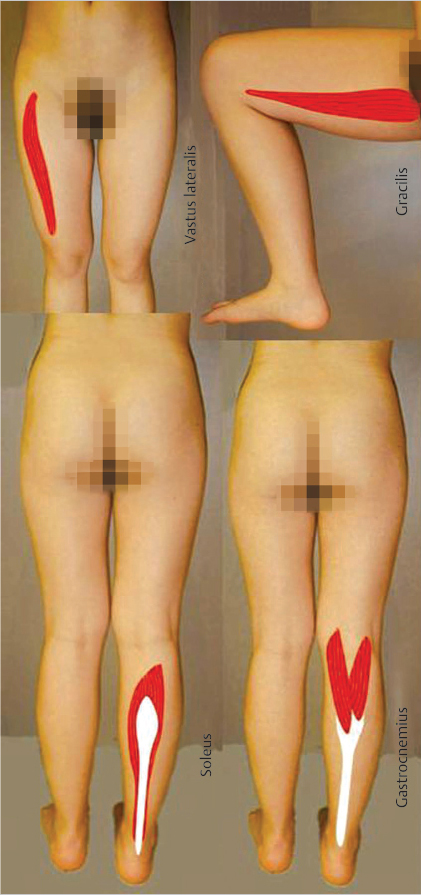
14 The Basic Practical Lower Limb Muscle Local Flaps
Summary
Outcome studies of functional lower limb salvage cite no difference if a muscle or cutaneous flap were used. Muscle flaps will never be obsolete, inarguably when, for example, a dynamic tissue transfer were indicated! Muscles are highly malleable, facilitating three-dimensional insetting. Since they are relatively nonbulky, the ultimate contour will not be obtrusive. Their vascular anatomy has few anomalies so that harvest of a muscle flap is a relatively simple process. Historically, the gracilis, gastrocnemius, and soleus muscles have been proven to be extraordinarily efficacious as local flaps in the lower extremity. The vastus lateralis muscle is invariably well known due to its intimate connection with the “workhorse” anterolateral thigh flap. Although many other lower limb muscle flap options exist, these four should necessarily be a part of the repertoire of every reconstructive surgeon.
Keywords: gracilis, medial and lateral gastrocnemius, soleus, vastus lateralis, muscle flaps
14.1 Introduction (▶ Fig. 14.1)
Many might say that muscle flaps have become obsolete1 now that there has been a paradigm shift in the selection of perforator flaps instead for lower extremity soft-tissue reconstruction.2 But the reconstructive matrix should remind us that the optimal reconstructive strategy must take into consideration more than just what will be the flap that will be chosen.3 There should be a realization that multiple factors are intertwined including the surgeon’s capabilities and preferences, the availability of technological resources, and proper evaluation of the patient’s own comorbidities and specific tissue requirements that must be completely understood.3,4
If the ultimate goal is functional limb salvage, note that outcome studies have shown no difference whether muscle or skin flaps are used.5,6 However, the number one absolute indication for selection of a muscle flap, that is inarguable, is if a dynamic transfer is indicated (see Chapter 20).1,7 Muscle flaps are highly malleable and relatively nonbulky, allowing easier contouring for three-dimensional insetting.1 The vascular pedicle for muscle flaps is more anatomically consistent, compared to perforator flaps where anomalies are always anticipated.1 This fact alone allows a more expeditious harvest of a muscle flap, which may be most important in the critically ill patient.1 In fact, in general the overall elevation of a muscle flap is comparatively simple. Usually, donor site morbidity is less, except for the loss of that muscle function; as always, a primary closure leaving a linear scar is possible.8 Muscle flaps are also important in combination with their corresponding perforator flap to capture all the advantages of chimeric flaps.9 Finally, if a perforator flap based on a musculocutaneous perforator is unsuccessful, the muscle that had the requisite intramuscular dissection by definition should have had enough function preservation that it could then be the back-up flap option.10,11
A step back in history to review the inaugural book on muscle flaps by Mathes and Nahai12 reveals the anatomy and how to use 22 intrinsic muscles as flaps in the lower limb. Over time, the majority have been proven to be too small or have segmental or variable blood supplies, and so they are unreliable for most of the lower extremity problems.13 For valid reasons, some flaps that many surgeons may prefer in their practices will not be discussed in this chapter. Is the distal-based peroneus brevis muscle flap always reliable?14,15 Will not the segmentally supplied sartorius muscle be at risk in the patient with significant peripheral vascular disease?16,17 Does not use of the rectus femoris muscle result in a loss of 15 degrees of knee extension, which would be problematic in the athlete?16 Are there not better alternatives to the rectus abdominis flap,18,19 such as the anterolateral thigh flap where the high risk of hernia or other abdominal wall morbidity can be altogether avoided?4,20 Is not the tensor fascia lata perforator flap without muscle better for the ambulatory patient?11 At least from a traditional standpoint, the gastrocnemius21 and soleus22 muscles as local flaps for the lower leg have long proven their value as basic flaps that must be part of the repertoire of every reconstructive surgeon. The gracilis muscle is a “workhorse” flap not just for the lower extremity,23 but also throughout the body, with special emphasis deserved for its dynamic versatility.24 The overwhelming adoption of the anterolateral thigh flap as the “ideal soft-tissue flap,”25 requires an intimate knowledge of the vastus lateralis muscle through which its perforators are closely aligned. For these reasons, this chapter is intended to provide enough that is basic to allow a fundamental understanding of the role of lower limb muscles for lower limb reconstruction.
References
[13] Hallock GG. Sagittal split tibialis anterior muscle flap. Ann Plast Surg. 2002; 49(1):39–43
[22] Hallock GG. Getting the most from the soleus muscle. Ann Plast Surg. 1996; 36(2):139–146
14.2 Chapter 14A: The Medial and Lateral Gastrocnemius Muscle Flaps
14.2.1 Introduction to the Medial and Lateral Gastrocnemius Flap
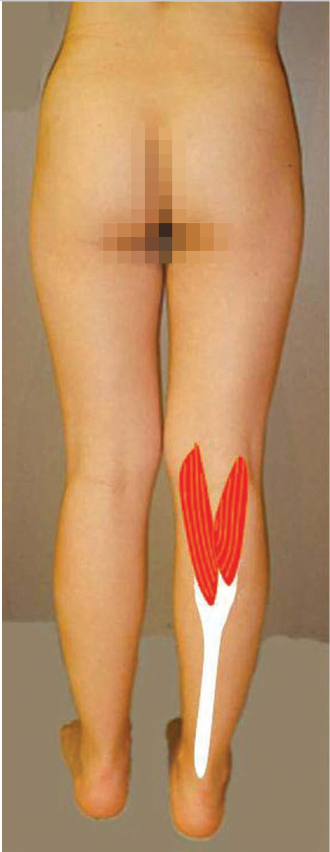
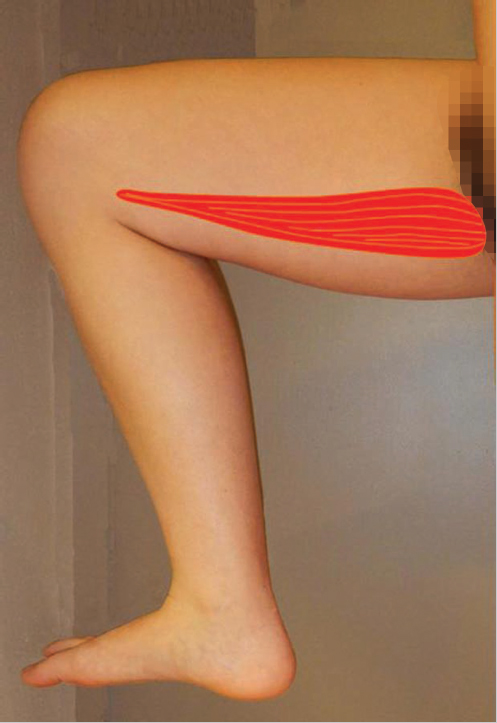
Over the past half century, many would say that the gastrocnemius (▶ Fig. 14.2) and soleus muscles have been the “dynamic duo” (▶ Fig. 14.3) when considering coverage of the lower leg.1 In actuality, the medial and lateral heads of the gastrocnemius muscle alone have been a “dynamic duo” and deserve their appellation as lower leg “workhorse” flaps.2,3 Long before this achievement, Stark4 had recognized the value of the pedicled gastrocnemius muscle for treatment of infected compound fractures, but only after similar repetitive reports by Barfod and Pers5 as well as Ger and Efron6 were surgeons convinced of their efficacy. McCraw et al7 defined their reliable musculocutaneous territory, and believed as such were more versatile since reach was significantly extended. The gastrocnemius muscle has even been used in cross-leg fashion as a regional flap.7,8 With the acquisition of microsurgical capabilities, this muscle soon proved useful under special circumstances as a free flap donor site for the more distal lower limb.9,10 Neurovascular anastomoses have even permitted dynamic transfers for forearm function restoration.9,11 Indeed, similarly an innervated pedicled gastrocnemius muscle flap has been attempted as an even simpler way to correct foot drop.3
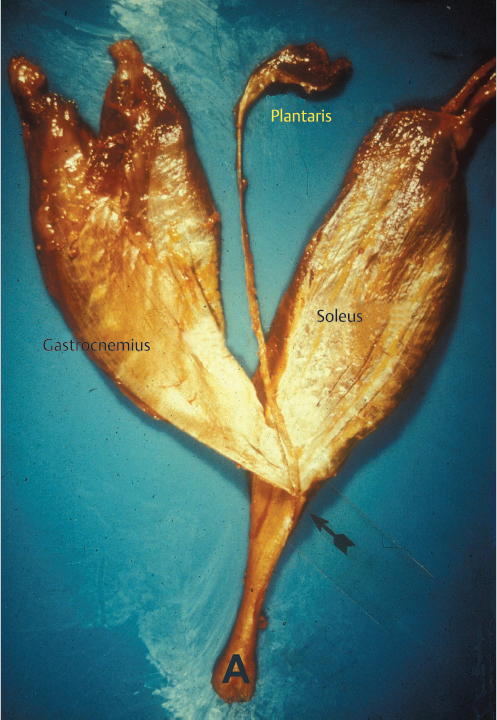
Fig. 14.3 The aponeuroses of the gastrocnemius and soleus muscles join to form the triceps surae (arrow), which continues as the Achilles (A) tendon. Note the plantaris tendon sandwiched between these two muscles.
The medial head of the gastrocnemius (MHG) muscle, since usually larger and longer (▶ Fig. 14.4),1,12,13,14 has traditionally been selected to correct problems of the distal thigh, region of the patella and knee joint, and proximal third of the leg itself (▶ Fig. 14.5).1,15 The less often used lateral head of the gastrocnemius (LHG) muscle still is important and a better choice for lateral knee or defects around the proximal fibula (▶ Fig. 14.6).3,16 The gastrocnemius muscle as a whole not only assists plantar flexion of the foot but also is a knee flexor and stabilizer.17,18 Yet the soleus muscle is a more powerful agonist with regard to ankle movement; so, many patients even after transfer of both gastrocnemius heads can ambulate normally, suggesting that either or both heads are expendable.2 However, Daigeler et al7 in an extensive review of their experience found that patients had some restrictions in basic activities like walking upstairs, standing, or jumping on tiptoes, and especially running—even if only a single head had been used as a flap.
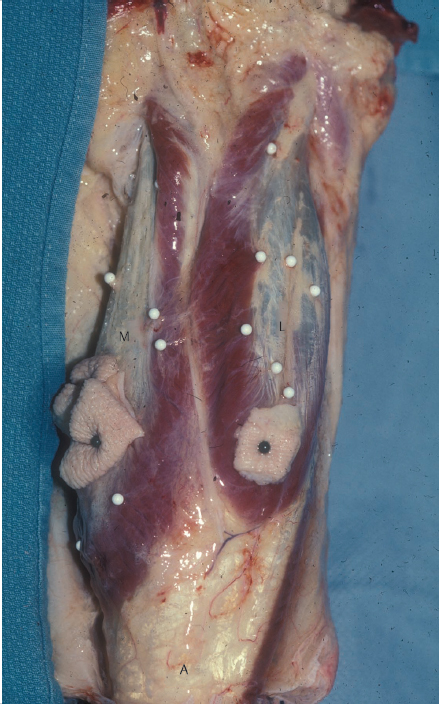
Fig. 14.4 In this posterior view of the gastrocnemius muscle in a fresh amputation specimen, as is not unusual, the muscle fibers of the medial head (M) have a more distal insertion than those of the lateral head (L) upon the aponeurosis that becomes the Achilles (A) tendon.
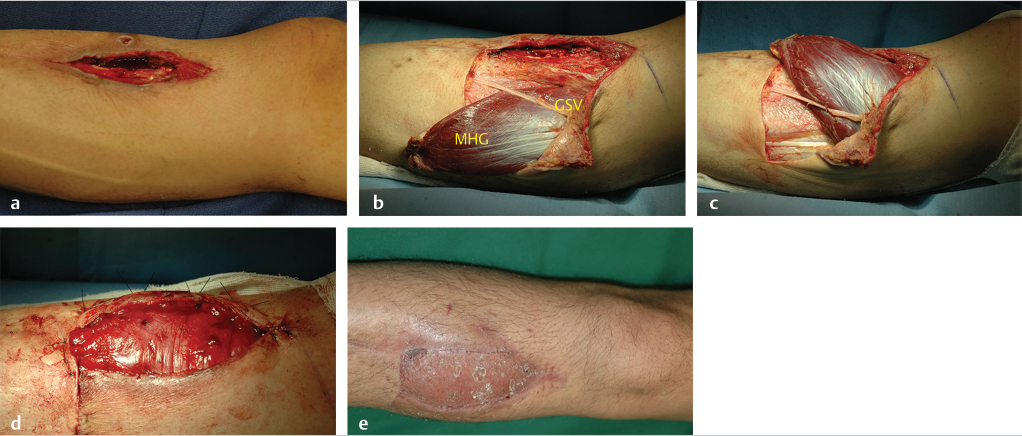
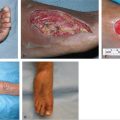
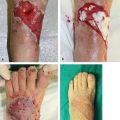
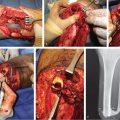
Fig. 14.5 (a) Chronic osteomyelitis of the right proximal tibia. (b) Transverse subfascial incision dropped from the distal margin of the open skin wound to the posterior midline (with preservation of the greater saphenous vein [GSV] and saphenous nerve) allowed access to the medial head of the gastrocnemius (MHG) muscle, taken with a short cuff of aponeurosis around the distal insertion of its muscle fibers. (c) MHG then rotated anteriorly under the GSV to fill the bony defect. (d) Perimeter of muscle inset as a safety factor underneath all the skin defect margins while covering the exposed bone in its entirety. (e) Healed skin grafted MHG flap.
14.2.2 Attributes and Detriments
Attributes
• Local option for distal-third thigh, knee region, and proximal-third leg coverage.
• Expendable.
• Straightforward harvest.
• Possible Achilles tendon repair.
• Minimal donor site deformity.
Detriments
• Not suitable for athlete.
• Musculocutaneous versions leave nonaesthetic donor site residue.
• Lateral head risks peroneal nerve palsy.
14.2.3 Anatomical Considerations
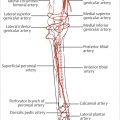
Literally the “belly of the calf,” the gastrocnemius muscle is the most superficial muscle of the posterior compartment of the leg.13 There are two distinct heads, each with a discreet neurovascular supply. The medial head originates from the popliteal surface of the femur just superior to the medial condyle, whereas the lateral head arises from the lateral epicondyle of the femur.3,18 The proximal portion of each head forms the borders of the lower triangle of the popliteal fossa.3 Distal to this, the two heads are fused together at a midline raphe that can be indistinguishable if not for the presence of the medial sural cutaneous nerve and accompanying branch of the lesser saphenous vein that traverse a groove between the two.18 The muscle fibers of each head again become distinct when they insert on the posterior aspect of its aponeurosis (▶ Fig. 14.4) that joins its counterpart from the soleus muscle to form the triceps surae (▶ Fig. 14.3). This conjoined aponeurosis becomes the Achilles tendon that ultimately inserts on the calcaneus. Most consider the medial head to be the larger and longer of the two,1,12,13,14 although Andejeklov et al19 in a recent anatomical study of fresh cadavers suggest that there is really no significant difference between them.
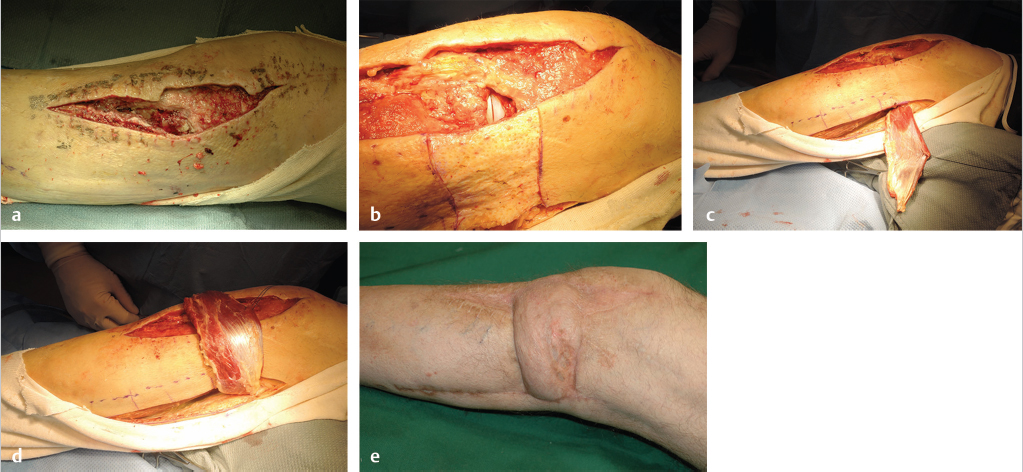
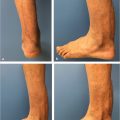
Fig. 14.6 (a) Debrided lateral parapatellar incision used for total knee replacement. (b) Implant exposed in lateral opening into joint capsule. Note area of skin de-epithelialization extending immediately inferiorly to the open joint to join the incision posteriorly used to harvest the lateral head of the gastrocnemius (LHG) muscle, which was selected since this head was the closest to the area needing coverage. (c) LHG withdrawn from incision that had proceeded longitudinally 2 to 3 cm below the fibula (marked by dotted line). (d) LHG transposed over the de-epithelialized skin to avoid the risk of compression in a subfascial tunnel in this thin individual, with tension-free coverage of the joint capsule. (e) Healed skin grafted LHG flap with prosthesis preservation. The donor site was closed primarily.
The arterial supply to each head of the gastrocnemius muscle always arises from the posterior surface of the popliteal artery just above the knee joint, but may bifurcate from a single common vessel or have double pedicles.9 Most often, however, there is a distinct solitary branch to each head, making each a type I muscle as defined by Mathes and Nahai.20 The caliber of these arteries at their takeoff is about 2 to 3 mm, and potential pedicle length is 2 to 5 cm.18,21 Soon after entering the hilum of each muscle, each source vessel bifurcates into two or more branches that run longitudinally throughout the length of the muscle,18 and is the basis for safely splitting the muscle.22 Communicating branches between the heads through the more distal fusion of the muscles will provide sufficient circulation such that one head can survive on that supplied by the other.23,24 Major musculocutaneous perforators from the longitudinal branches typically are found in the more proximal half of the muscle.25 Numerous venae comitantes accompany the arteries until eventually emptying into the popliteal vein.18,21
Motor innervation of the gastrocnemius muscle is by a distinct branch from the medial popliteal nerve that accompanies the dominant vascular pedicle into the hilum of each head.18 According to Taylor et al,26 this is a type III mode of innervation, since having multiple branches from the same nerve trunk to the gastrocnemius muscle as a whole, but an argument could be made that each head if considered individually would have a solitary unbranched nerve entering that muscle, and thus be a type I muscle. Especially when assessing the use of the LHG, it must be understood that the common peroneal nerve will be found near the head and neck of the fibula superficial to that muscle and deep to the biceps femoris.12
14.2.4 Anatomical Variants and Potential Pitfalls
Musculocutaneous Flaps
Perforasomes
The perforasome for each head of the gastrocnemius muscle approximately corresponds to the dimensions of that component. For the MHG muscle, this would stretch from the posterior midline to the medial tibial border, in contradistinction to the LHG muscle that would proceed from the posterior midline to the lateral border of the fibula.7 Any distal extension beyond the point of insertion of the MHG or LHG muscle at the triceps surae will be safest if the Achilles tendon sheath or loose layer overlying that tendon and more proximally the involved muscle is kept attached to the subfascial layer under the cutaneous component of this composite flap,27 as otherwise that portion would behave like a random flap where survival of a length-to-width ratio greater than 1:1 would be unlikely.21 As a musculocutaneous flap, McCraw et al,7 even without inclusion of the tendon sheath, found that the cutaneous portion of the MHG musculocutaneous flap could extend up to 5 cm above the medial malleolus, or the LHG up to 10 cm proximal to the lateral malleolus. This has provided enhanced reach of these flaps even to the mid-third of the tibia. Agarwal et al28 after a series of delay tactics used an LHG musculocutaneous flap to even reach the midlateral femur.
Donor Site Morbidity
Although there may be some minor ways to improve the appearance of the usually bulky musculocutaneous flap,29 the donor site for this variation is aesthetically unacceptable to most,2,13,18,22 and rightfully so if the Achilles tendon itself would become exposed. Pico et al30 also found that there was an increased risk of peripheral edema and cutaneous sensory loss compared to a muscle flap alone. Elghamry27 avoided the latter concern by using a “myotendon sheath flap” that had no cutaneous component. This had a shorter reach than the musculocutaneous flap, but sufficed sometimes also for midleg coverage.
Split Flaps
Since the respective sural arteries at the least bifurcate soon after entering each head of the gastrocnemius muscle, segmental vertical splitting of either head about a longitudinal branch is possible.22 This allows transfer of a smaller muscle portion if adequate, the creation of multiple flaps for multiple defects, or retention of a segment to allow some function preservation. The most proximal division of these segments requires an awareness of the branching pattern of each head’s source artery, as that could be in the proximal third or not even until reaching the upper half of the muscle. Inadvertent division of a branch puts at risk devascularization of that segment.
Inferior-Based Flaps
Numerous vascular bundles or branches communicate as collaterals across the raphe between the two fused heads of the gastrocnemius muscle.23,24 If only the most inferior branch or two were kept intact, one muscle head could be elevated independent of its dominant pedicle and turned over for more distal reach to the mid-tibia.24,31 That head would then survive via perfusion through these branches originating from the contralateral head. Incorrect estimation of potential flow through these small connecting branches puts at risk ischemia of the transposed muscle head if inaccurate.
Chimeric Flaps
A cutaneous flap based on a musculocutaneous perforator of either head of the gastrocnemius muscle can be simultaneously elevated with the muscle flap.3,32 Perforators of the LHG are often unreliable, so the medial head will more often be used with a medial sural artery perforator (MSAP) flap.32 The separate pedicle to the MSAP flap can be lengthened as far as necessary as long as branches to the muscle component remain. The muscle, since more malleable, can then be used for fill or three-dimensional contouring, followed by independent insetting of the perforator flap to restore skin coverage.
Achilles Tendon Repair
Linton33 has described using the combined attachment of the gastrocnemius muscle heads to the triceps surae with an extension of this aponeurosis for Achilles tendon reconstruction. This required a V-Y advancement of essentially a large island musculocutaneous flap, which might be limited in just how far advancement is possible. Another issue is how much donor Achilles tendon would be available, although the plantaris tendon under the gastrocnemius muscle, since now exposed, could be used as a reinforcing graft.
Extending Flap Reach
Even for the traditional role of providing coverage around the knee, reach of either gastrocnemius muscle head may be difficult. A simple way to extend the length and even width of the muscle flap would be to make incisions into or remove the investing fascia of the muscle.2 Splitting the muscle into smaller segments may extend reach,22 but would be limited by exactly where the circulation enters that segment, which is often uncertain. An inferior-based flap based on communicating branches from the contralateral head may also be precarious.24 Division of the pes anserinus may allow some more minor proximal transposition of the MHG with limited function disturbance.15 Rotating the LHG under the common peroneal nerve may similarly extend reach.1 Probably the most reliable and best way to enhance reach is to take down the origin of each head, with subsequent dissection and release of the vascular pedicle, albeit with some risk of its injury.1,15,34 Several analyses of the arc of rotation of the muscle heads have shown that a 30 to 50% increase in reach can be expected if this was done.15,34
Function Sequela
Sacrifice of either head of the gastrocnemius or even both as a muscle flap has traditionally been considered not to be an issue.2 The muscle(s) is(are) therefore theoretically expendable. Yet Daigeler et al17 in their review of several hundred cases showed some unexpected long-term findings, as confirmed by others.30 Exercise-dependent activities like jumping, standing on tiptoes, and walking upstairs were restricted. Only 42% of their patients had no problems running.17 Maximum plantar flexion force at the ankle joint was 25% weaker than that of the unoperated leg.17 The take-home message is that every muscle has a function, and, therefore, there should always be a consideration to save the muscle if possible.
Peroneal Palsy
If the LHG muscle is the selected flap, an awareness of the location and proximity to the common peroneal nerve is essential during flap harvest. Even only raising the nerve to allow passage of the LHG underneath can result in a transient peroneal palsy. Daigeler et al17 had a 4.5% incidence of peroneal palsy when the LHG was used as a muscle flap, which was sometimes permanent.
14.2.5 Flap Design
The defect requiring reconstruction will dictate the flap design, beginning with which head should be selected. Except in the obese individual, having the patient preoperatively stand on their tiptoes with the ankle joint maximally plantar flexed usually allows visualization of the outline of each head as they insert into the triceps surae. The muscle chosen will usually be the longer and that most likely to reach the defect.
Donor site morbidity is limited if only muscle is taken as the flap, but composite, chimeric, or other variations are possible; an overview on these anatomical variants has already been given in the preceding section. Both muscle heads and their vascular sources are readily approached using a stocking seam incision that is marked to crisscross the popliteal crease before continuing vertically distally along the midportion of the calf up to the triceps surae.34 This is easiest with the patient in the prone position, but most parapatellar and proximal third leg defects are more amenable to having the patient in the supine position, which also simplifies the administration of anesthesia and nursing staff preparation. Depending on the margins of the defect (▶ Fig. 14.5), a curvilinear line starting at the knee joint and continued longitudinally to the level of the triceps surae about 2 cm posterior to the medial posterior border of the tibia will, in this position, most readily allow access to the medial head. The lateral head will require a similar line drawn (▶ Fig. 14.6), but instead 2 cm posterior to the lateral border of the fibula.
14.2.6 Flap Harvest
Proper markings for harvesting the gastrocnemius as a muscle flap will depend on the location of the defect, patient position on the operating table, and which head will be utilized, as described in the preceding section on flap design. In the majority of cases, the patient can be in the supine or lateral decubitus position to allow independent elevation of either head only, which will minimize donor site functional morbidity. Use of a mid-thigh tourniquet with the leg exsanguinated will provide the clearest assessment of anatomical details.
Medial Gastrocnemius
For proximal tibia and medial knee region defects, the medial head since usually longer will have the best reach (▶ Fig. 14.4). The patient, if supine on the table, should have the hip flexed 70 degrees, thigh abducted, and knee flexed where comfortable (▶ Video 14.1). A somewhat curved then longitudinal incision is made 2 to 3 cm below the posteromedial border of the tibia, extending from the medial knee joint to the calf where the muscle fibers inserting into the triceps surae often can be marked preoperatively with the patient standing on their tiptoes. The incision is immediately made through the deep fascia, which is kept with the overlying subcutaneous tissues. Usually, a rim of fat along the medial border of the MHG distinguishes it from the more anterior soleus muscle. A relatively bloodless subfascial dissection, except for a few musculocutaneous perforators that must be ligated or coagulated, extends superficial to the muscle to be ceased near the posterior midline when the paired medial sural cutaneous nerve and accessory branch of the lesser saphenous vein are found. The latter structures are important, as they will continue proximally superficial to the raphe where the medial and lateral heads have fused together.
The most distal extent of the medial head will be distinguished by the oval shape of its muscle fibers that have inserted onto the triceps surae (▶ Fig. 14.4). Preceding proximally along its anterior border, finger dissection in the loose areolar plane beneath it will separate it from the underlying soleus muscle as needed. More careful elevation begins distally by sharp incision of a few centimeters of the cuff of the triceps surae aponeurosis about its muscle fiber insertion (▶ Fig. 14.5), which facilitates traction during handling while being a tough material that will hold sutures during the eventual flap insetting.
With traction on this cuff, the only restraint for more proximal mobilization of the muscle is its decussation with the lateral head along the raphe. Muscle fibers must be sharply divided while being careful to protect the medial sural cutaneous nerve. Once the inferior triangle of the popliteal fossa is reached, this separation is easier as the two heads once again become distinct entities.
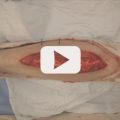
The amount of proximal dissection stops once the defect can be reached without tension. Although transverse incisions in the muscle fascia will provide some additional stretch,2 this cannot be counted on. If the pes anserinus is obstructing flap rotation, it can be divided with marginal functional impairment.15 Pulling it under or repairing the pes anserinus can be detrimental if it compresses the underlying muscle. A definite method to enhance reach is the division of the muscle origin from the popliteal surface of the femur.15,34 The more anterior vascular pedicle may be nearby, so this can be a risky and not-so-easy a maneuver. Begin by severing the most aponeurotic appearing portion seen at the origin, and as proximal as possible, while always being wary of the vascular hilum on the muscle undersurface. Once the origin has been completely divided, this becomes an island flap allowing easier identification and freeing of the vascular pedicle. Pico et al30 suggested dividing the medial sural nerve as well, as the denervated muscle is less likely to dehisce acutely from muscle spasms, atrophies eventually to minimize bulging and results in a better contour deformity, and eliminates pain that otherwise could occur during plantar flexion of the foot. The islanded muscle may reach up to 30 to 50% farther to allow the desired insetting,15,34 best done with the knee joint extended to ensure a tensionless closure regardless of the patient position. Direct closure of the donor site incision should be possible.
Video 14.1 Medial Head Gastrocnemius Muscle Flap. https://www.thieme.de/de/q.htm?p=opn/cs/20/7/12265265-ad3519f8
Video 14.2 Lateral Head Gastrocnemius Muscle Flap. https://www.thieme.de/de/q.htm?p=opn/cs/20/7/12265264-026e8167
Lateral Gastrocnemius
For lateral knee or proximal fibular region defects, the LHG will have better reach (▶ Video 14.2). With the patient in the supine position, or if better the lateral decubitus position with the contralateral side down, the LHG is approached by a longitudinal incision 2 to 3 cm posterior to the fibula, which should extend from the lateral knee joint to its point of insertion onto the triceps surae (▶ Fig. 14.6). Subsequent dissection is almost identical, albeit in mirror image fashion, to that as previously described for the MHG. A major difference that must be appreciated is that near the head and neck of the fibula the common peroneal nerve must always be identified and protected before proceeding with any more proximal LHG dissection. Additional reach may be sufficient by carefully pulling the muscle under the common peroneal nerve without excess traction on the latter before insetting. This approach will also prevent a compression neuropathy if it were to be left superficially. Of course, even more reach is possible if the LHG origin is released from the lateral epicondyle of the femur. If possible, division of the lateral sural nerve will result in a denervated muscle with the advantages as discussed already for the MHG.30 Again, insetting with the knee extended will limit the risk of lower limb hyperextension causing flap dehiscence. Direct closure of the donor site incision should be possible.
14.2.7 Postoperative Care Protocols
Usually a suction drain left to collapse the void created by removal of either head of the gastrocnemius is a reasonable routine. Especially if the origins remain intact, or the muscle has been transposed across the knee joint, a posterior splint with the knee extended is essential until healing appears satisfactory, to prevent inadvertent knee joint flexion or hyperextension causing flap dehiscence. Care should be taken to also keep the ankle joint in a neutral position to prevent a subsequent equinus deformity of the foot.
14.2.8 Conclusion
The gastrocnemius muscle has two heads with distinct neurovascular pedicles that allow either or both to be used as a flap for reliable coverage of the distal thigh, knee, or proximal-third leg regions. Although considered expendable, there can sometimes be a considerable functional deficit left afterward, preventing common exercise activities. Design and harvest of either head as a flap is straightforward, as the muscle has a superficial location in the calf with a fairly reliable anatomy. Use as a musculocutaneous flap has virtually been abandoned primarily because of aesthetic concerns at the donor site and recipient site contour irregularities. Although possible to use as a regional or free flap donor site, the greatest efficacy in using the gastrocnemius muscle remains as a local pedicled flap.
References
[1] Hallock GG, Morris SF. Skin grafts and local flaps. Plast Reconstr Surg. 2011; 127(1):5e–22e
[25] Hallock GG. Anatomic basis of the gastrocnemius perforator-based flap. Ann Plast Surg. 2001; 47(5):517–522
14.3 Chapter 14B: The Gracilis Local Muscle Flap
Geoffrey G. Hallock