Key Words
alveolar bone graft, oronasal fistulae, cleft lip and palate, midface growth, ilium, iliac crest, cancellous bone graft, cortical bone graft
Synopsis
Cleft lip and palate provides a four-dimensional reconstructive challenge, encompassing all tissue types and extending across the age spectrum from infancy into adulthood. This chapter focuses on alveolar bone grafting, a component of cleft reconstruction typically performed in early adolescence. The goals of alveolar bone grafting are to establish a solid bony foundation for the erupting adult dentition, to stabilize the maxilla into a single stable arch, and to simultaneously close remaining soft tissue defects such as oronasal fistulae. Although the ideal timing and technique for bone grafting remain controversial, this chapter will review the most commonly advocated techniques, with a step-by-step guide to performing the procedure.
Clinical Problem
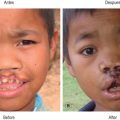
The treatment of children with cleft lip and palate extends beyond their initial cleft repairs in infancy and includes a stepwise staged reconstruction. This is true throughout the world, including in developing countries. The defect in complete cleft lip and palate is a complex three-dimensional malformation that includes soft tissue as well as bony disruption ( Fig. 3.5.1 ). Failure of fusion of the medial and lateral palatine processes causes a bony defect that can extend from the pyriform rim and along the nasal floor. Oronasal clefts can present as a range of deformities ( Fig. 3.5.2 ), and no two clefts are exactly alike. Depending on the anomaly, the surgical timeline may include cleft lip repair, cleft palate repair, cleft lip revisions, cleft palate revisions and speech surgery, alveolar bone grafting, rhinoplasty, and possible orthognathic surgery. The number of procedures for children with a complete cleft lip and palate may be seven or more by age 18.
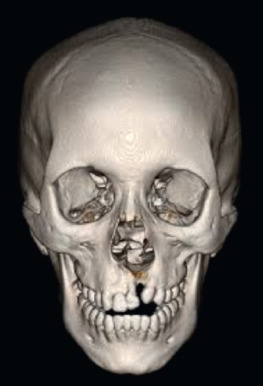
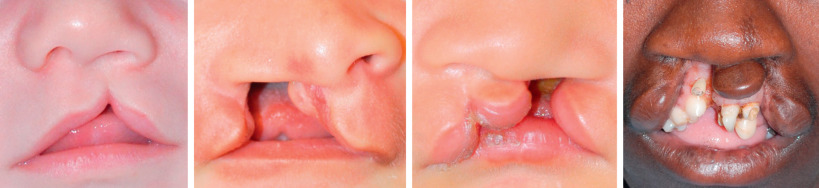
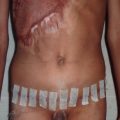
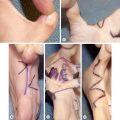
Although the cleft lip and palate are ideally repaired within the first year of life, children with an alveolar cleft will require bone grafting in early adolescence. Alveolar bone grafting supports the erupting adult dentition, stabilizes the maxillary arch, and allows for closure of oronasal fistulae. There are many potential donor sites for corticocancellous bone including the ilium, the tibia, the mandibular ramus, and the cranium. Although each arguably has pros and cons, the most commonly used is the ilium, and we will limit ourselves to a discussion of this donor site for the purposes of this chapter.
Children with cleft lip and palate can have a number of dental and orthodontic anomalies that may challenge the reconstructive surgeon and orthodontist. Typically, children will have asymmetrical delays in dental eruption as well as crowding; maxillary arch collapse; lingual crossbite; supernumerary, ectopic, or missing teeth; and oronasal or labionasal fistulae. Coordinated treatment with a pediatric dentist, orthodontist, and plastic surgeon optimizes outcomes while ideally minimizing the number of surgical interventions.
Most children with cleft lip and palate are non-syndromic, but hundreds of syndromes have been found to present with cleft lip and palate as part of their phenotype. These syndromes should be identified and understood before surgery because there are added surgical risks.
Pre-Operative Management
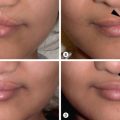
Examination of the child by a multidisciplinary team allows a coordinated approach to comprehensive care and surgical correction. When preparing for alveolar bone grafting, the surgeon should also carefully inspect the lip and palate repairs, because scar revision or dermal/fat-grafts can be combined with the alveolar bone graft operation. These revisions often provide the only visible gains from the perspective of the child and family, because alveolar bone grafting is an otherwise painful but “invisible” procedure. There is some controversy as to whether alar base position improves significantly with bone grafting, but if it does, these changes are subtle. Position of the alveolar segments, fistulae, and quality and number of teeth should all be noted. The orthodontist can assist by assuring adequate maxillary arch width with pre-operative expansion and alignment of the alveolar segments, in preparation for bone grafting.
The gingiva, labial mucosa, and palatal mucosa adjacent to the cleft should also be assessed, because this soft tissue will be needed to cover the bone graft and close fistulae. Supernumerary teeth can be removed several weeks before bone grafting to allow the gingiva and mucosa to heal. Primary teeth within the recipient site may need to be removed before bone grafting or at the time of the procedure to enable graft placement. The ideal setup is when the teeth are healthy, there is adequate or redundant soft tissue, and the bony alveolar segments are well aligned with a narrow cleft gap of 2 to 3 mm.
Historically, there has been some controversy as to the ideal timing for bone grafting. Immediate bone grafting at the time of lip or palate repair in infancy, also known as primary bone grafting, has largely been abandoned given concerns about midfacial growth restriction as well as graft take. A number of studies have suggested that alveolar bone grafting at mixed dentition does not affect midfacial growth and has optimal graft take. Although some reports have suggested that initial gingivoperiosteoplasty at the time of lip repair in infancy prevents the need for bone grafting, many studies have shown that a bony deficit persists, which requires bone grafting during mixed dentition (one should note that grafting at the time of mixed dentition is often referred to as “secondary bone grafting” but is not meant to imply a second attempt at the procedure). Periapical radiographs provide information on the canine development, which will assist in timing of surgery. Grafting when the canine root is two-thirds formed on radiographs allows the tooth to erupt into graft, functionally loading the graft and increasing graft take. For this reason, early bone grafting in mixed dentition, before canine eruption, is felt to be better than grafting after the canine has erupted. This is typically between 7 and 12 years of age.
Once the orthodontist has prepared the child for bone grafting with palatal expansion and alignment of the alveolar segments, and the surgical timing has been coordinated with canine eruption, the child can be prepared for surgery. For non-syndromic children, no special pre-operative assessment is required. Syndromic children may require careful airway evaluation, cardiac clearance, and cervical spine precautions, depending on their condition. A general anesthetic with endotracheal intubation is standard.
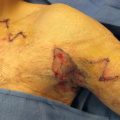
The operative setup should include two surgical fields: one sterile field for harvesting bone and one sterile field for the alveolar recipient site in the relatively “dirty” oral cavity. For harvesting bone, osteotomes 4 to 8 mm, a mallet, curettes, and malleable retractors facilitate the procedure. Bupivacaine can be used to infiltrate the donor and recipient sites, and studies have shown improved post-operative analgesia if a resorbable sponge, such as Gelfoam soaked in bupivacaine, is packed into the donor site. Preparing the recipient site requires a 15 blade, Cottle elevator, lip retractors, and absorbable suture such as 4-0 chromic gut or polyglactin 910 (e.g., Vicryl). Both sites should have a suction setup, whereas electrocautery is typically required only at the donor site. Some centers make use of a tooth-borne surgical splint, fashioned from a pre-operative mold, secured to the surgical brackets or bonded to the teeth to protect the graft site post-operatively. This is more critical after bilateral procedures, to support the unstable premaxilla, than it is for unilateral procedures, where the alveolar segments are relatively stable.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree