© Springer-Verlag Berlin Heidelberg 2017
Nina Dragicevic and Howard I. Maibach (eds.)Percutaneous Penetration Enhancers Drug Penetration Into/Through the Skin10.1007/978-3-662-53270-6_1818. Retardation Strategies for Sunscreen Agents
(1)
Galderma Spirig, Egerkingen, Switzerland
(2)
University Hospital Zürich, Department of Dermatology, Zürich, Switzerland
(3)
University Hospital Basel, Department of Dermatology, Petersgraben 4, CH-4031 Basel, Switzerland
Keywords
Sunscreen filterSunscreen formulationPercutaneous absorptionVehicle effectAbsorption enhancing effectAbsorption retarding effectTopical bioavailability18.1 Introduction
There is overwhelming evidence indicating that human skin is damaged in different ways by exposure to sunlight. Of the solar radiation reaching the earth’s surface, the ultraviolet (UV) component (290–400 nm) is a major factor leading to skin pathologies that range in severity from inflammatory responses, cutaneous photoaging, dendritic keratitis to various types of skin cancer (Pathak 1991; Ziegler et al. 1994; Hochberg and Enk 1999; Surber et al. 2012). The increasing knowledge of the deleterious effects of sunlight has promoted the widespread use of topical sunscreen preparations (Hayden et al. 1998; Green et al. 1991), which contain chemicals that absorb, reflect, or scatter UV radiation (Patel et al. 1992) and are thereby highly effective skin protectants. Organic sunscreen agents are compounds that decrease the intensity of UV light reaching the epidermal strata by absorbing radiation (typical electron promotion from a lower- to a higher-energy molecular orbital). The activated sunscreen molecule dissipates the excess energy in the form of heat, by fluorescence, phosphorescence, interaction with neighboring molecules, or by undergoing photochemical modifications (Broadbent et al. 1996). Particulate sunscreens present a physical barrier between the incident radiation and the epidermis, scattering or reflecting the radiation. However, to be effective, these agents must remain on or in the outermost layers of the skin, the stratum corneum (SC). One major drawback of current sunscreen formulations is that they are constantly lost from the skin surface by abrasion from clothing, sweating, or swimming, requiring frequent reapplication for continued effectiveness. Moreover, several of the chemical sunscreens currently on the market exhibit irritancy and sensitization reactions after absorption into the dermal strata in predisposed individuals, often causing immunological problems (Deflandre and Lang 1998; Dromgoole and Maibach 1990; Mariani et al. 1998). A significant improvement in sunscreen technology would be the development of systems that retard the penetration of the chemical into the skin and bind the agent in the stratum corneum so that minimal loss occurs by diffusion or abrasion. The degree of sunscreen penetration depends strongly on the physicochemical properties of the active compound and of the nature of the vehicle in which the sunscreen is applied, that is, polarity of the ingredients that form the vehicle and the filters particle size (Benech-Kieffer et al. 2000). Therefore, the development of suitable formulations that prevent penetration of the sunscreen into the skin is a challenge for developers. Some of the few vehicular penetration retardation strategies being researched for sunscreens are reviewed below.
18.2 Cyclodextrins and Photostability
Cyclodextrins are cyclic, toroidal-shaped oligosaccharides with a hydrophilic external surface and a hydrophobic central core. They are capable of incorporating appropriately sized, nonpolar compounds or some lipophilic moiety of a molecule into their apolar cavities, forming non-covalent inclusion complexes (Rajewski and Stella 1996; Loftsson and Brewster 1996). This type of molecular encapsulation can lead to changes in some of the physical and chemical properties of the included substance, such as the enhancement of stability to air and light and apparent aqueous solubility (Rajewski and Stella 1996; Loftsson and Brewster 1996; Uekama et al. 2003). Moreover, cyclodextrin complexation can affect the topical availability of applied drugs, either increasing or decreasing their penetration into or permeation through the skin (Rajewski and Stella 1996; Loftsson and Masson 2001). Butyl-methoxydibenzoylmethane (BM-DBM) is a widely used filter that provides protection against UVA radiation in the 320–400 nm range. However, BM-DBM experiences marked photodegradation (Schwack and Rudolph 1995; Scalia et al. 1998; Tarras-Wahlberg et al. 1999; Chatelain and Gabard 2001; Damiani et al. 1999; Scalia et al. 2002), forming highly reactive photolytic products that are exposed to the living tissues of the epidermis and dermis following percutaneous permeation. Scalia et al. (1998, 2002) have demonstrated that the degree of decomposition and free radical formation upon exposure of BM-DBM to simulated sunlight were reduced by complexation with hydroxypropyl-13-cyclodextrin (HP-13-CD). The effects of HP-13-CD and sulfobutylether-13-cyclodextrin (SBE7-13-CD) on in vitro human skin penetration and retention of the sunscreen agent BM-DBM were investigated by Simeoni et al. (2004). They reported that approximately 14–16 % of the applied dose of BM-DBM penetrated into the skin tissue; however, no sunscreen was detected in the dermis and in the receptor phase. The greater proportion (84.6–95.5 %) of the absorbed UV filter was localized in the SC with no significant differences between uncomplexed and complexed BM-DBM. Notable levels (2.3 % of the applied dose) of the sunscreen agent accumulated in the epidermis from the preparation containing free BM-DBM. The epidermal concentration of the UV filter was markedly reduced (0.7 % of the applied dose) by complexation with SBE7-13-CD, whereas HP-13-CD had no effect. The results demonstrated that complexation of BM-DBM with SBE7-13-CD attained high sunscreen levels at the skin surface where its action is most desirable and produced lower concentrations of the active in the epidermis.
18.3 Transcutol®
Ethoxydiglycol (Transcutol® CG, Gattefosse, France) is a hydgroscopic liquid that is freely miscible with both polar and nonpolar solvents. Transcutol® has been recognized as a potential transdermal permeation enhancer due to its nontoxicity, biocompatibility with skin, and excellent solubilizing properties (Godwin et al. 2002). However, Transcutol® has also been reported to increase the skin accumulation of topically applied compounds without a concomitant increase in transdermal permeation (Ritschel et al. 1991; Panchagnula and Ritschel 1991). It is theorized that this depot effect is created by a swelling of stratum corneum intercellular lipids, without alteration of their multiple bilayer structure. The expanded lipid domain is then able to retain drugs (especially lipophilic compounds) to form the depot, with a simultaneous decrease in transdermal permeation.
Godwin et al. (2002) studied the influence of Transcutol® CG concentrations in sunscreen formulations on the transdermal permeation and skin accumulation of the UV absorbers 2-hydroxy-4-methoxybenzophenone (oxybenzone) and 2-octyl-4-methoxycinnamate (cinnamate).
When formulated alone, both these lipophilic sunscreens have been shown to permeate through the skin and enter the systemic circulation (Treffel and Gabard 1996). In their study, the concentration of the UV absorber was held constant at 6 % (w/w) for all vehicle systems while the concentration of Transcutol® CG was varied from 0 to 50 % (w/w). The data demonstrated that both UV absorbers exhibited an increase in skin accumulation with increasing concentrations of Transcutol® CG. Skin accumulation of oxybenzone was significantly (P < 0.05) greater than that of cinnamate for all formulations investigated. However, no significant differences were found in the transdermal permeation of oxybenzone or cinnamate for any of the formulations tested. The results of this study demonstrate that the inclusion of Transcutol® CG in sunscreen formulations appears to increase the skin accumulation of the UV absorbers oxybenzone and cinnamate, without a concomitant increase in transdermal permeation. Their data support the theory of the formation of an intracutaneous depot for both oxybenzone and cinnamate when formulated with Transcutol® CG. These observations have to be confirmed in vivo. As the accumulation occurs not only in the epidermis but also in deeper layers of the skin the potential bioavailability remains unknown.
18.4 Encapsulation Structures
Colloidal drug carriers, including submicron emulsions, nanospheres, nanocapsules, liposomes, and lipid complexes, have been attracting increasing interest as drug delivery vehicles. These encapsulation systems have been evaluated for the intravenous administration of lipophilic drugs, as improved parenteral formulations, and as systems for site-specific drug delivery such as tumors (Allemann et al. 1993). In general, two techniques have been used for the preparation of nanocapsules based on biodegradable polymers: the emulsification-diffusion technique (Quintanar-Guerrero et al. 1996) and the solvent displacement procedure (Fessi et al. 1988; AI Khouri Fallouh 1986). The ideal medium in which an active ingredient is incorporated must provide not only the necessary solubility but also maintain contact between the active ingredient and the skin. The nature of the colloidal carrier and the effects of size and surface charge influence penetration and permeation of UV filters into the skin (Zeevi et al. 1994; Treffel and Gabard 1996; Gupta et al. 1999).
Alvarez-Roman et al. (2001) investigated the optimization of a solvent displacement method for poly(ε-caprolactone) nanocapsules, using the lipophilic UV filter, octyl methoxycinnamate (OMC) as the oil core. In addition, these researchers evaluated the influence of polysorbate 85 (Tween 85) and poloxamer 188 (Pluronic F 68) as filter-stabilizing agents, the OMC loading capacity, and the photoprotective potential of the formulations. The OMC-nanocapsule-gel preparation resulted in a significantly better (P < 0.05) protection against UV-induced erythema than a simple OMC gel. Sunscreen effectiveness implies that the sunscreens adhere to the skin more efficiently as a protective film. These results suggest that the nanoparticles are able to cover more efficiently the skin surface due to their high specific surface area. Sunscreen nano-capsules, therefore, show good potential as improved skin retention vehicles.
Liposomes and emulsions are formulated from biocompatible excipients and can easily be produced on a large scale. Compared to liposomes and emulsions, solid particles afford protection of incorporated active compounds against chemical degradation and allow more flexibility in modulating the release of the compound. The advantages of solid particles, emulsions, and liposomes were, therefore, combined by the development of solid lipid nanoparticles (SLNs) (Müller et al. 2002), produced by simply exchanging the liquid lipid (oil) of the emulsions by a solid lipid.
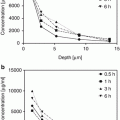
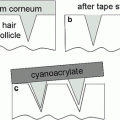
Wissing and Müller (2002a) compared an SLN and a conventional o/w emulsion carrier system for the sunscreen oxybenzone, by studying the in vitro rate of release with a membrane-free model and static Franz diffusion cells. They reported that the release rate could be decreased by up to 50 % with the SLN formulation. Penetration of oxybenzone into stratum corneum on the forearm in vivo was also investigated by a tape stripping method. In congruity with the in vitro data, it was shown that the active release rate could be decreased by 30– 60 % with SLN formulations. In all test models, oxybenzone penetrated into the skin less rapidly and to a lesser extent than from conventional emulsions. The authors concluded that using SLN as a carrier system offers two main advantages. SLNs act as physical sunscreens on their own; therefore, the concentration of molecular sunscreen agents can be decreased while maintaining the formulation sun protection factor. Moreover, SLNs are able to provide a sustained release carrier system, enabling the sunscreen to remain longer at its site of action on the surface of the skin.
Similar results were obtained by Wissing et al. (2002b) when they compared the efficacy of a conventional o/w emulsion and crystalline lipid nanoparticles (CLN) incorporating the sunscreen benzophenone-3. This in vitro study based on the TransporeTM test (3M, USA) (Diffey and Parr 1996) showed that the amount of molecular sunscreen can be decreased by up to 50 % while maintaining the UV protection efficacy, simply because of the particulate nature of the CLN structures.
Nanocapsules (NC) have been introduced as a new generation of carriers for cosmetics and UV blockers for use on human skin and hair. Jimenez et al. (2004) compared the porcine skin permeation of a lipophilic sunscreen, OMC, from different emulsions and encapsulated sunscreen-poly(ε-caprolactone) nanocapsules. Their results showed that the use of NC emulsions decreases the permeation of OMC through pig skin when compared with equivalent w/o and o/w emulsions. NC emulsions are, therefore, novel vehicle-type dispersion systems and can be used advantageously as sunscreen carriers to lower permeation of the active through the skin.
A very promising approach is to entrap the organic UV absorbers in a sol-gel silica glass shell. The product is commercialized under the trade name of Eusolex® UV Pearls™ (Merck, Germany) and is promoted under the slogan Sunglasses for the Skin™. The UV absorber, which is usually an oil or oil soluble compound, constitutes about 80 % of the capsule weight. The products are manufactured as an aqueous dispersion, containing 35 % (by weight) UV absorber(s). The particle size is about 1 micron on average and they do not tend to agglomerate; therefore, they give a “pleasant skin feeling”. The refraction index of the particles is small enough that they appear transparent when applied topically. The glass microcapsules were shown to effectively retain the encapsulated UV absorbers in a series of stress experiments. These include temperature, drying, pressure (e.g., spraying), and incubation with emollients, tensides, and other raw materials, which could have the potential to extract the capsules content. All experiments clearly demonstrated the stability of the capsules. Additionally, different skin penetration experiments showed the maintenance of sunscreen actives on or near the surface of the skin compared to non-encapsulated material.
The encapsulated sunscreens obtained through the Sol-Gel glass-making technology is currently the most promising technology for significantly improved product characteristics. The direct contact between the sunscreen filters and the body tissues is suppressed – the filter is no longer bioavailable. Products with UV Pearls are therefore suited for products for sensitive skin. In addition, the UV Pearls give the product a pleasant sensory feel (Pflücker et al. 2002).
18.5 Physical Properties of Organic Particulate UV Absorbers
Most of the UV filters in use are oil-soluble and, consequently, are incorporated into the oil phase of sunscreen emulsions; however, even solubility in the oil may be problematic. UV absorbers that are poorly soluble in oils and relatively insoluble in water may be micronized to form aqueous dispersions of ultrasmall particle size. The protective performance of these particles depends on size, as both absorption and scattering play a role in the attenuation of UV light. To this end, it is desirable to achieve particle sizes in the submicrometer range.
Herzog et al. (2004a) generated microparticles of a benzotriazole derivative in the 0.16–40 μm range by milling particles in the presence of a dispersing agent. The UV absorption increases with a decrease in particle size, while the light scattering shows a maximum at a certain particle size. These researchers investigated the UV-attenuating properties of particulate organic absorbers as a function of particle size, with special emphasis on the differentiation between absorption and scattering functionalities of the particles (Herzog et al. 2004b). The efficiency of the UV extinction of the dispersion increases with decreasing particle size down to a maximum extinction at a particle size of 80 nm, and the UV extinction decreases for particles smaller than 80 nm indicating an optimum at 80 nm. It was found from reflection spectroscopic measurements that scattering accounts for about 10 %, and absorption 90 %, of the UV-attenuating effect of the particles.
18.6 Inorganic Materials
Micronized TiO2 particles with a diameter of about 15 nm are used in sunscreens as physical UV filters. These particles are suspected to be absorbed through the stratum corneum into the epidermis or dermis via intercellular channels, hair follicles, and sweat glands. This penetration is undesirable because of the risk of damage to DNA and RNA by the photocatalytic effects of the TiO2 after absorption of UV light (Dunford et al. 1997). Furthermore, the particles can activate the immune system and accumulations of these particles in the skin can decrease the threshold for allergies (Granum et al. 2001). The function of the stratum corneum as a barrier against dermal uptake of ultrafine particles was the subject of several investigations, which came to different conclusions concerning the penetration depth of the particles (Pflücker et al. 2001; Tan et al. 1996). Researchers have investigated a number of other ultrafine, inorganic particles, including ceria (CeO2) (Yabe and Sato 2003) and zinc oxide, for efficacy as UV-protectants. Most of these agents are ideal for cosmetic applications because they are relatively transparent to visible light, but have excellent ultraviolet radiation absorption properties, and appear transparent on the skin. However, many of these chemicals exhibit high photocatalytic potential after UV activation (Cai et al. 1991). This reactivity can be lowered by coating of the particles (with amorphous silica for example) or by doping with a metal ion possessing lower valence and larger ionic size.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree