Peripheral nerve sheath tumors (PNSTs) are represented by a group of soft tissue tumors that are composed of one or more cell types such as Schwann cells in the case of schwannoma, perineurial cells in the perineurioma, or a mixture of neural elements including neurites, Schwann cells, and perineurial cells in the neurofibroma (NF) or hybrid PNSTs.
2 Granular cell tumor (GCT) is considered a Schwannian neoplasm in almost all cases except for the congenital epulis arising from the gingival soft tissues of an infant. Other neural lesions presenting in the skin and underlying soft tissues in children include the neural lipofibromatous hamartoma (NFH), ectopic meningothelial hamartoma, and neuromas of various types that are either developmental anomalies or a disordered reactive-regenerative process.
NEUROFIBROMA
NF was the most common of the PNSTs presenting in the skin and subcutis of children and represented 75% of cases in our series (
Table 24-1). Unlike most cutaneous NFs in adults that are typically sporadic and solitary, the clinical context in children is quite the contrary, with multiple tumors in superficial and deep soft tissue sites; these patients all have NF type 1 (NF1).
3 In general, the cutaneous NFs in NF1 are just one feature in which café au lait spots and axillary freckles are also seen.
4,
5 Less frequently, NFs are seen in NF type 2 (NF2), but with many fewer lesions, and are often more subtle in their clinical presentation.
6,
7,
8 Schwannomas arising in the skin and soft tissues are seen in the setting of NF2 or schwannomatosis.
9,
10 The salient features of NF1,
NF2, and schwannomatosis are summarized in
Table 24-2. Virtually all of those with NF1 develop NFs, and 50% of children with NF1 have at least one, if not more, NF by the age of 10 years. In children under 10 years of age, NFs often have exclusive plexiform features.
NF1 is caused by mutation in the
NF1 gene that encodes the tumor suppressor protein neurofibromin.
11 Specifically, NFs are characterized by biallelic inactivation of the
NF1 gene in a subpopulation of Schwann cells within the lesion.
11 NF2 is caused by a loss of function of the
NF2 gene that encodes the cell membrane protein and tumor suppressor protein merlin.
12 Schwannomatosis is caused by a mutation in the
SMARCB1 gene.
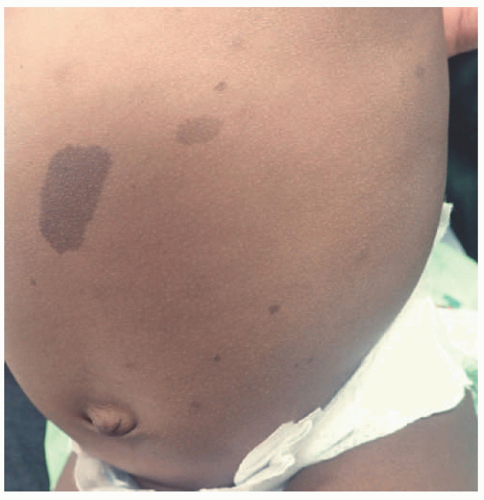
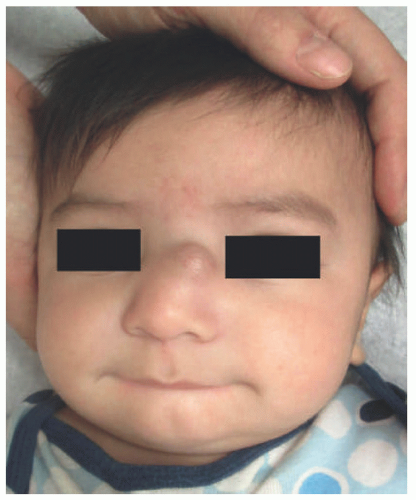
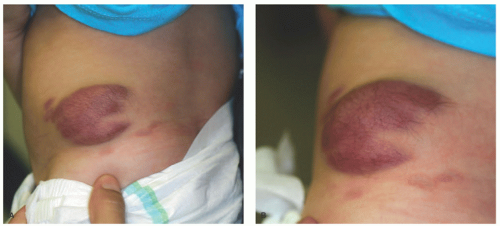
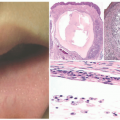
13NFs present as protuberant to pedunculated, flesh-colored to pinkish papules or nodules (
Figure 24-1). They are soft on palpation and can demonstrate the “buttonhole sign” in which the lesion can be invaginated into the subcutis with pressure and reappear after release of pressure.
14In the setting of NF1, NFs occur in young adults and continue to develop over time.
4 Patients can have tens, hundreds, or even thousands of cutaneous NFs.
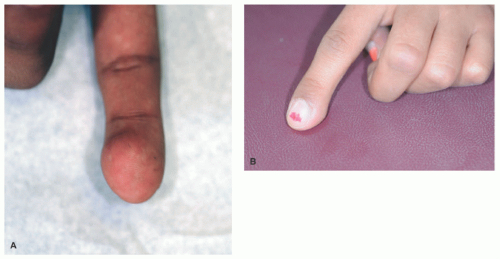
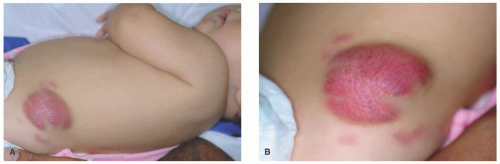
4 Cutaneous NFs occur in 99% of patients with NF1. Two or more NFs or one plexiform NF (
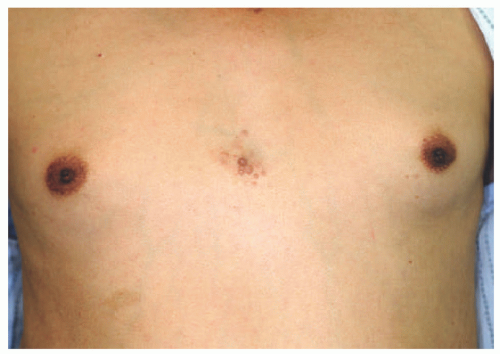
Figure 24-2) is included in the diagnostic criteria for NF1. Other diagnostic criteria, of which the patient needs two to confirm the diagnosis, are six café au lait spots greater than 5 mm in diameter in prepubertal children and greater than 15 mm in postpubertal children (
Figure 24-3); freckling in the axillary or inguinal region; optic glioma; two or more iris hamartomas (Lisch nodules); a distinctive osseous lesion (sphenoid dysplasia or thinning of long bones); and a first-degree relative with NF1.
15 Approximately onethird of NF1 patients develop plexiform NFs.
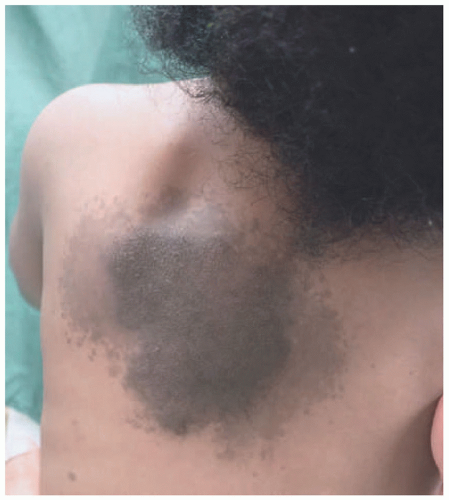
11,
16Plexiform NFs are clinically distinct from cutaneous NFs as they are generally present at birth (congenital). They are associated with overlying hyperpigmentation and/or hypertrichosis and are often described as “bag of worms” on palpation.
16 Lesions are diffuse and can grow into large, disfiguring masses.
17In the setting of NF2, NFs are significantly less common. NF2 is characterized by the development of acoustic nerve schwannomas, meningiomas, ependymomas, and ocular abnormalities. Studies have reported 70% of patients with NF2 have skin tumors, although only 10% have greater than 10 cutaneous lesions.
8 Cutaneous tumors in NF2 are most commonly schwannomas, although NFs do occur. Schwannomatosis has significant clinical overlap with NF2.
13Pathologically, the sporadic, solitary NF is characterized by a circumscribed dermal nodule, typically measuring 1 cm or less and with a differential diagnosis that includes a dermal melanocytic nevus. A pale-staining, uniform spindle cell proliferation has an eosinophilic background, with only a hint of stromal mucin. Scattered mast cells are present
within the stroma. Nuclear pleomorphism and sclerosis are two uncommon features.
18,
19 It is uncommon to encounter a solitary NF in an individual less than 20 years of age.
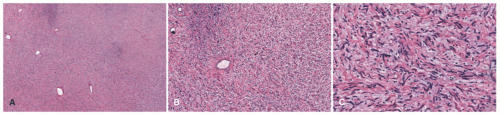
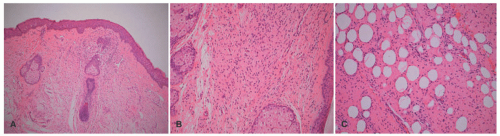
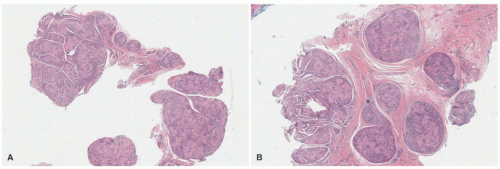
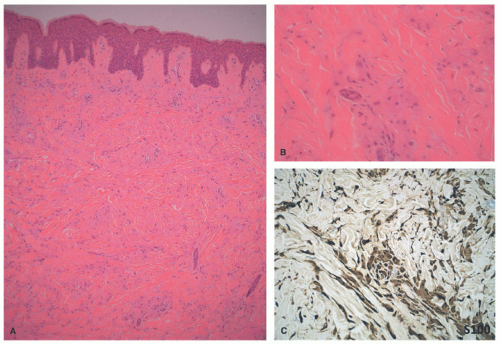
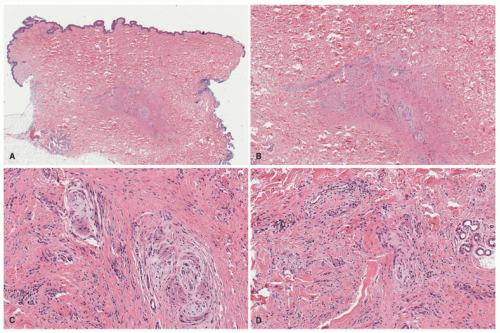
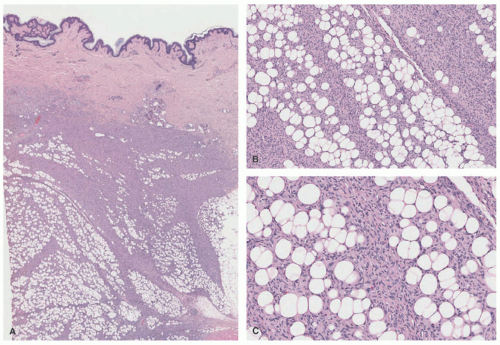
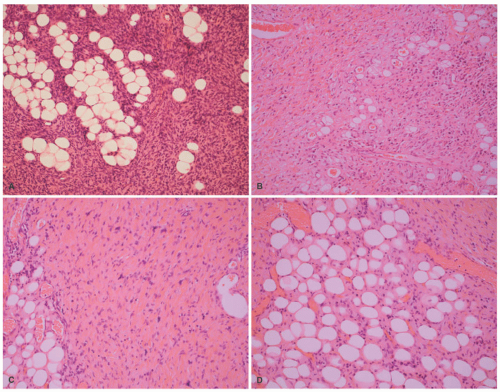
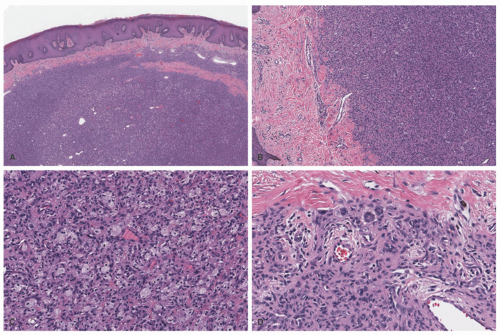
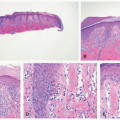
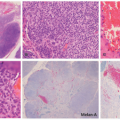
A contrasting histologic pattern of dermal and subcutaneous involvement is present in the NF1-associated NF where the tumor has a diffuse and/or plexiform appearance with growth through the dermis and replacement by bland spindle cells in a pale-staining eosinophilic stroma (
Figure 24-4A-C). Adnexal structures in the dermis are surrounded, but not displaced, by the proliferation. The plexiform profiles may be present as one or more bundles of spindle cells within the background of diffuse NF or as separate distinct profiles in the dermis and/or subcutis (
Figure 24-4A-C). Within the dermis, the short to fusiform spindle cells have elongated, bland nuclei; nuclear enlargement with hyperchromatism is uncommon in the dermalbased NFs, but may be seen in the deep soft tissue NFs in older children and young adults.
20 When the subcutis is involved as it often is, the growth is usually contiguous with a dermal component and the fat is overgrown in a manner similar to dermatofibrosarcoma protuberans (DFSP) and some of the fibrous tumors of childhood (
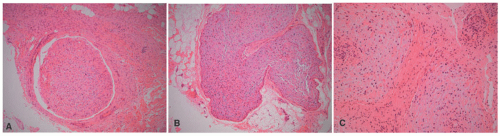
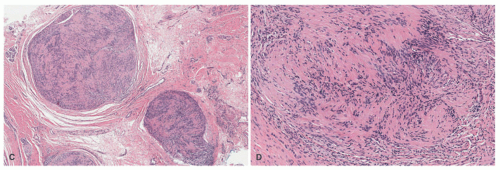
Figure 24-4A-C). The plexiform pattern of NF is characterized by multiple circumscribed bundles of pale-staining spindle cells in a myxoid background (
Figure 24-5A-C). The contours of the plexiform bundles are sharply demarcated as distinct profiles in the deep dermis and subcutis. Nuclear enlargement and atypia are uncommon in the more superficial plexiform NFs but, if present, should not be viewed as indicative of sarcomatous transformation (
Figures 24-5C and
24-6).
Other features found in NFs in the setting of NF1 include floret-type giant cells with similarities to those seen in the pleomorphic lipoma (rare in children) and giant cell fibroblastoma (GCF; typically presenting in children).
21 Melanin pigmentation is yet another uncommon histologic finding.
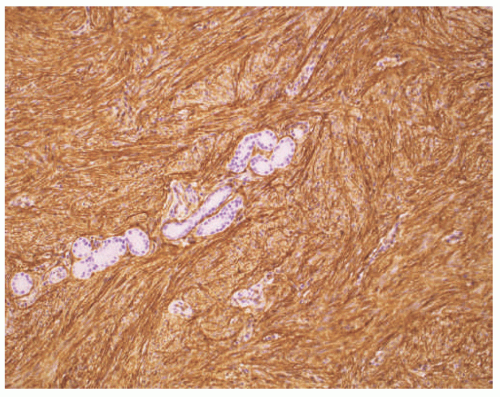
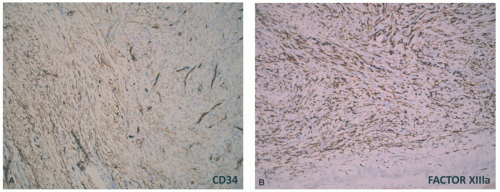
22The differential diagnosis of NF in the skin or subcutis includes the following in the presence of the diffuse, infiltrating pattern: infantile subcutaneous fibromatosis (ISF; lipofibromatosis), fibrous hamartoma of infancy (FHI), DFSP (including the pigmented Bednar tumor), cellular neurothekeoma (CNTK), and congenital melanocytic nevus with neurotization. Various immunohistochemical approaches can be applied with a combination of smooth muscle actin (mature fibrous areas of ISF and FHI), S-100 protein (NF and neurotized nevus), and CD34 (DFSP and NF, diffuse staining and immature nodules in FHI), collagen type IV (NF), and Melan-A (neurotized nevus and melanocytes in NF).
23 An additional challenge in the diagnosis
of diffuse NF is the recently reported neural ISF-like tumor because it expresses CD34 and S-100 protein and has a recurrent
NTRK1 fusion.
24
SCHWANNOMA
Schwannoma is a purely Schwann cell neoplasm that occurs sporadically as a solitary mass in any number of anatomic sites or as multiple tumors in NF2, schwannomatosis, and Carney complex.
25 A review of the files of one of us (LPD) revealed that only 10% of schwannomas presented in the first two decades of life
1; a schwannoma in an individual less than 20 years old is likely to have NF2 or schwannomatosis.
9,
26 Cutaneous manifestations in NF2 occur less commonly in children with NF2 than with NF1.
27 Cutaneous schwannomas or cortical and posterior subcapsular or capsular cataracts are more common as initial presentations in prepubertal children with NF2 in contrast to adolescents and adults whose clinical presentation is more often related to the bilateral VIII nerve schwannomas.
28,
29 However, congenital-infantile NF2 may present with bilateral acoustic neuromas or schwannomas with or without cutaneous schwannomas.
30 It is estimated that 70% of those with NF2 develop one or more skin tumors, generally less than 10.
8
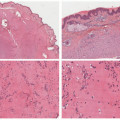
Schwannoma presents as one or more (uncommon) soft to firm nodular mass(es) measuring 1 to 2 cm in the dermis or subcutis. Approximately one-third of tumors are accompanied by pain and tenderness, unlike most NFs, and there is a predilection for the extremities where the tumor can measure in excess of 5 cm.
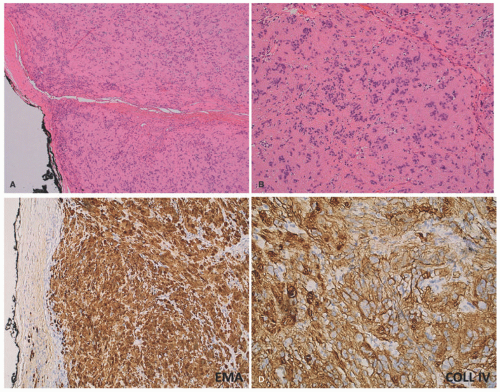
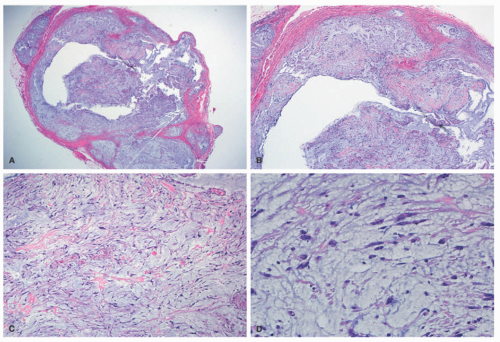
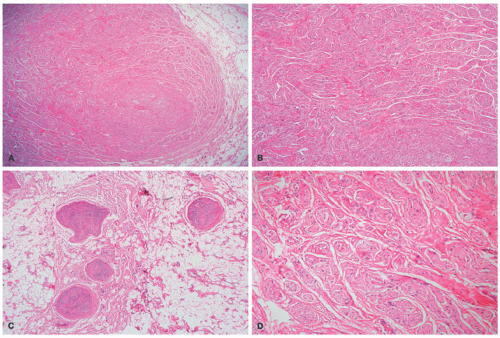
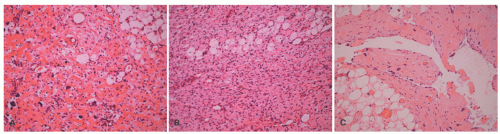
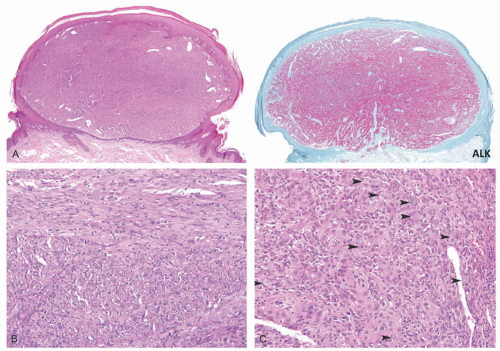
16Pathologically, the schwannoma is a well-circumscribed, encapsulated spindle cell neoplasm to be differentiated from the myriad of other similarly appearing related and unrelated tumors (
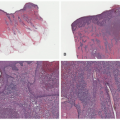
Figure 24-7). In those schwannomas whose capsule is not apparent, the cellular variant with or without mitotic activity, nuclear enlargement, and hyperchromatism or one of the several less common variants including the plexiform (
Figure 24-8), myxoid-reticular (
Figure 24-9), and glandular subtypes can be a histopathologic challenge.
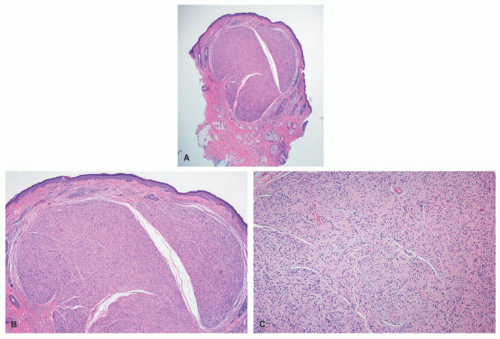
When a schwannoma presents in a child, it is often in the setting of NF2 and may present in the subcutis and less often in the dermis alone, with the exception of the plexiform schwannoma (PS) with its predilection for the skin and underlying soft tissues.
31 This tumor is seen in children and is associated with NF2 and schwannomatosis.
32,
33,
34 PS is often cellular and displays some nuclear pleomorphism and mitotic activity (5 or less per high-power field [HPF]); these features in the context of PS are not indicative of malignancy because this tumor behaves in an entirely benign fashion (
Figure 24-8). Unlike the conventional schwannoma, a capsule is not present in most PSs. The encapsulated nature of PS may be inapparent.
The small, superficial schwannoma in a young individual is often moderately cellular and is composed of uniform fusiform cells often, but not always, without Verocay bodies, hyalinized vessels, macrophages, and myxoid or Antoni type B stroma.
In addition to the cellularity of the PS, the cellular variant of schwannoma is a problematic tumor because it must be differentiated from the malignant PNST (MPNST), an extremely rare tumor in the skin or subcutis of a child even in the presence of NF1.
35 Retained nuclear expression for SOX-10 and H3K27me, and diffuse expression of S-100 protein, collagen type IV, and calretinin all favor a schwannoma including the PS rather than MPNST.
20,
36,
37,
38Melanotic schwannoma (MS) is a rare variant that typically presents in the paraspinal location, but not to the exclusion of other sites including the skin.
39 An MS should strongly suggest the likelihood of Carney complex.
40,
41 The cause of Carney complex in most patients is a mutation in the
PRKAR1A gene, a tumor suppressor gene.
42,
43 PRKAR1A expression is lost in 35% to 40% of sporadic and Carney complex-associated psammomatous MSs.
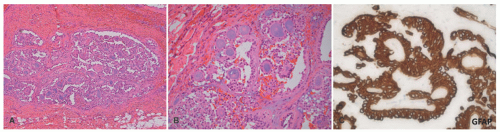
44Psammomatous MSs most commonly present on the posterior spinal nerve roots, alimentary tract, bone, and rarely skin.
40,
45 Tumors can range between 1.4 and 7.0 cm, with an average of 3.2 cm. As lesions are most commonly deep, patients generally present with pain, muscle weakness, dysesthesias, and numbness.
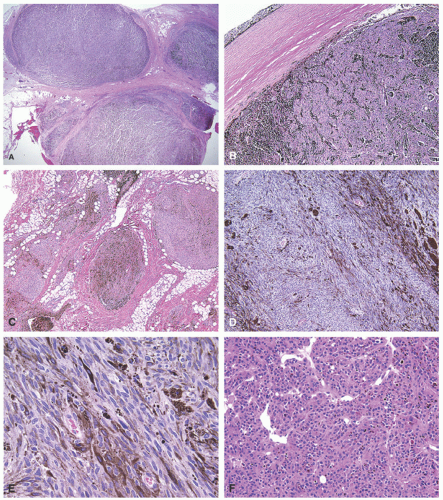
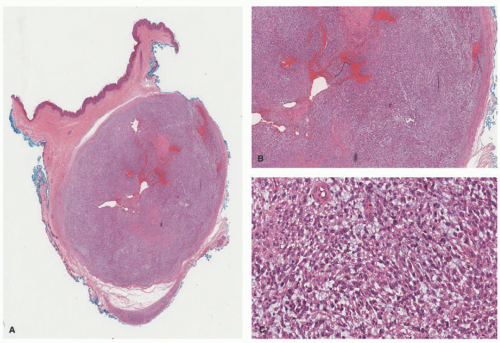
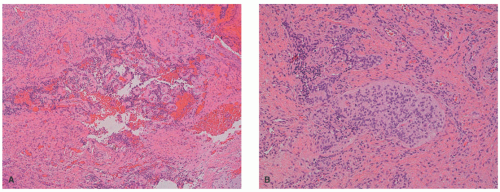
44 The behavior of melanotic psammomatous schwannomas is unpredictable, with metastases reported in 13% to 42% of patients (
Figure 24-10).
44This heavily pigmented spindled to epithelioid neoplasm can have substantial nuclear atypia and may have psammomatous calcifications. In the absence of the latter findings, metastatic melanoma is an
unavoidable consideration, but in the context of a child, it is very unlikely. Schwannoma may have a predominant epithelioid cellular composition or may resemble a neuroblastoma.
46,
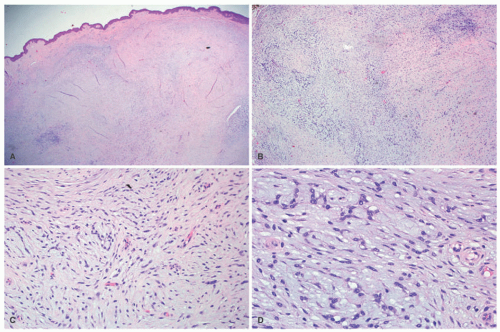
47Epithelioid schwannoma (
Figure 24-11) presents in the skin and/or subcutis, and only a minority of cases occur in children, usually in older children and adolescents.
46,
48 The challenge is differentiating the ES from the menu of other epithelioid neoplasms ranging from epithelioid sarcoma (ES) to hemangioendothelioma. A single cell pattern with the features of a classic schwannoma focally in those tumors with trabecular to nodular profiles of epithelioid cells in a myxohyaline, myxoid, or dense collagenous stroma is the spectrum of microscopic features.
46,
48,
49,
50,
51,
52,
53,
54 In addition to the characteristic immunophenotype of the schwannoma, a CD34
+ population of spindle cells may also be present.
Schwannomatosis is the “third” neurofibromatosis that is characterized by multiple, nonvestibular schwannomas in contrast to NF2.
1 Germline mutations in either
SMARCB1 or
LZTR1 tumor suppressor genes are found in almost 90% of familial cases (representing 15%-25% of all cases) and in 40% of sporadic cases (representing 75%-85% of all cases). Somatic mutations in both
NF2 alleles have been detected in the schwannomas.
55 In the presence of a germline mutation in
SMARCB1, the risk of malignant rhabdoid tumor (MRT) may also exist.
56,
57
PERINEUROMA
Perineuroma (PN) is the third type of PNST that is composed of spindle cells with the immunophenotype of pia-arachnoidmeningeal cells with expression of vimentin and epithelial membrane antigen.
58 This tumor is well circumscribed, arises in the subcutis and dermis, has an intraneural localization in some cases, and has a predilection for the extremities. PN occurs more commonly in adults, but is seen in children, usually over 10 years of age.
59 Delicate fusiform cells resembling Schwann cells are arranged in fascicles and/or whorls of loosely arranged
spindle cells. A myxoid background or plexiform growth may be prominent in some cases (
Figure 24-12).
2 Dense sclerosis is a feature in some cases. Epithelial membrane antigen reactivity is essential because the PN may also express CD34, smooth muscle actin, and S-100 protein (less than 10%).
58
Hybrid PNST is a neoplasm that has challenged the concept of the single lineage nerve sheath tumor with the combination of patterns and the overlapping immunophenotype of the three major PNSTs.
60,
61,
62 These tumors have a preference for the subcutis and dermis of adults more commonly than older children. The PN and schwannoma hybrid is one of the more frequent combinations. Other neural tumors in the skin and superficial soft tissues include both neoplasms, reactive proliferations, and possibly a hamartoma or localized overgrowth process.
GRANULAR CELL TUMOR
GCT is a neoplasm with a broad distribution of primary sites, with the skin as the most common location in children where 60% of lesions are found with a predilection for the head and neck region.
1 GCTs may be congenital, known by a variety of different names including congenital GCT, congenital epulis, congenital myoblastoma, or Neumann tumor.
63 Their incidence is suggested to be 6 per million children per year.
63 GCTs occur in adults between 20 and 60 years of age.
64 Females are affected more commonly than males in both congenital and adult lesions.
63,
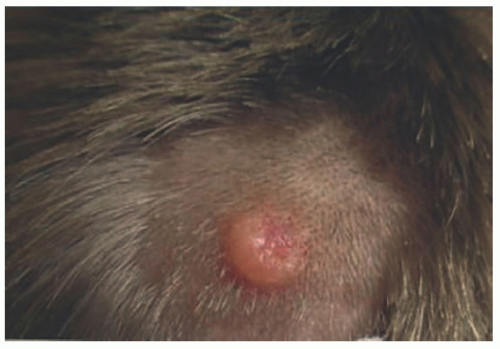
64GCTs arise most commonly as solitary asymptomatic, pale, or yellowish smooth nodules, rarely larger than 3 cm (
Figure 24-13).
64,
65 There is a predilection for the head and neck region. The congenital forms typically present on the gum pads.
63 In adults there is a predilection for the oral cavity, especially the tongue, and, when present in the skin, the upper extremities and upper torso.
64,
66,
67Because of almost universal S-100 protein immunopositivity, GCT is considered a Schwann cell-derived tumor except in the case of the congenital granular cell epulis arising in the oral-maxillofacial region.
68,
69 Multifocal congenital GCTs and familial GCTs are other reported occurrences.
70,
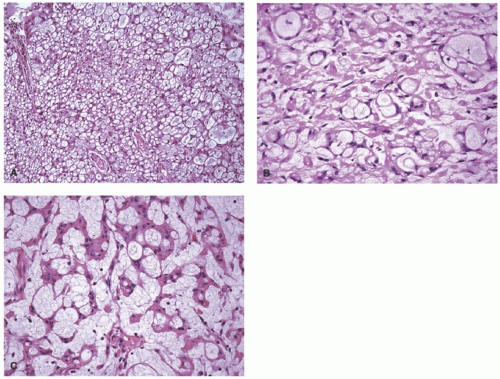
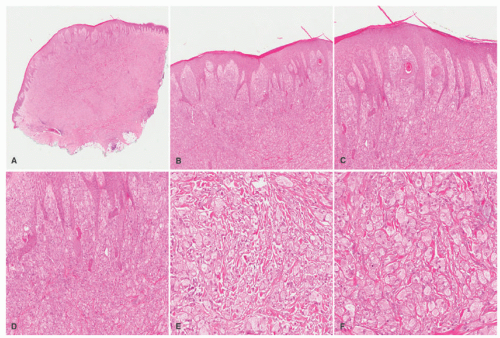
71 Both circumscribed and infiltrating growth patterns of granular cells with abundant eosinophilic granular cytoplasm with somewhat larger eosinophilic bodies are present in the biopsy or excision. In the GCT with infiltration of
the dermis, the small groups of granular cells blend into the background of dermal collagen (
Figure 24-14). The nuclei of the tumor cells are rounded to ovoid with fine to coarse chromatin, but infrequently the nuclei may appear larger and hyperchromatic with an inconspicuous nucleolus. The granules are lysosomes that account for CD68 immunopositivity, in addition to characteristic S-100 protein reactivity (
Figure 24-15). The congenital granular tumor or epulis has a densely cellular appearance as it forms a nodule or mass. With the rare exception, these tumors are nonreactive for S-100 protein.
NEUROMA
Neuroma is a generic designation for several lesions of neoplastic, hamartomatous, and traumatic nature.
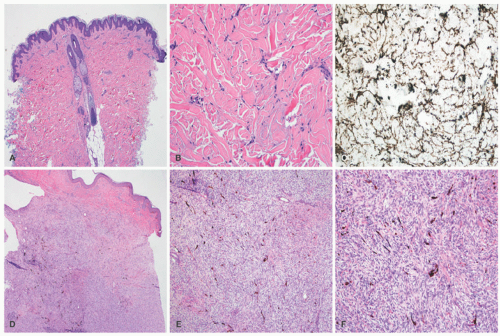
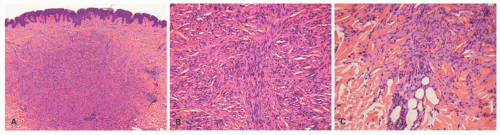
Palisaded encapsulated neuroma (PEN) is a likely neoplasm presenting in the dermis or submucosa of the oral cavity, but it occurs in other sites such as the palms and soles in children.
77,
78,
79 Only 10% of cases occur in children. It has been suggested that a histogenetic relationship may exist between PEN and the mucocutaneous neuromas of multiple endocrine neoplasia 2B (MEN2B).
80 A solitary flesh-colored papule or nodules, generally measuring less than 2 cm, is the clinical presentation. A uniform, spindle cell population arranged in fascicles is surrounded by a delicate fibrous capsule (
Figure 24-17). A contiguous peripheral nerve is present in some cases. As in the case of a schwannoma, the spindle cells are diffusely positive for S-100 protein, but unlike the latter tumor, neurofilament protein and epithelial membrane antigen are positive.
77,
80
Mucocutaneous neuroma of MEN2B is composed of smaller and less expansile circumscribed individual bundles of Schwann cells than the PEN. When these lesions initially develop in a young child on the tongue, palate, pharynx, and lip, they may be the first manifestation of MEN2B.
81,
82,
83 The multiple neural bundles are separated by a stromal background. The site of the neural bundles is somewhat circumscribed, but in the lip with progressive enlargement, the entire submucosa is occupied by these formations. Like the PEN and
schwannoma, the spindle cells are uniformly S-100 protein positive and a peripheral capsule, if present, is at least focally positive for epithelial membrane antigen.
Traumatic neuroma occurs in the axilla of infants after neonatal brachial plexus palsy or the hand or foot in an older child after laceration of a digital nerve (
Figure 24-18). A background of fibrosis (scar) contains numerous small peripheral nerve twigs in a more or less circumscribed focus. A mixture of axons, Schwann cells, and perineurial cells is the composition of the neuroma (
Figure 24-19). A variation
of the traumatic neuroma occurs after ligature amputation of a rudimentary digit in polydactyly, whereas this complication is avoided with surgical excision.
84,
85
NEUROGLIAL HETEROTOPIA
Neuroglial heterotopia constitutes a rare category of anomalies presenting as a mass, most commonly in children during the first decade of life. Not entirely surprising, these
masses have a predilection for the head and neck region, but not exclusively so.
90 Those lesions presenting in the skin and subcutis often come to the attention of the pediatrician and dermatologist, whereas a nasal or oronasopharyngeal lesion is more likely seen by the pediatrician. All of these lesions have a common histogenesis of a presumed extracranial sequestration of neuroectodermal tissue.
Nasal glioma (NG), a misnomer in terms of its “glioma” designation, is seen in 1:20 000 to 40 000 live births as an intranasal mass at or before 2 years of age (
Figure 24-21).
91 Unlike the encephalocele, NG lacks an intracranial component and is regarded as a so-called sequestered encephalocele. A firm mass is composed of dense fibrous stroma with embedded variably sized islands of more or less mature neuroglial tissue, which may contain an isolated neuron.
Other sites of heterotopic neuroglia are found in and around the oral cavity, oropharynx, and various other soft tissue sites in the head and neck region. Scalp lesions are potentially problematic when there is an underlying defect through the cranium.
92 An isolated nodule of neuroglial tissue with or without meningothelial elements or choroid plexus is the spectrum of microscopic findings.
93,
94,
95,
96,
97,
98Myxopapillary ependymoma (MXPE) and rests are known to present as an extramedullary cutaneous and/or subcutaneous nodule or mass in the sacrococcygeal region of children.
99,
100,
101 This anatomic site is the embryonic consequential location of the primitive streak so that it is not entirely surprising that heterotopic nephrogenic rests, teratoma, and other anomalies present as a mass lesion, as an anal skin tag, or as an incidental microscopic finding (
Figure 24-22).
102,
103,
104,
105,
106 These embryonic rests may be identified in a pilonidal sinus or skin tag.
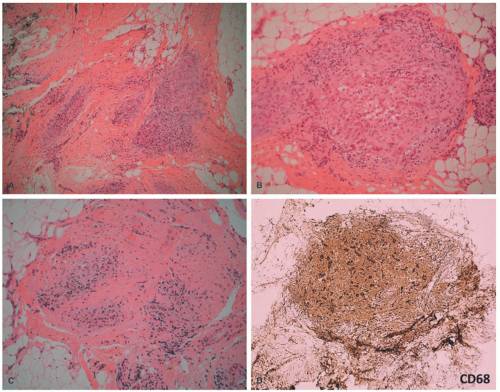
107 The focus of the MXPE usually measures 1 cm or less, is well circumscribed, and is located in the dermis or more often in the subcutis. Papillary profiles with myxohyaline stroma supporting the papillae are the microscopic findings (
Figure 24-23A-C).
Rudimentary meningocele (meningothelial hamartoma) is a lesion that may present with alopecia, tufted hairs, or soft mass of the scalp (20% or more of cases) in a young child.
108 Other sites include the spine and forehead. The age at clinical presentation ranges from the newborn period to later childhood.
109,
110,
111 The dermis and/or
subcutis is the location of a circumscribed focus of collapsed, irregular spaces with an inconspicuous lining and/or a small nest(s) of compact, bland-appearing round cells, calcifications (psammomatous in some cases), and giant cells. A solid glial nodule may be present in some cases to provide a clue to neurogenic process of some kind. The giant cells and a pseudoinfiltrative pattern may suggest GCF or a vascular anomaly.
112,
113 The spaces are immunoreactive for vimentin and epithelial membrane antigen and nonreactive for CD31.