Introduction
Antibiotics alone, or in combination with other therapeutic strategies, have been empirically used in hidradenitis suppurativa (HS) with variable success, resulting only in temporary symptom relief in the disease process. Although very few randomized controlled studies have been performed to confirm their efficacy, antibiotics are currently recommended worldwide as first-line therapy by several guidelines issued from scientific dermatological societies.
Rationale for the Use of Antibiotics in Hidradenitis Suppurativa
Role of Bacterial Dysbiosis in the Pathophysiology of Hidradenitis Suppurativa
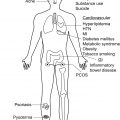
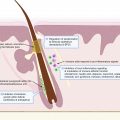
The pathogenesis of HS has not been fully elucidated, but is associated with genetic, immunological, hormonal, environmental, and microbiological factors. The recently unraveled microbiology of HS suggests abnormal host-microbiome cross-talks, bacterial pathogenicity, and dysbiotic variations according to the clinical severity of the disease ( Fig. 16.1 ). The persistence of bacteria within highly inflammatory lesions probably results from both an immune skin defect in HS and bacterial pathogenic properties.
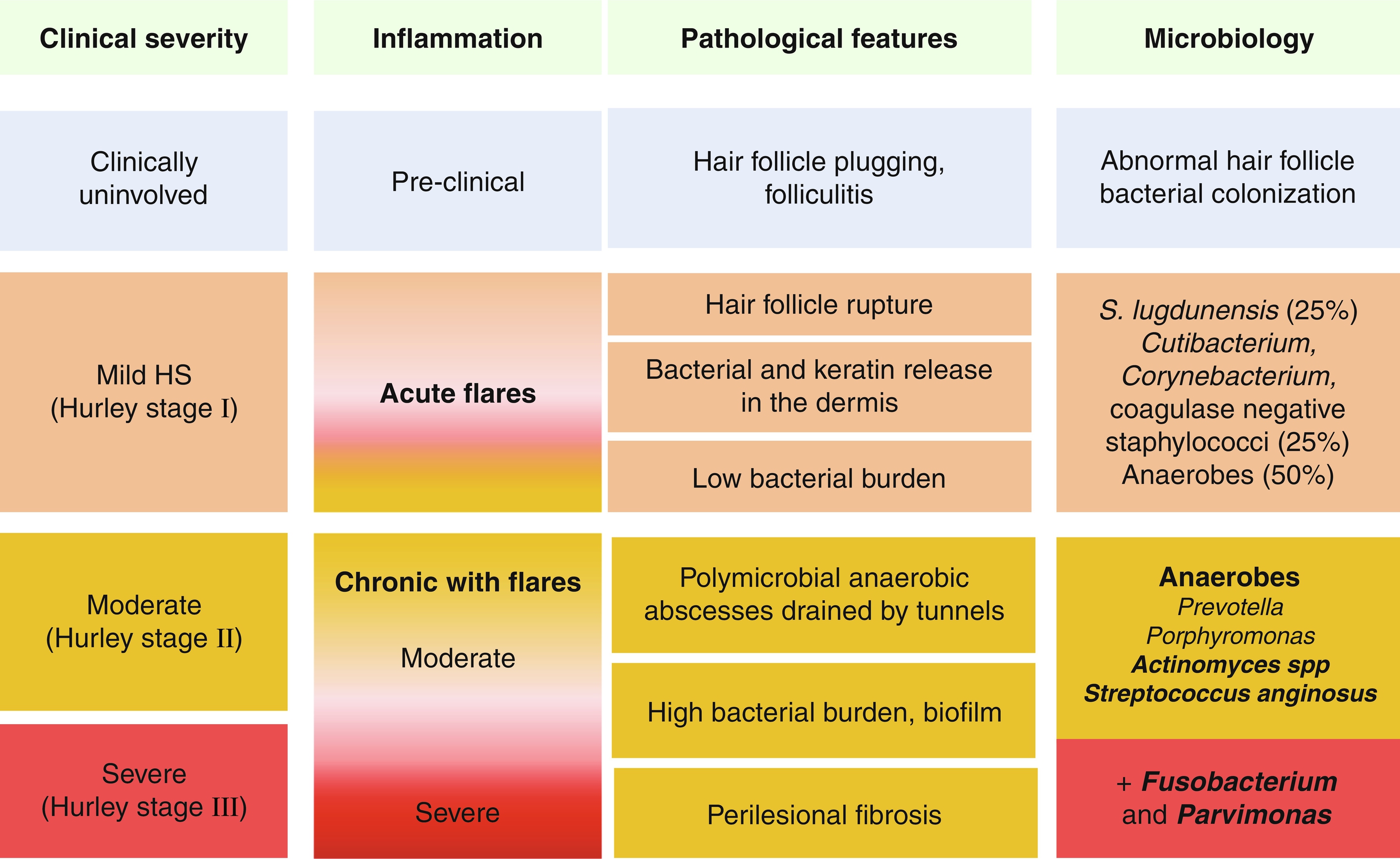
Considering the results of pathological studies, the initial pathologic process results from hair follicle (HF) plugging, which, in case of rupture, leads to the release of keratin debris and bacteria within the dermis. Extensive bacteriological studies of HS lesions have shown that mild HS lesions (Hurley stage I nodules and abscesses) are associated with low virulent skin colonizers such as Cutibacterium (formerly Propionibacterium ) avidum , and coagulase negative staphylococci, including the virulent Staphylococcus lugdunensis and Corynebacterium spp. Anaerobes are associated with 50% of Hurley stage I lesions. Some patients develop more severe and chronic lesions with scarring (Hurley stage II and III). The microbiology of these lesions, as assessed before antibiotic treatments, is constituted of anaerobes, with a predominance of Prevotella and Porphyromonas , two gram-negative anaerobic rods. Parvimonas and Fusobacterium are associated with Hurley stage III lesions.
Chronic HS lesions are associated with bacterial biofilms, which are made of dormant bacteria surrounded by a gelatinous substance. Biofilms are associated with chronic infections in humans, reflecting the inability of the immune system to clear these bacteria. Indeed, biofilms are notably difficult to treat using antibiotics, which are poorly active on dormant bacteria and may not diffuse properly into biofilms. Biofilm presence may explain constant relapses after antibiotic treatments, usually occurring in previous scars.
The role of fungibiomes has not been specifically studied in HS using new generation sequencing technology. In our experience, Candid a spp. can be grown on culture media used for prolonged bacterial cultures of HS suppurative lesions, but always in patients having received prolonged and potent antibiotic course, with clinical signs of candidiasis and rarely in patients naïve from any antibiotic treatment. Moreover, systemic treatment of candidiasis does not improve HS. Therefore, a role for fungal microbiome in HS pathogenesis seems unlikely.
Arguments for a Rational Use of Antibiotics in Hidradenitis Suppurativa
HS is a chronic dermatological disease with a significant negative impact on quality of life due to unmet needs. Continuous or repeated intermittent antibiotic courses are regularly employed in 70% of HS patients by general practitioners as well as specialists, with few validated alternative and satisfactory treatment options.
- •
Systemic nonsteroidal antiinflammatory drugs (NSAIDs) and corticosteroids are typically used as supportive care to decrease symptoms such as pain, but no controlled studies have proved their efficacy and harmlessness.
- •
Antitumor necrosis factor (TNF)-alpha agents are only approved for moderate and severe HS, but not for mild cases. Moreover, anti-TNF-alpha agents such as adalimumab provide significant improvement (reaching HiSCR) in only 50% of the patients and, although rarely, they can cause worrisome/significant side effects.
- •
Surgery, although a cornerstone treatment of HS, cannot always be performed due to the multiplicity, size, and/or localization of lesions, as well as lack of access to surgical experts specialized in HS procedures. Recurrences in the surgical margins can occur in up to 50% of cases.
Although antibiotics may be useful in the management of HS, their long-term risks include Clostridioides difficile infections and the emergence of resistance.
Antibiotics in Hidradenitis Suppurativa: Literature Review
A search through PUBMED, performed from 1950 to 2020, using the MeSH terms “hidradenitis suppurativa,” “acne inversa,” and “antibiotic” resulted in 311 references. We excluded reviews and articles that were not in English. Parameters analyzed in our report included design, primary outcome, number of patients, initial severity, disease severity assessment, treatments, adverse events, follow-up, and limitations.
Single Oral Antibiotics
Tetracyclines
- •
A randomized trial compared the efficacy of oral tetracycline 500 mg bid versus topical clindamycin (1%) in 46 Hurley stage I and II patients, 34 patients being evaluable. Primary outcome was improvement in a composite score consisting of subjective and objective data at month 3. Compared to baseline, there was a 30% to 40% improvement as assessed by the number of nodules or abscesses in both groups with no statistical difference. Considering the low number of patients, the high number of patients lost to follow-up, and the absence of placebo control group, the results of this study (which was performed in the late 1990s) are difficult to interpret.
- •
In a recent retrospective multi-center descriptive study performed in adolescents, Riis et al. reported the efficacy of tetracycline in 16/32 patients with a median age of 16 and an interquartile range of 3, but no details were specified on the disease severity, associated treatments, or outcome measures.
- •
A retrospective study compared the outcomes of 10 patients who were treated with doxycycline 200 mg/d (7 Hurley stage I, 2 Hurley stage II, and 1 Hurley stage III) and of 35 patients treated with the rifampin-clindamycin combination (7 Hurley stage I, 19 Hurley stage II, and 9 Hurley stage III). Hidradenitis suppurativa clinical response (HiSCR) was achieved at month 3 in 60% of patients treated with doxycycline and in 46% of patients treated with the rifampin-clindamycin combination.
- •
Armyra et al. reported a series of 20 HS patients treated with minocycline 100 mg/d combined with colchicine 1mg/d for 6 months, followed by 6 months of colchicine alone. Hurley staging and PGA (Physician Global Assessment) score were used. The series consisted of 1 Hurley stage I, 15 Hurley stage II, and 4 Hurley stage III patients. Primary objective of clinical improvement was obtained in all subjects. No patients achieved complete remission. The part played by colchicine alone and minocycline alone in the management of HS cannot be ascertained from this study.
Trimethoprim-Sulfamethoxazole
Trimethoprim-sulfamethoxazole (TMP-SMX) is commonly used in the long term to prevent Pneumocystis jiroveci pneumonia in HIV and immunosuppressed patients (80/400 mg/d or 160/800 mg/d) and has a good safety profile. In one retrospective and one uncontrolled prospective study totaling 56 patients conducted by the same researchers, TMP-SMX was given as maintenance treatment to prevent flares (80/400 mg/d under 80 kg, 160/800 mg/d above 80 kg) after clinical remission of all lesions. Notably, no new sites affected with HS were observed during the 6-month follow-up in the retrospective study and during the 9-month follow-up in the prospective study. A limited number of relapses occurred only in scars, suggesting bacterial persistence of biofilms, and these were easily controlled by pristinamycin +/– metronidazole prescribed for 3 weeks. These findings suggest that a low maintenance treatment with an appropriate antibiotic such as TMP-SMX may limit relapses and prevent HS progression in new areas.
Dapsone
Dapsone is known for its antimicrobial properties in leprosy and is additionally used as an antiinflammatory agent in several dermatological diseases. Most reports of dapsone use in HS involve a combination of dapsone with different drugs (anti-androgens, anti-TNF agents, cyclosporine, metformin, liraglutide, and finasteride). With these combination treatments, clinical improvement of HS has been reported using various non-validated criteria. A G6PD deficiency should be ruled out before starting dapsone and regular monitoring for anemia and methemoglobinemia is recommended during this treatment.
Combination of Oral Antibiotics
Clindamycin-Rifampin
- •
Mendonça et al. first reported the efficacy of the clindamycin-rifampin (CR) combination (600mg/d each) in a retrospective study of 14 HS patients without any information on clinical severity. Remission (not defined) was achieved in 8 patients “between 1 and 4 years” after a single course of 10 weeks and in 2 other patients after adding minocycline 100 mg/d; 4 patients stopped the combination because of diarrhea.
- •
Gener et al. reported a retrospective series of 116 patients treated with the CR combination (600 mg daily for each antibiotic). Out of 116, only 70 patients were evaluable (51 Hurley stage I, 57 Hurley stage II, and 8 Hurley stage III). Median Sartorius score dropped from 28 to 19, with only 8 patients in complete remission (modified Sartorius 0). Adverse effects were predominantly diarrhea; follow-up data were not available.
- •
Van der Zee et al. reported a retrospective series of 34 HS patients (4 Hurley stage I, 20 Hurley stage II, and 10 Hurley stage III) treated with different dosing and duration of the CR combination. Assessment relied on global evaluation of inflammation by physician, remission being defined as at least 75% of improvement from baseline. A total of 16/34 patients obtained “remission,” 12 patients only had a partial response, and 6 had no improvement, or worsening. Nine patients stopped treatment due to diarrhea. Relapses without maintenance antibiotics occurred within 5 months.
- •
Bettoli et al. reported a prospective series of 23 moderate to severe patients with 20 evaluable patients. No remission was observed, but an improvement was noted in 17/20 subjects, with a median Sartorius score decreasing from 132 to 71.
- •
Dessinioti et al. reported a prospective series of 26 patients (4 Hurley stage I, 16 Hurley stage II, and 6 Hurley stage III) treated with the CR combination (600 mg each daily) for 12 weeks starting with 5 day-intravenous (IV) administration of clindamycin 900 mg/d. Response was defined as an improvement of > 50% in nodules and abscesses count. Clinical improvement was noted in 19/26 patients. During the subsequent one year follow-up in 17 patients, 10 had relapses within a 4-month median without maintenance treatment.
- •
Marasca et al. reported a retrospective series in moderate to severe HS patients with no reported Hurley staging, comparing 30 subjects on CR combination (rifampin being given alone for the first week at 600 mg daily, then adding clindamycin 600 mg daily) with 30 patients on adalimumab for 10 weeks. Clinical outcomes were an improvement in modified Sartorius, HiSCR and HIDRAdisk scores at week 10, in comparison to baseline. Only 10/30 patients achieved HiSCR in the antibiotic combination group, with a decrease in modified Sartorius from 69 to 58. Further follow-up was not reported.
Rifampin is never used alone because of a high risk of emergence of resistant mutants during treatment. The CR combination should theoretically prevent this phenomenon. However, rifampin is a strong inductor of P450 cytochrome, which can result in an increased metabolism of clindamycin. In 10 patients using the oral combination, a dramatic decrease of clindamycin plasma level was observed at day 10 (up to 82% of the peak concentration, undetectable trough levels). Increasing dosage of clindamycin did not help to improve its level. These interactions and clinical findings were subsequently confirmed in another study. Therefore, it is likely that the CR combination is in fact a monotherapy of rifampin after 10 days of use. Even though rifampin may work in mild HS, because of a low bacterial load and the highly bactericidal activity of rifampin, it should ideally be avoided as a single agent in the management of HS.
The above findings led to reconsideration of the long-term utility of the CR combination in HS. Caposiena Caro et al. prospectively compared the efficacy of the CR combination to clindamycin alone in 2 groups of 30 HS (11 Hurley stage I, 30 Hurley stage II, and 19 Hurley stage III) patients. HiSCR at week 8 was achieved in 57% and 63% of patients, respectively. During the 1-year follow-up, 8 and 10 patients of the two groups respectively remained in remission without a maintenance treatment.
In a recent retrospective multicenter Spanish study on 509 HS patients treated with the CR combination, 26.5% stopped the CR combination before week 10, mostly due to gastro-intestinal intolerance. C. difficile infections are rarely reported in HS patients with this combination and prevalence was identical to a large non-HS control group in a recent study.
Clindamycin-Ofloxacin
This alternative was proposed to spare rifampin use in HS. Delaunay et al. reported a retrospective series of 65 HS patients (21 Hurley stage I, 21 Hurley stage II, and 23 Hurley stage III) treated at two different centers using a weight-based regimen of ofloxacin 200 to 400 mg/d and clindamycin 600 to 1800 mg/d for 3 months. Clinical response was assessed after 3 months as complete, partial (reduction without disappearance), or none (stability or worsening). Twenty-two (34%) patients achieved clinical remission and 38 (58%) reported partial improvement.
Rifampin, Moxifloxacin and Metronidazole
Join-Lambert et al. reported a retrospective series of 28 HS patients (6 Hurley stage I, 10 Hurley stage II, and 12 Hurley stage III) treated with the combination of rifampin (10 mg/kg/d), moxifloxacin (400 mg/d), and metronidazole (500 mg tid) for 6 weeks, followed by 1 to 3 months with only rifampin and moxifloxacin. Remission, defined by absence of any inflammation in scars, was observed in 6/6 Hurley stage I and 8/10 Hurley stage II subjects within 2 to 4 months. Of note, severe patients received a 2-week induction of IV ceftriaxone and oral metronidazole, and after clinical remission, patients were given TMP-SMX for maintenance treatment. Adverse events included gastro-intestinal side-effects, candidiasis, reversible tendonitis, and increase of liver enzymes. During the follow-up under TMP-SMX maintenance, seven patients had no relapse within 6 months and seven experienced relapses occurring only in previously involved areas (scars containing biofilms). The same team reported a prospective study of 28 severe Hurley stage I HS patients using this treatment strategy. The median-modified Sartorius score dropped from 14 to 0 at week 12, with 75% being in remission. The median number of flares dropped from 21/year to 1/year during the 12-month follow-up with TMP-SMX maintenance treatment.
Intravenous Beta-Lactam Antibiotics
The efficacy of once-daily intravenous (IV) broad-spectrum beta-lactams has been reported in both a 3 to 6 week induction or when used as a rescue treatment strategy in moderate and severe HS.
Ceftriaxone in Combination with Oral Metronidazole
In the retrospective study of Join-Lambert et al. (see above) on the moxifloxacin-rifampin-metronidazole combination for moderate (Hurley stage II) and severe (Hurley stage III) HS, patients received a 2-week induction course of IV ceftriaxone 1g daily associated with metronidazole (500 mg 3 times daily orally). The efficacy of the induction treatment was not specifically reported in this study, the primary endpoint being the percentage of clinical remissions at week 12. However, all patients were clinically improved with this partial induction regimen at the 6-week visit as assessed by the size and number of active lesions (authors’ personal data).
In a prospective study, Delage et al. reported a small series of 17 Hurley II HS patients treated with a 3-week IV induction of ceftriaxone 1g daily associated with metronidazole (500 mg 3 times daily orally), followed by 3 weeks of the combination of rifampin (10 mg/kg/d), moxifloxacin, (400 mg/d), and metronidazole (500 mg tid) for 3 weeks, then by 6 weeks of only rifampin and moxifloxacin, then cotrimoxazole for maintenance. The primary endpoint was remission at week 12, which was obtained in 11 patients (65%) and subsequently in 3 other patients during the 1-year follow-up. Many mild side effects were reported.
Ertapenem
Join-Lambert et al. reported a retrospective series of 30 severe (17 refractory Hurley stage II and 13 Hurley stage III) HS patients treated with an IV ertapenem induction (1g daily) through a PICC-line for 6 weeks, followed by the combination of oral rifampin, moxifloxacin, and metronidazole. Median-modified Sartorius improved from 49 to 19 and median pain level dropped from 6/10 to 0 at week 6. At the 6-month follow-up, 59% (16) of the patients who underwent oral consolidation after ertapenem course were in remission. In patients who stopped treatment after ertapenem, Sartorius score stopped improving or returned to baseline.
Braunberger et al. reported a retrospective series of 36 severe and refractory HS patients treated with ertapenem (28 evaluable) along with anti-TNF-alpha agents biotherapy in 4 patients and systemic corticosteroids in 5 patients. Seventy-one percent were very satisfied or satisfied, with a quality of life notably improved in 86%. Although based on subjective criteria, such a level of satisfaction from patients with any treatment within 6 weeks has rarely been reported so far in HS.
Chahine et al. reported a case of severe and refractory HS with a high suspicion of septicemia, which was dramatically improved with ertapenem.
Based on unpublished and personal communications (Hamzavi et al. and Nassif et al.), IV ertapenem as a bridge to surgery for 2 to 3 weeks prior has been successfully used to control severe/refractory Hurley stage III HS. Also, in some patients, IV ertapenem has been used to control symptoms until patients are able to use biologic therapy such as anti-TNF-alpha agents.
Antibiotic Resistance in Hidradenitis Suppurativa
Mechanisms of Acquired Resistance in Bacteria
Mutation of the antimicrobial target gene is the most rapid mechanism of acquired resistance. Risk of resistance among different antibiotic families is variable and compounded by the bacterial bioburden within HS lesions. Rifampin, when used alone, is particularly prone to select resistant mutants.
High-level resistance against clindamycin and tetracycline are usually mediated by chromosomal insertion of mobile genetic elements carrying resistance genes such as erm and tet genes, respectively. Transfer of mobile genetic elements is favored by low antibiotic concentrations and prolonged treatments.
Depending on the bacterial species, beta-lactam resistance usually occurs by target mutation, resistance gene acquisition (beta-lactamases, target variants), or impermeability due to porin loss or efflux. Compared to fluoroquinolones and rifampin, the risk of mutant selection is much lower using betalactam antibiotics. However, it can occur with repeated antibiotic treatments.
Impact of Low Antibiotic Dosages on the Emergence of Antimicrobial Resistance
In dermatology, antibiotics are often given as “immunomodulating” treatments for prolonged periods and in low dosages to reduce adverse events. However, bacterial resistance emerges quickly with low antibiotic concentrations.
Antimicrobial Resistance in Hidradenitis Suppurativa
Bacteria associated with HS are intrinsically susceptible to a wide range of antibiotics ( Table 16.1 ). We have little data on antimicrobial resistance in HS. However, recent reports indicate an increase in antibiotic resistance, particularly to clindamycin and, to a lesser extent, rifampin. In addition, clindamycin resistance in anaerobes has emerged in the community (10% to 30% depending on the species). This may account for antibiotic failures in HS.
| Antibiotics | Aerobes | Anaerobes | ||||||
|---|---|---|---|---|---|---|---|---|
| Family | Name | CoNS | SLU | Cor | Str | Cocci | G + rods | G- rods |
| Betalactam antibiotics | Amoxicillin | V | V | S | S | S | S | S |
| Amoxicillin + clavulanic acid | V | S | S | S | S | S | S | |
| (IV) Piperacillin + tazobactam | V | S | S | S | S | S | S | |
| (IV) Ceftriaxone | V | S | S | S | S | S | V | |
| (IV) Ertapenem | V | S | S | S | S | S | S | |
| Macrolides | Clindamycin | V | V | V | V | V | V | V |
| Pristinamycin a | S | S | S | S | S | S | S | |
| Tetracyclines | Doxycycline | V | S | S | S | S | S | S |
| Minocycline | S | S | S | S | S | S | S | |
| Fluoroquinolones | Ofloxacin | V | S | V | S | V | V | V |
| Levofloxacin b | V | S | V | S | V | V | V | |
| Moxifloxacin b | V | S | S | S | S | V | S | |
| Other | Trimethoprim-sulfamethoxazole | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| Rifampin | S | S | S | S | S | S | S | |
| Metronidazole | R | R | R | R | S | V | S | |
| Linezolid | S | S | S | S | S | S | V | |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree