, and the general public. The EHSF, for example, has pioneered an in-person HS School for HS patients and their families that has been well received.
- •
Foundations can also promote, develop, and support clinical, epidemiology, genetics, basic science, and translational research through grants programs. For example, the HSF offers the Danby HSF Research Grants every year to help investigators generate pilot data to apply for larger funding with research institutes. Foundations can provide infrastructure investments in programs such as registries and biobanks to facilitate future research projects.
- •
Consensus efforts, such as defining HS lesion morphology, clinical treatment algorithms, treat-to-target paradigms, clinical trials guidance, and practice gaps, can be promoted and conducted by foundations.
- •
Another important role for foundations is patient advocacy, directed by patients who can guide priorities and lead in this area, supported by healthcare provider allies.
- •
Foundations can provide much-needed support to patients and families through partnership or sponsorship of support groups. For example, the US-based Hope for HS works closely with the HSF to promote the development of local support groups.
- •
There is also a potential role for an umbrella international HS Foundation consortium to bring together these national foundations, raise awareness of HS across the world, share processes, and collaborate on activities to reach shared goals.
Patient-Industry-Academia-Physician-Researcher Collaborations
- •
The International Dermatology Outcome Measures (IDEOM) group established the Hidradenitis SuppuraTiva cORe outcomes set International Collaboration (HISTORIC). This collaborative effort has determined core domains to measure in HS clinical trials: pain, physical signs, HS-specific quality of life, global assessment, progression of course, and symptoms. The priority is improved outcome measures for clinical trials, but these instruments will also be extremely useful for clinical care.
- •
Collaborations between patients, physicians, and researchers also enable education regarding each groups’ perspectives and priorities. Global VOICE was an international patient survey conducted by HS physicians’ clinics. Another example of a town hall style educational meeting about HS with clinicians, researchers, patients advocates, and patients is archived on the Clinical Directors Network website.
- •
There are also numerous opportunities for research collaborations. This includes individual principal investigators developing projects with industry partners and industry supporting larger registry and biobanking projects.
- •
Patient involvement in foundation and research priority setting, education of providers (such as at scientific meetings), and clinical trial design, can also be beneficial for all. Ideally, diverse HS patients are represented in these collaborative projects.
HS Registry and Biobanks
- •
Registries and biobanks offer powerful opportunities to address many of the gaps and goals discussed.
- •
The first HS registry established was the European Hidradenitis Suppurativa Registry (HSR). The mission of this registry is to establish a unique database with a large prospective cohort of patients with HS. This registry is designed to collect data on the current status and follow-up of patients with HS.
- •
The US has recently initiated the Hidradenitis Suppurativa Prospective Observational Registry and Biospecimen Repository (HS PROGRESS). The mission of HS PROGRESS is to facilitate HS research through collaboration between investigators, clinicians, patients, and industry in order to improve the lives of people living with HS. Specific goals include:
- •
To develop a longitudinal cohort of HS patients in all stages of disease to understand HS clinical course.
- •
To collect biospecimens from HS patients for ongoing and future studies.
- •
To establish a cohort of consented patients who can be contacted for future clinical studies.
- •
To develop an infrastructure to support pilot projects that can lead to NIH funding.
- •
- •
Other international groups have started an Italian registry and a Scandinavian registry for HS (HISREG).
- •
Additional registry efforts are important to expand our knowledge of HS beyond Europe and US populations. Analysis of aggregate data across different registries will also be informative to understand HS in different populations.
- •
Patients can be recruited to registries from HS and multi-specialty clinics, leading to important real-world data and experience that will direct optimal clinical care and can help generate research questions.
Research Funding
For all these endeavors, research funding is critical. Some sources of funding include the following:
- •
Foundations. For example, the HSF offers Danby HSF Research Grants every fall. Other programs are in the planning stages for endeavors such as translational research and mentoring grants.
- •
Federal funding. NIH/NIAMS has offered a Funding Announcement for R21 and R01 grants specifically for HS translational research (excluding clinical trials).
- •
Industry has partnered with principal investigators at academic centers to perform translational studies to identify therapeutic targets and biomarkers.
- •
Philanthropic contributions and grassroots campaigns are future sources to be investigated for research funding.
Conclusion
We have presented major obstacles to optimal care of HS patients and suggested initiatives and endeavors to address these gaps. These activities will enable us to achieve the goals of preventing disease progression, optimizing current management, personalizing care, and developing novel therapeutics, with the ultimate goal of attaining permanent remission and cure. Mechanisms to drive and enhance these goals are underway, including active international HS Foundations, numerous collaborations between major stakeholders, formations of HS registries and biobanks, and an improving research funding landscape. From neglect to awareness, we are confident that the future of HS is finally looking promising.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree