Key Words
dermatome, donor site, epithelialization, graft, harvest, mesh, thickness, wound
Synopsis
Although the field of plastic surgery has advanced significantly in its ability to provide “like for like” coverage of wounds with various flaps, skin grafting still provides a facile, effective, and expeditious method of wound coverage. In the appropriate patient and with the proper technique, complications are minimal. The greatest limitations to skin grafting include donor site pain and appearance, need for specialized equipment, and variable cosmetic appearance at the reconstructed site.
Clinical Issues
Presentation
Skin is the largest organ in the human body. It provides insulation, sensation, and a barrier for the body from the external environment. Although the body has an inherent ability to heal open wounds, wounds to the deep dermis lose regenerative potential because of the lack of skin appendages present. This is because hair follicles contain pluripotent stem cells that are activated when a wound is created. As a result, deep wounds require longer time to closure, are at a higher risk of infection, and have a poorer cosmetic, and potentially functional, outcome.
Etiology
The importance of attaining more expedient wound closure depends on the etiology of the wound (i.e., cancer resection, trauma, vascular compromise), the complexity, patient comorbidities that may preclude wound healing (i.e., diabetes, cancer, radiation or chemotherapy, malnutrition), and resultant complications if wound closure is not attained quickly (i.e., presence of an underlying implant or prosthesis, nutritional loss from large wounds).
Associated Conditions
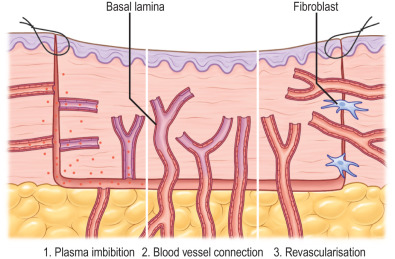
Skin grafts are avascular patches of skin that are transferred to an open wound. Skin grafting allows for coverage of large open wounds quickly and with minimal morbidity. Grafting takes advantage of the body’s ability to regenerate partial-thickness skin defects by borrowing from normal skin that can regenerate quickly and by giving to large wounds that otherwise would take significant time to heal. This procedure is possible because the body can quickly revascularize the graft over the course of 3 to 7 days before necrosis occurs. Revascularization occurs via a graduated process: The graft is initially nourished by oxygen diffusion from the wound bed in the first 24 hours, with revascularization occurring in the subsequent 24 to 48 hours ( Fig. 2.1.1 ). Thus any clean wound with a well-vascularized bed can accept a skin graft. Wounds with denuded bone, tendon, or exposed implants will not accept a graft.
Management
Key Area of Concern
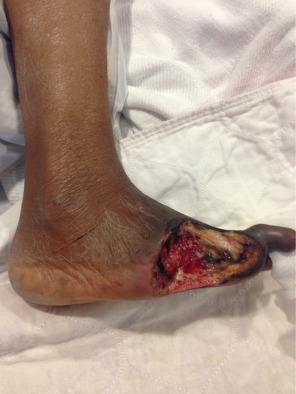
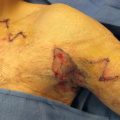
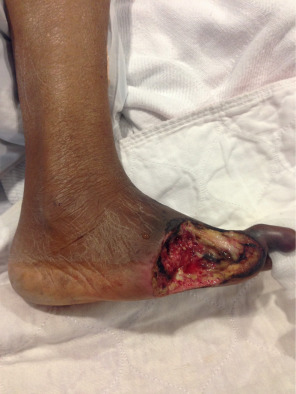
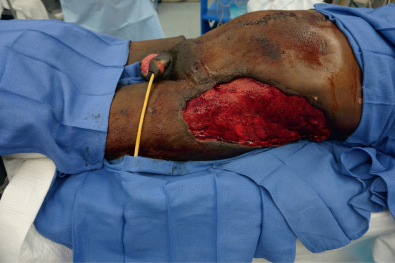
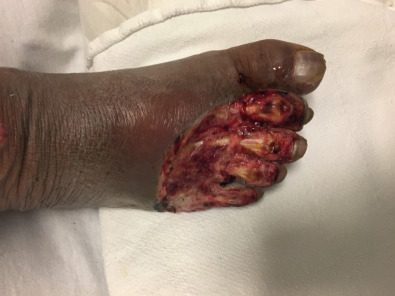
Before reconstruction with a skin graft, the surgeon must ensure that the wound is appropriate for skin grafting and is ready to accept a graft. Wounds with poorly perfused beds and high bacterial load will not accept a skin graft. The skin edges surrounding the wound should be “refreshed” by removing any intervening tissue that may be partially epithelialized. Any necrotic tissue or fibrinous exudate at the base of the wound should be debrided, because these physically block new vessels from growing into the graft ( Fig. 2.1.2 ). In addition, the wound should be copiously irrigated to decrease the bacterial load. If the wound appears clean but does not have a granulating bed, granulation tissue can be induced with negative pressure therapy or simple dressing changes ( Fig. 2.1.3 ). While healthy paratenon and periosteum generally have adequate perfusion to sustain a graft, denuded tendon or bone does not. Granulation tissue can be induced over small areas of tendon or bone with negative pressure therapy or dermal substitutes ( Fig. 2.1.4 ). Additionally, non-viable or desiccated tendon can be debrided to create a proper wound bed. Calvarial bone can be burred down to induce punctate bleeding and can ultimately be prepared to accept a skin graft.
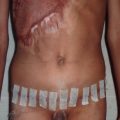
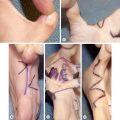
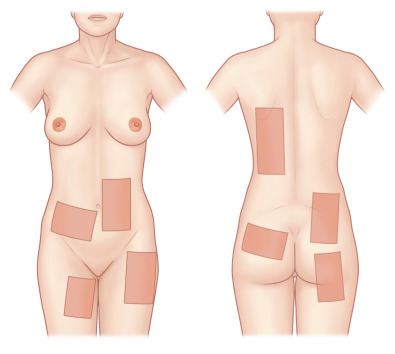
Subsequently, the surgeon must determine what type of graft will be utilized and what the donor site will be. Skin grafts serve a functional purpose of wound coverage as well as an aesthetic purpose in restoring anatomical form to a region. Functionally, the surgeon must determine the size of the area to be covered and the effect of contracture on the reconstructed area. The options for skin graft types are split-thickness and full-thickness grafts. Split-thickness grafts include the epidermis and a thin layer of dermis. Split-thickness grafts are less resistant to long-term wound contracture, but can be harvested in large amounts because the donor sites retain skin appendages and can re-epithelialize on their own. The most common locations for split-thickness skin graft harvest are the lateral thighs and trunk ( Fig. 2.1.5 ). Full-thickness grafts, in contrast, have less long-term wound contracture but are limited in size. Because the entire thickness of dermis is harvested with a full-thickness graft, all skin appendages are removed, precluding the donor site from healing on its own. Full-thickness graft donor sites should be small enough to be closed primarily. Thus full-thickness grafts are generally used in esthetically demanding areas, such as the face. Areas in which wound contracture would result in poor functional outcome may need full-thickness grafts (i.e., digits) or “thick” split-thickness grafts (i.e., joints). Considering that the dermis provides strength and stability to the skin, the thinner the graft, the less stable and more prone to breakdown it will be. Importantly, thicker skin grafts have a longer distance for nutrient diffusion and thus should not be used in wound beds with tenuous blood supply.
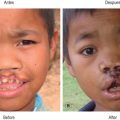
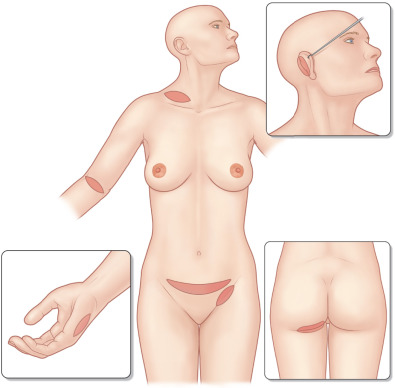
Esthetically, the surgeon must try to match the color and texture of the skin in the region, and again must take into consideration the effects of wound contraction. Skin from the same area, contralateral side, or a similar area will generally give the best color and texture match. For wounds of the face, full-thickness supraclavicular, pre- or posterior auricular, or upper eyelid grafts are good options ( Fig. 2.1.6 ).
Management
Key Area of Concern
Before reconstruction with a skin graft, the surgeon must ensure that the wound is appropriate for skin grafting and is ready to accept a graft. Wounds with poorly perfused beds and high bacterial load will not accept a skin graft. The skin edges surrounding the wound should be “refreshed” by removing any intervening tissue that may be partially epithelialized. Any necrotic tissue or fibrinous exudate at the base of the wound should be debrided, because these physically block new vessels from growing into the graft ( Fig. 2.1.2 ). In addition, the wound should be copiously irrigated to decrease the bacterial load. If the wound appears clean but does not have a granulating bed, granulation tissue can be induced with negative pressure therapy or simple dressing changes ( Fig. 2.1.3 ). While healthy paratenon and periosteum generally have adequate perfusion to sustain a graft, denuded tendon or bone does not. Granulation tissue can be induced over small areas of tendon or bone with negative pressure therapy or dermal substitutes ( Fig. 2.1.4 ). Additionally, non-viable or desiccated tendon can be debrided to create a proper wound bed. Calvarial bone can be burred down to induce punctate bleeding and can ultimately be prepared to accept a skin graft.