Chest surgery
Chest-wall contouring is an important, early surgical step for transmen and may help to facilitate their transition. The goals of chest surgery include the aesthetic contouring of the chest by removal of breast tissue and excess skin, reduction and repositioning of the nipple-areola complex when necessary, release of the inframammary crease, liposuction of the chest, and, when possible, minimization of chest scars and preservation of nipple sensitivity.
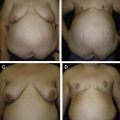
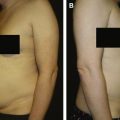
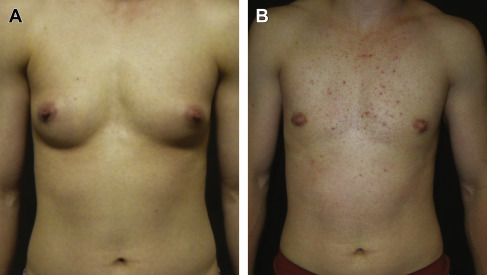
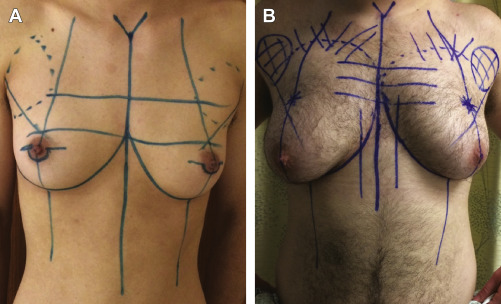
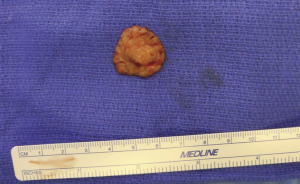
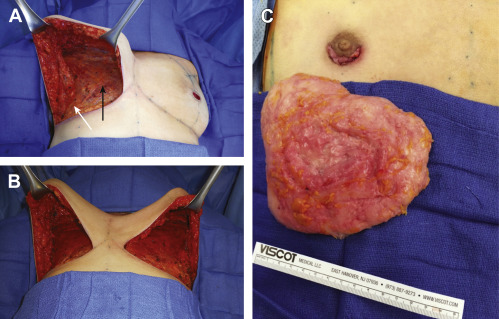
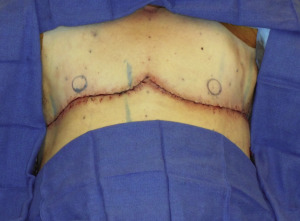
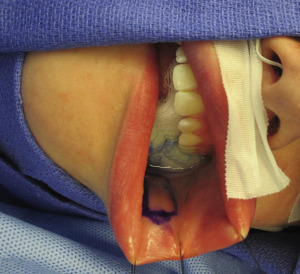
Chest surgery in transmen presents an aesthetic challenge because of breast volume, breast ptosis, nipple-areola size and position, degree of skin excess, and potential loss of skin elasticity. Breast binding, commonly performed by transmen, may lead to the loss of skin elasticity, thereby necessitating significant amounts of skin removal ( Fig. 6.1 ). Several surgical methods are used, and the choice of technique depends on the skin quality and elasticity, the degree of breast ptosis, and the position of the nipple-areola complex. In addition, preservation of subcutaneous fat on the mastectomy skin flaps, preservation of the pectoralis and serratus fascia, release of the inframmary crease and sternal attachments, and contouring of the lateral chest wall are also important components of chest surgery and chest wall contouring. Preoperative breast imaging (ie, ultrasound or mammogram) is performed based on patient age, personal and family history, and physical examination.
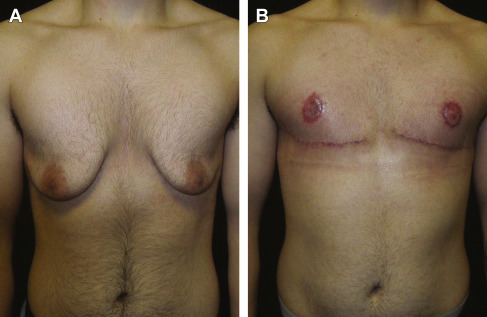
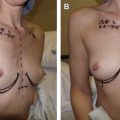
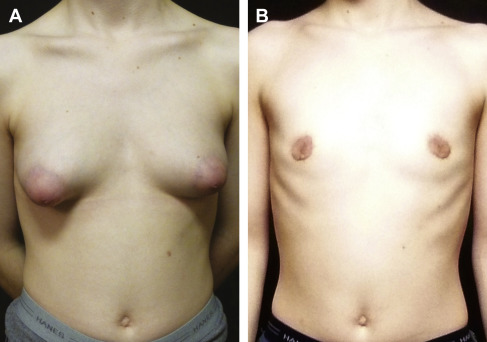
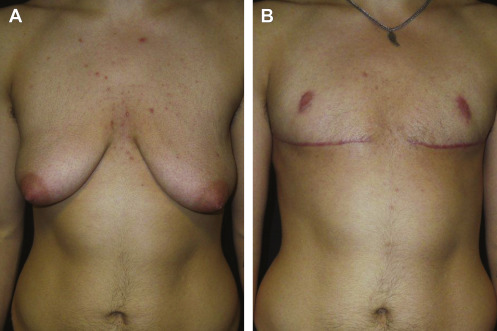
Small, nonptotic breasts can often be treated with periareolar incisions (“limited incision”). In these cases, the nipple may be reduced by a wedge resection of the lower pole, but the areola is not repositioned. A small amount of tissue is left beneath the nipple-areola to preserve viability ( Fig. 6.2 ). Circumareolar (“purse-string”) or vertical incisions with free nipple-areola grafts can be used for larger breasts with mild ptosis requiring smaller amounts of skin removal ( Fig. 6.3 ). Finally, traditional transverse inframammary crease incisions (“double incision”) with free nipple-areola grafts may better serve individuals with larger breast volumes and increased breast ptosis requiring large amounts of skin removal ( Fig. 6.4 ). Nipple reduction is commonly used, often in conjunction with free nipple-areola grafts ( Fig. 6.5 ). In addition, liposuction is frequently used for discontiguous undermining of the inframammary crease and lateral chest wall. Generally, free nipple-areola grafts, as opposed to the maintenance of the nipple-areola complex on a dermoglandular pedicle, are the preferred technique for nipple transposition so as to prevent residual breast fullness.
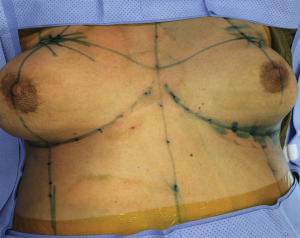
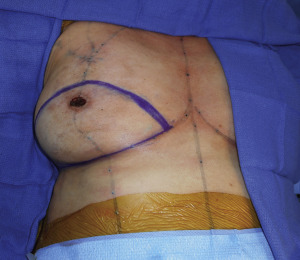
As with genital surgery, hormone therapy is discontinued 2 weeks before surgery. The patient is marked in the upright position. The relevant reference points include the inframammary crease, the midbreast meridian, the lateral border of the pectoralis major muscle, the axillary tail of the breast, midline, and the anticipated position of the nipple-areola complex ( Fig. 6.6 ).
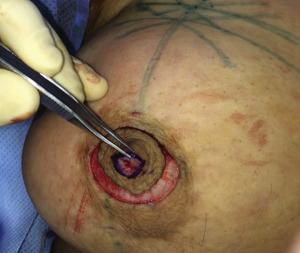
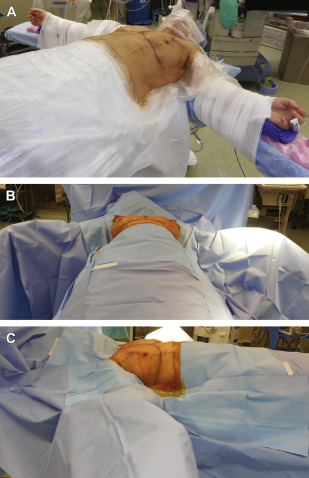
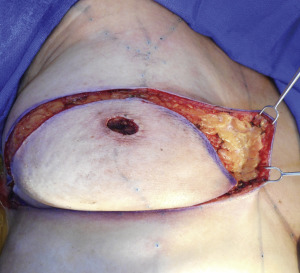
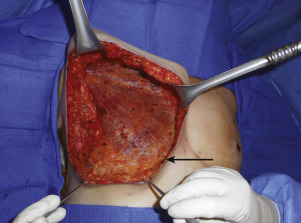
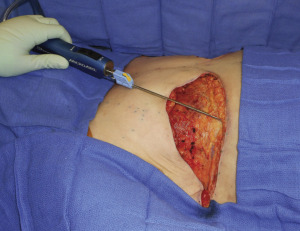
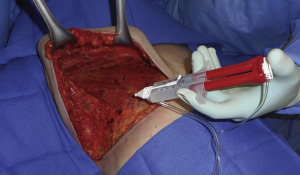
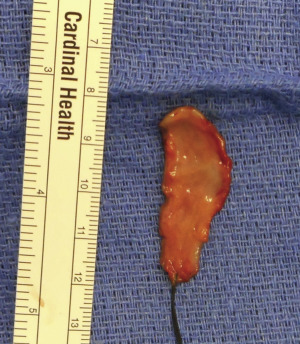
Before surgery, sequential compression devices are placed and intravenous antibiotics are administered. Following induction of general anesthesia, chemoprophylaxis for venous thromboembolism is administered subcutaneously (either fractionated or unfractionated heparin depending on institutional policies). With each of the operative techniques, the patient is positioned supine, with the arms abducted on foam rests with flexion at the elbow, and a lower-body forced-air warming blanket is placed. Draping should allow access to the surgical field from both above and below the upper extremity ( Fig. 6.7 ). In addition, the patient is positioned and secured in anticipation of raising the back of the operating table in order to assess symmetry and guide placement of the nipple-areola grafts. The reference points are tattooed with methylene blue to aid with intraoperative identification ( Fig. 6.8 ). If free nipple-areola grafts are to be performed, the nipple-areola complex is resized to a diameter of 2.5 to 3.0 cm. In addition, if a nipple reduction is required, a wedge resection of the lower pole of the nipple is performed ( Fig. 6.9 ). The nipple-areola grafts are harvested, defatted, and placed in moist gauze sponges for later use ( Fig. 6.10 ). When a skin resection is planned, regardless of the technique, the anticipated area of skin resection is incised in order to facilitate breast removal ( Fig. 6.11 ). The skin flaps are developed at the junction between the breast tissue and the subcutaneous fat ( Fig. 6.12 ). The inframammary crease is undermined approximately 1 to 2 cm ( Fig. 6.13 ). The medial dissection of the breast stops at the sternal border, and the perforating branches of the internal mammary artery are preserved, if possible. The lateral dissection proceeds subcutaneously over the serratus muscle, with preservation of the serratus fascia, and the breast is reflected off of the chest wall, with preservation of the pectoralis fascia ( Fig. 6.14 ). Liposuction of the inframammary crease, the lateral chest, and the axillary tail is performed ( Fig. 6.15 ). Fibrin sealant is placed in the cavity ( Fig. 6.16 ); the patient is sat upright, and the skin flaps are tailored and inset ( Fig. 6.17 ) in a layered fashion over a 15-French closed suction drain. Following resection, the breast tissue is marked and sent to pathology for routine examination.
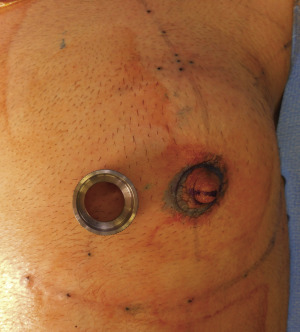
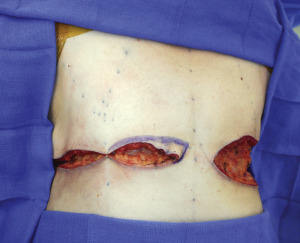
In positioning the nipple-areolar complex, clinical judgment is used. In general, the nipple-areola is positioned medial to the lateral border of the pectoralis major muscle, approximately 1 to 2 cm above the inferior insertion of the pectoralis major muscle ( Fig. 6.18 ). The nipple-areola is inset on a dermal bed, and a bolster dressing is placed ( Fig. 6.19 ).
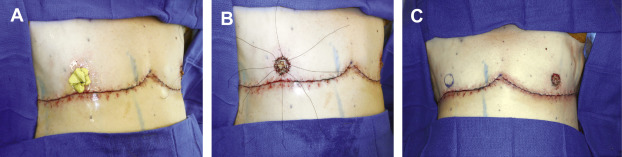
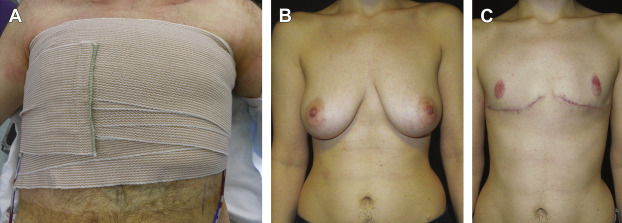
An elastic compression wrap remains in place for 4 to 5 days ( Fig. 6.20 A). At the time of the initial dressing change on postoperative day 4 or 5, the bolster is removed, and local care with a topical antimicrobial ointment is initiated. The compression wrap is continued for 3 weeks after surgery, followed by a compression shirt for an additional 3 weeks ( Fig. 6.20 B, C).
Secondary revisions related to the mastectomy scar and/or the nipple-areolar complex are not uncommon. Additional complications include, but are not limited to, hematoma, seroma, infection, delayed healing, loss of nipple grafts, asymmetry, and underresection and overresection of tissue leading to chest wall contour abnormalities.
Genital surgery for transmen
The goal of genital surgery in transmen may range from clitoral release (metoidioplasty), with or without urethral lengthening, to creation of a penis, capable of sexual penetration. Similar to genital surgery for transwomen, transmen undergoing genital surgery discontinue their hormone therapy 2 weeks before surgery and complete a bowel preparation the day before surgery. Before surgery, sequential compression devices are placed and intravenous antibiotics are administered. Following induction of general anesthesia, chemoprophylaxis for venous thromboembolism is administered subcutaneously (either fractionated or unfractionated heparin depending on institutional policies); the patient is positioned in the lithotomy position, and bony prominences are padded. For metoidioplasty procedures, the arms are abducted on foam rests with flexion at the elbows; an upper body forced-air surgical warming blanket is placed, and an indwelling urinary catheter is inserted under sterile conditions after the patient is prepared and draped. For phalloplasty procedures, a 325-mg aspirin suppository is administered, and the upper extremity chosen as the donor site is placed on a hand table, with a tourniquet applied to the upper arm.
Most transmen undergoing genital surgery will have undergone a hysterectomy and oophorectomy at least 3 months before either metoidioplasty or phalloplasty. For hysterectomy and oophorectomy procedures, either a laparoscopic or a vaginal approach is preferred. However, access for the vaginal route may be limited in nulliparous patients on testosterone therapy.
Metoidioplasty
Metoidioplasty, described in 1996 by Hage, is an alternative to microsurgical or pedicled-flap phalloplasty in transmen. The term, coined by Laub, is based on the Greek prefix meta -, relating to change, and aidoio , relating to the genitals. The procedure entails lengthening the hormonally hypertrophied ( Fig. 6.21 ) clitoris by release of the suspensory ligament and resection of the ventral chordee. In order to allow for urination while standing, the natal female urethra is lengthened with the use of labia minora, vaginal musculomucosal flaps, and buccal mucosal grafts. Although metoidioplasty may permit urination while standing, it does not allow placement of an erectile device, and it is unlikely that individuals undergoing metoidioplasty will be able to engage in penetrative intercourse. In addition, scrotal reconstruction following metoidioplasty positions the scrotum in a lateral, rather than an anterior, position. As such, patients may describe their testicles as being positioned “between their legs” rather than “in front of their legs.” The metoidioplasty procedure is less complex compared with phalloplasty, avoids the disadvantage of additional scarring at remote donor sites, and may be converted to a phalloplasty at a later date, if desired. For some transmen, these considerations are sufficient to warrant the choice of metoidioplasty over phalloplasty.
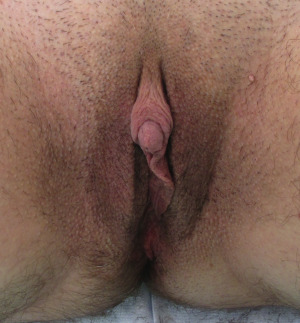
The operative technique involves removal of the female genitalia with vaginectomy and colpocleisis (see vaginectomy/colpocleisis in the later phalloplasty section) concomitant with clitoral release, urethral lengthening, and scrotoplasty. A distally based, anterior vaginal wall flap may be used in conjunction with a vestibular lining flap in order to reconstruct the proximal neourethra. On the ventral aspect of the clitoris, the urethral plate is divided and dissected from the clitoral bodies. The division of the chordee allows for the release of the ventral clitoral curvature, permitting straightening and lengthening of the clitoris. A suprapubic tube is placed for temporary urinary diversion.
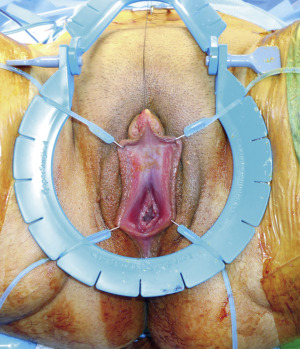
The patient is positioned in the lithotomy position, and a lone star retractor is used for exposure of the urethra, clitoris, labia minora, and vagina ( Fig. 6.22 ). An access incision, designed as a rectangular vestibular lining flap, is created between the native urethra and the virilized clitoris ( Fig. 6.23 ). The vestibular lining flap is formed by 2 parallel incisions beginning at the native urethral meatus and extending along the vestibular lining toward the clitoris. Approximately one-third of the distance between the urethra and clitoris, a transverse incision communicates the 2 parallel incisions. The 2 parallel incisions are then extended to the clitoris and represent the medial incisions for the subsequent left and right labia minora flaps ( Fig. 6.24 ).
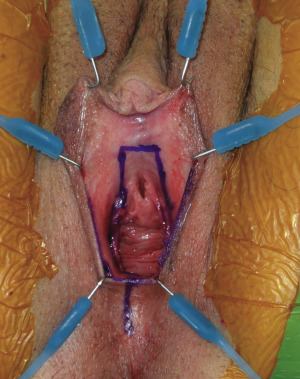
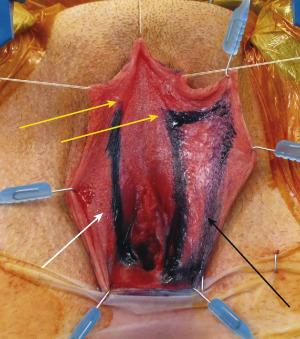
The parallel incisions for the vestibular lining flap continue caudally, from the native urethral meatus, and extend intravaginally. These incisions, located along the anterior vaginal wall, are contiguous with the vaginal wall flap ( Fig. 6.25 ). The vaginal flap is elevated as a musculomucosal flap, with its base at the urethral meatus ( Fig. 6.26 ). These 2 flaps, the vestibular lining flap and the anterior vaginal wall flap, will be sutured to each other, forming the proximal urethra ( Fig. 6.27 ).
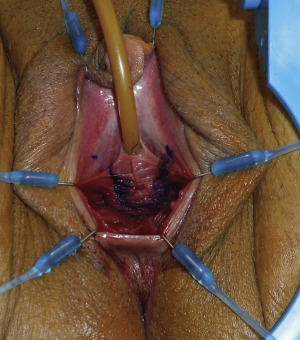
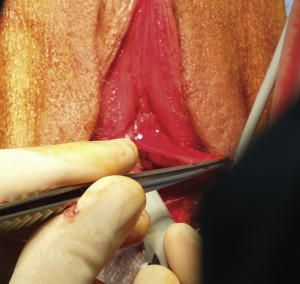
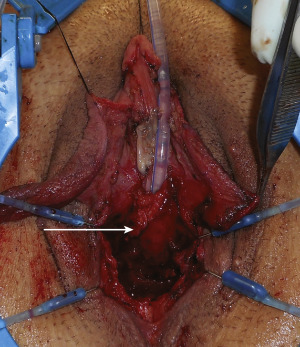
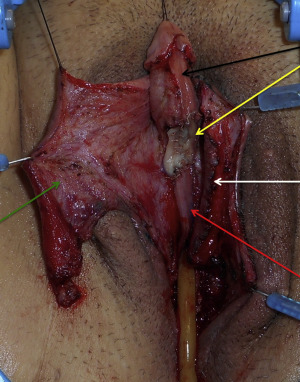
The lateral and dorsal aspects of the corporal bodies are exposed through the aforementioned vestibular incisions and an incision located at the junction of the glans clitoris and the clitoral hood ( Fig. 6.28 ). The plane of dissection follows the fascial layer covering the corporal bodies so as not to injure the vascular pedicle to the labia minora flaps. The skin is elevated to the pubic symphysis, and the suspensory ligament is identified. Division of the suspensory ligament remains at the discretion of the surgeon and depends on the length of the clitoris. The suspensory ligament may be left intact if there is adequate clitoral length, because division of this ligament may destabilize the clitoris. During this dissection, care is taken to preserve the clitoral neurovascular bundles located on the dorsum of the corporal bodies at approximately the 11 and 1 o’clock positions ( Fig. 6.29 ).
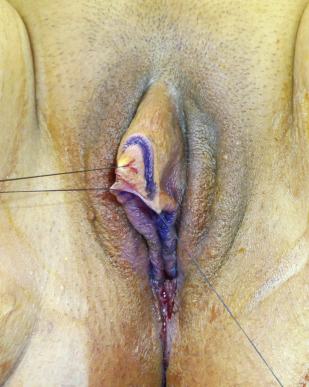
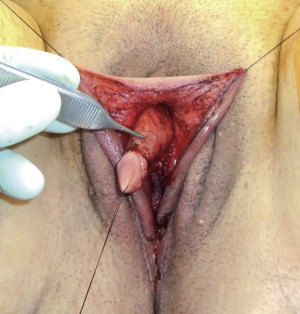
Once the corporal bodies are anatomically defined, the ventral chordee is divided. Division of the chordee from the underlying corporal bodies allows straightening and advancement of the clitoris ( Fig. 6.30 ).
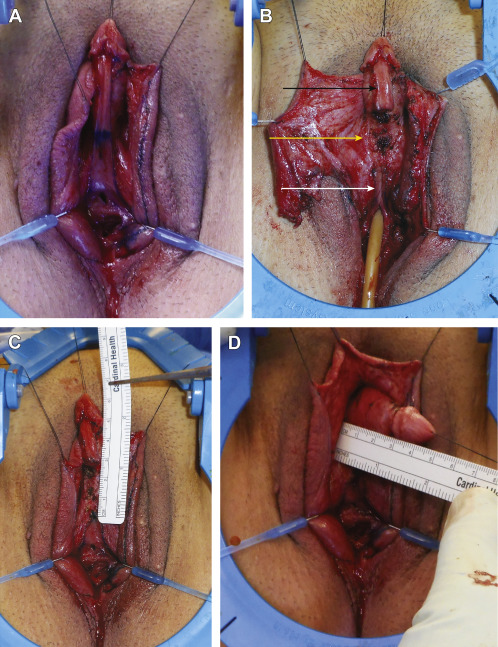
The remainder of the urethral reconstruction is performed with bilateral labia minora flaps. The first flap is developed from the inner aspect of one side of the labia minora (left), and a second flap is developed from the contralateral labia minora (right) ( Fig. 6.31 ). The 2 transposition flaps will overlap, creating a layered closure around the urethra. The left labia minora flap is outlined as a rectangle on the inner surface of the left labia minora, with the flap’s medial border delineated by the previous vestibular incision. The outer, cutaneous, surface of the left labial flap is de-epithelialized. The contralateral right labial flap is elevated to allow subsequent mobilization over its left-sided counterpart. The cutaneous surface of the right-sided flap will be left intact, because this forms the ventral skin of the penile shaft ( Fig. 6.32 ).
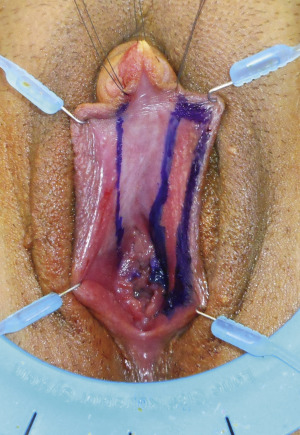
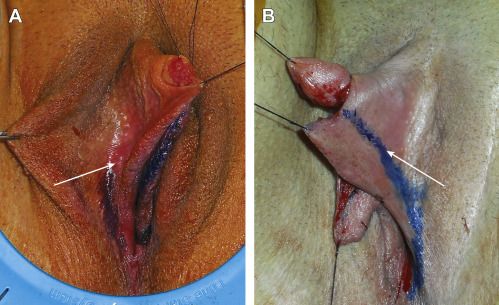
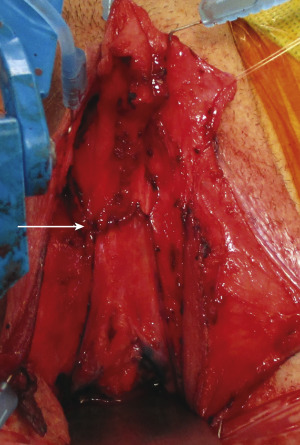
Construction of the urethra commences with anchoring of the distal margin of the vestibular lining flap to the corpora cavernosa ( Fig. 6.33 ). A buccal mucosa graft is harvested, defatted, and sutured to the ventral corporal bodies with multiple quilting sutures ( Figs. 6.34 and 6.35 ). The dimensions of the buccal graft correspond to the size of the defect created by the resection of the ventral chordee and typically measure approximately 1.5 cm × 6 cm ( Fig. 6.36 ). Creation of the proximal urethra is completed by suturing the vaginal wall flap to the vestibular lining flap ( Fig. 6.37 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree