Spongiotic, Psoriasiform, and Lichenoid Dermatoses
Laura E. K. Gifford
Emily J. Osier
Charlene W. Oldfield
Judith V. Williams
Alejandro A. Gru
ATOPIC DERMATITIS
Definition and Epidemiology
Atopic dermatitis is a chronic, relapsing inflammatory skin condition defined by pruritus.1 The most widely used diagnostic criteria specify that a patient must have pruritus with an eczematous dermatitis of typical morphology and distribution, with a chronic and relapsing course. Additional diagnostic features include xerosis and additional signs of atopy such as asthma, allergic rhinitis, and food allergies.2
Atopic dermatitis affects between 10% and 27% of all children.3,4 Initial presentation is in the first year of life in approximately 60% of patients,3 with the vast majority, approximately 90%, presenting prior to 5 years of age.1 Although many of these patients will outgrow their atopic dermatitis by adulthood, it persists in up to 30% into adulthood. A small percentage of affected adults initially develop atopic dermatitis during adulthood.1 Persistent atopic dermatitis has been associated with comorbid atopic conditions and with a family history of atopy.4
Etiology
The etiology of atopic dermatitis is complex and is mediated by a combination of genetic and environmental factors. An estimated 70% of patients have a family history of atopy (atopic dermatitis, asthma, allergic rhinitis).1 A recent genome-wide association study identified 136 genetic variants associated with atopic disease, including genes involved in epidermal barrier formation, immune dysregulation, and inflammation.5,6 The Th2 cytokines, IL-4, IL-5 IL-13, IL-31, and CCL18, are often implicated, whereas some variants are associated with Th 1 cytokines IL-17, IL-22, and IL-23.7
Among the most well-characterized genetic risk factors for atopic dermatitis is filaggrin mutation. Loss-of-function mutations in this gene are seen in 10% to 50% of patients with atopic dermatitis.5 Affected patients have decreased epidermal production of the profilaggrin protein, an important contributor to the hydration and barrier functions of the epidermis. Other proteins and lipids contributing to the hydration and barrier of the epidermis include loricrin, involucrin, ceramides, and cholesterols.5 The resultant defective epidermal barrier increases water loss, allergen sensitization, and bacterial colonization with Staphylococcus aureus.8 All of these factors, in combination with immune system variations, result in the development of atopic dermatitis.8
Up to 80% of patients with atopic dermatitis are colonized with S. aureus, compared with only 10% of nonatopic children.9 The production of exotoxins by S. aureus influences the adaptive immune system, specifically increasing IL-22 and IL-31.5 Increased IL-4, IL-13, and IL-22 levels potentiate colonization with S. aureus.5 A decrease in the antimicrobial peptides, cathelicidin, and human beta-defensin-3, part of the innate immune system, further contributes to increased rates of S. aureus colonization.10
Clinical Presentation
The diagnosis of atopic dermatitis is based, in part, on a typical distribution. For newborns and infants, this includes the face, neck, and extensor extremities, and in older
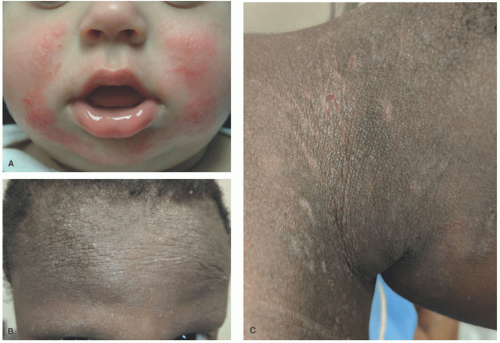
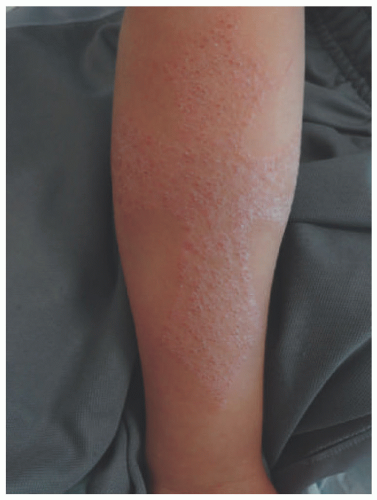
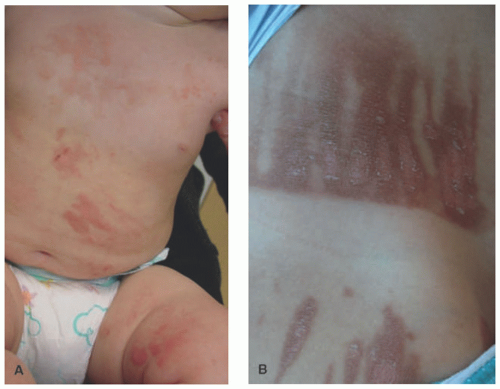
children and adults, the antecubital and popliteal fossae are commonly involved. The groin and axillae are typically spared.2 Morphologically, the acute phase can present with vesicles and weeping plaques. Over time, the plaques become more lichenified and scaly (Figure 3-1).1 Pruritus often leads to excoriations, erosions, and crusting. Variants include a papular variant resembling a lichenoid eruption and a follicular variant.11 Associated clinical findings include cheilitis, nipple dermatitis, keratosis pilaris, hyperlinear palms, ichthyosis, follicular accentuation, facial pallor, and white dermatographism.2 Asthma, allergic rhinosinusitis, and/or food allergies affect 70% to 80% of patients with atopic dermatitis.12 This “atopic march” starts with atopic dermatitis, because the impaired epidermal barrier allows exposure and sensitization to both airborne and foodborne allergens.12
children and adults, the antecubital and popliteal fossae are commonly involved. The groin and axillae are typically spared.2 Morphologically, the acute phase can present with vesicles and weeping plaques. Over time, the plaques become more lichenified and scaly (Figure 3-1).1 Pruritus often leads to excoriations, erosions, and crusting. Variants include a papular variant resembling a lichenoid eruption and a follicular variant.11 Associated clinical findings include cheilitis, nipple dermatitis, keratosis pilaris, hyperlinear palms, ichthyosis, follicular accentuation, facial pallor, and white dermatographism.2 Asthma, allergic rhinosinusitis, and/or food allergies affect 70% to 80% of patients with atopic dermatitis.12 This “atopic march” starts with atopic dermatitis, because the impaired epidermal barrier allows exposure and sensitization to both airborne and foodborne allergens.12
Histologic Findings
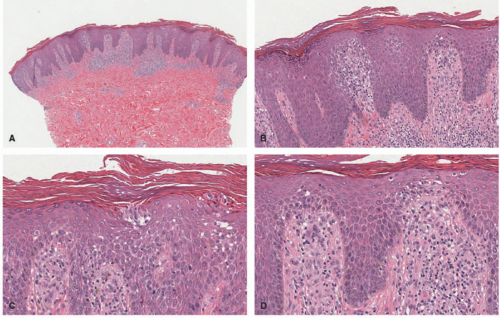
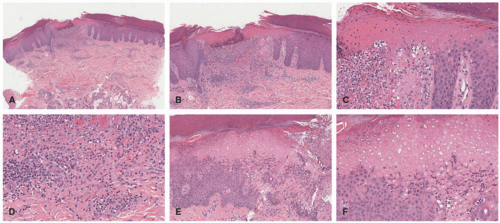
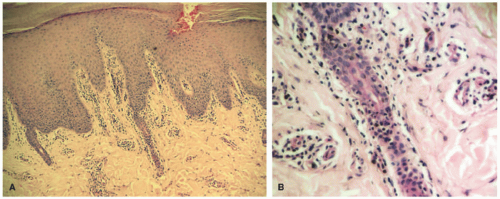
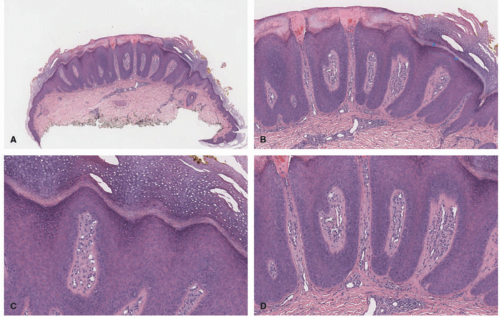
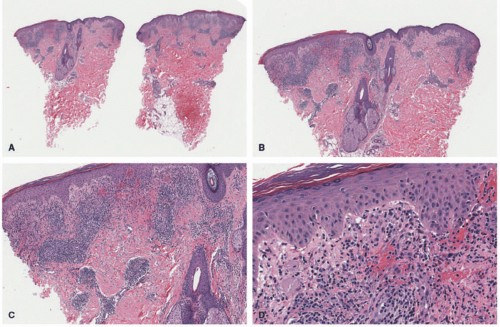
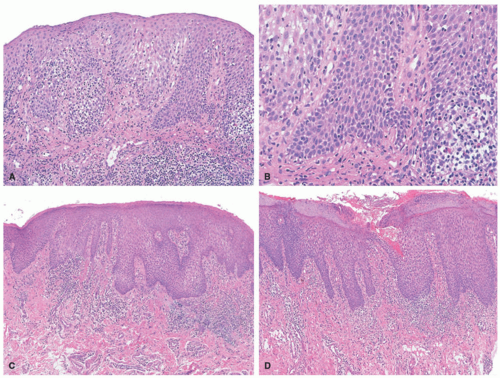
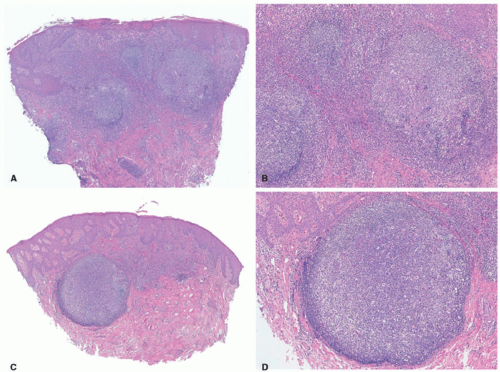
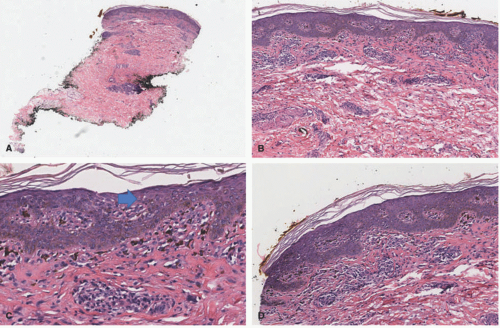
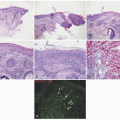
In the acute phase, the changes are usually subtle. Focal areas of parakeratosis overlie an epidermis with a mild degree of spongiosis. Scattered intraepidermal collections of Langerhans cells can be present. Within the dermis, there is a superficial and perivascular lymphocytic inflammatory infiltrate with variable numbers of scattered eosinophils (Figure 3-2). Follicular spongiosis can sometimes be seen, a finding that has been encountered more frequently in dark-skinned individuals.13,14,15
In the chronic phase, the findings are typically nonspecific and correspond to those of a “chronic spongiotic dermatitis.” There is epidermal hyperplasia with psoriasiform changes and larger areas of hyperkeratosis with parakeratosis (Figure 3-3). Lichen simplex chronicus (LSC)-like changes can be seen, with thick orthokeratotic hyperkeratosis, a thickened granular cell layer, and sometimes a zone of stratum lucidum at the base of the cornified layer. Dermal eosinophils can sometimes be seen, and a slight increased number of mast cells are found.
Differential Diagnosis
The histologic differential diagnosis for the spongiotic pattern is broad and requires adequate clinicopathologic correlation. Id reaction, allergic contact dermatitis (ACD), spongiotic drug eruptions, and nummular dermatitis may present with identical histologic features and should be included as histologic possibilities. Seborrheic dermatitis (SD) is also frequently encountered in children and can also be a mimicker. The location of the lesions, the presence of plasma cells, and perifollicular parakeratosis can be used to separate those apart.13,14,15 Other conditions featuring spongiosis include pityriasis rosea and dermatophyte
infection. Pityriasis rosea (PR) features mild spongiosis and mounds of parakeratosis, and dermatophyte infection may feature clusters of intraepidermal or intracorneal neutrophils. Routine performance of periodic acid-Schiff (PAS) or Grocott-Gomori’s methenamine silver (GMS) staining in all biopsies of spongiotic dermatitis is common practice in some dermatopathology laboratories.
infection. Pityriasis rosea (PR) features mild spongiosis and mounds of parakeratosis, and dermatophyte infection may feature clusters of intraepidermal or intracorneal neutrophils. Routine performance of periodic acid-Schiff (PAS) or Grocott-Gomori’s methenamine silver (GMS) staining in all biopsies of spongiotic dermatitis is common practice in some dermatopathology laboratories.
CAPSULE SUMMARY
ATOPIC DERMATITIS
Atopic dermatitis is a very common chronic, relapsing inflammatory skin condition characterized by pruritus, eczematous dermatitis, and a typical distribution. Affected children often outgrow atopic dermatitis, but there is a large percentage of patients in whom it persists in, or develops during, adulthood. The histologic findings are those of a spongiotic dermatitis, and clinical correlation is required to differentiate these findings from other primary spongiotic disorders such as Id reactions, ACD, and nummular dermatitis.
IRRITANT CONTACT DERMATITIS
Definition and Epidemiology
Irritant contact dermatitis (ICD) is an eczematous response to substances that are inherently irritating to the skin.16 It does not require sensitization and may occur in anyone.16
Etiology
ICD is an “outside-in” dermatitis resulting from cytotoxic effects of irritants on the skin.19 Contributing factors include the biology of the underlying skin, the location on the body, the strength of the irritant substance, and the duration of exposure. The homeostasis of the stratum corneum affects permeability and susceptibility to irritants.20 Regional risk factors affecting this homeostasis include occlusion, maceration, and perspiration. Environmental factors, such as cold and humidity, both disrupt the homeostasis of the stratum corneum and may act as primary physical irritants.20,21
The irritant itself contributes to the speed and severity of the dermatitis; strong irritants act quickly, whereas weak irritants can require cumulative exposures.19 Common irritants in children are soaps, bleach, detergent, disinfectants, solvent, acids, alkalis, bubble baths, foods, saliva, urine, feces, and metal salts.16,19 Generally, alkalis are more caustic than acids.20
Clinical Presentation
Because of universal susceptibility for ICD, the presentation is highly variable and eruptions can be polymorphous.16 Clinical findings include erythema, scale, edema, vesicles, erosions, fissuring, ulceration, folliculitis, and even bulla.19,20 Areas are often sharply demarcated.19 The most common sites of involvement are exposed areas, such as the hands and face, which are open to irritant exposure. The exception in children is the diaper area.20,22,23
The perioral region is frequently involved in infants and toddlers because of saliva and foods. Teething, pacifier use, and sucking on fingers are common predisposing factors.16 Common culprit foods include citrus, carrots, shrimp, spinach/kale, corn, radish, mustard, garlic, onion, and pineapple.16,19 Saliva exposure through lip licking is a common culprit in older children and adults.
Diaper or napkin dermatitis is most commonly caused by an ICD to urine and feces.23 Basic excrement alters the normally acidic local pH, resulting in irritation, which is compounded by fecal proteases, lipases, and bacteria. Diaper dermatitis is typically found on the convex surfaces and perianally.23 The gluteal cleft and inguinal creases are typically spared, although the perianal area can erode with prolonged contact with irritants.23 The occlusive nature of the diaper/napkin amplifies the irritant effect.22
Different common irritants tend to cause ICD in different body locations. The dorsal hands, for example, are a hot spot for handwashing and hand-sanitizer ICD. In contrast, less than 4% of foot dermatitis is attributable to ICD, whereas nearly half of cases are caused by immunologically mediated ACD.24 Shin guards, as worn in soccer and hockey, can cause both ICD related to trapped moisture and friction and ACD related to the materials from which the guard is made.25,26 Infants with involvement of the bilateral elbows, upper posterior thighs, lower lateral legs, and a band-like distribution on the occipital scalp may develop ICD caused by sweat trapping related to shiny nylon-like car seat material in warmer months, with negative patch testing for relevant allergens.27 Plaques on the posterior thighs and lower buttocks have been reported as pediatric positional sitting dermatitis, an ICD secondary to sitting cross-legged.28 Pustular ICD on the eyelids has been reported in an 8-year-old because of dexpanthenol, a known irritant in cosmetics.29
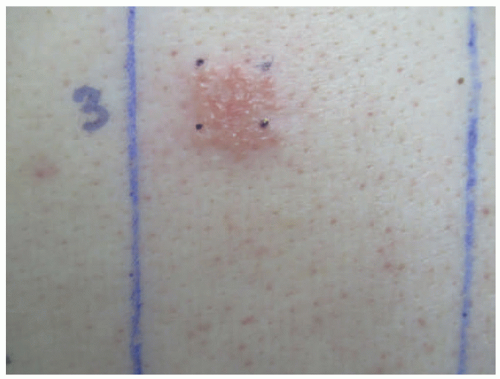
Many unique irritants have been reported in children. Epidemics of fiberglass dermatitis have been reported in classrooms related to desks and airborne irritation.30 The methylphenidate patch commonly used for attention deficit hyperactivity disorder is a relatively common cause of ICD, which can be controlled by rotating patch sites (Figure 3-4).31 Black henna, a common cause of ACD to paraphenylene diamine, has also been reported to cause ICD, with secondary postinflammatory hypopigmentation in a 10-year-old.32 Other reported causes include topical lidocaine/prilocaine (EMLA), green walnut (Juglans regia) shell and husk, and rove beetle (Paederus) toxin.33,34,35,36
Histologic Findings
The pathologic findings depend on the potency of the irritant and the extent of exposure. Strong irritants can cause confluent epidermal necrosis with “ghost” nuclei caused by the loss of nuclear basophilia, either within the superficial epidermis or its full thickness. Epidermal spongiosis is a common finding. Dermal eosinophils may be present (Figure 3-5).37,38
Differential Diagnosis
In general, the differential diagnosis for ICD overlaps with that of spongiotic dermatitis, as discussed previously for atopic dermatitis. The presence of epidermal necrosis should prompt a consideration of interface dermatitis as well. However, in IDC, the injury results from external factors, and so vacuolar degeneration of the basal keratinocytes is absent. Single necrotic keratinocytes at the junction may also reflect wound healing from trauma induced externally.
CAPSULE SUMMARY
IRRITANT CONTACT DERMATITIS
ICD is a very common result of exposure to compounds that are inherently irritating and cytotoxic to the skin. Although adults are often affected by occupational exposures, infants are at high risk because of a weaker epidermal barrier. Presentations include erythema, scale, edema, vesicles, erosions, fissure, ulceration, folliculitis, and even bulla. The histologic changes are those of a spongiotic dermatitis with the presence of necrotic keratinocytes that result from the external damage.
ALLERGIC CONTACT DERMATITIS
Definition and Epidemiology
ACD is an immunologically mediated eczematous dermatitis that results from contact sensitization with a lowmolecular-weight chemical or metal ion and subsequent reexposure leading to a type IV delayed hypersensitivity reaction.39,40 Approximately 20% of people experience contact
sensitization to cutaneous or mucosal exposures, with up to 18% going on to develop ACD.39,40,41,42 Up to 42% of children with atopic dermatitis also have ACD.43
sensitization to cutaneous or mucosal exposures, with up to 18% going on to develop ACD.39,40,41,42 Up to 42% of children with atopic dermatitis also have ACD.43
Etiology
ACD is a type IV delayed hypersensitivity reaction occurring on the mucosa or skin.39,42,43 Asymptomatic contact sensitization occurs 10 to 15 days after initial exposure to an allergen, with subsequent exposure eliciting a reaction after 72 to 96 hours.39,40
Contact sensitizers are polar, low-molecular-weight substances.39,40,41 After penetration of the epidermal barrier, sensitizers bind with endogenous proteins, creating a complex that activates the innate immune system.39,41 Pattern recognition receptors including Toll-like receptors 2 and 4 are activated, resulting in the activation of dendritic cells.39,41 Mast cells, neutrophils, and natural killer cells support the sensitization response.41 Activated dendritic cells migrate through the lymphatics to activate both regulatory and effector T cells in lymph nodes. The balance between these subsets determines susceptibility to future allergic response versus the development of tolerance. Susceptible individuals are characterized by a Th2 skewed response and subsequent inflammation.39,44
Although mild atopic dermatitis may be a risk factor for developing ACD, particularly to nickel,45 some studies suggest that patients with moderate to severe atopic dermatitis may have decreased rates of ACD related to alterations in the Th1/Th2 balance.43 The most frequent contact allergens for children, after Toxicodendron species (poison ivy, oak, and sumac),46 are nickel, fragrance, Balsam of Peru, bacitracin, formaldehyde, cocamidopropyl betaine, propylene glycol, wool alcohol, lanolin, and bronopol.43
Clinical Presentation
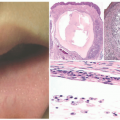
Morphologically, ACD may appear similar to atopic dermatitis. Patients develop pink to erythematous, pruritic, scaly eczematous papules and plaques. Vesicles may be present acutely, whereas lichenification and fissures may be seen with chronic exposures.45,47 The diagnosis should be suspected when the eruption shows linearity, sharp cutoffs, odd/geometric shapes, or other atypical distributions of eczematous dermatitis indicating that something external interacted with the skin.45,47 For example, several cases of ACD to para-phenylenediamine, a dye added to make “black” henna temporary tattoos, have been reported, with the pattern of dermatitis appearing in the shape of the tattoo (Figure 3-6).48 Another classic example presents with linear arrays of bullae on the lower legs, after brushing by poison ivy during a hike, caused by exposure to the potent contact allergen urushiol. Other typical patterns of ACD include nickel allergy on the abdomen where belts and buckles come in contact with the skin (Figure 3-7),45 diaper wipe dermatitis to methylchloroisothiazolinone or methylisothiazolinone, presenting with recalcitrant diaper dermatitis,49 and shin guard dermatitis to adhesives and rubbers.25 Without an obvious external exposure pattern, the diagnosis of ACD can be challenging to differentiate from atopic dermatitis, because many patients are affected with both.47 Additionally, children are more likely to have a scattered and generalized pattern of dermatitis.43,45,50
Systemic contact dermatitis is a generalized, diffuse presentation of dermatitis after a noncutaneous exposure to an allergen.51,52 The exposure is typically by ingestion but can be by parenteral exposure. Balsam of Peru is a tree resin
used in fragrances with significant cross-reactivity with tomato, cinnamon, clove, and orange peel. Ingestion of these foods has been implicated in several cases of systemic contact dermatitis.51,52 Nickel in the diet, in the form of chocolate, oats, legumes, canned foods, and other items, can lead to systemic contact dermatitis as well and often improves with a nickel-free diet.53
used in fragrances with significant cross-reactivity with tomato, cinnamon, clove, and orange peel. Ingestion of these foods has been implicated in several cases of systemic contact dermatitis.51,52 Nickel in the diet, in the form of chocolate, oats, legumes, canned foods, and other items, can lead to systemic contact dermatitis as well and often improves with a nickel-free diet.53
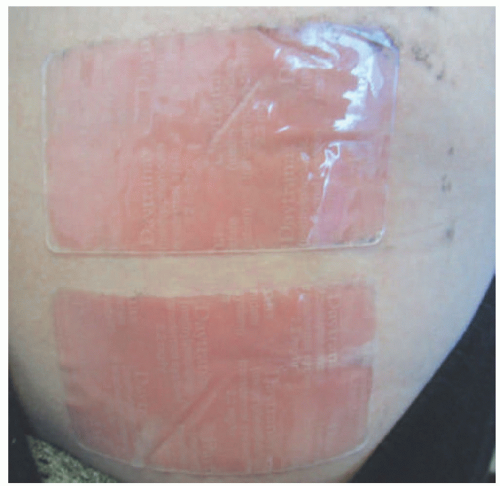
The diagnosis of ACD is made by patch testing. This process involves applying known contact allergens to the skin for 48 hours under occlusion and evaluating for a localized, inflammatory response such as induration or vesiculation at 48 and 96 hours (Figure 3-8).54 Correlation of patch testing results with clinical findings is important to determine the relevance of positives, because testing may reveal prior sensitization to substances with which patients do not regularly come into contact.
Histologic Findings
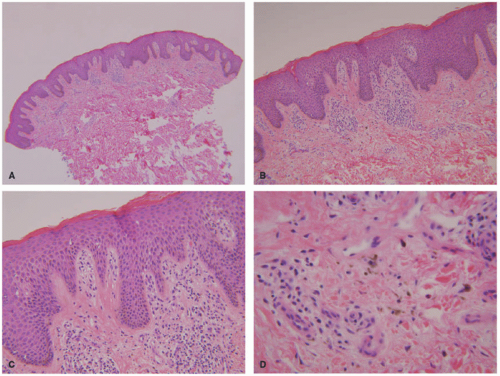
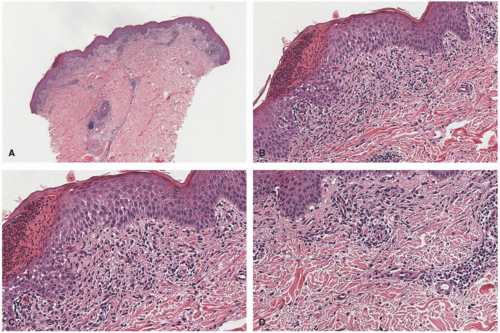
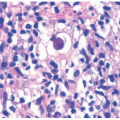
The main pathologic change in ACD is spongiotic dermatitis with frequent epidermal Langerhans cells microabscesses, typically accompanied by a superficial perivascular lymphocytic inflammatory infiltrate with eosinophils. In very florid cases, spongiosis can lead to intraepidermal vesiculation as seen in dyshidrotic eczema (seen on volar skin) and severe acute ACD (as frequently seen in exposure to poison ivy). The dermal eosinophils can also extend into the surface epidermis (eosinophilic spongiosis) (Figure 3-9). The inflammation does not extend to the deeper portions of the biopsy. Scattered dermal neutrophils can also be present, in addition to superficially papillary dermal edema. Chronic photoallergic contact dermatitis, in which contactants
become allergenic only after exposure to sunlight, can show deep perivascular inflammation. Variants of ACD include urticarial (eg, latex), systemic, pustular (eg, cement), purpuric (eg, textile dyes), granulomatous (eg, gold), lymphomatoid (eg, nickel), and chronic dermatitis with prominent postinflammatory hypopigmentation (leukodermic forms).38,55,56
become allergenic only after exposure to sunlight, can show deep perivascular inflammation. Variants of ACD include urticarial (eg, latex), systemic, pustular (eg, cement), purpuric (eg, textile dyes), granulomatous (eg, gold), lymphomatoid (eg, nickel), and chronic dermatitis with prominent postinflammatory hypopigmentation (leukodermic forms).38,55,56
Differential Diagnosis
The histologic differential diagnosis is identical to that of atopic dermatitis, as described earlier, and includes atopic dermatitis, Id reaction, nummular dermatitis, and other causes of spongiotic dermatitis.
CAPSULE SUMMARY
ALLERGIC CONTACT DERMATITIS
ACD is an eczematous dermatitis resulting from a type IV delayed hypersensitivity reaction to a substance to which an individual has been previously sensitized. It may appear clinically similar to atopic dermatitis, presenting with pink to erythematous, pruritic, scaly papules and plaques overlying an erythematous base. Vesicles may be present acutely, whereas lichenification and fissures may be seen in chronic exposures. The pathologic findings include epidermal spongiosis and dermal eosinophils accompanying superficial dermal perivascular lymphocytic inflammation.
SEBORRHEIC DERMATITIS
Definition and Epidemiology
Seborrheic dermatitis (SD) is an eczematous dermatitis that is typically seen in infancy and may reappear starting in adolescence.16 Onset of infantile SD most commonly occurs during the 3rd and 4th weeks of life (with a range of 2-10 weeks of age) and peaks in incidence around 3 months of age.57 Resolution can sometimes be seen as quickly as 3 to 4 weeks after onset, but findings may persist until 8 to 12 months of age. In adolescence, onset is typically seen around puberty.16
Etiology
Although sebaceous gland involvement has been implicated in its development given its distribution in sebaceous gland-rich locations, the exact etiology of SD is unclear and varies with age of onset. Pityrosporum ovale (Malassezia ovalis) is a contributor to SD of adolescence and adulthood, but its role in infantile disease has been debated.58,59,60 The strong association between infantile SD and the later development of atopic dermatitis61 has led many to consider them on a clinical spectrum.62
Clinical Presentation
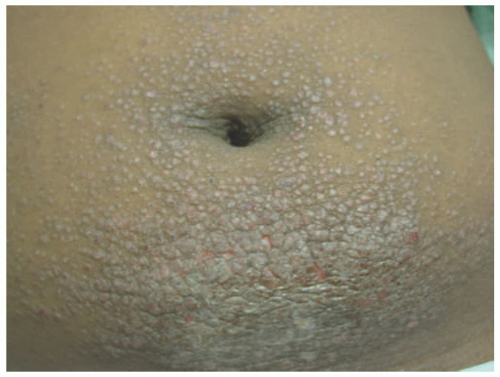
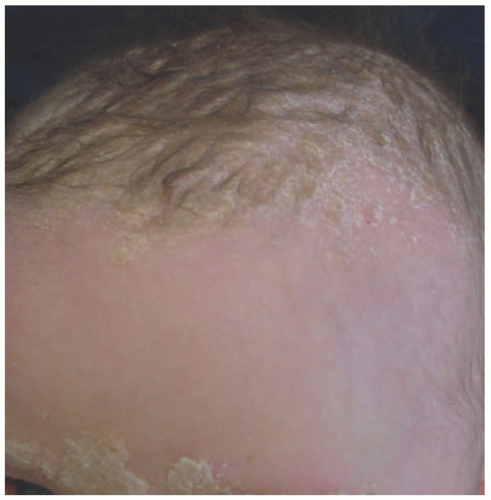
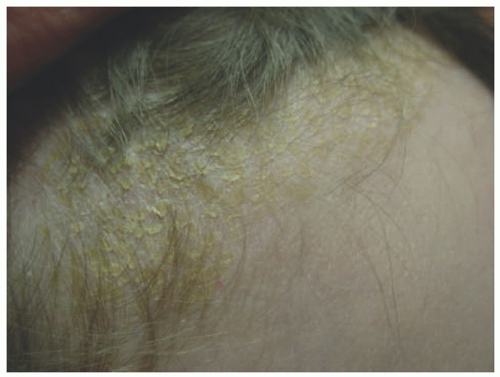
Infantile SD begins as thin dry scaling of the scalp, commonly referred to as “cradle cap” (Figure 3-10). Although isolated scalp involvement is most commonly seen, the diaper area may be affected, with the development of well-marginated, salmon-colored plaques with thick, yellow-brown, greasy crust, and may be complicated by bacterial and candidal infections.16 Involvement may progress to a more typical “seborrheic” distribution including the forehead, ears, eyebrows, nose, occiput, and intertriginous and flexural areas. Pruritus, if present, is usually mild.16 Adolescent SD is similar in presentation to adult disease and is characterized by fine, dry scalp desquamation, commonly referred to as dandruff (Figure 3-11). Inflammation of varying degrees commonly affects the skin in a seborrheic distribution.16 Unlike infantile disease, the eyes can also be affected, where blepharitis presents with erythema and scaling of the lid margins, and tends to be more chronic and recurrent in nature.16
Histologic Findings
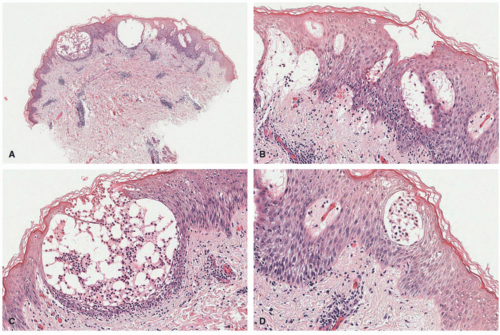
SD shows characteristically a spongiotic or psoriasiform and spongiotic pattern in the epidermis. The classic feature associated with SD is the presence of parakeratosis in the outflow tracts of the hair follicles (also called “shouldering” parakeratosis). Small clusters of neutrophils in the epidermis can be seen, often within the stratum corneum. The epidermis shows psoriasiform elongation of the rete pegs and focal hypogranulosis in most cases. Mild intraepidermal spongiosis is noted (Figure 3-12). The latter is more typically seen in the areas around the hair follicles. Papillary dermal edema and superficial vascular dilatation can
also be noted. Within the dermis, there is a superficial to mid-dermal, perivascular but more typically perifollicular chronic inflammatory infiltrate composed of lymphocytes, histiocytes, and scattered neutrophils. Plasma cells can also be seen. Colonization by Pityrosporum yeast forms is present within the follicular keratin and stratum corneum.63
also be noted. Within the dermis, there is a superficial to mid-dermal, perivascular but more typically perifollicular chronic inflammatory infiltrate composed of lymphocytes, histiocytes, and scattered neutrophils. Plasma cells can also be seen. Colonization by Pityrosporum yeast forms is present within the follicular keratin and stratum corneum.63
Differential Diagnosis
The main differential diagnosis is between SD and psoriasis. Psoriasis shares the epidermal changes seen in SD. However, broader areas of parakeratosis and hypogranulosis are features more typical in the setting of psoriasis. SD also has a larger amount of intraepidermal spongiosis. Other forms of spongiotic dermatoses (previously described) should also be included in the differential diagnosis.
PITYRIASIS ALBA
Definition and Epidemiology
Pityriasis alba (PA) is a mild eczematous eruption that typically results in residual postinflammatory hypopigmentation.16 Its overall incidence in the pediatric population ranges from 1.9% to 5.2%, but is more frequently seen in children with darker skin types. Age of onset is typically between 3 and 16 years.64
CAPSULE SUMMARY
SEBORRHEIC DERMATITIS
SD is an eczematous dermatitis that, within the pediatric population, is typically seen in infancy and adolescence. Although typically isolated to the scalp in infants (cradle cap), it may appear on other typical seborrheic locations such as the face, chest, and intertriginous areas, as well as the diaper area. Histologically, it is characterized by a spongiotic or psoriasiform and spongiotic pattern in the epidermis. The classic feature associated with SD is the presence of parakeratosis in the outflow tracts of the hair follicles (also called “shouldering” parakeratosis).
Etiology
The exact etiology of PA is unclear. It has been postulated that PA is a minor form of atopic dermatitis20,65,66 and is associated with overexposure to light,67,68 personal hygiene practices such as long and frequent bathing and mechanical exfoliation,68,69 microbiologic factors such as Malassezia furfur, S. aureus, and Propionibacterium acnes64 colonization, and even low serum copper levels.70
Clinical Presentation
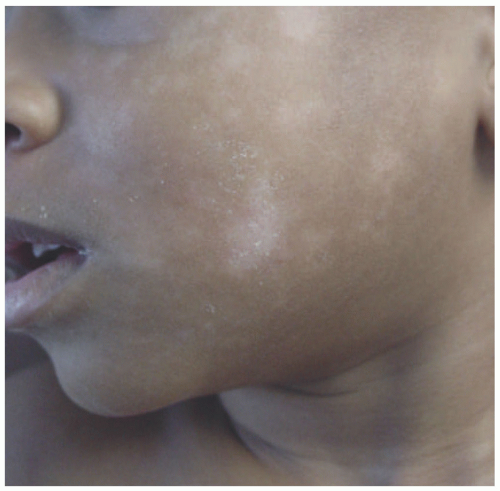
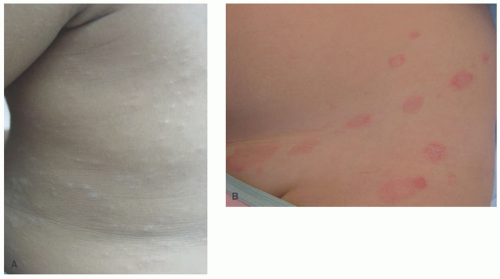
Clinically, PA is considered to be a chronic condition with multiple stages typically involving the upper body, most notably the face. Initially, erythematous changes with uplifted edges may be seen, which, upon resolution, leave hypopigmentation and fine scale (Figure 3-13). Lesions can be associated with pruritus and range in size from 0.5 to 5
cm that may resolve spontaneously or persist for months to years, and can recur.64,69,71
cm that may resolve spontaneously or persist for months to years, and can recur.64,69,71
Three clinical variants of PA have been described—classic (CPA), extensive (EPA), and pigmenting (PPA). CPA is the most common form, presenting with well-defined hypopigmented patches of the lips, chin, and cheeks with scaling borders in primary school-aged children.64 Of the three variants, CPA has the shortest course and is most responsive to treatment.72 EPA is similar in presentation, but the individual lesions tend to be symmetric and larger (>2.0 cm) and involve the neck, trunk, shoulders, and extensor surfaces of upper extremities in teenagers and young adults, particularly females.73,74 PPA is the least common variant and is notable among non-Caucasian individuals of the Republic of South Africa and the Middle East.75,76 In fact, there are no documented cases within the Caucasian population. It presents with approximately 1.5 cm bluish patches with a surrounding hypopigmented halo typically involving the forehead and cheeks of females during childhood and adolescence.64,75,77,78 PPA has been associated with mycotic infections and can coexist with CPA.75
Histologic Findings
The pathologic findings are relatively nonspecific. There is a mild degree of epidermal spongiosis with hyperkeratosis and areas of parakeratosis. Follicular plugging can be sometimes present. Atrophic sebaceous glands have been reported in some patients. Within the dermis, there is a superficial and perivascular lymphocytic infiltrate with focal exocytosis into the epidermis. The more characteristic finding is the presence of decreased melanin pigment in the epidermis, with normal numbers of melanocytes. Residual melanocytes within the affected areas demonstrate a decreased number of melanosomes on ultrastructural studies.73,79,80,81
Differential Diagnosis
The main clinical differential diagnoses are vitiligo and tinea versicolor. Histologically, the presence of superficial scale is a useful clue to distinguish PA from vitiligo (which is usually nonscaling). In addition, vitiligo shows absence of melanocytes within the hypopigmented patches (as opposed to the more preserved number of cells in PA). Pityriasis versicolor can be easily distinguished from PA on the basis of the presence of fungal organisms in the stratum corneum with the use of PAS or GMS stains.
CAPSULE SUMMARY
PITYRIASIS ALBA
PA is a mild eruption, often associated with residual postinflammatory hypopigmentation, most typically involving the upper body, most notably the face, and presenting as variably dyspigmented patches with fine scale. The pathologic findings are relatively nonspecific and include a mild degree of epidermal spongiosis with hyperkeratosis and areas of parakeratosis. The more characteristic finding is the presence of decreased melanin pigment in the epidermis with a relative sparing of the number of intraepidermal melanocytes.
JOB SYNDROME
Definition and Epidemiology
Job syndrome, or autosomal dominant hyperimmunoglobulinemia E syndrome (AD-HIES), is a rare primary immunodeficiency disorder characterized by elevated immunoglobulin E (IgE) (>2000 IU/mL), eczematous eruptions, and abscesses and sinopulmonary infections.16 Onset of cutaneous symptoms can be seen as early as the neonatal period or infancy, with recurrent infections beginning as early as 3 months of age.16
Etiology
Job syndrome is associated with mutations in STAT3, which is integral to multiple cytokines’ signal transduction, particularly Th17 and CD4. These mutations result in a failure of Th17 and CD4 differentiation, leading to increased susceptibility to various infections such as S. aureus,82,83,84,85 mucosal candidiasis,86,87,88,89 and lung and cutaneous infections.89,90 Nonimmunologic abnormalities such as supernumerary teeth, delayed tooth eruption, and craniosynostosis are thought to be a consequence of impaired IL-11 signaling secondary to missense mutations in Arg296Trp, resulting in an impaired stimulation of STAT3.91
Clinical Presentation
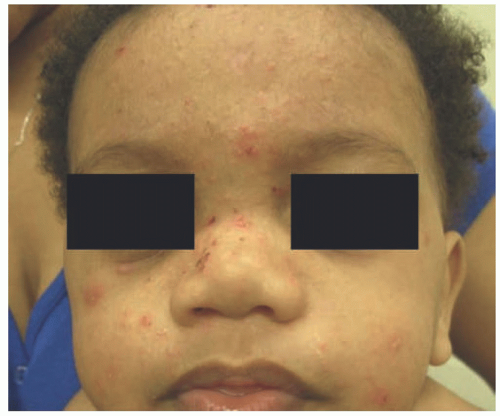
Clinically, Job syndrome can have a wide phenotypic presentation because of the pleiotrophic effects of STAT3. Immunologic abnormalities can present within the first few weeks of life and even at birth. Typically, this is seen as an eczematous or pustular eruption of the face and scalp,92,93 which can progress to an eczematoid dermatitis (Figure 3-14). S. aureus is implicated as a driving force behind the eruption.94,95 Recurrent sinopulmonary infections are commonly seen, with the majority of patients experiencing at least one episode of pneumonia and more than 50% experiencing three or more episodes. Causative organisms can include S. aureus, Streptococcus pneumoniae, and less frequently, Haemophilus species.96,97,98 Subsequent aberrant healing, in the form of pneumatoceles and bronchiectasis, is seen in up to 75% of patients.95 Causative agents are similar to those associated with cystic fibrosis, making treatment difficult and resulting in significant morbidity and mortality.95,99 Fungal infections, specifically chronic oral candidiasis, are seen in up to 80% of patients.95 Lastly, “cold abscesses,” or boils and furuncles lacking in erythema and warmth, are an almost universal feature of Job syndrome.100 Patients with Job syndrome also have a higher incidence of malignancy, specifically non-Hodgkin lymphoma.101,102,103
Nonimmunologic features, including craniofacial, musculoskeletal, dental, and vascular abnormalities, can also be seen. Characteristic facies present in late childhood and early adolescence, including increased interalar distance, prominent forehead and chin, coarse skin, and facial asymmetry.96,104 Musculoskeletal abnormalities include scoliosis, joint hyperextensibility, early development of degenerative joint disease, and minimal trauma fractures.96,98 Oral and dental abnormalities, such as retention of primary teeth (often three or more), are seen in approximately 70% of patients,95 and almost all patients demonstrate a high-arched palate with central ridging and fissuring of the palate and buccal mucosa.105 Vascular abnormalities have been more recently identified and can include aneurysms of middle-sized arteries, lacunar infarcts, and hypertension in the absence of atherosclerosis.95
Histologic Findings
The pathologic findings in Job syndrome are those of a spongiotic dermatitis with dermal and/or intraepidermal eosinophils. There is hyperkeratosis with areas of parakeratosis and a small serum crust. There is epidermal acanthosis and spongiosis. A superficial and perivascular lymphocytic infiltrate is present, which sometimes extends to the epidermis (exocytosis). In some cases, an eosinophilic pustular folliculitis (resembling the eosinophilic folliculitis in newborns) can be seen. At the time of birth, many patients show a pustular eruption that is more pronounced in the face and contains abundant eosinophils on the biopsy. However, such changes resolve rapidly, and the more classic spongiotic pattern develops.92,106
Differential Diagnosis
As with other forms of spongiotic dermatitis, the differential diagnosis overlaps with that of eczema, contact dermatitis, atopic dermatitis, and others (previously discussed). Eosinophilic folliculitis can also be considered in the differential diagnosis, particularly in those acute presentations after birth.
CAPSULE SUMMARY
JOB SYNDROME, OR AUTOSOMAL DOMINANT HYPERIMMUNOGLOBULINEMIA E SYNDROME
AD-HIES is a rare primary immunodeficiency disorder characterized by elevated IgE (>2000 IU/mL), recurrent eczematous eruptions, abscesses, and sinopulmonary infections. Immunologic abnormalities can present within the first few weeks of life and even at birth. Typically, this is seen as an eczematous or pustular eruption of the face and scalp, which can progress to an eczematoid dermatitis. The pathologic findings in Job syndrome are those of a spongiotic dermatitis with dermal and/or intraepidermal eosinophils.
JUVENILE PLANTAR DERMATOSIS
Definition and Epidemiology
Juvenile plantar dermatosis (JPD) is a very common scaling and fissuring dermatitis of infancy and childhood, localized to the distal aspects of the plantar surfaces.16,107 It is seen
more commonly among atopic children16,107,108 and can be associated with hyperhidrosis.16 Onset is generally between 3 and 14 years old.107
more commonly among atopic children16,107,108 and can be associated with hyperhidrosis.16 Onset is generally between 3 and 14 years old.107
Etiology
Clinical Presentation
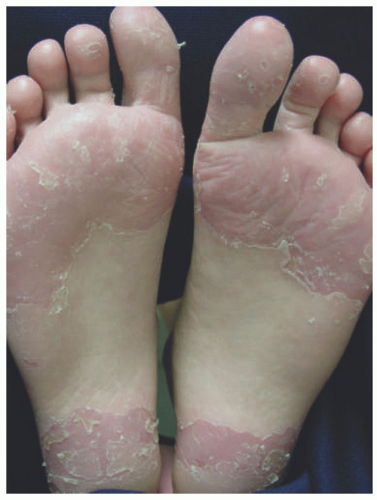
Clinically, JPD appears as symmetric, shiny erythematous plantar skin and toes, particularly of the great toes (Figure 3-15). There is notable sparing of the arches and interdigital web spaces. Scaling and fissuring can be seen, and if chronic, can become lichenified.16,107 There are reports of similar cutaneous changes of the fingertips in 5% of patients with palmar hyperhidrosis.16 JPD can persist for several years if left untreated, and flaring can be precipitated by heat, sweating, or friction secondary to socks and/or occlusive footwear.16,107
Histologic Findings
The pathologic findings are those of a subacute or chronic spongiotic dermatitis. Some patients can also have a spongiotic and psoriasiform pattern. A distinctive feature of JPD is the presence of perieccrine lymphocytic inflammation (Figure 3-16). The inflammation of the acrosyringium or the eccrine sweat duct at its epidermal starting edge tends to be present in the most typical clinical picture of JPD.109,110
Differential Diagnosis
The differential diagnosis includes other entities with spongiotic tissue reactions. The presence of perieccrine inflammation can also be seen in lichen striatus. However, the blaschkoid distribution of the clinical lesions and the presence of interface changes in lichen striatus can easily distinguish those conditions.
CAPSULE SUMMARY
JUVENILE PLANTAR DERMATOSIS
JPD is a very common scaling and fissuring dermatitis of infancy and childhood, localized to the distal aspects of the plantar surfaces of the feet. Clinically, JPD will manifest as symmetric, shiny erythematous plantar skin and toes, particularly of the great toes. There is notable sparing of the arches and interdigital web spaces. The pathologic findings are those of a subacute or chronic spongiotic dermatitis. A distinctive feature of JPD is the presence of perieccrine inflammation.
LICHEN SIMPLEX CHRONICUS
Definition and Epidemiology
Lichen simplex chronicus (LSC) (also known as atopic or circumscribed neurodermatitis) is a chronic, pruritic dermatosis localized to the areas of habitual rubbing, scratching, and, less commonly, picking.16,111
The incidence of LSC increases with age, and patients are most likely to be adolescents or adults.16,112 Pediatric LSC may have a slight male predominance.111 When present in children, LSC is usually caused by underlying pruritic skin disease, such as atopic dermatitis, or by a tendency toward repetitive behaviors.20,113 Anxiety disorders, such as obsessive-compulsive disorders and autism spectrum disorders, have characteristic repetitive behaviors that are potentially self-injurious; these behaviors often arise out of psychological distress and predispose patients to LSC.20,113,114
Etiology
LSC represents reactive epidermal hypertrophy in response to repetitive focal skin trauma or manipulation.20
Clinical Presentation
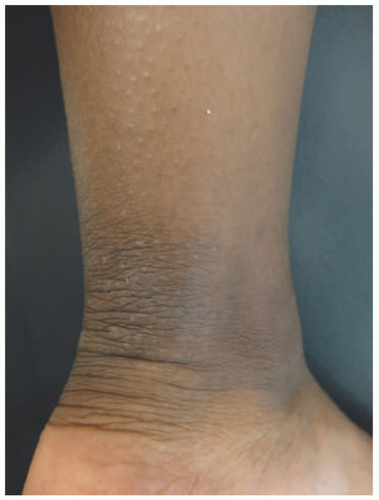
Early LSC has erythematous, slightly edematous, scaling plaques with accentuated skin markings.16 Over time, these evolve into well-demarcated, dry, thickened plaques with a leathery appearance (Figure 3-17).16,20,111 They can be hyperpigmented or hypopigmented over time, even developing a white hue in moist areas.16,20,111 Excoriation may be noted.111 LSC can arise de novo in normal skin or in areas of primary pruritic dermatoses, such as atopic dermatitis, SD, psoriasis,
or lichen planus (LP). LSC is usually located in areas that are easily reached and can be inconspicuously scratched: the neck, wrist, ankles, hands, forearms, and pretibial shins.16,20 When involving the anogenital region, LSC is usually symmetric and favors the labia majora, scrotum, or perianal area.111 The diagnosis is made clinically on the basis of lichenified plaques with severe pruritus and a history of scratching/rubbing.16,111
or lichen planus (LP). LSC is usually located in areas that are easily reached and can be inconspicuously scratched: the neck, wrist, ankles, hands, forearms, and pretibial shins.16,20 When involving the anogenital region, LSC is usually symmetric and favors the labia majora, scrotum, or perianal area.111 The diagnosis is made clinically on the basis of lichenified plaques with severe pruritus and a history of scratching/rubbing.16,111
Histologic Findings
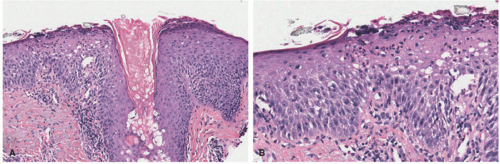
LSC and prurigo nodularis (“picker’s nodules”) are considered as two ends of the spectrum of clinical lesions that range from a plaque (LSC) to a nodule (PN). Pathologically, there is compact hyperkeratosis, often without significant parakeratosis (Figure 3-18). The epidermis is acanthotic, with irregular or regular elongation of the rete pegs in a psoriasiform pattern. A mild degree of epidermal spongiosis can be noted. The granular cell layer is thickened, and there is usually a zone of stratum lucidum between the epidermis and the stratum corneum. In cases of continuous exogenous trauma, scattered apoptotic keratinocytes can be present. There is superficial papillary dermal fibrosis with vertically oriented collagen bundles extending from the superficial vascular plexus to the epidermis. A mild superficial perivascular lymphohistiocytic inflammatory infiltrate is seen, with or without dermal eosinophils. Neural hypertrophy has been described in LSC. LSC typically has a flat epidermal surface. Its counterpart PN shows a gradient of crescendo/decrescendo acanthosis. Epidermal excoriation can also be seen.115,116,117,118,119
Differential Diagnosis
The differential diagnosis includes psoriasis, tinea infection, LP, epidermal nevus, and verruca vulgaris. LSC and psoriasis can share the epidermal acanthosis and regular elongation of the rete. However, as opposed to LSC, the granular cell layer is markedly diminished in psoriasis. In addition, clusters of neutrophils in the epidermis and stratum corneum are seen in psoriasis, but not in LSC. LP can share the hyperkeratosis and thickening of the granular cell layer seen in LSC, but the epidermis shows more irregular acanthosis (“sawtooth” pattern). In addition, the band-like infiltration of lymphocytes obscuring the dermal-epidermal junction and interface changes are not seen in LSC. Verruca vulgaris may also be considered. Warts have viral cytopathic changes, columns of parakeratosis, and intracorneal hemorrhage. Epidermal nevi can also look similar to LSC, but lack the dermal fibrosi of LSC. Tinea can easily be excluded using special stains (which are often advocated to be performed in all cases of LSC).
CAPSULE SUMMARY
LICHEN SIMPLEX CHRONICUS
LSC (also known as atopic or circumscribed neurodermatitis) is a chronic, pruritic dermatosis localized to the areas of habitual rubbing, scratching, and picking. It typically presents with erythematous, slightly edematous, scaling plaques with accentuated skin markings. Pathologically, LSC shows psoriasiform acanthosis, hypergranulosis, a stratum lucidum, and superficial dermal fibroplasia with vertical orientation to the epidermis.
POLYMORPHOUS LIGHT ERUPTION
Definition and Epidemiology
Polymorphous light eruption (PMLE) is the most common idiopathic photosensitive disorder. It is characterized by recurrent cutaneous reactions following exposure to natural or artificial ultraviolet (UV) radiation. Sensitivity occurs commonly to the UVA and/or UVB spectrum,120 but occasionally to visible light as well.121 These reactions, usually seen in spring or early summer, develop minutes to hours after exposure and can last for 1 to 2 weeks afterward.
PMLE has a slight female predominance and is more common in the 2nd to 3rd decades of life. It has a higher incidence in populations living at higher latitudes and elevations. Juvenile spring eruption, a subset of PMLE, is seen in children between 5 and 12 years of age and occurs more often in boys than in girls.
Etiology
The etiology of PMLE has not been fully elucidated. Proposed mechanisms include a delayed-type hypersensitivity reaction to endogenous cutaneous photo-induced antigens and abnormal UV-induced immunosuppression.122 Photohardening studies suggest that decreased levels of regulatory T cells, mast cells, and epidermal Langerhans cells may play a role in the pathogenesis of PMLE.123
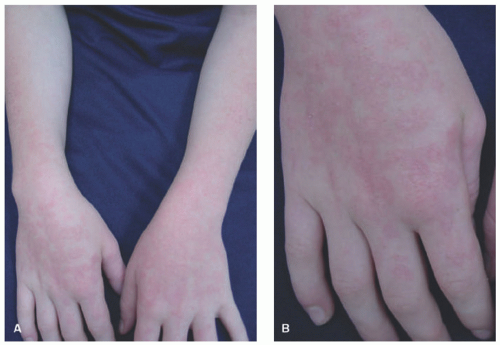
Clinical Presentation
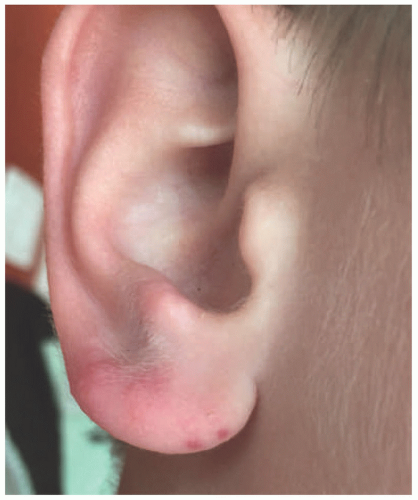
PMLE, as the name implies, may present with a variety of findings, from erythematous nonscarring papules or vesicles to urticarial or erythema multiforme (EM)-like papules or plaques on exposed surfaces (Figure 3-19). Pruritus is also variable, ranging from none to severe. In children, it often appears as an eczematous eruption with small papules on the face, progressing to other sun-exposed areas. Individuals with darker skin types may present with a lichen nitidus-like eruption.124 Juvenile spring eruption, considered a subset of PMLE, consists of edematous papules and vesicles, mainly on the helices of the ears but occasionally on the dorsal surfaces of the hands
or elbows (Figure 3-20).125 It can occur 8 to 24 hours after UV exposure and is often pruritic. Like PMLE, the lesions resolve without scarring, usually within 2 weeks.
or elbows (Figure 3-20).125 It can occur 8 to 24 hours after UV exposure and is often pruritic. Like PMLE, the lesions resolve without scarring, usually within 2 weeks.
Histologic Findings
PMLE is pathologically characterized by a superficial and deep, relatively dense, predominantly chronic lymphocytic inflammatory infiltrate largely devoid of eosinophils and plasma cells (Figure 3-21). Depending on the type of clinical lesion, other histopathologic findings can be noted. Urticarial lesions and blisters are accompanied by marked superficial dermal edema. Eczematous or papulovesicular lesions show epidermal spongiosis. Purpuric lesions frequently have extravasated red blood cells. In children, the histologic findings of juvenile spring eruption are considered by some to be on the spectrum of PMLE.126,127,128,129,130
Differential Diagnosis
Actinic prurigo (AP) may present with psoriasiform epidermal changes, in addition to a deeper and denser inflammatory infiltrate when compared with PMLE. The inflammation can have eosinophils and reactive lymphoid follicles. Photoallergic contact dermatitis should be considered in the differential diagnosis if spongiosis and eosinophils are present. Acute radiation dermatitis and phototoxic dermatitis show scattered single necrotic keratinocytes in the mid-epidermis (sunburn cells) and junctional vacuolar alteration (interface changes). Lupus erythematosus can also enter the differential diagnosis and can be differentiated by the presence of superficial and deep, perivascular, and periadnexal inflammation with increased dermal mucin.
CAPSULE SUMMARY
POLYMORPHOUS LIGHT ERUPTION
PMLE is the most common idiopathic photosensitive disorder. It is characterized by recurrent cutaneous reactions following exposure to natural or artificial UV radiation. In children, it often appears as an eczematous eruption with small papules on the face, progressing to other sun-exposed areas. Individuals with darker skin types may present with a lichen nitidus-like eruption. PMLE is pathologically characterized by a superficial and deep, relatively dense, and predominantly chronic, lymphocytic inflammatory infiltrate.
ACTINIC PRURIGO
Definition and Epidemiology
AP also known as hydroa aestivale and Hutchinson summer prurigo, is a chronic photodermatosis that is less common than PMLE. It usually begins in childhood and can last for many years.131 Although patients with AP appear to have a higher percentage of first-degree relatives with PMLE,132 it appears to be a distinct photosensitive disorder. Both UVB and UVA have been implicated, but reactivity to UVB appears to be more common.133
AP occurs mainly in Native Americans and Mestizo Indians from North, Central, and South America, although it can be seen in other parts of the world. Human leukocyte antigen (HLA) DRB1*0407 is found in 60% to 70% of patients with AP, but in only 4% to 8% of unaffected individuals.134 HLA DRB1*0401 has been shown in up to 20% of affected individuals. Although both AP and PMLE can occur in the same individual, no HLA associations have been found with the latter. AP is more common in females.
Etiology
The pathogenesis of AP has yet to be determined, but an immunologic mechanism seems likely, given known HLA associations. AP lesional skin shows proliferative responses to autologous skin antigens in vitro.135 Response to thalidomide in AP patients correlates with a decrease in TNF-α production and an increase in IFN-γ-positive CD3+ peripheral blood mononuclear cells.136 A recent study suggests the role of a type IV hypersensitivity-like reaction, indicated by the presence of IgE, eosinophils, and mast cells in tissue from AP patients.137
Clinical Presentation
AP lesions present on sun-exposed areas but may involve covered areas as well, especially the buttocks. The lesions appear as erythematous and often crusted papules, nodules, and lichenified plaques, which can heal with pitting or linear scars, perhaps exacerbated by scratching because of intense pruritus (Figure 3-22). Ocular or lip involvement can be seen in 30% to 50% of affected patients. Cheilitis usually involves mainly the lower lip and may be the sole manifestation of the disease.138 Ocular findings include conjunctivitis, photophobia, and exudate.139
Histologic Findings
The pathologic findings of AP are variable and relatively nonspecific. There is hyperkeratosis with ortho- or parakeratosis. The epidermis shows regular acanthosis, focal or multifocal spongiosis, and thickening of the basal membrane. There is a dense perivascular lymphocytic infiltrate in the superficial to mid-dermis, with papillary dermal edema. Eosinophils, melanophages, and extravasated erythrocytes can be seen (Figure 3-23).
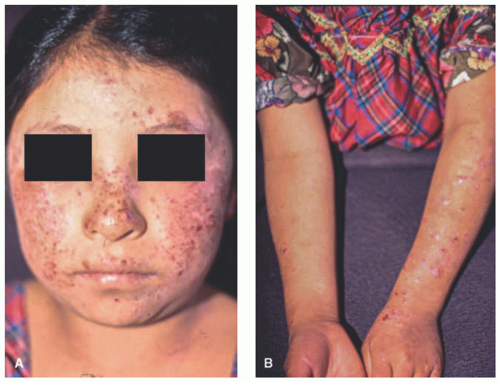 FIGURE 3-22. Actinic prurigo. Crusted and eroded erythematous papules and plaques on sun-exposed areas of the face and arms (A and B). |
The pathologic findings on the biopsies of cheilitis vary depending on where the biopsy is taken from and at what phase of the disease the lesion has been biopsied. The biopsies from the lower vermillion show more typical changes that appear to be more specific for this condition. In the acute phase, there is acanthosis, spongiosis, and eosinophilia, along with dermal edema, congested vessels, and a lymphocytic infiltrate. Chronic lesions show an ulcerated epithelium with a dense lymphohistiocytic infiltrate and the presence of well-formed lymphoid follicles. The lymphoid follicles have been interpreted as a relatively specific finding for the diagnosis of AP cheilitis and have been termed “follicular cheilitis.” The follicles can show germinal center formation and coalesce into irregular shapes but maintain a discernible polarized mantle and marginal zone (Figure 3-24).
The affected conjunctivae show epithelial hyperplasia alternating with atrophy, vacuolization of the basal layer, and dilated capillaries in the dermis. Similar to mucosal lip biopsies, lymphoid follicles can be seen.138,140,141
Differential Diagnosis
The most important pathologic differential diagnosis is with cutaneous lymphoid hyperplasia (CLH) and low-grade B-cell lymphomas. Follicular cheilitis and CLH have similar histologic changes. As opposed to AP, CLH typically presents as a solitary lesion on the face, with a predilection for females and Caucasian patients. The lips are not typically involved by CLH. Low-grade B-cell lymphomas include cutaneous marginal zone lymphomas and primary cutaneous follicle center lymphomas. The latter is not typically a disease of children and shows poorly formed follicles that lack germinal centers. BCL-2 can be coexpressed in 25% of the neoplastic cells. Cutaneous marginal zone lymphomas are the more common lymphomas in children, also occurring as solitary lesions in the trunk and extremities. The follicles tend to be atrophic and not reactive as in AP. Demonstration of clonality (by polymerase chain reaction [PCR] or in situ hybridization) can also help in distinguishing an
inflammatory (polyclonal) from a neoplastic (clonal) condition. Atypical marginal zone hyperplasias can also be included in the differential because they sometimes occur in children. They are characterized by clonal lambdarestriction by immunohistochemistry or in situ hybridization studies and lack a clonal population of cells by PCR analysis of the IGH gene.
inflammatory (polyclonal) from a neoplastic (clonal) condition. Atypical marginal zone hyperplasias can also be included in the differential because they sometimes occur in children. They are characterized by clonal lambdarestriction by immunohistochemistry or in situ hybridization studies and lack a clonal population of cells by PCR analysis of the IGH gene.
CAPSULE SUMMARY
ACTINIC PRURIGO
AP, also known as hydroa aestivale and Hutchinson summer prurigo, is a chronic photodermatosis that usually begins in childhood and can last for many years. Clinically, it presents on sun-exposed areas but may involve covered areas as well, especially the buttocks. Lesions appear as erythematous and often crusted papules, nodules, and lichenified plaques, which heal with pitting or linear scars. Ocular or lip involvement can be seen in 30% to 50% of affected patients. The presence of follicular cheilitis, lymphoid follicles in affected areas of the lip, appears to be a distinctive histologic finding of this condition.
PHOTOTOXIC REACTIONS
Definition and Epidemiology
Phototoxic reactions occur when light activates a drug or chemical agent (Table 3-1). UVA (340-400 nm) is most often the cause, with rare cases attributable to UVB or visible light. When activated, these agents or their metabolites are converted into products that can cause direct injury to keratinocytes, resulting in an inflammatory response in the skin that is noted minutes to hours after exposure. Phototoxicity is the main mechanism of systemic drug photosensitivity.142,143 Topical psoralens (from medications or furocoumarin-containing plants) also produce a phototoxic reaction. Prior sensitization to the agent is not required because the reaction is not immune mediated.
Phototoxic reactions are relatively common, but their prevalence is difficult to determine because many cases may not be recognized or reported as such. At several photodermatology centers, phototoxicity was the diagnosis in 5% to 16% of patients.144,145 Not all patients exposed to the same drug exhibit a phototoxic response, and there is variability in the severity and extent of involvement.146 Emerging drugs with phototoxic potential include voriconazole and BRAF inhibitors. A recent review of pediatric patients treated with voriconazole at a children’s hospital over a 10-year period showed the incidence of phototoxicity to be 20%, increasing to 47% in patients treated for 6 months or longer.147
TABLE 3-1. Phototoxic medications | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Etiology
When phototoxic compounds absorb photons from radiation, a variety of reactive oxygen-intermediate compounds are formed, such as free oxygen radicals, superoxide anions, and hydrogen peroxide, which then activate proinflammatory pathways. The result is direct injury to cell membranes and cytoplasmic structures, or even nuclear DNA (as is the case with psoralens), which can lead to cell death.
Clinical Presentation
The most common presentation of a phototoxic reaction is sunburn-like erythema in exposed areas within minutes to several hours after light exposure. There may be associated stinging or burning. Edema, vesicles, or blistering may occur, followed by healing with desquamation and occasionally hyperpigmentation.
Phytophotodermatitis is the most common phototoxic reaction in children and is caused by furocoumarin-containing fruits and plants (Table 3-2).16 When there is
contact with skin followed by exposure to sunlight, erythema, and sometimes vesicles or blisters, occurs, often in a streaky pattern that reflects the areas of contact with the agent (Figure 3-25). Hyperpigmentation eventually appears in the involved areas. Phytophotodermatitis can be mistaken for ACD, cellulitis, and even child abuse.148
contact with skin followed by exposure to sunlight, erythema, and sometimes vesicles or blisters, occurs, often in a streaky pattern that reflects the areas of contact with the agent (Figure 3-25). Hyperpigmentation eventually appears in the involved areas. Phytophotodermatitis can be mistaken for ACD, cellulitis, and even child abuse.148
TABLE 3-2. Drugs implicated in drug-induced hypersensitivity syndrome | ||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||
Pseudoporphyria is another phototoxic reaction that can be seen in up to 11% of children who are taking nonsteroidal anti-inflammatory agents, especially naproxen sodium for arthritis.149,150,151 These patients present with lesions that clinically resemble porphyria cutanea tarda with fragile skin, tense bullae, and erythema with erosions that crust, often healing with milia and scarring. Involvement is usually seen a few months after starting the drug, and occurs in sun-exposed areas that are subject to mild trauma, especially the backs of the hands. Unlike true porphyria, there are no porphyrin abnormalities in the urine, stool, serum, or red blood cells. Similar reactions have been noted with other medications, including antibiotics and antifungals such as tetracyclines, ciprofloxacin, voriconazole, systemic retinoids, diuretics, metformin, dapsone, imatinib, and cyclosporine.150
Histologic Findings
Pathologically, phototoxic reactions show a mild degree of epidermal spongiosis with scattered apoptotic keratinocytes (“sunburn” cells), which are often present at all levels of the epidermis. There is a lymphohistiocytic infiltrate with a superficial dermal perivascular distribution and associated exocytosis (Figure 3-26). Eosinophils are not frequently seen.152,153,154
Differential Diagnosis
The differential diagnosis includes graft-versus-host disease (GVHD), EM, and other interface dermatitides (lupus, dermatomyositis, etc.). PMLE may also be considered. GVHD may have overlapping histopathologic changes, but can be easily differentiated on the basis of the clinical history and presentation. Lupus erythematosus shows interface changes
and a superficial and deep, perivascular, and periadnexal infiltrate with increased dermal mucin. Dermatomyositis shares similar histologic findings, including the presence of interstitial dermal mucin, but typically shows epidermal atrophy. PMLE lacks apoptotic keratinocytes and often has deep perivascular inflammation and dermal edema that are not frequent in phototoxic reactions. Photoallergic dermatitis classically shows abundant dermal eosinophils and more prominent spongiosis.
and a superficial and deep, perivascular, and periadnexal infiltrate with increased dermal mucin. Dermatomyositis shares similar histologic findings, including the presence of interstitial dermal mucin, but typically shows epidermal atrophy. PMLE lacks apoptotic keratinocytes and often has deep perivascular inflammation and dermal edema that are not frequent in phototoxic reactions. Photoallergic dermatitis classically shows abundant dermal eosinophils and more prominent spongiosis.
CAPSULE SUMMARY
PHOTOTOXIC REACTIONS
These occur when light activates a drug or chemical agent after systemic or topical exposure. The most common presentation of a phototoxic reaction is sunburn-like erythema in exposed areas within minutes to several hours after light exposure and may be associated with stinging or burning. Edema, vesicles, or blistering may occur, followed by healing with desquamation and occasionally hyperpigmentation. Pathologically, phototoxic reactions show a mild degree of epidermal spongiosis with scattered apoptotic keratinocytes (“sunburn” cells).
DRUG-INDUCED HYPERSENSITIVITY SYNDROME
Definition and Epidemiology
Drug-induced hypersensitivity syndrome (DIHS) has been known by many names since it was described almost 70 years ago in patients taking certain anticonvulsants. Initially described as phenytoin hypersensitivity syndrome, the most familiar name is Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS). Aromatic anticonvulsants still predominate as the precipitating drugs, as do sulfonamides, but multiple other medications have been implicated (Table 3-2). DIHS consists of a spectrum of clinical and laboratory features that include fever, exanthem, lymphadenopathy, facial edema, eosinophilia, and internal organ involvement. DIHS is distinguished from other drug reactions and syndromes by its longer latency until onset of findings and the relative lack of mucous membrane involvement.
In children, the frequency of DIHS has been reported to be 27.6%155 with a mean age of about 9 years.156 It occurs almost equally in males and females. In some ethnic groups, specific HLA allele types have been linked to a predisposition to severe drug hypersensitivity reactions, including
DIHS, with exposure to phenobarbital, carbamezepine, allopurinol, dapsone, and abacavir.157,158,159
DIHS, with exposure to phenobarbital, carbamezepine, allopurinol, dapsone, and abacavir.157,158,159
Etiology
Although the exact mechanism has not yet been elucidated, the etiology of DIHS appears to be a manifestation of immune responses to one or more pathogenic events involving defects in drug metabolism, eosinophil production, and viral reactivation. Patients with genetic polymorphisms in genes encoding drug-metabolizing enzymes in the cytochrome P450 system accumulate toxic arene oxide and hydroxylamine metabolites, which then evoke a complex hypersensitivity response by the immune system.147,160,161 The presence of large numbers of eosinophils as part of the immune response in DIHS can cause direct tissue damage upon release of cytotoxic granule proteins.158,162
Reactivation of human herpesviruses HHV-6, HHV-7, Epstein-Barr virus (EBV), and cytomegalovirus (CMV) has been found in patients with many cutaneous adverse drug reactions,163 but it appears that reactivation of HHV-6 is found almost exclusively in patients with DIHS.164 The massive expansion of regulatory T cells that occurs in the acute stages of DIHS may have the effect of inducing viral reactivation.165 In children with drug hypersensitivity syndrome, patients who showed HHV-6 positivity had a more severe disease course than those children who were HHV-6 negative.166
Clinical Presentation
The clinical features of DIHS are similar in children and adults and usually begin on average 6 weeks after beginning the culprit medication, with a range of 2 to 8 weeks. It often starts with features similar to those of a viral prodrome with malaise, pharyngitis, and fever. Lymphadenopathy as well as arthralgia may be noted. A morbilliform rash will follow in 75% of patients accompanied by edema of the face, especially the periorbital area (Figure 3-27). The rash can be follicular based and becomes pruritic as it advances down the trunk and extremities. The development of vesicles, tense bullae, and targetoid-like lesions can occur and may suggest Stevens-Johnson syndrome (SJS), but mucous membrane involvement is not a hallmark of DIHS and is usually found only when there is progression to diffuse erythroderma.156,160,167
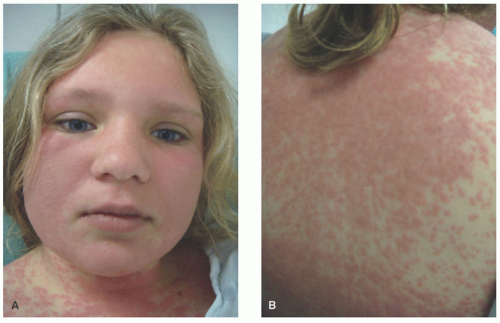 FIGURE 3-27. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia. Coalescing erythematous, edematous papules and plaques (A and B). Note the marked facial swelling. |
Laboratory evaluation is key to making the diagnosis of DIHS in children as it is in adults. The incidence of organ system involvement is similar regardless of age, with the most common finding being peripheral eosinophilia (>2.0 × 109 eosinophils/L) in 50% to 80% and atypical lymphocytosis in 72% of patients. The liver is the most commonly affected visceral organ, with involvement in 50% to 90% of cases. Although rare, progression to fulminant hepatitis with liver necrosis may occur and carries a mortality of 10%. The kidney is affected in 11% to 16% of patients, especially when allopurinol is the inciting agent. Pulmonary involvement is more likely with abacavir exposure and may include interstitial pneumonitis, pleuritis, and rarely acute respiratory distress syndrome.160,167 Myocarditis, meningitis, and encephalitis have been reported, but are rare.
Involvement of the skin and other organs can last for weeks to months. Autoimmune disorders, including thyroiditis, type 1 diabetes mellitus, and pancreatitis, can occur 2 to 3 months after resolution of DIHS.167,168 It has been
postulated that a gradual loss of regulatory T-cell function may increase the risk of developing subsequent autoimmune disease.165 Patients should be monitored for these disorders beginning several months after resolution of DIHS.
postulated that a gradual loss of regulatory T-cell function may increase the risk of developing subsequent autoimmune disease.165 Patients should be monitored for these disorders beginning several months after resolution of DIHS.
Histologic Findings
The pathologic findings seen in DRESS are relatively nonspecific and do not differ significantly from other medication-induced hypersensitivity reactions (for a detailed explanation of histologic findings in association with specific medications, see Chapter 8). In general, biopsies taken from DRESS show a superficial and deep, perivascular lymphocytic inflammatory infiltrate with abundant dermal eosinophils. Epidermal spongiosis can be present, and scattered apoptotic keratinocytes are seen in a minority of cases (Figure 3-28). In some circumstances, the number of dermal eosinophils can be quite low, despite the presence of peripheral blood eosinophilia. The diagnosis of DRESS is strictly based on specific clinical criteria, because the pathologic findings are relatively nonspecific. The group of drug-induced hypersensitivity reactions in association with anticonvulsants (eg, phenytoin, carbamazepine, etc.) can potentially mimic a T-cell lymphoma.169,170,171,172,173
Differential Diagnosis
Drug reactions may present with nearly every histologic inflammatory reaction pattern in the skin. In disorders characterized by superficial perivascular inflammation such as spongiotic, interface, or psoriasiform patterns, the presence of deep inflammation and eosinophils is often invoked as evidence to include the differential diagnosis of a drug reaction.
CAPSULE SUMMARY
DRUG-INDUCED HYPERSENSITIVITY SYNDROME
DIHS to medications (also known as DRESS) is a spectrum of clinical and laboratory features that includes fever, exanthem, lymphadenopathy, facial edema, eosinophilia, and internal organ involvement. Following viral prodrome-like fever and malaise, a morbilliform eruption occurs in 75% of patients accompanied by edema of the face, especially the periorbital area. The pathologic findings seen in DIHS are relatively nonspecific and do not differ significantly from other medication-induced hypersensitivity reactions.
PITYRIASIS ROSEA
Definition and Epidemiology
PR is a common, self-limited inflammatory dermatosis that is usually seen in children.174 It has a classic distribution and morphology, and evidence suggests a viral etiology. It occurs most commonly between the ages of 10 and 35, with 50% of cases in patients under 20 years of age.16,174 Although less than 10% of cases occur in children under 10 years, it has been reported in those as young as 3 months of age.16 It affects males and females equally.174
Etiology
Clinical Presentation
Classically, PR begins with a herald patch on the trunk: an asymptomatic, pink erythematous scaling patch with slight central depression.174,182,183 The herald patch, seen in 17% to 76% of cases, can grow to several centimeters in size and precedes the rest of the eruption by at least 2 days. The secondary eruption appears 2 to 84 days after the herald patch with crops of thin-to-nonsubstantive, faint salmon-pink, scaling, oval-to-oblong plaques oriented along Langer lines of skin cleavage (Figure 3-29).174,180,183 These give the characteristic “Christmas tree” appearance. Each lesion can have a “peripheral collarette scaling pattern.”183 PR resolves in an average of 6 to 8 weeks, but can last up to 5 months.174,180 Some children (5%-69%) present with a prodrome: sore throat, headache, anorexia, nausea, malaise, fever, or upper respiratory infection symptoms.16,176,180 The eruption may be slightly pruritic.174 There are several noted variants of PR, and an atypical presentation is not uncommon.176 Inverse PR is characterized by predominant facial and extremity over truncal involvement.174 African-American children with PR are more likely to have facial and scalp involvement (30%), an inverse presentation, papules (33%), and postinflammatory pigment alteration (77%).184 Papular and vesicular PR are variants that are more common in children.180 Vesicular PR can have intense pruritus and is diffuse. Other rarer variants include EM like, purpuric/hemorrhagic with oral hemorrhagic ulcers, urticarial, and unilateral PR.174,176,180
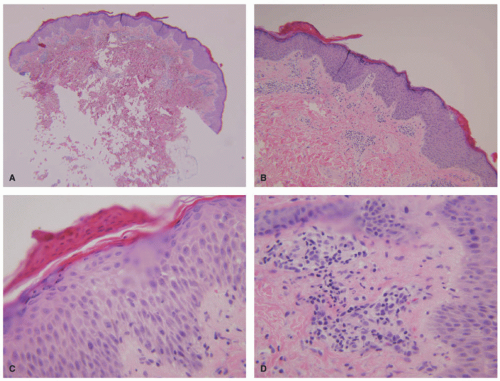
Histologic Findings
There is epidermal spongiosis with occasional mounds of parakeratosis. Prominent lymphocyte exocytosis is typical (Figure 3-30). Within the dermis, there is a perivascular lymphocytic infiltrate with associated extravasation of red blood cells. Scattered single or multiple apoptotic keratinocytes can be seen, particularly in biopsies taken from the “herald patch,” but frank interface dermatitis is uncommon. Atypical presentations of PR may show a slightly more intense dermal inflammatory infiltrate with papillary dermal edema, more exocytosis, and extensive basal vacuolar alteration. Dyskeratotic cells are also seen. A vesicular form of PR, characterized by foci of more extensive spongiosis, is rare, but occurs with a higher frequency in the pediatric age group, particularly in those with darker skin color.184,185,186,187
Differential Diagnosis
PR is typically diagnosed on the basis of clinical findings alone without the need for a biopsy. Classic examples of PR cannot always be differentiated reliably on a histologic basis from other forms of spongiotic dermatoses. If the epidermal changes are focal, the findings in PR might be indistinguishable from the superficial variant of erythema annulare centrifugum (EAC). Because the biopsies taken from EAC may sometimes exhibit multiple foci of parakeratosis with spongiosis, including intraepidermal vesiculation, clinicopathologic correlation is necessary to differentiate the two entities. Lymphocyte exocytosis, scattered apoptotic keratinocytes, and extravasated red blood cells are also features present in pityriasis lichenoides and morbilliform viral exanthems. Pityriasis lichenoides show more prominent interface changes than PR. In pityriasis lichenoides et varioliformis acuta (PLEVA), areas of epidermal necrosis are seen, features never encountered in PR. Morbilliform viral exanthems show a sparse lymphocytic infiltrate with or without extravasated red blood cells, mild vacuolar changes, minimal spongiosis with lymphocyte exocytosis, and no parakeratotic mounds.
CAPSULE SUMMARY
PITYRIASIS ROSEA
This is a common, self-limited inflammatory dermatosis that is usually seen in children. Classically, it begins with a herald patch on the trunk: an asymptomatic, pink-erythematous scaling patch with slight central depression, followed by appearance 2 to 84 days later of crops of thin, faint salmon-pink scaling, oval-to-oblong plaques oriented along Langer lines of skin cleavage. Histologically, there is epidermal spongiosis, with occasional mounds of parakeratosis. Prominent lymphocyte exocytosis is typical. Within the dermis, there is a perivascular lymphocytic infiltrate with associated extravasation of red blood cells.
PSORIASIS
Definition and Epidemiology
Psoriasis is a common, chronic, immune-mediated inflammatory dermatosis.188,189,190 It has a relapsing and remitting
course and may affect the skin, nails, and joints. Approximately 2% to 3% of the global population has psoriasis, and one-third of those cases begin in childhood.189,191 The age-and sex-adjusted annual incidence of pediatric psoriasis is 40.8/100 000, which has more than doubled since 1970.191,192 Although psoriasis is not thought to have a gender predominance, some studies show a female predilection in children.189 The mean age at onset is 8-11 years old.192
course and may affect the skin, nails, and joints. Approximately 2% to 3% of the global population has psoriasis, and one-third of those cases begin in childhood.189,191 The age-and sex-adjusted annual incidence of pediatric psoriasis is 40.8/100 000, which has more than doubled since 1970.191,192 Although psoriasis is not thought to have a gender predominance, some studies show a female predilection in children.189 The mean age at onset is 8-11 years old.192
Etiology
The risk for psoriasis is a complicated interplay between genetic and environmental factors. About 30% of children have a first-degree family member with psoriasis, and in children with onset before 16 years, this can be up to 71%.188,189,193 Many genes have been associated with psoriasis, including the following: PSOR1, CARD14/PSOR2, IL 12-B9, IL-13, IL-23R, HLABw6, PSORS6, STAT2/IL-23A, TNFAIP3, and TNIP1.16,188 CARD14 mutations have specifically been associated with familial psoriasis vulgaris and familial generalized pustular psoriasis.194 Many cases of pustular psoriasis have IL-36 receptor antagonist mutations.189
The innate and adaptive immune systems mediate psoriatic inflammation, but Th1 and Th17 cells predominate.16,195 Proinflammatory cytokines (TNF-α, IFN-γ, IL-1β, and IL-6) activate dendritic cells to express IL-12 and IL-23. These subsequently trigger the dominant Th1 and Th17 cell responses. Th1 and Th17 cytokines (TNF-α, IFN-γ, IL-17, IL-22, IL-23) then stimulate keratinocytes and perpetuate the inflammation and immune activation of psoriasis.16 A recent study examining the T-cell and cytokine milieu in pediatric psoriatic plaques revealed the increased prevalence of IL-22-secreting T cells more so than IL-17-secreting T cells, which contrasts with adult psoriasis.195 Additionally, disease severity has been shown to correlate with increased levels of circulating Th-17 and T regulatory cells.196
Clinical Presentation
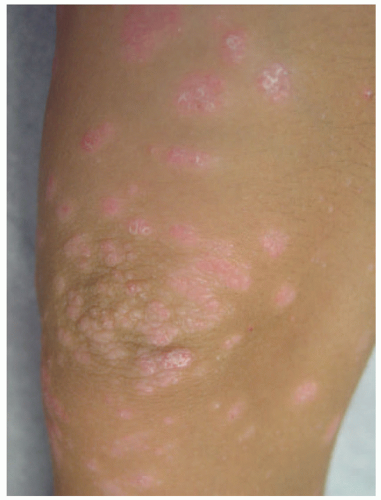
Although pediatric psoriasis can present with all of the same variations as adults, the morphology, distribution, and symptoms of pediatric psoriasis can vary.189 Children are affected by significant psychosocial impact and adverse effects on the quality of life.189 Additionally, pediatric psoriasis is more likely to be preceded by stress, trauma, and pharyngitis, and it exhibits the koebnerization phenomenon (development of psoriasis at sites of trauma such as scratching).190,193 The most common form of pediatric psoriasis is chronic plaque type (73.7%), with salmon-pink, sharply demarcated, silver-white scaling, flat-topped papules and plaques (Figure 3-31).192 They are usually symmetrically distributed on the extremities or scalp at presentation (46.8%-57.3%).189,192,193 Plaques in childhood tend to be smaller and thinner than in adults.189 Although the scalp (46.8%-57.3%) and extremities are the most common sites of presentation, flexural surfaces are involved more frequently in children than in adults, with potential for maceration.189,192,193 Facial involvement, particularly periorbital, is seen more frequently in children, even in the absence of other areas of involvement.16,189
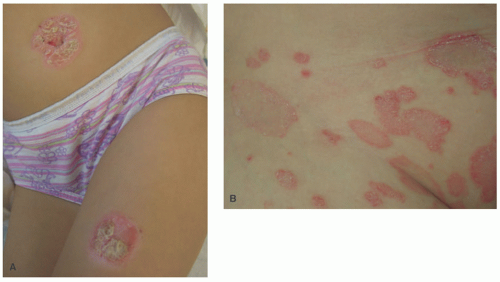
At 44%, guttate psoriasis is the second most common presentation in children. It presents with smaller, droplet-like papules and plaques and has a more acute onset than plaque
psoriasis (Figure 3-32). It is an often-triggered β-hemolytic Streptococcal infection of the pharynx or perineum.16,189 If guttate psoriasis progresses to chronic plaque type (40%), it can herald a more severe disease course.16,189,190 Young children and infants may present with recalcitrant diaper dermatitis that has sharp demarcation, maceration, and involvement of the inguinal folds.189 Nail changes are seen in 40% of children and are more common in boys—these changes include pitting, oil spots, onycholysis, subungual hyperkeratosis, onychodystrophy, and splinter hemorrhage.189,190 Nail changes may not correlate with arthritis.190
psoriasis (Figure 3-32). It is an often-triggered β-hemolytic Streptococcal infection of the pharynx or perineum.16,189 If guttate psoriasis progresses to chronic plaque type (40%), it can herald a more severe disease course.16,189,190 Young children and infants may present with recalcitrant diaper dermatitis that has sharp demarcation, maceration, and involvement of the inguinal folds.189 Nail changes are seen in 40% of children and are more common in boys—these changes include pitting, oil spots, onycholysis, subungual hyperkeratosis, onychodystrophy, and splinter hemorrhage.189,190 Nail changes may not correlate with arthritis.190
Pustular psoriasis is composed of superficial sterile pustules, is quite rare (1%-5.4% of childhood psoriasis), and usually presents before 5 years of age (Figure 3-33).189 Although pustular psoriasis is more common in adult patients, the von Zumbusch (febrile, arthralgias) and annular variants are more common in children.189 Other infrequently encountered forms of pediatric psoriasis include the following: erythrodermic (1.4%), inverse (affecting intertriginous sites), palmoplantar, isolated facial, Blaschkolinear, glossitis, and congenital.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree