Skin Disease in Acute and Chronic Immunosuppression: Introduction
|
Impairment of the body’s immune system results from a variety of causes, including natural aging, ultraviolet radiation, diabetes, malnutrition, cancer, and iatrogenic suppression. While few skin conditions appear solely in immunocompromised individuals, clinical presentations may be morphologically atypical, follow unusual clinical courses, or prove harder to treat than in individuals with intact immunity. This chapter focuses on dermatologic manifestations in immunosuppressed patients without human immunodeficiency virus (HIV) disease, predominantly in those with immunosuppression induced by drugs, conditions surrounding solid organ and bone marrow transplantation, and hematologic malignancy. Skin manifestations of HIV disease are described in Chapter 198. Other chapters cover graft-versus-host disease (see Chapter 28), skin signs associated with primary immunodeficiency disorders (see Chapter 143), and detailed side effects of medications, including corticosteroids, cancer chemotherapeutic agents, immunosuppressants, and cytokines (see Chapters 224, 227, 233, and 234). The salient clinical features particularly associated with immunosuppression are emphasized here.
While a variety of inflammatory skin diseases and paraneoplastic processes occur in the setting of immunosuppression, infections, and malignancy are most commonly seen and are discussed herein. When approaching an immunocompromised patient, it is helpful to determine the time frame of the immune loss as well as the specific immune defect. This chapter is divided into two major subsections based on this concept: acute immunosuppression and chronic immunosuppression. When patients are acutely immunosuppressed, usually from iatrogenic ablation of the immune system or from acute leukemia, infections occur that are normally controlled by innate immunity, which typically involve neutrophils and macrophages. In chronically immunosuppressed individuals, such as organ transplant patients and those taking corticosteroids on a long-term basis, T-cell function is impaired, and diseases will often be similar to those observed in HIV disease. Thus, it is helpful to understand the underlying immune defects associated with the medical conditions of each patient (Table 29-1), because it helps to focus the history taking and physical examination toward skin manifestations of specific pathogens.
Immune Defect | Usual Conditions | Common Bacterial Pathogens | Common Viral Pathogens | Common Fungal Pathogens |
|---|---|---|---|---|
Defective cell-mediated immunity | Organ transplantation, metastatic cancer, Hodgkin disease, glucocorticoid, or cyclosporine therapy | Listeria, Salmonella, Nocardia, Mycobacterium avium-intracellulare, M. tuberculosis, Legionella | Cytomegalovirus, herpes simplex virus, varicella zoster virus | Candida, Cryptococcus, Histoplasma, Coccidioides |
Defective humoral immunity | Multiple myeloma, chronic lymphocytic leukemia | Streptococcus pneumoniae, Haemophilus influenzae, Neisseria meningitidis | Enteroviruses | — |
Neutropenia | Cancer chemotherapy, acute leukemia, adverse drug reaction | Aerobic Gram-negative bacteria; Staphylococcus aureus, Streptococcus viridans, Staphylococcus epidermidis | Herpes simplex virus | Candida, Aspergillus |
Defective neutrophil function | Chronic granulomatous disease, myeloperoxidase deficiency | Catalase-positive bacteria: S. aureus, Escherichia coli | — | Candida |
Hyposplenism | Splenectomy, hemolytic anemia | S. aureus, Streptococcus | — | Candida |
Defective complement components | Congenital or acquired deficiencies | S. pneumoniae (C2, C3, C5 alternate), H. influenzae (C2, C3, alternate), S. aureus (C5), Enterobacteriaceae (C5), Salmonella(alternate), N. meningitidis (C6–C8) | — | — |
Skin barrier disruption | Intravascular catheters, decubitus ulcers, burns | Staphylococcus, M. fortuitum, Gram-negative bacteria, anaerobes | — | Candida, Aspergillus, Mucor |
In ill-immunosuppressed patients, disease often manifests in the skin. Appropriate evaluation and diagnosis of skin lesions are critical to the overall health of these individuals, because the skin is often a window to more severe systemic illness. In particular, unusual presentations of infection with typical pathogens and infections with rare opportunistic pathogens are common in these patients. Diagnosis is also made more difficult by the variety of organisms that share similar morphologies and the wide variety of morphologic presentations of a single organism (Table 29-2). This makes prompt clinical evaluation and extensive use of skin biopsy and culture necessary to make an accurate diagnosis and initiate prompt treatment to obviate significant morbidity and mortality.
Organism | Ecthymatous Lesions | Morbilliform Eruption | Vesicles | Erythemas (Cellulitic Patches and Plaques) | Ulcers |
|---|---|---|---|---|---|
Bacteria | |||||
Pseudomonas aeruginosa | X | X | |||
Streptococcus viridians | X | X (facial) | |||
Staphylococcus sp. | X | X | |||
Aeromonas hydrophilia | X | ||||
Nocardia spp. | X | ||||
Vibrio vulnificus | X | X | |||
Fungi | |||||
Aspergillus sp. | X | X (hemorrhagic) | X (necrotic) | ||
Zygomycetes organisms | X | X (facial) | X (necrotic) | ||
Fusarium sp. | X | ||||
Cryptococcus neoformans | X | X | |||
Histoplasma capsulatum | X | X (mucosal) | |||
Coccidioides immitis | X | ||||
Viruses | |||||
Herpes simplex virus | X | X | X | ||
Varicella zoster virus | X | X | X | ||
Cytomegalovirus | X (mucosal) |
Acute Immunosuppression
The prototype of an acutely immunosuppressed patient needing dermatologic evaluation is the neutropenic patient undergoing chemotherapy around the time of hematopoietic transplantation. Pancytopenia and neutropenia in particular predispose to invasive infections caused by gram-negative and -positive bacteria and the fungal organisms Candida and Aspergillus.1 These complications from many cancer therapies often pose a more immediate threat to survival than the malignancy itself. In the past two decades, overall mortality due to infection among patients undergoing hematopoietic transplantation has decreased significantly with the use of better prophylaxis and nonmyeloablative regimens, but still represents an ongoing risk to survival. The causes of infection-related death have remained relatively stable, with death due to bacterial infections being the most common (36%), followed by deaths due to infection by viruses (31%), fungi (28%), and parasites (5%).2,3 Infections in the acute period following solid organ transplantation are less opportunistic and tend to reflect the usual nosocomial pathogens associated with surgical procedures and hospitalization.4
Bacteria are responsible for most infections during acute neutropenic episodes. Empiric antimicrobial therapy for fever and neutropenia was first introduced in the 1970s when 60%–70% of infections were due to Gram-negative bacteria such as Escherichia coli, Pseudomonas aeruginosa, and Klebsiella species. Dramatic shifts have occurred since that time, such that over 50% of bacterial infections in cancer patients are now caused by Gram-positive organisms, and 75%–80% in patients that are bacteremic.5,6 The use of indwelling intravascular catheters, medications predisposing to mucositis, and prophylactic fluoroquinolones are all thought to play a role in the shift to Gram-positive organisms, such as coagulase-negative staphylococci, Staphylococcus aureus, Enterococcus species, and viridians group streptococci.7 The emergence of drug-resistant organisms, including methicillin-resistant Staphylococcus aureus and vancomycin-resistant Enterococcus, as well as polymicrobial infections also complicates the situation.6
Routine cellulitis from staphylococcal and streptococcal organisms is a common manifestation of skin infection in the acutely immunocompromised host. Muted clinical signs and symptoms can be found in this population, so care must be taken to rule out deeper involvement as occurs in necrotizing fasciitis.8 Bacteremia may result from skin and soft-tissue infections such as folliculitis, furuncles, and wound infections. Bone marrow transplant patients and other patients with neutropenia are prone to streptococcal bacteremia and may develop facial flushing, a widespread erythematous, petechial or purpuric eruption of macules and papules, and desquamation of the palms and soles.9 Staphylococcal scalded-skin syndrome, which typically occurs in children (see Chapter 177), can occur in immunosuppressed adults.10
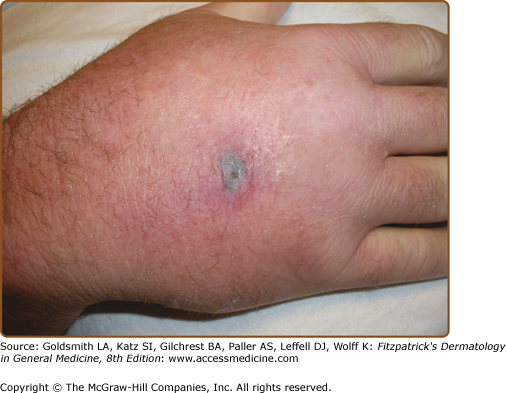
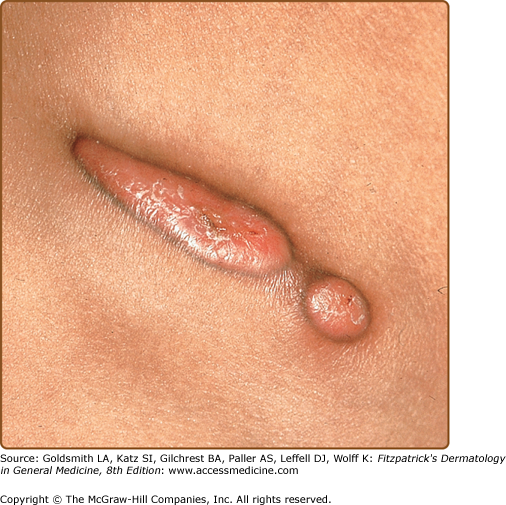
Ecthyma gangrenosum is one of the more specific clinical signs of bacteremia and is characterized by a painful erythematous to dusky nodule or plaque that rapidly develops a central pustule or hemorrhagic vesicle, followed by necrosis (Fig. 29-1). The groin, perianal area, and axillae are the most common locations. There may be one or many lesions. Classically described in patients with Pseudomonas septicemia, it is now recognized that other bacterial and fungal organisms, including S. aureus, Aeromonas hydrophilia, Serratia marcescens, K. pneumoniae, E. coli, Aspergillus, and Mucor species, can also cause similar lesions.11,12 Necrosis is secondary to underlying focal vasculitis, which can be observed in skin biopsy specimens. Diagnosis is made by culture of the organism from skin or blood.
Patients with neutropenia, cystic fibrosis, or extensive burns are particularly susceptible to systemic P. aeruginosa infection (see Chapter 180).13 The mortality rate of P. aeruginosa bacteremia in transplant patients is high at upwards of 40%.14 Other cutaneous manifestations of P. septicemia may appear initially as grouped vesicles, cellulitis, subcutaneous nodules, petechiae, purpura, or folliculitis.15 Progression to ulcerative and necrotic lesions that are more characteristic of ecthyma gangrenosum may occur. Primary cutaneous infection, usually at the site of a medical procedure, can also cause ecthyma gangrenosum-like lesions. As is common with other infections in neutropenic patients, primary lesions can lead to bacteremia and should be treated aggressively.
In the acute transplant setting, invasive fungal infections are less common than bacterial infection, but cause much greater mortality. Mortality rates range from 40% to close to 100%, especially when treatment is delayed.16 Prolonged neutropenia is a significant risk factor and recovery from disseminated fungal infections is rare unless neutropenia resolves. Candidiasis and aspergillosis represent the two most common invasive fungal infections that occur in patients who are undergoing cytotoxic chemotherapy or stem cell transplantation or who have acute myeloproliferative disorders.17 However, they are not unique to the neutropenic patient and are encountered in settings such as surgical and neonatal intensive care units, and in patients with cell-mediated immune dysfunction such as those undergoing long-term immunosuppression after solid organ transplantation. Additional risk factors for opportunistic fungal infection include hyperalimentation, antibiotic use, hyperglycemia, corticosteroid use, and central venous catheter use. Other fungal organisms causing infection in hosts with acute neutropenia include Trichosporum species, Fusarium species, and organisms in the Zygomycetes class.18
Candidiasis (see Chapter 189) remains the most common opportunistic fungal infection worldwide, although its role in invasive infections is changing in certain immunosuppressed populations.19 Candida species still account for more than half of invasive fungal infections in solid organ transplant recipients, but aspergillosis has become more common in hematopoietic stem cell transplant recipients.20–22 Historically, most candidal infections were due to Candida albicans, but there has been an emergence of other organisms in recent years, including C. glabrata, C. krusei, C. parapsilosis, and C. tropicalis.17 In certain populations of patients with hematologic malignancy or stem cell transplantation, non-C. albicans species now predominate, so awareness of local and regional patterns of infection is important.20,23
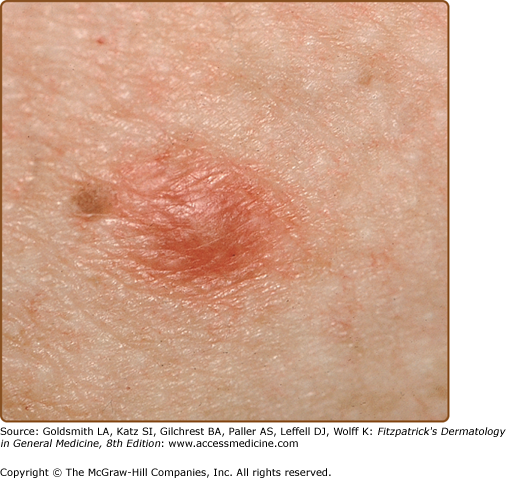
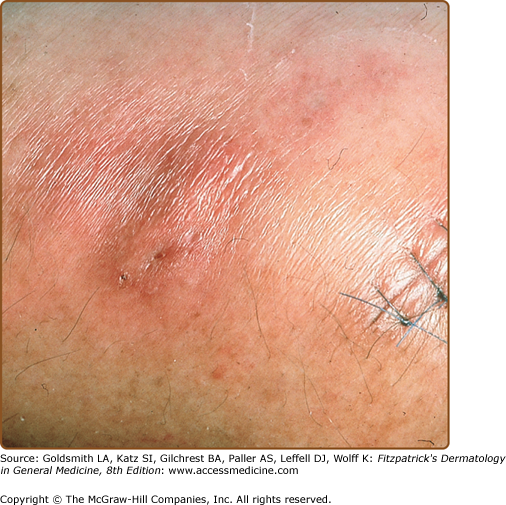
The classic triad of fever, myalgias, and erythematous skin lesions in a septic patient not responding to antibiotic therapy is highly suggestive of disseminated candidiasis. Fungi may seed numerous organs, causing myositis, meningitis, endocarditis, pneumonitis, cerebritis, esophagitis, bursitis, osteomyelitis, arthritis, and endophthalmitis. Cutaneous lesions are present in only 5%–10% of individuals with disseminated candidiasis.15,24 Lesions are characteristically painless, nonblanching, discrete, erythematous macules, papules, or nodules (Fig. 29-2) and may develop central purpuric, pustular, or necrotic changes. Involvement is usually generalized, but occasional patients have very few lesions limited to the proximal extremities. The major clinical differential diagnosis includes infections caused by other opportunistic pathogens and drug eruptions. Histologically, periodic acid-Schiff-positive yeast forms are seen in the dermis, usually in association with vascular damage and mild inflammation. Candida can be grown from sterile skin lesion samples in approximately 50% of patients. Tissue scrapings from dermal skin can be obtained at the time of biopsy with a scalpel or small-diameter curette for slide examination and can be an invaluable aid in making a rapid diagnosis in an acutely ill patient.
The treatment of choice for presumed disseminated candidiasis is usually intravenous liposomal amphotericin B, although the new class of echinocandins are also being evaluated.25,26 Culture results are again important since C. glabrata, C. albicans, C. tropicalis, and C. parapsilosis are showing resistance to fluconazole, and C. krusei is naturally resistant.22 Newer azoles, including voriconazole and posaconazole, are effective against Candida species, although breakthrough infections with resistant C. glabrata have already been reported with voriconazole (see Chapter 232).27
While aspergillosis remains the second most common cause of opportunistic fungal infection in immunosuppressed patients as a whole, it has now surpassed Candida as the most common cause of invasive fungal infection in hematopoietic stem cell transplant patients and certain hematologic malignancies.15,16,20 Incidence rates vary in different immunosuppressed groups, but may reach 25% in acute leukemia and organ transplant patients.28 Persistent neutropenia and neutrophil dysfunction are risk factors for disseminated infection. Infection rates are also high for patients undergoing allogeneic stem cell transplantation, and risk factors in this group are expanded to include immunosuppression for graft-versus-host disease prophylaxis, graft-versus-host disease itself, and other infectious diseases, especially cytomegalovirus (CMV) infection. Invasive infection with Aspergillus was classically seen during acute periods of neutropenia, but shifts in conditioning regimens and other strategies to promote earlier engraftment have led to infections after 30–40 days posttransplantation.20 This observation emphasizes that immune defenses other than those mediated by granulocytes are important for protection against invasive fungal infections, and against Aspergillus infections in particular.
The incidence of invasive aspergillosis is also increasing in nonclassic immunocompromised hosts such as critically ill patients in the intensive care unit. Environmental factors also clearly contribute to the development of aspergillosis, especially in primary cutaneous disease. These include hospital construction (which increases spore counts in ventilation systems), the use of indwelling catheters (which provide portals of entry for organisms), and contamination of tape and arm boards used to secure catheters. Mortality rates have improved with the introduction of newer antifungal agents, but remain higher than 50% in stem cell and organ transplant recipients.28,29
A. fumigatus is the most common cause of disseminated infections, although emerging strains of A. flavus, A. niger, and A. terreus are accounting for more disease.20 Reports suggest that A. flavus is associated most commonly with primary cutaneous disease.30 A. terreus is more likely to be resistant to amphotericin B, and multiple tri-azole resistant A. fumigatus has been described.28
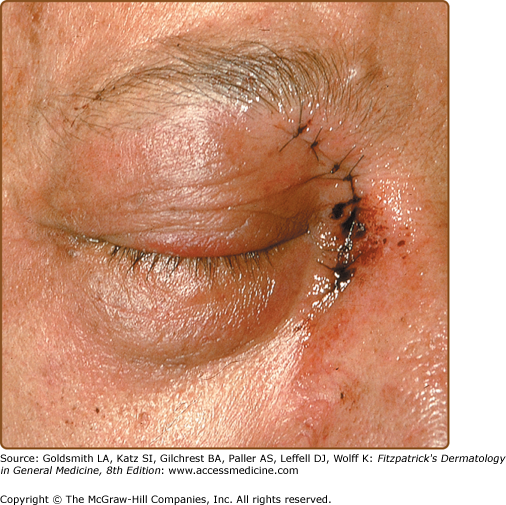
Primary cutaneous aspergillosis often develops at paronychial locations, sites of intravenous catheters, or under areas of occlusion. Lesions initially appear as small cellulitic areas and progress quickly to necrotic ulcers with black eschars (Fig. 29-3) due to the angioinvasive nature of the organism. In patients with Aspergillus sinusitis, necrotic ulcers with black eschars can occur in the anterior nares and on the nasal septum, palate, and skin overlying the nasal bridge. MRI may be useful in diagnosing the underlying sinusitis, and prior treatment with amphotericin B does not exclude the diagnosis as surgical treatment may be needed in this setting.31–33
Pulmonary, and less often primary cutaneous or sinus, infection can easily become invasive and lead to disseminated disease in immunocompromised hosts. Patients with disseminated aspergillosis often present with unremitting fever despite antibiotic use. The central nervous system, heart, kidneys, and gastrointestinal tract may also be involved. Cutaneous manifestations of disseminated aspergillosis are uncommon, occurring in only 5%–10% of patients.30 Lesions begin as single or multiple painful, erythematous papules, nodules, or plaques. They rapidly expand and develop central hemorrhagic vesicles or bullae, then eschar. In tissue sections, diagnosis can be made by demonstration of nonpigmented septated hyphae that branch at acute angles. Blood culture results often are not positive or reliable because Aspergillus is found commonly as a laboratory contaminant. Voriconazole has become the first-line agent for treatment of invasive aspergillosis. Alternatives include caspofungin, liposomal amphotericin B, itraconazole, and posaconazole.29 Surgical removal of isolated lesions of primary cutaneous aspergillosis can be attempted, although this may not necessarily prevent secondary disseminated infection in patients with persistent neutropenia.
Zygomycosis is the third most common opportunistic fungal infection in immunosuppressed hosts, and may account for closer to 50% of invasive fungal infections in certain populations such as renal transplant patients.18 The term zygomycosis is used to describe a group of fungal infections caused by ubiquitous Zygomycetes found in soil and decaying matter. Infections in humans are mostly caused by the order Mucorales (mucormycosis) and include the genera of Mucor, Rhizopus, Absidia, Rhizomucor, and Cunninghamella. The term zygomycosis is now preferred over mucormycosis because it is broader and more relevant when organisms are not identifiable. Like aspergillosis, zygomycosis is rare in individuals without underlying immunodeficiency or predisposing conditions. Host defenses usually prevent the germination of spores unless the inoculation is too great, as in trauma or surgical wounds. Chronic medical conditions that affect macrophage function, such as diabetes or corticosteroid-induced immunosuppression, lead to an inability to inhibit spore germination, and these patients are at increased risk of infection. Additional risk factors besides immunosuppression include iron overload, burns, intravenous illicit drug use, and malnourishment. Recently, the use of voriconazole in immunosuppressed patients with presumed or diagnosed aspergillosis may account for part of the increase in zygomycotic infections.27
Primary infection can occur by inhalation, by direct inoculation into damaged skin, or by ingestion. Patients with prolonged neutropenia present most often with pulmonary disease and dissemination. The mortality rate in these individuals is very high, approaching 100%.34 Diabetic patients with sustained hyperglycemia and metabolic acidosis are predisposed to primary rhinocerebral (66%) and pulmonary (16%) infections.35 Malnutrition and gastrointestinal disease predispose patients to primary gastrointestinal tract infection. Wounds and burn injuries predispose to primary cutaneous infection. Each type of primary infection can lead to hematogenous spread and disseminated infection of numerous organs (especially the brain).
The clinicopathologic hallmarks of cutaneous zygomycosis are vascular invasion, ischemic infarction, and necrosis, which result in painful erythematous nodules and plaques that ulcerate rapidly and form central black eschars.34 Clinical manifestations of primary cutaneous disease can range from necrotic papules to cellulitis, to subcutaneous nodules with rapid extension and dissemination especially in neutropenic patients.36 Rhinocerebral zygomycosis typically begins with facial edema and erythema (Fig. 29-4), bloody nasal discharge, and ulceration of the palate or nasal septum. Within a few days, necrotic skin lesions, headache, focal neurologic defects, exophthalmos, and altered vision develop and can progress to seizures, stupor, coma, and death. Disseminated disease from a noncutaneous primary site infrequently presents with skin findings.36
Diagnosis of zygomycosis is usually made by demonstration of nonseptated hyphae (with branching at right angles) within infected tissue. The treatment of choice for disseminated disease is lipid preparations of intravenous amphotericin B and surgical debridement. Some advocate the addition of posaconazole. If possible, reversal or removal of underlying predisposing conditions should be attempted.18
Fusarium is a filamentous mold found in soil and plants belonging to the fungal group of hyalohyphomycoses. Disseminated infections are found in severely immunocompromised individuals, whereas immunocompetent patients have localized lesions at areas of skin breakdown. Neutropenic patients are particularly susceptible to infection and rapid dissemination.31 The source of infection in patients undergoing acute immunosuppressive therapy is often the skin, especially from cellulitis developing at the site of onychomycosis, local trauma, or insect bites. Nasal sinuses are another source of primary infection that can lead to dissemination following acute immunosuppression.18 In disseminated disease, patients present with multiple painful erythematous papules and nodules, some with central necrosis. Lesions are often at different stages of development, and a specific presentation of papules evolving into target-like lesions with a ring of normal-appearing skin and an outer rim of erythema has been observed.31 Skin lesions in disseminated disease often precede fungemia and are found in approximately 75% of patients making dermatologic evaluation valuable.8 The mortality rate in patients who are persistently neutropenic is about 80%, compared with 30% in patients whose immune systems recover. Disease in solid organ transplant recipients may occur later than in patients with hematologic malignancies. Newer tri-azole antifungals such as voriconazole have some efficacy against infection with Fusarium species, for which treatment options have traditionally been limited. Surgical resection of localized skin infection is useful. Granulocyte transfusions may also play a role in treatment.18
Trichosporon beigelii, a yeast-like organism that causes white piedra in the tropics, may produce acute systemic infection in immunosuppressed patients, most commonly in the setting of neutropenia.31 Trichosporon is an emerging pathogen in organ transplant recipients as well.18 Patients with disseminated trichosporonosis are acutely ill. They may have fever, hypotension, pulmonary infiltrates, renal involvement, and hepatosplenomegaly. Skin lesions occur in 30% of patients and appear similar to cutaneous lesions of disseminated candidiasis (multiple red papules that may ulcerate). Definitive diagnosis is made by culture, and the treatment of choice is fluconazole or itraconazole; amphotericin B resistance is common.18
Viral infections are predominantly associated with defects in cellular immune function and are not typically expected to cause problems in patients whose main immunologic defect is neutropenia.4 The most common viral infection that occurs in patients who are undergoing induction chemotherapy for lymphoma or an acute leukemia or who are in the first few weeks after a hematopoietic stem cell transplantation is reactivation of latent herpes simplex virus (HSV) infection (see Chapter 193).37 Clinical presentations in acutely immunosuppressed patients include an increased severity of oral mucositis, intraoral ulcers outside of the gingival margin, and necrotizing gingivitis. Pneumonitis can occur from either contiguous spread from the oropharynx or from viremia.37 Antiviral prophylaxis with acyclovir is very effective in preventing disease during chemotherapy and following hematologic and solid organ transplantation. When disease does occur in transplant patients, approximately 10% of cases are resistant to acyclovir because of a mutation in the gene coding for thymidine kinase, which is the enzyme required for efficacy of acyclovir, valacyclovir, and famciclovir (see Chapter 231).38 The treatment of choice in these patients is foscarnet, although reports of resistance to both agents is increasing.37,39,40
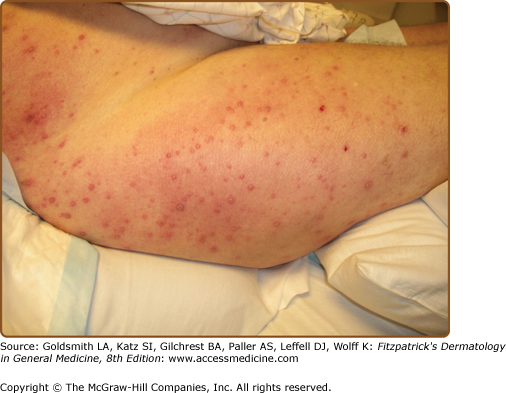
Reactivation of varicella zoster virus (VZV) (see Chapter 194) usually occurs 3 months or longer after transplantation and is relatively rare in the acutely immunosuppressed patient. VZV infection in adults with leukemia or after solid organ transplantation is rarely primary, but more often represents reactivation of latent virus. In this setting, patients are at increased risk for both skin and systemic dissemination of virus (Fig. 29-5).41 Before the use of antiviral prophylaxis in bone marrow transplantation, disseminated primary varicella or zoster infection was associated with mortality rates of 30%.42
Chronic Immunosuppression
Patients with chronic immunosuppression include those that are iatrogenically immunosuppressed because they are taking medications that impair the immune system and those with chronic diseases that are associated with immune dysfunction, such as diabetes mellitus. Moreover, individuals with cancer often have immune system defects before aggressive cytotoxic, radiation, or surgical therapy.43 For example, tumors can secrete immunosuppressive factors (e.g., transforming growth factor-β1 and interleukin-10) or induce T-cell anergy, which help them evade normal immune responses and lead to further systemic immunosuppression.
The population of patients taking long-term immunosuppressive medications is growing as solid organ transplantation becomes a therapeutic option for many human diseases and the survival of patients in the short and long term has improved. These individuals require lifelong therapy with immunosuppressive drugs to maintain function of the transplanted organ. Cyclosporine, tacrolimus, sirolimus, prednisone, mycophenolate mofetil, azathioprine (see Chapters 227 and 233), and the newer agents daclizumab and basiliximab are the drugs used most commonly to prevent graft-versus-host disease, predominately by inhibiting cell-mediated immunity (i.e., T-cell function).44 Humoral immunity (i.e., B-cell function) remains relatively intact in these patients. Thus, opportunistic diseases in most transplant patients are dominated by viral and fungal infections, intracellular bacterial infections, and virus-associated malignancies—conditions that are controlled predominantly by cell-mediated immune mechanisms in immunocompetent hosts.
Atypical mycobacteria (see Chapter 184) are ubiquitous organisms found in soil and water. The most common organisms in this group include Mycobacterium marinum, M. chelonae, M. fortuitum, M. abscessus, M. kansasii, M. haemophilum, and M. avium-intracellulare. Before the epidemic of acquired immunodeficiency syndrome (AIDS), most cases occurred in persons with underlying pulmonary disease. However, nontuberculous mycobacterial infections after transplantation are increasing in frequency and severity, with the highest incidence (up to 5%) seen in recipients of hematopoietic stem cell transplants. One-third present with catheter-related infections, although skin lesions are rare in this population. Skin involvement is the most commonly reported manifestation of nontuberculous mycobacterial infections in solid organ recipients except lung and heart transplant recipients, who are more likely to have pulmonary involvement. One-third of these patients have localized or disseminated cutaneous disease and the rapid growing species M. chelonae, M. fortuitum, and M. abscessus are most commonly isolated.45,46 Median time to infection varies depending on the type of transplant, ranging from 4 months posttransplant in stem cell recipients, to 30 months in heart recipients.
Atypical mycobacterial infections in the skin are characterized by diverse morphologies, including reddish brown nodules and plaques (eFig. 29-5.1), abscesses (Fig. 29-6), and ulcers.47 M. avium-intracellulare and M. haemophilum commonly cause disseminated infection, which can involve the lungs, lymph nodes, liver, spleen, bone marrow, and skin. Organisms can be identified by special stains or by culture of specimens from affected skin. Specific antimycobacterial antibiotic treatment regimens are complex and depend on the mycobacterial species, results of sensitivity testing, extent and severity of disease, and presence or absence of underlying immune defects.45,46