1 Prevention of Future Skin Cancer Nonmelanoma skin cancer (NMSC) is the most common malignancy in humans. More than 3.5 million NMSCs in more than 2 million people are diagnosed in the United States annually.1 In fact, the incidence of skin cancer is higher than new cases of breast, prostate, lung, and colon cancer combined.2 Melanoma incidence rates have been increasing over the past three decades. Most recent data suggest an increase of 2.7% from 2006 to 2010.2 In 2014, melanoma is estimated to be the third and fourth most common cancer in men and women, respectively.2 The total estimated cost of treatment for skin cancer (including melanoma, basal cell carcinoma [BCC]), and squamous cell carcinoma [SCC]) is $8.1 billion each year in the United States.3 Aside from treatment, cost due to lost workdays and restricted-activity days is estimated at $106.2 million annually.2 The high incidence of skin cancer—in addition to the cost and toll on society—is astounding considering that an estimated daily application of only sun protection factor (SPF) 15 in the first 18 years of life would reduce the lifetime incidence of NMSC by 78%.4 These statistics highlight the importance and beneficial effects of skin cancer prevention. Dermatologists have an opportunity to play an active role in the prevention of skin cancer. Primary prevention, as defined by the United States Preventive Services Task Force (USPSTF), involves measures provided to individuals to prevent the onset of a targeted condition, in this case skin cancer.5 Primary prevention methods in general have included counseling patients, involving other physicians, participating in community outreach programs, and endorsing school sun-safety programs and policy. Chemoprevention is also considered a primary preventive measure and will be discussed in greater detail later in this chapter.5 Since NMSC and melanoma share the risk factors of exposure to ultraviolet (UV) radiation and history of severe sunburns, following primary preventive measures aimed at UV protective habits can maximize great outcomes regarding skin cancer prevention. Skin cancer prevention can be enhanced by dermatologists asking patients if they are protecting themselves from the sun and counseling them about the importance of sun protection. The National Ambulatory Medical Care Survey found that only 1.6% of dermatologists reported mentioning sunscreen during patient visits.6 The ability to counsel and distribute educational pamphlets gives dermatologists the opportunity to change a patient’s perspective and understanding of skin cancer. During dermatologic examinations, it would be important to discuss close monitoring and personal protective clothing with patients who have exposure to known chemical risk factors for melanoma such as coal, tar, pitch, creosote, arsenic, radium, and pesticides. It is important to educate patients during visits about skin cancer warning signs and morphology for both melanoma and NMSC. Cheng et al found 59% of self-detected melanomas in situ had an established dermatologist, and many patients attributed this to education obtained during a dermatology appointment.7
1.1 Introduction
1.2 Role of Dermatology
1.2.1 Primary Prevention
Sponsor | URL | Information |
American Academy of Dermatology | Basic dermatology curriculum for physicians and medical students | |
The Skin Cancer Foundation | Educational links and helpful tips for skin cancer prevention | |
The Skin Cancer Foundation | Ideas for getting involved in fundraising, advocacy, schools, and your community | |
American Academy of Dermatology | Links to find a dermatologist, free cancer screening, and educational information | |
The American Cancer Society | http://www.cancer.org/healthy/morewaysacshelpsyoustaywell/acs-skin-cancer-prevention-activities | Skin cancer awareness campaigns and education resources |
The American Cancer Society | http://www.cancer.org/healthy/toolsandcalculators/quizzes/sun-safety/index | Quiz on sun safety |
Centers for Disease Control and Prevention | http://www.cdc.gov/cancer/dcpc/resources/features/SkinCancer/ | Facts about skin cancer, educational links, and sun-safety tips |
National Council on Skin Cancer Prevention | News, ideas to get involved, advocacy, and educational links |
Source: http://www.skincancerprevention.org.
Skin cancer prevention can be even more effective in a multi-disciplinary approach. Dermatologists have the opportunity to enlist the help of fellow physicians, especially primary care providers, to emphasize the importance of the basics of skin cancer prevention including sun and tanning bed avoidance, as well as regular sunscreen use to their patients. The American Academy of Dermatology (AAD) has created a free basic dermatology curriculum that is available to the public ( Table 1.1).8 This curriculum is aimed toward physicians and students. Learning modules include actinic keratosis (AK), SCC, BCC, evaluation of pigmented lesions, and melanoma. These modules are very helpful given they provide instruction and guidance on when to assure or refer the patient. The USPSTF conducted a systematic review of evidence on the effectiveness – of behavioral counseling to prevent skin cancer and found in primary care settings where counseling was given there was a decrease in intentional tanning and an increase in sun-protective behaviors.9 Another finding of this review was that appearance-focused messages were the most successful at reducing late-adolescent women’s intent to pursue indoor training.9 Following this review, in May 2012, the USPSTF recommended counseling fair-skinned children, adolescents, and young adults aged 10 to 24 years about minimizing their exposure to UV radiation to reduce their risk for skin cancer.9 The National Ambulatory Medical Care Survey found that only 0.07% of patient visits had mention of sunscreen and only 0.9% of patients with a history of skin disease were counseled on the importance of sunscreen.6 The results of this survey demonstrate that dermatologists and other health care providers can improve efforts to increase awareness of the importance of skin cancer prevention.
Table 1.1).8 This curriculum is aimed toward physicians and students. Learning modules include actinic keratosis (AK), SCC, BCC, evaluation of pigmented lesions, and melanoma. These modules are very helpful given they provide instruction and guidance on when to assure or refer the patient. The USPSTF conducted a systematic review of evidence on the effectiveness – of behavioral counseling to prevent skin cancer and found in primary care settings where counseling was given there was a decrease in intentional tanning and an increase in sun-protective behaviors.9 Another finding of this review was that appearance-focused messages were the most successful at reducing late-adolescent women’s intent to pursue indoor training.9 Following this review, in May 2012, the USPSTF recommended counseling fair-skinned children, adolescents, and young adults aged 10 to 24 years about minimizing their exposure to UV radiation to reduce their risk for skin cancer.9 The National Ambulatory Medical Care Survey found that only 0.07% of patient visits had mention of sunscreen and only 0.9% of patients with a history of skin disease were counseled on the importance of sunscreen.6 The results of this survey demonstrate that dermatologists and other health care providers can improve efforts to increase awareness of the importance of skin cancer prevention.
Another responsibility of dermatologists is to promote skin cancer prevention and awareness in our communities. National surveys found that only 3 out of 10 adults practice routine sun protection themselves, and approximately 60% of parents applied sunscreen and 25% used shade to protect their children.10 The results of this survey demonstrate the need to raise community awareness of skin cancer by participating in national or local sun protection campaigns. For example, the American Cancer Society has a national skin cancer awareness campaign called “Slip! Slop! Slap! and Wrap.” This slogan promotes slipping on a shirt, slopping on sunscreen, slapping on a hat, and wrapping on sunglasses to protect the eyes. The American Cancer Society has also designated the Friday before Memorial Day as “No Fry Day.” Pool Cool is a sun-safety education program with eight brief sun-safety lessons taught during regular swimming lessons for children and parents.11 Pools that use this program have been found to have better protected pool environments and better sun protection habits among swimmers and lifeguards that result in fewer sunburns.3 A randomized controlled trial conducted in Australia showed that a population screening that included melanoma awareness campaign led to a decrease in thickness of the melanomas diagnosed during the campaign.12,13 A study done in 1999 evaluating the previous 20 years of primary health prevention programs in Australia found a very large shift in knowledge, attitudes, beliefs, and behaviors about sunlight exposure and suntans.14 Participation and support of national campaigns, such as these, could increase the community’s awareness and application of sun-safety.
Studies of immigrants to Australia point to sun exposure during childhood and adolescence as a major risk factor for developing NMSC.15,16 Sunburns in children are a risk factor for development of skin cancer later in life, and it has been shown that building healthier sun protection habits at an early age can lead to better sun protection as adults.17,18 Dermatologists and other health care providers have the opportunity to promote formation of policy in local schools to educate students on healthy sun protection habits. Dermatologists and other health care providers can have a positive impact in school policy by encouraging the creation of school sun-safety strategies such as allowing students to apply sunscreen at school, having play equipment in shaded areas, scheduling recess to avoid peak sun intensity, and encouraging students to wear sun-protective clothing such as hats, long-sleeved shirts, and pants. Sun-safety curriculum has also been shown to play an important role in skin cancer prevention. Such curriculums have been shown to be effective in school-aged children as early as preschool.19 A randomized trial found that interactive multimedia programs in conjunction with teacher instruction improved sun-safety in primary school-aged children.20 Arizona was among the first to require skin cancer prevention as part of their health education curriculum. Since 2004, Arizona has implemented SunWise in grades K-8 in all public schools.21 SunWise is a health and environmental education program that offers participants free materials to promote cross-curricular learning about sun-safety, UV radiation, and ozone science. The SunWise program has had success in raising awareness and increasing sun-safety behaviors.22 This program has also been shown to prevent approximately 11,000 skin cancer cases among participants resulting in saving $4 for every $1 spent.23 Involvement in endorsing local school policy and curriculum that affects younger individuals has great potential to further decrease the incidence of skin cancer.
Patients with previous solar damage and/or existing precancerous lesions can be treated by several means to prevent future skin cancer. The risks and benefits of the preventive treatment should be discussed and agreed upon with the patient to encourage compliance and awareness. The subsequent sections will discuss various methods of prevention and their respective efficacy. Treatments include cryotherapy, light therapy, topical medications, lasers, and chemical peels.
1.2.2 Chemoprevention
Chemoprevention is considered as primary prevention by the USPSTF.5 The subject of chemoprevention is extensive and multiple methods deliver impressive efficacy.24 For the purposes of this chapter, chemoprevention is the treatment of AKs. AKs account for 5.2 million office visits per year and $920 million in treatment.25 The progression of AKs to primary invasive or in situ SCC was found to be 0.6% at 1 year and 2.57% at 4 years.26 Dodson et al estimated a 10-year risk of AKs transforming to SCC to be around 6.1 to 10.2%.27 Hypertrophic AKs measuring less than 1 cm in diameter located on the dorsum of the hand, wrist, or forearm in white patients were found to have a transformation rate of 50%.28 Criscione et al found that approximately 65% of primary SCC and 36% of BCC were originally diagnosed as AKs.26 Treatment can be directed at single or multiple AKs. If multiple less well-defined AKs are present over large areas, then field therapy can be very beneficial. Field therapy targets clinically visible and subclinical AKs. Ultimately, the choice of the method of chemoprevention should be individualized to each patient.
Cryotherapy
Cryotherapy is the treatment of choice for individual AK lesions.29 A multicenter study found a 67.2% clearance rate 3 months after treatment when lesions were frozen for at least 2 seconds, and greater than an 80% clearance rate when AKs were frozen for at least 10 seconds.30 Krawtchenko and colleagues reported 68% clinic clearance and 32% histological clearance 6 weeks after one or two treatments lasting 20 to 40 seconds for each lesion.31 At a 12-month follow-up, 6% of patients who were initially cleared remained clear,31 in contrast to a study done by Lubritz and Smolewski that reports a cure of 98.8% treated lesions with up to 8.5-year follow-up.32 Adverse effects include redness, blistering, crusting, oozing, and ulceration.29 Thai et al reported hypopigmentation resulted in up to 29% of patients treated with a freeze time greater than 20 seconds.30 In a Cochrane review, cryotherapy was found to be significantly inferior to 5-fluorouracil (5-FU) and aminolevulinic acid (ALA) photodynamic therapy (PDT).24
Photodynamic Therapy
PDT involves the selective accumulation of photosensitizing agents that generate reactive oxygen species when exposed to light, which causes selective apoptosis in premalignant and/or malignant cells. ALA and methyl aminolevulinic acid (MAL) are topical photosensitizing agents that are applied for a certain period prior to exposure to blue or red light. A Cochrane review found that neither the topical photosensitizing agent nor the type of light produced significantly different results.24 The review did find that longer incubation periods of 4 hours with ALA resulted in better outcomes compared to shorter incubation times of 0.5, 1, and 2 hours.24 In a randomized clinical trial, PDT showed a 14% better chance of complete clearance compared to cryotherapy after 3 months.33 A single application of 5-ALA patches applied 4 hours prior to light exposure resulted in 89% clearance 12 weeks after treatment.34 At the 12-month follow-up, this same treatment regimen was associated with 24% recurrent lesions.34 Common adverse events include photosensitivity, pain, erythema, and pruritus.33 PDT was found to produce a better cosmetic outcome than cryotherapy and 5-FU.24 Other than cosmetic outcome, PDT is a good choice for patients where compliance may be an issue due to its single application.
5-Fluorouracil
5-FU decreases cell proliferation and induces cell death by interfering with deoxyribonucleic acid (DNA) and ribonucleic acid (RNA) synthesis in cells with high mitotic rates.24 A Cochrane review found that the efficacy of 0.5 and 5% 5-FU was similar when directly compared.24 The review also found that 5-FU efficacy was similar to PDT and superior to cryotherapy.24 A review of studies demonstrated an average complete clearance of 52%.35 Treatment with 5% 5-FU applied twice a day for 4 weeks showed an initial clearance of 96% and a histological clearance of 67%.31 At 12-month follow-up, 35% of the initially clear patients remained clear.31 The Cochrane review found that patients treated with 5-FU treatment experienced substantial skin irritation, but in general was not cause for withdrawal from treatment.24 Possible adverse effects of 5-FU are erythema, erosions, edema, and dryness at the site of application.36,37 The recommended treatment and duration of 5-FU varies with strength and location of application, but usually is between 1 and 4 weeks.
Diclofenac
Diclofenac is a nonsteroidal anti-inflammatory drug in a 2.5% hyaluronic acid gel vehicle. Although the precise mechanism is not understood, the current hypothesis is that the inhibition of cyclooxygenase-2 (COX-2) leads to a decrease in prostaglandin synthesis and may be responsible for inhibition of cell differentiation, induction of apoptosis, alteration of cell proliferation, and inhibition of angiogenesis.38,39,40,41 A meta-analysis of three studies that included 364 patients found cumulative and total lesion scores of zero in 50% of patients treated for 90 days and 33% clearance of patients treated for 60 days.42 A study of six organ transplant patients reported three out of the six completely cleared clinically and histologically after applying diclofenac 3% twice a day for 16 weeks, while the rest achieved at least 30% clearance.43 Akarsu et al reported a complete clearance of 19.1% 24 weeks after starting diclofenac twice daily for 12 weeks, and reported no withdrawals from the study secondary to adverse effects.44 Common adverse effects included itching, scaling, dryness, rash, and erythema, and rarely paresthesia and hyperesthesia.44 One disadvantage of diclofenac is the lengthy treatment period of twice daily application for 60 or 90 days.
Imiquimod
Imiquimod is an immune modulator that enhances innate and acquired immune responses resulting in antiviral and antitumoral activity.45 A meta-analysis of four studies and 393 subjects showed an average complete clearance of 70% in patients treated with 5% imiquimod cream.35 Application of 5% imiquimod three times weekly for 4 weeks and repeating this regimen if initial lesions did not clear after a 4-week rest period produced a histological and clinical clearance of 73 and 86%, respectively, 8 weeks after final treatment.31 Other studies report a complete clearance rate ranging from 48.3 to 57.1%.46,47,48 At a 12-month follow-up, 86% of patients who were initially clear showed sustained clearance.31 Patients with initial clearance were found to have a 25% recurrence rate when applying imiquimod three times per week and a 47% recurrence rate with twice a week application at 16 months.49 Krawtchenko et al also reported that 81% of the patients treated with imiquimod had an excellent cosmetic outcome.31 Complete clearance rates are higher with 5% imiquimod versus 3.75% imiquimod following 8 weeks of treatment.50,51 A Cochrane review found treatment with 5% imiquimod resulted in more participant withdrawal secondary to adverse effects than treatment with 2.5 and 3.75% imiquimod.24 Common adverse events were erythema, scabbing, and flaking ( Table 1.2).47
Table 1.2).47
Chemical Peels
Chemical peels are another option for treating widespread AKs. A variety of chemical peels can be used to treat AKs and should be selected according to the histological level or severity of skin pathology in order to achieve optimal results.61 Medium-depth chemical peels such as 70% glycolic acid with 35% trichloroacetic acid (GA-TCA) and Jessner’s solution with 35% trichloroacetic acid (JS-TCA) penetrate to the papillary dermis and can be used to treat AKs.57 The exfoliative effect of chemical peels stimulates new epidermal growth and collagen formation.61 Chemical peels have shown to alter the epidermis restoring a more normal pattern of columnar cells with increased polarity.61 JS-TCA and GA-TCA at day 60 post peel showed a hyperplastic epidermis and an expanded papillary dermis.57 This study also reported 7% of JS-TCA peels and 15% of GA-TCA peels developed new elastic fibers. In a split face study, JS-TCA was found to produce equivalent results when compared to 5-FU.53 Subjects in this study had a 75% reduction in AKs and experienced no recurrence of AKs for at least 1 year. In addition to treating AKs, Abdel-Daim et al suggest chemical peeling with glycolic acid, salicylic acid, and trichloroacetic acid could serve tumor prevention by removing photodamaged cells.62 This was shown in a study done by Hantash et al which demonstrated a 40-fold lower rate of developing subsequent NMSC after treatment with 30% trichloroacetic acid peel compared to 5-FU and Carbon dioxide (CO2) laser.54 This study also comments on the excellent cosmetic results produced by trichloroacetic acid peels. JS-TCA and GA-TCA peels resulted in erythema, crusting, and swelling within 24 hours with crusting and swelling resolving by day 7.57 Medium- and deep-depth peels have been associated with pigmentary changes for dark-skinned individuals, infections, allergic reactions, improper healing, hypersensitivity, and disease exacerbation.55,56
Lasers
Different ablative laser systems are used to treat AKs. CO2 lasers work by collateral heat which causes collagen synthesis and the sealing of blood vessels and nerve endings leading to less pain and bleeding, whereas Erbium:yttrium aluminum garnet (Er:YAG) lasers work with high water absorption resulting in little collateral heat.63 Re-epithelialization and resolution of erythema following Er:YAG treatment occurred in 5 to 8 days and 3 to 6 weeks, respectively.58 In comparison, re-epithelialization and resolution of erythema following CO2 laser treatment occurred on an average of 7 days and 6 weeks, respectively.64 Jiang et al treated five patients with a mean of 24 lesions per patient with the Er:YAG laser, resulting in a 93% reduction in AKs.58 In a retrospective study, Iyer et al treated 24 patients with an Er:YAG or CO2 laser or a combination of the two lasers, resulting in a 94% reduction of AKs.60 Also in a retrospective study of 25 patients treated in the same manner, Ostertag et al found 80% of patients remained lesion free for 1 year,59 which is similar to the 87% clearance reported by Iyer et al.60 On average, 64% of recurrences occurred 32.7 months after treatment.59 This study also failed to identify a statistical difference in the apparatus used in relation to the recurrence. Histological studies 3 months after Er:YAG treatments showed reversal of epidermal actinic damage, as well as an increase in collagen formation and decreased elastosis in the superficial papillary dermis.58 Ablative laser treatment is believed to provide prophylaxis against future keratosis and cancers.58,59,60,65 Immunohistochemistry analysis 4 weeks after treatment with a CO2 laser found a decrease in expression of p53 and Ki-67.63 A study reported a decrease in the p53 mutation 6 months after CO2 laser treatment, and the authors concluded this treatment theoretically could decrease the risk of malignant transformation.66 Ablative laser techniques offer dramatic improvement of actinic damage of the skin.59 A comparison study of 5-FU and Er:YAG laser resurfacing found laser resurfacing to be more effective in relation to recurrence rates.67 The most common side effects of ablative lasers are infection, hypopigmentation, hyperpigmentation, acne, milia, scar formation, and atrophic and/or easily bruising skin.59 Since Er:YAG lasers cause less tissue damage, they have fewer associated side effects such as scarring and pigmentary changes, along with a decreased recovery period.58
Cyclooxygenase-2 Inhibitors
Epidemiological studies suggest COX-2 may be involved in the pathogenesis of NMSC.68,69,70,71,72,73,74,75 COX-2 is not constitutively expressed in most tissues,76 but can be induced by UV radiation.70,73,77 Celecoxib has been shown to be a selective inhibitor of COX-2.78 Animal models have shown treatment with celecoxib inhibits the development of UV-induced premalignant skin papillomas in mice, which are believed to correspond to AKs in humans.68,69,70,71,72,73 A randomized, double-blind, placebo-controlled trial with oral celecoxib over 11 months in 240 subjects with biopsy-proven NMSC or AK reported a decrease of 58% of NMSC in the treatment group, but failed to show a decrease in AKs.79 Tang et al performed a trial in which patients with basal cell nevus syndrome received celecoxib or a placebo, and found that the treatment group had 26% fewer new BCCs compared to the placebo group at the end of 2 years.80
Antioxidants
Selenium is necessary for the functioning of glutathione peroxidase,81 which acts to reduce DNA-damaging hydroxyl-free radicals.82 Increased dietary selenium in mice has been shown to provide protection from UV-induced skin tumors.83 A prospective study of 485 adults found patients with 1.3 to 2.8 μmol/L of selenium had a decreased incidence of both BCC and SCC.84 However, no significant effect to the incidence of BCC or SCC was found in a randomized controlled trial of patients who were treated with 200 µg/d of selenium for a mean of 4.5 years and a total follow-up of 6.4 years.85 A Cochrane review that included this study found the selenium group was 17% more likely to develop NMSC, in particular SCCs.81 The review also reported a 35% reduction in the risk of other cancer in the treatment group.
Beta carotene has provitamin A (retinol) activity, and is an antioxidant that may reduce free radical damage of DNA after UV exposure.81 Beta carotene supplementation is believed to work by reducing immunosuppression induced by UV radiation.86 In a study of 1,805 patients with a recent history of NMSC, patients received 50 mg of beta carotene daily for up to 5 years without a significant effect on the incidence of new NMSC.87 Oral vitamin A supplements, however, have been shown to significantly reduce the 5-year probability of developing a new SCC.88
1.2.3 Dietary Modifications
Unsaturated fatty acids are major targets for free radical attack81; therefore, it is possible that decreased dietary fats could reduce free radical attack and carcinogenesis.89 A series of animal studies showed a high level of dietary fats intake markedly shortened the time between UV exposure and tumor appearance, and increased the number of tumors that developed.90 Based on diet alone, patients on a higher fat diet were found to be at 4.7 times greater risk for developing AKs during a 2-year period than those on a lower fat diet.89 A comparison study of normal diet and a reduced fat diet over 24 months in 115 people with a history of no more than 2 NMSC found a trend toward significant reduction in the incidence of NMSC at 2 to 5 years.91 Jaax et al further analyzed the results of the study and found a significant difference of new NMSC in subjects on a low-fat diet compared to a normal diet.92
1.2.4 Chemoprevention in Organ Transplant Recipients
Rates of developing AKs and Bowen’s disease in organ transplant recipients (OTR) are 50 to 100 times higher than the general population.93 OTRs are 250 and 10 times more likely to develop SCC and BCC, respectively, than the general population.94 SCC and BCC account for more than 90% of skin cancers in transplant recipients.95,96,97,98 The incidence of skin cancer in renal transplant patients increases with number of years posttransplantation, starting at 7% after 1 year, and increasing to 45% after 11 years, and 70% after 20 years.97 In addition, the incidence of skin cancer is related to age of transplantation with a mean of 8 years posttransplant for those who received transplants at approximately 40 years of age,99 and only 3 years for patients who received transplants after the age of 60.95 Skin cancer development is proportional to the level of immunosuppression.97 Significantly lower CD4 counts have been associated with OTRs with cutaneous carcinomas compared to those without skin lesions.100 SCCs appear to be more aggressive in OTRs.101,102,103,104 This point is demonstrated in a study of cardiac transplant recipients where 27% of all deaths 4 years post-transplant were attributed to skin cancer.105 These studies emphasize the importance of taking preventative measures in this high-risk population to significantly impact morbidity and mortality rates.
Systemic Retinoids
Retinoids are vitamin A derivatives that work by inhibiting cell growth and inducing normal differentiation in malignant cells.106 Although indications for systemic retinoids are not conclusive, it has been suggested that retinoids are most effective in patients with a history of multiple NMSCs.107 Acitretin is a retinoid and has been shown to be effective in the prevention of SCC in renal transplant recipients with a history of NSMC.108 A double-blinded placebo-controlled study demonstrated 42% reduction in premalignant lesions and a 78% risk reduction in developing NMSC within the first year of treatment of acitretin.109 In a crossover study, George et al reported an average increase of 42% of SCCs during drug-free period compared to acitretin period.108 Studies have shown a statistically significant decrease in skin cancers with a robust effect in patients with multiple prior NMSCs.109,110 Systemic low-dose retinoids have been shown to significantly reduce the mean number of SCCs for up to 3 years.111 Systemic retinoids can cause cheilitis, headache, rash, photosensitivity, dry eyes, palmoplantar desquamation, epistaxis, elevate triglycerides, “stickiness,” aggravation of existing musculoskeletal symptoms, nail changes, gastrointestinal side effects, and pruritus.108 A systematic review of oral retinoids found that headaches, rash, musculoskeletal symptoms, and hyperlipidemia were the most common causes of withdrawal from treatment.112 No significant difference in adverse effects was found when comparing 0.4 mg/kg/d acitretin for 12 months versus 0.4 mg/kg/d for 3 months, then 0.2 mg/kg/d for the remaining 9 months.113 Retinoids are effective only during treatment.111 A “rebound” effect on the development of SCCs when acitretin is discontinued has been reported,107,108,109,111 and is most marked in patients taking retinoids longer than 12 months.111 Continued use of systemic retinoids can accelerate osteoporosis, calcification of tendons and ligaments, osteophytes, and bone spurs around joints.114,115
Besides recommending treatments to prevent the transformation of precancerous lesions, dermatologists also perform skilled physical exams that allow for early detection of skin cancers and educate patients on mole mapping and self-skin exams (SSE).
1.2.5 Secondary Prevention
The USPSTF defines secondary prevention as preventive measures that identify and treat asymptomatic persons who have already developed risk factors or preclinical disease, but in whom the condition has not become clinically apparent.5 Dermatologists consistently perform secondary prevention methods in their practice by performing skin examinations, mole mapping, and teaching patients how to perform skin self-examinations. Since Breslow thickness is the strongest prognostic indicator,116 it is important to detect melanoma at the earliest stage possible.
Dermatologists have a positive impact on early detection of melanoma with an increase in incidence of melanoma and a decrease in mortality.117 This study found that the increase in incidence was mostly due to an increase in the diagnosis of stage I cancers.117 Authors attributed this finding to the ease of access to health care due to the high ratio of dermatologists per capita and increased public awareness.117 In addition, patients with an established dermatologist were more likely to be given a diagnosis of melanoma in situ and to have thinner invasive melanoma.7
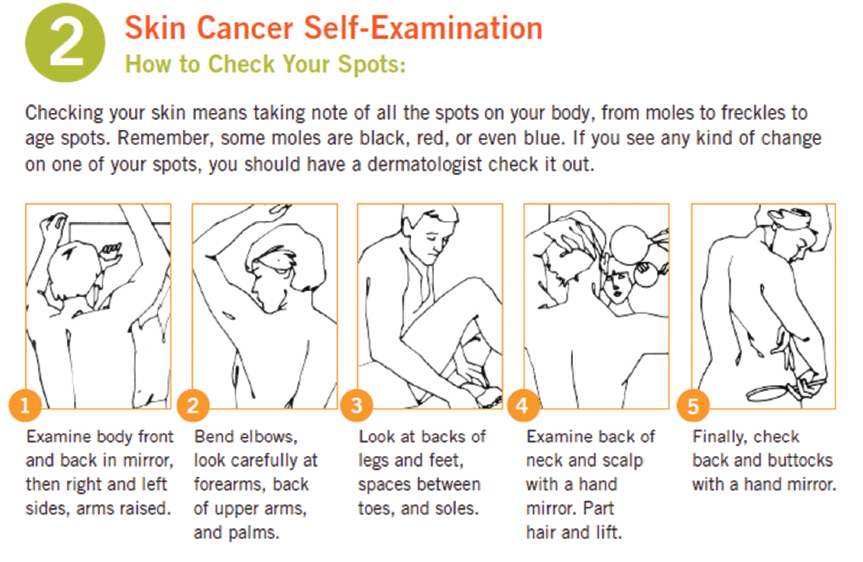
Although currently the USPSTF does not recommend SSE for the general population,118 there is evidence that supports SSEs, especially in high-risk groups. Risk factors for melanoma include personal history of melanoma (5–8%),119,120 family history of melanoma in two or more family members,121 numerous nevi and presence or history of atypical/dysplastic nevi,122,123,124 fair skin (fourfold),125 red hair and blue eyes (twofold),126 history of NMSC (twofold to threefold),127,128 history of sunburns or excessive UV light exposure (twofold),129 and indoor tanning (twofold to threefold).130 The National Comprehensive Cancer Network (NCCN) recommends SSE in patients with a history of melanoma and NMSC.37,131,132 It is estimated that 75% of melanomas are detected by patients themselves or by spouses, friends, or other lay persons.133,134,135,136,137 A retrospective cross-sectional study reported patients detected 41.7% of melanomas in situ, 51% of melanomas, and 62.4% of invasive melanomas.7 A study of 816 melanoma patients found patients who conducted SSE were associated with early diagnosis.134 In a separate study, patients who conducted SSE were found to have significantly thinner melanomas.138 In a large study of 1,062 melanoma patients, 19% had a recurrence that was most often self-detected.139 This study also observed that self-detection independently predicted survival. A literature review showed a wide range of sensitivity and specificity for SSE ranging from 25 to 88% sensitivity and 57 to 97% specificity.140 A randomized controlled trial showed an increase in accuracy and specificity of SSEs when patients received a leaflet with photos of nevi, seborrheic keratoses, and early melanomas.141  Fig. 1.1 is taken from the AAD’s website demonstrating the appropriate method to perform a SSE.142 The SSE can create a feeling of joint responsibility with the dermatologist in early detection of melanoma.143 It is recommended that SSEs be performed frequently at first to become familiar with their nevi and then monthly (
Fig. 1.1 is taken from the AAD’s website demonstrating the appropriate method to perform a SSE.142 The SSE can create a feeling of joint responsibility with the dermatologist in early detection of melanoma.143 It is recommended that SSEs be performed frequently at first to become familiar with their nevi and then monthly ( Table 1.3).143
Table 1.3).143
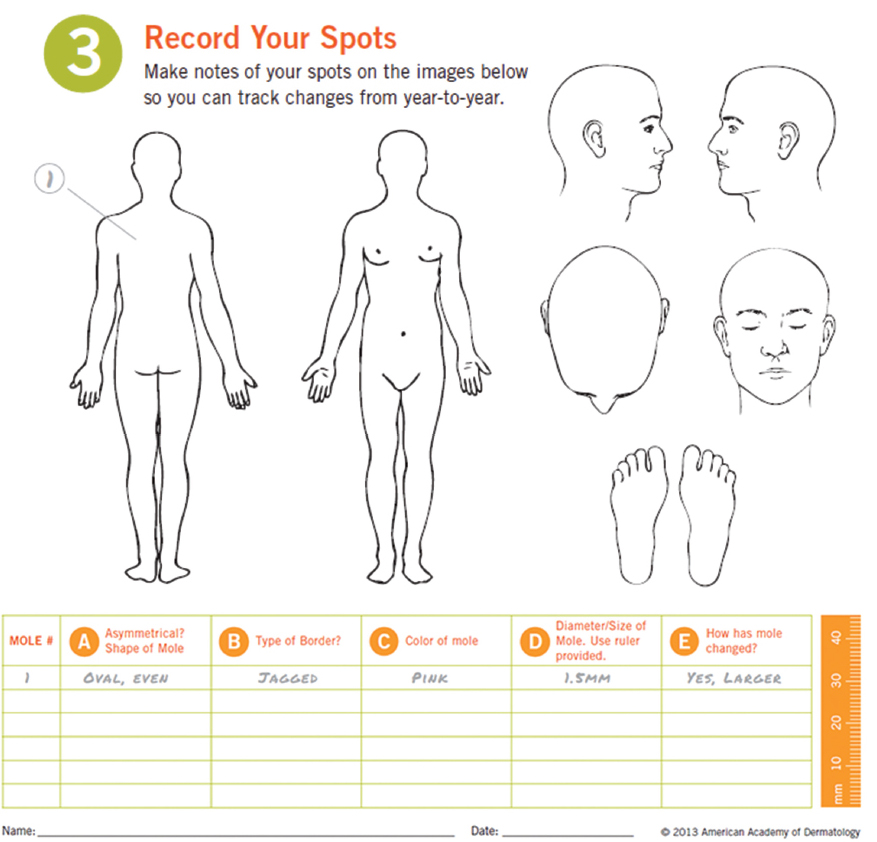
Patients with multiple nevi are at increased risk for developing melanoma compared to the general population. A study found a relative risk of 1.6 for patients with 11 to 25 nevi, 4.4 for 26 to 50 nevi, 5.4 for 51 to 100 nevi, and 9.8 for patients with more than 100 nevi.147 Patients with atypical nevi are also at high risk. The same study reported relative risks of 3.8 and 6.3 for patients with 1 to 5 atypical and more than 6 atypical nevi, respectively.147 It is estimated that 22 to 50% of melanomas show nevus origin.123,148,149 Mole mapping is especially helpful in these individuals. Simply drawing a mole on a mole diagram improved accuracy of reporting new lesions on SSE.150 The ADD has provided an excellent mole map with a lesion log that is demonstrated in ( Fig. 1.2).142 Other studies have demonstrated that the use of digital photography increases the diagnostic accuracy of SSEs.150,151,152 Total nevus number usually peaks by the third decade of life and declines in the seventh or eighth.153 With this in mind, dermatoscopic monitoring can be performed for concerning lesions in younger individuals and total body photography to monitor for new lesions in the elderly.143
Fig. 1.2).142 Other studies have demonstrated that the use of digital photography increases the diagnostic accuracy of SSEs.150,151,152 Total nevus number usually peaks by the third decade of life and declines in the seventh or eighth.153 With this in mind, dermatoscopic monitoring can be performed for concerning lesions in younger individuals and total body photography to monitor for new lesions in the elderly.143
1.3 When to Treat
1.3.1 Nonmelanoma Skin Cancer
The primary goal of treatment of BCC is the cure of the tumor along with maximal preservation of function and cosmesis. In order to achieve this goal, each case should be individualized based on treatment cure rate, preservation of function and cosmesis, and patient’s preference. Tumors with low-risk characteristics and uncomplicated high-risk patients can be effectively treated by dermatologists. Indications, risks, and benefits for different treatment modalities will be discussed later in the book, so this section will focus on characteristics of low-risk tumor characteristics found in  Table 1.4.
Table 1.4.
Fig. 1.1 Self-skin exam. (Reproduced with permission from the American Academy of Dermatology.142 Copyright ©2015. All rights reserved.)
Table 1.3 Risk factors for cutaneous melanoma
Risk factor | Odds ratio | Information |
Personal history of melanoma |
| Standardized incidence ratio 4.5–25.6 |
Family history of melanoma | 1.8 |
|
11–50 common moles > 2 mm | 1.7–1.9 |
|
55–100 common moles > 2 mm | 3.2–3.7 |
|
> 100 common moles > 2 mm | 7.6–7.7 |
|
1–4 atypical nevi | 1.6–7.3 | Atypical moles: ill-defined or irregular border; irregular pigmentation; diameter > 5 mm; erythema (blanchable in lesion or at edge); accentuated skin markings |
Reported growth of a mole | 2.3 |
|
Giant congenital melanocytic |
| Relative risk range 239–1,224 for nevi > 20 cm in diameter extra cutaneous as well as cutaneous melanoma |
Skin that does not tan easily | 1.98 |
|
Light-colored Skin | 1.40–1.42 |
|
Light-colored eyes | 1.55–1.60 |
|
Red or light-colored hair | 1.4–3.5 |
|
Presence of actinic lentigines | 1.9–3.5 |
|
History of nonmelanoma skin cancer |
| |
Female |
| Female:male standardized incidence ratio is 1.3:1 |
| Melanoma is rare in childhood and adolescence but risk increases with age | |
Affluence |
| Relative risk approximately of 3 for least deprived compared to most deprived on Carstairs deprivation category |
Unusually high sun exposure | 2.6 |
|
Indoor tanning |
| A single session can increase the risk of melanoma by 20%144 |
Occupational exposure to coal tar, pitch, creosote, arsenic, or radium145 |
|
|
Pesticide use more than two times per year145 |
|
|
Source: Modified from Scottish Intercollegiate Guidelines Network 2004.146
Fig. 1.2 Mole map and log example. (Reproduced with permission from the American Academy of Dermatology.142 Copyright ©2015. All rights reserved.)
Table 1.4 Characteristics of low-risk recurrence
Basal cell cancer | Squamous cell cancer142 |
Area < 20 mm on trunk and extremities (excluding pretibia, hands, feet, nail units, and ankles) | Area < 20 mm on trunk and extremities (excluding pretibia, hands, feet, nail units, and ankles) |
Area < 10 mm on cheeks, forehead, scalp, neck, and pretibia | Area < 10 mm on cheeks, forehead, scalp, neck, and pretibia |
Area < 6 mm on central face, eyelids, eyebrows, periorbital, nose, lips, chin, mandible, preauricular and postauricular skin/sulci, temple, ear, genitalia, hands, and feet (location independent of size may constitute high risk in certain clinical settings). | Area < 6 mm on central face, eyelids, eyebrows, periorbital, nose, lips, chin, mandible, preauricular and postauricular skin/sulci, temple, ear, genitalia, hands, and feet (location independent of size may constitute high risk in certain clinical settings). |
Primary tumor | Primary tumor |
Not a site of prior radiation therapy | Not a site of prior radiation therapy or chronic inflammatory process |
Immunocompetent | Immunocompetent |
Well-defined borders | Well-defined borders |
Nodular, superficial, keratotic, infundibulocystic, and fibroepithelioma of Pinkus | Well or moderately differentiated on pathology |
Absence of perineural involvement | Absence of perineural or vascular involvement |
| Slow-growing tumor |
| Absence of neurologic symptoms |
| Thickness < 2 mm or Clark levels I, II, III |
Source: Modified from National Comprehensive Cancer Network 2014.131
Note: Risk factors are also increase likelihood metastasis.
1.3.2 Melanoma
Understanding when to treat or when to refer melanoma can be difficult since there are no hard and fast rules. Dermatologists can perform a wide local excision on lesions with a Breslow depth of less than 1 mm and less aggressive histological features. Ulceration and mitoses are also important to consider. A T1b melanoma can be referred to a general surgeon for sentinel lymph node biopsy and/or a medical oncologist for further work-up. Tumor, node, metastasis (TNM) staging criteria are listed in  Table 1.5.154
Table 1.5.154
1.4 When to Refer
1.4.1 Nonmelanoma Skin Cancer
The NCCN recommends that complicated high-risk tumors, regional recurrence, or the development of distant disease be managed by a multidisciplinary tumor board.131 Characteristics of high-risk basal and squamous cell tumors can be found in  Table 1.6. Metastasized SCC to regional lymph nodes has been associated with lymphovascular invasion, poor differentiation, and perineural invasion.155 In addition, patients with regional nodal involvement are at a significant increased risk for recurrence and mortality.156 The NCCN recommends patients with palpable lymph nodes or suspicious lymph nodes on imaging receive a fine needle or core biopsy, and that negative findings be confirmed by re-biopsy and/or re-evaluation.131 In the rare instance of metastasized cutaneous BCC, the NCCN suggests that a trial of vismodegib may be helpful even in the case of metastatic basosquamous carcinoma.131 In the case of meta-static SCC, the NCCN recommended cisplatin monotherapy, cisplatin plus 5-FU, epidermal growth factor inhibitors, or clinical trials.131
Table 1.6. Metastasized SCC to regional lymph nodes has been associated with lymphovascular invasion, poor differentiation, and perineural invasion.155 In addition, patients with regional nodal involvement are at a significant increased risk for recurrence and mortality.156 The NCCN recommends patients with palpable lymph nodes or suspicious lymph nodes on imaging receive a fine needle or core biopsy, and that negative findings be confirmed by re-biopsy and/or re-evaluation.131 In the rare instance of metastasized cutaneous BCC, the NCCN suggests that a trial of vismodegib may be helpful even in the case of metastatic basosquamous carcinoma.131 In the case of meta-static SCC, the NCCN recommended cisplatin monotherapy, cisplatin plus 5-FU, epidermal growth factor inhibitors, or clinical trials.131
Table 1.5 AJCC TNM criteria for cutaneous melanoma
Tis | Thickness | Ulceration status/mitosis |
T1 | N/A | N/A |
| ≤ 1 mm | a: w/o ulceration and mitosis < 1/mm2 |
|
| b: with ulceration and mitoses ≥ 1/mm2 |
T2 | 1.01–2 mm | a: w/o ulceration |
|
| b: with ulceration |
T3 | 2.01–4 mm | a: w/o ulceration |
|
| b: with ulceration |
T4 | > 4 mm | a: w/o ulceration b: with ulceration |
N classification | Number of metastatic nodes | Nodal metastatic mass |
N0 | 0 nodes | N/A |
N1 | 1 node | a: Micrometastasisa |
|
| b: Macrometastasisb |
N2 | 2–3 nodes | a: Micrometastasisa |
|
| b: Macrometastasisb |
|
| c: In-transit met(s)/satellite(s) without metastatic nodes |
N3 | 4 or more metastatic nodes, or matted nodes, or in-transit met(s)/satellite(s) with metastatic node(s) |
|
M classification | Site | Serum LDH |
M0 | 0 sites | N/A |
M1a | Distant skin, subcutaneous, or nodal metastases | Normal |
M1b | Lung metastases | Normal |
M1c | All other visceral metastases | Normal |
Abbreviation: AJCC, American Joint Committee on Cancer; LDH, lactate dehydrogenase; TNM, tumor, node, metastasis.
Source: Modified from Balch et al 2010.154
Table 1.6 Characteristics of high-risk recurrence
Basal cell cancer | Squamous cell cancer |
Area ≥ 20 mm on trunk and extremities (excluding pretibia, hands, feet, nail units, and ankles) | Area ≥ 20 mm on trunk and extremities (excluding pretibia, hands, feet, nail units, and ankles) |
Area ≥ 10 mm on cheeks, forehead, scalp, neck, and pretibia | Area ≥ 10 mm on cheeks, forehead, scalp, neck, and pretibia |
Area > 6 mm on central face, eyelids, eyebrows, periorbital, nose, lips, chin, mandible, preauricular and postauricular skin/sulci, temple, ear, genitalia, hands, and feet (location independent of size may constitute high risk in certain clinical settings) | Area > 6 mm on central face, eyelids, eyebrows, periorbital, nose, lips, chin, mandible, preauricular and postauricular skin/sulci, temple, ear, genitalia, hands, and feet (location independent of size may constitute high risk in certain clinical settings) |
Recurrent tumor | Recurrent tumor |
Site of prior radiation therapy | Site of prior radiation therapy or chronic inflammatory process |
Immunosuppressed | Immunosuppressed |
Poorly defined borders | Poorly defined borders |
Pathology showing morpheaform, basosquamous (metatypical), sclerosing, mixed infiltrative, or micronodular features in any portion of the tumor | Poorly differentiated on pathology |
Perineural involvement | Adenoid (acantholytic), adenosquamous (showing mucin production), or desmoplastic subtypes |
| Rapidly growing tumor |
| Neurologic symptoms |
| Thickness or Clark level ≥ 2mm or IV, V |
| Perineural or vascular involvement |
Source: Modified from National Comprehensive Cancer Network 2014.131
1.4.2 Melanoma
In general, tumors with a Breslow depth of greater than 1 mm should be referred to a surgeon for sentinel lymph node biopsy. It is also important to discuss a general surgeon referral with patients who have T1b cancers and/or a Breslow depth between 0.75 and 1 mm with 1 or more mitoses/mm2. A medical oncologist is an important part of patient care for those with stage II melanoma that exhibits aggressive features such as micrometastases and a deep Breslow depth, or greater than stage III cancer.
1.5 Follow-Up
1.5.1 Nonmelanoma Skin Cancer
It is estimated that 30 to 50% of patients with NMSC will develop another NMSC within 5 years.157 In addition to a 10-fold increased risk of developing a subsequent NMSC compared to the general population,158 they are also at increased risk for developing cutaneous melanoma.159 Nearly 70 to 80% of all cutaneous SCC recurrences develop within 2 years of initial treatment.160 Consequently, it is important to closely follow these patients.
NCCN recommendations for nonmelanoma skin cancer
• Complete skin exam every 6–12 months for life in patients with a history of BCC.
• Complete skin and regional lymph node exam every 3–12 months for 2 years, then every 6–12 months for 3 years, then annually for life for patients with a history of local SCC.
• Complete skin exam with regional lymph node exam every 1–3 months for 1 year, then every 2–4 months for 1 year, then every 4–6 months for 3 years, then every 6–12 months annually for life for patients with regional SCC.
• Patient education on sun protection and self-skin examination regardless of type of skin cancer.
Abbreviations: BCC, basal cell carcinoma; SCC, squamous cell carcinoma.
Source: Modified from National Comprehensive Cancer Network 2014.131
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree