10 Operative Lymphadenectomy Malignant melanoma is the cutaneous malignancy that most often mandates an operative lymphadenectomy. The presence or absence of a positive lymph node in the draining basin is one of the most important prognostic factors in this disease. The rationale for a sentinel lymph node biopsy (SLNB) is discussed elsewhere in this text. For melanoma patients with positive SLNBs, lymph node dissection of the entire nodal basin is currently the standard of care in the United States. Other cutaneous malignancies for which a sentinel node biopsy and lymphadenectomy may be performed include Merkel’s cell carcinomas,1,2 high-risk squamous cell carcinomas (SCCs),3 cutaneous apocrine adenocarcinoma,4 and digital papillary adenocarcinomas.5 The concept of SLNB, as a minimally invasive technique to assess nodal involvement, was developed in 1977 by Cabanas, for the management of penile SCC.6 In 1992, Morton et al applied the same concept to the management of cutaneous melanoma, with the objective to reduce the morbidity related to lymph node dissection in these patients. The SLNB protocol is currently applied to other cutaneous malignancies as well.7 The assumption is that early detection of tumor cells in lymph nodes leads to a better prognosis by assisting in appropriate management by aiding the expeditious removal of other diseased lymph nodes in the affected basin. It also hones the indications for lymphadenectomy, by limiting the procedure only to those situations where an involved node has been definitively identified. This helps to limit the number of lymphadenectomies performed, consequently limiting the morbidity that they entail. In cutaneous melanomas, a completion lymphadenectomy performed for a positive SLN will reveal that only a small percentage of the dissection specimens examined will harbor more involved nodes—that is, nodes other than the sentinel node(s) containing malignant deposits. A representative figure of 15% was quoted by Elias et al from the Massachusetts General Hospital.8 Thus, there is scope for further refinement of this particular intervention. One idea is to use the SLNB as a stand-alone therapeutic procedure. This concept is currently being tested out in clinical trials. The concept of lymphoscintigraphy began with cadaveric studies in the 1800s when Cruickshank used intradermal mercury injections to demonstrate lymphatic drainage patterns.9 It was later discovered that lymphatic drainage is not as dogmatic and predictable as once thought. Although it is possible, based on population studies, to predict which basin a particular cutaneous zone will drain into, a great deal of variability exists, especially in the head/neck and trunk.10 Lymphoscintigraphy allows noninvasive preoperative identification of the primary nodal basin for cutaneous neoplasms.11 This imaging modality is especially valuable in lesions that have the potential to drain to several nodal basins. For example, lesions of the lower extremity can drain to the popliteal, femoral, inguinal, or iliac regions. In a similar fashion, lesions of the anterior scalp can drain to the intraparotid lymph nodes or levels I to V of the anterior cervical chain. In current practice, lymphoscintigraphy is performed by injecting a radiolabeled substance into the intradermal space around the tumor site so that the tracer can be taken up into the lymphatic system. The ideal radiolabeled injectable substance should have the following characteristics12: • It locates and demonstrates the sentinel lymph node without migrating further into the lymphatic chain. • It rapidly clears from the injection site to optimize ease of imaging the draining nodal basin. • The radiotracer should have a high detection rate but a short half-life and low particulate emission to minimize radiation dosage to health care personnel. Radiopharmaceuticals used for lymphoscintigraphy are principally technetium (Tc)-based labels and include 99m-Tc antimony sulfide, 99m-Tc rhenium sulfur colloid, and 99m-Tc albumin nanocolloid. In the United States, 99m-Tc sulfur colloid (50–2,000 nm particle size) is the most popular radiopharmaceutical. Typically, the filtered form of the compound is used. The use of a 0.2-μm filter eliminates particles larger than 200 nm, which allows for optimal uptake and transmission to the primary nodal basin for the lesion ( Identification of a sentinel node, however, does not mandate a lymphoscintigram. Vital dyes (isosulfan blue, methylene blue) have been used to duplicate the results of their radioactive counterparts. In this case, however, the identification of the sentinel lymph node cannot be performed in a noninvasive manner since the process of identification requires making an incision and visually identifying the appropriately stained node. A combination of 99m-Tc sulfur colloid and a blue dye has been reported to yield the best detection of sentinel nodes.13 A recent innovation has been the use of fluorescent imaging in the detection of sentinel lymph nodes.14,15,16 Indocyanine green (ICG), a tricarbocyanine dye with a molecular weight of 751.4Da, is the most commonly used compound for this purpose. ICG has been widely used for a variety of human studies, most commonly in ophthalmology, where it helps in delineating the retinal vasculature. In the context of sentinel node biopsies, the dye is injected in a peritumoral fashion analogous to its radioactive counterpart. The dye track and sentinel node are subsequently visualized using special imaging devices. The principle of fluorescence imaging is based on illuminating the target tissue with light at the excitation wavelength (□750–800 nm) while observing it at longer emission wavelengths, that is, greater than 800 nm. An additional benefit is that the green color of the dye can provide visual confirmation of the node ( Fig. 10.1 The injection site (small arrow) and the uptake in the axilla. Concurrent use of isosulfan blue confirmed the presence of the sentinel node. Note blue lymphatic vessel leading into node. Fig. 10.2 The injection site of ICG in front of the ear (held by forceps) and the uptake in the node adjacent to the skin hook. The visualized node is also stained green. A variety of other techniques such as quantum dots, receptor binding agents, and nanospheres have been used to visualize sentinel nodes on an experimental basis but have not yet been used routinely in the clinical arena. As previously mentioned, the sulfur colloid is injected intradermally to mimic lymphatic drainage of the primary lesion. Using a 25-gauge needle, the tracer is injected to surround the lesion or on both sides of the center of the incisional scar (if the primary tumor has already been excised) in four to six sites. Dynamic and static images are then obtained, 1 to 2 hours after injection, such that all potential nodal basins within the field are visualized. Each static image collection time should last approximately 5 to 10 minutes so that faintly labeled lymph nodes can be detected. The static images are obtained in standard views to allow adequate anatomic localization. Markers can be used to identify stable anatomic landmarks such as the ear canal or clavicle. The addition of SPECT/CT (single-photon emission computed tomography/computed tomography) allows tracer imaging to be superimposed on to the anatomic information obtained by CT and can be useful in head and neck primary melanomas.17 It is worthwhile to consider the timing of preoperative lymphoscintigraphy given that it can be obtained either days in advance of the proposed sentinel node biopsy or on the day of the procedure. Given the logistical issues described earlier regarding timing delays of image acquisition, it can be difficult to schedule lymphoscintigraphy on the day of the procedure. Obtaining imaging in advance of the operative intervention allows for review and confirmation with the interpreting nuclear medicine specialist. The other side of this argument is that a vast majority of these studies are straightforward and do not require detailed analysis. The typical practice is to inject the patient with the radionucleotide on the day of surgery. This obviates loss of the signal and any false positives that might result from the migration of the label into secondary or tertiary lymph nodes. Drainage patterns that do not conform to the norm are significantly more common in the head/neck and trunk.18,19 Interval nodes refer to the presence of nontraditional nodes situated in line of the drainage path between the site of the tumor and the main (traditional) drainage basin. In the head and neck, these patterns include drainage to the postauricular nodes from the anterior forehead or drainage to the occipital nodes from the scalp. Truncal melanomas may drain to the triangular intermuscular space, interpectoral nodes, interval nodes over the costal margin, or even retroperitoneal nodes. The epitrochlear and popliteal regions are a part of the normal lymphatic drainage patterns in the upper and lower limb, respectively, but their involvement is seen in less than 1% of cases ( As with the lymphoscintigram, the radiopharmaceutical is injected in four to six sites intradermally in a peritumoral location. This injection, depending on the institution, is done either by radiologists in the nuclear medicine suite or in the operating room by the surgeon. The uptake in the appropriate basin(s) is confirmed by a Geiger counter prior to the start of the procedure. Occasionally, this may take longer than 5 minutes, especially in the elderly with lower extremity melanomas. The vital dye, if used, can be injected in the operating room, after the induction of anesthesia to prevent unnecessary discomfort to the patient. In the interests of patient comfort, our practice is to inject the dye and the radiopharmaceutical after the induction of anesthesia. The site of the biopsy is included in the operating field and is appropriately prepared. If access to the node involves nerve stimulation, the anesthesiologist is informed, so that appropriate short-acting muscle relaxants are used, if indicated. We use prophylactic antibiotics that are administered immediately prior to the start of the procedure. Fig. 10.3 A subungual melanoma draining to epitrochlear node (smaller inferior dark circle) and a high axillary node (large dark circle) seen below left clavicular marker. The epitrochlear node contained metastatic melanoma. The experience of the last two decades has shown that the “hottest” node, that is, the node with the highest amount of radioactivity, does not translate into a guarantee that the malignant deposit will be found within it. In other words, a less radioactive lymph node may well be the abode of tumor cells. In 2008, Chung et al established 10% rule for SLN for breast cancer.20 These authors suggest that the removal of all the nodes with > 10% of radioactivity of the “hottest node” minimizes the false-negative rate of SLNB. The same criteria have been applied for SLNB in melanoma. Some other criteria used include radioactivity of 10 × background and even 3 × background.11 The sensitivity after removal of three “hot nodes” is 93 to 99% and that after removal of four nodes is 100%.21 The false-negative rate is 1 to 10% using the combined method (dye plus radionucleotide).22 Nonvisualization of sentinel lymph nodes may occur in some cases. In our experience, this has usually been seen in elderly men in the head and neck region. The proximity of the sentinel node site to the primary tumor may obscure the visualization of the sentinel node ( The presence of previous flap surgery may alter the flow pattern of the draining lymph vessels and thus yield false-negative results. In an analogous problem, the presence of a large scar (from an ill-advised attempt at an excisional biopsy) can generate misleading results if the entire length of the scar is included in the injection of the radiopharmaceutical. Under these circumstances, injecting the central part of the scar, the presumed site of the primary lesion, can yield satisfactory results ( Fig. 10.4 Intense radioactivity at site of injection of right mandibular melanoma obscuring sentinel node (left). Low-power histological section depicting resected skin at the 11 o’clock position and the rim of the lymph node (blue) at the 4 o’clock position. Occasionally, anatomic or surgical constraints may dictate whether a sentinel node is accessible ( The presence of a rich lymphatic network in and around the parotid gland complicates the harvest of sentinel nodes in the head and neck region.23 The preauricular lymph nodes, situated in the sulcus between the external auditory meatus and the parotid gland, are usually small and are therefore difficult to detect. This difficulty increases when the sentinel node is detected within the substance of the parotid gland. Based on traditional teaching, the recommended method of harvesting these nodes is via a superficial parotidectomy with a formal identification of the facial nerve in order to minimize the chances of damaging it. Another option is to directly access the sentinel node in the parotid using a classic facelift incision and carefully dissecting through the substance of the gland, using a nerve stimulator if necessary, to harvest the node.24 At our center, we have used both methods and currently prefer the technique of directly harvesting the sentinel node without a formal parotidectomy. In our series of 58 patients, we did not encounter a single case of facial nerve palsy. Interestingly, a survey of Head and Neck surgeons emphasized the wide variability in approaches to this problem, with as many as 48% of the surgeons surveyed opting for a superficial parotidectomy.25 It must be emphasized that the parotid sparing approach for sentinel node harvest should only be undertaken by experienced surgeons, failing which a parotidectomy is always the safer option. Frozen sections, in general, are not recommended for the analysis of sentinel nodes in melanomas.26 These require a special expertise on the part of the examining pathologists which may not be universally available. The presence of benign nevus cells in the subcapsular region that may be mistaken for cells of metastatic melanoma further heightens the degree of uncertainty during such analyses. A final consideration for lymphoscintigraphy in melanoma (and other malignancies) is how lymphatic drainage and therefore radiotracer localization may be affected by regional meta-static disease. There is evidence that micrometastases do not affect mapping techniques or SLNB identification. Additionally, there are some studies that demonstrate normal or near-normal lymphatic drainage patterns despite lymph node metastases. Sentinel node biopsies, generally speaking, are very safe procedures. Seromas and hematomas probably constitute the most common problems. To limit the occurrence of the former problem, we are liberal in the use of drains in our practice. Wound-healing problems such as infection and wound dehiscence are similarly rare. There have been reports of lymphedema occurring after the harvest of sentinel nodes alone but these situations, too, are extremely rare. Planning for operative lymphadenectomy begins with thorough history and physical exam at the initial encounter with the patient. Neck, groin, or axillary bulges, masses, or discomfort can be the first presentation of a clinically positive regional lymph node in a patient with melanoma and rarely in other cutaneous cancers. Physical exam should include examination of all possible drainage basins based on tumor location. Factors in this exam might be of considerable importance in the conduct of these procedures. Some examples include the presence of previous scars that might modify placement of incisions or impact the vascularity of poorly placed ones, problems affecting the cervical spine or the shoulder which might limit optimal positioning and exposure of the operative site of the patient, or the presence of inguinal of femoral hernias that might go unnoticed or unrecognized leading to possible damage in the dissections of this area. Certain common features apply to the dissection of the major lymph nodal basins, that is, cervical, axillary, and inguinofemoral and pelvic basins. • Positioning is of utmost importance. A poorly positioned patient can significantly add to operating time. • Communication with the anesthesia team is critical if nerve testing is to be performed so that long-acting paralytic agents are not administered. • The resection of large tumor masses that may result in significant bleeding should necessitate a preoperative type and cross-match of blood, if this is not routinely performed. • Along the same lines, if an unusually long or bloody procedure is anticipated, the preoperative placement of a Foley catheter may be helpful. This is mandatory in patients with pelvic or groin dissection to prevent inadvertent damage to the bladder, especially in the elderly. Fig. 10.8 Melanoma (circled) in a patient with history of spinal fusion with rib harvest from left chest wall and a midline incision over the spinous processes for Harrington’s rod placement. Lymphoscintigram revealed drainage to paravertebral nodes. An attempt at biopsy was abandoned owing to difficulty of access under Harrington’s rods. • Sequential compression devices are routinely used on the lower extremities to prevent deep vein thrombosis. • Postoperatively, one must be vigilant about lymphedema, especially with the lower limb, pelvic, and groin dissection and to a lesser degree in the upper limb. The importance of this preoperative discussion cannot be overemphasized. The prophylactic use of controlled pressure stockings may be of some benefit in high-risk patients, such as the obese ones. • The adequacy of the lymphadenectomy can be judged using the number of nodes harvested during the procedure. Thus, Spillane and colleagues from the Sydney Melanoma Unit offer the following benchmarks.27 Axillary dissections should aim for a minimum of 10 nodes, inguinal and ilioinguinal dissections for a minimum of 7 nodes, and cervical lymph node dissections involving 4 or more levels should aim for a minimum of 20 nodes. The cervical lymph nodes are the most common initial site of regional metastases for melanomas of the head and neck. As mentioned previously, patterns of lymphatic spread can generally be predicted based on the location of the primary tumor. The anterior scalp, ear, and face typically drain first to the intraparotid node. These structures as well as the anterior neck can also drain to the cervical nodes including levels I to V ( The posterior scalp and posterior neck typically drain to level V cervical nodes as well as the retroauricular and suboccipital node. The discussion of head and neck lymphadenectomy is incomplete without a mention of SCCs of the lip and cheek. SCCs account for nearly 95% of lip cancers. The size of the primary tumor is one of the most significant predictors for prognosis.28 For example, T2 lesions have a 5-year survival rate of approximately 90%, while T3 lesions have a 40 to 60% survival rate at 5 years. Regional metastases to the lymphatic system seem to occur less frequently in SCCs of the lip as compared to other SCCs of the head and neck. Lower lip SCCs metastasize to the submandibular nodes in 70 to 90% of cases of regional spread.28 Isolated nodal spread is present in 75% of regional metastases for SCCs of the lip, and 25 to 45% of these cases involved the submental, level I nodes. SCCs of the upper lip typically spread to the submandibular or lower intraparotid nodes. Fig. 10.9 Division of nodal groups by subzones in both lateral (left) and anterior view (right). Level I: Submental and submandibular nodes. This group includes the lymph nodes located within the submental triangle, bounded by the anterior belly of the digastric muscle and the hyoid bone. The submandibular group includes the lymph nodes located within the boundaries of the anterior and posterior bellies of the digastric muscle, the stylohyoid muscle, and the body of the mandible. The submandibular gland, which is located within this cervical space, should be removed when this nodal group is included in the resection. IA, submental nodes; IB, submandibular nodes. Level II: Upper jugular nodes. This group contains the lymph nodes located around the upper third of the internal jugular vein and the spinal accessory nerve. It goes from the level of the skull base superiorly to the level of the inferior border of the hyoid bone and carotid bifurcation inferiorly. The posterior boundary is the posterior border of the sternocleidomastoid muscle, and the anterior boundary is the lateral border of the sternohyoid and stylohyoid muscles. IIA, upper jugular nodes anterior to the XI nerve; IIB, upper jugular nodes posterior to the XI nerve. Level III: Middle jugular nodes. This group includes the lymph nodes located around the middle third of the internal jugular vein. The boundaries of this space are the inferior border of the hyoid bone and the carotid bifurcation superiorly, the inferior border of the cricoid cartilage, and the junction of the omohyoid muscle with the internal jugular vein inferiorly, the posterior border of the sternocleidomastoid muscle posteriorly, and the lateral border of the sternohyoid muscle anteriorly. Level IV: Lower jugular nodes. This nodal group contains the lymphatic structures located around the lower third of the internal jugular vein. Its boundaries are the inferior border of the cricoid cartilage and the omohyoid muscle superiorly, the clavicle inferiorly, the posterior border of the sternocleidomastoid muscle posteriorly, and the lateral border of the sternohyoid muscle anteriorly. Level V: Posterior triangle. This group includes the lymph nodes located along the transverse cervical artery and lower half of the spinal accessory nerve as well as the supraclavicular lymph nodes. The boundaries are the anterior border of the trapezius muscle posteriorly, the posterior border of the sternocleidomastoid muscle anteriorly, the clavicle inferiorly, and the convergence of the sternocleidomastoid and trapezius muscles superiorly. VA, lymph nodes in the posterior triangle located above the level of the inferior border of the cricoid cartilage; VB, lymph nodes in the posterior triangle located below the level of the inferior border of the cricoid cartilage. Level VI: Anterior compartment. This level contains the pre- and paratracheal nodes, precricoid (Delphian) node, perithyroidal nodes, and the lymph nodes along the recurrent laryngeal nerves. The boundaries are the hyoid bone superiorly, the suprasternal notch inferiorly, and the carotid arteries laterally.
10.1 Operative Lymphadenectomy
10.1.1 Introduction
10.2 Sentinel Node Biopsy: Technical Aspects
10.3 Lymphoscintigraphy
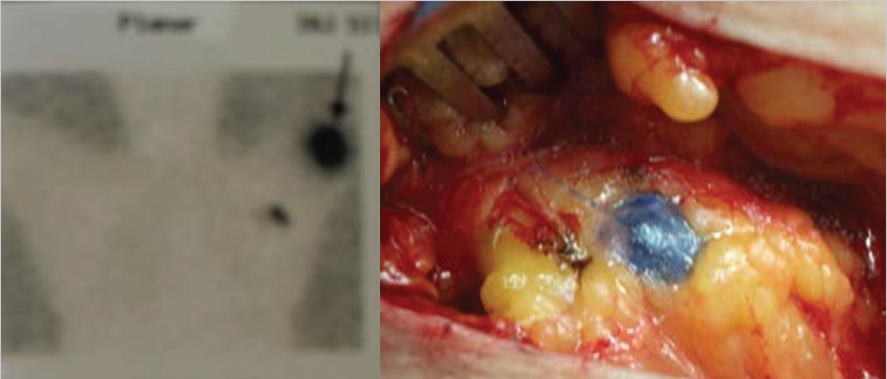
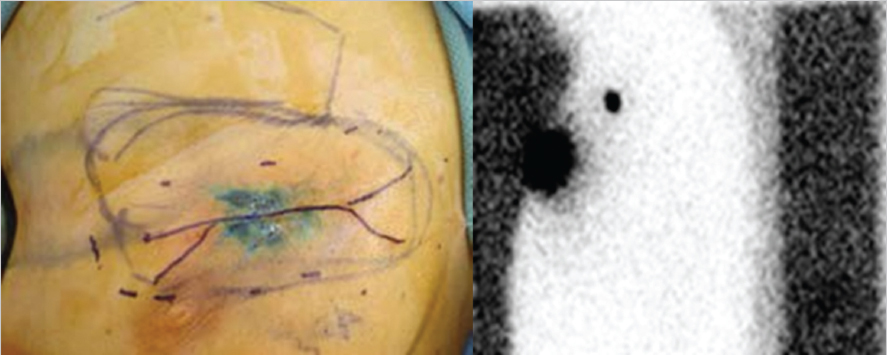
 Fig. 10.1).
Fig. 10.1).
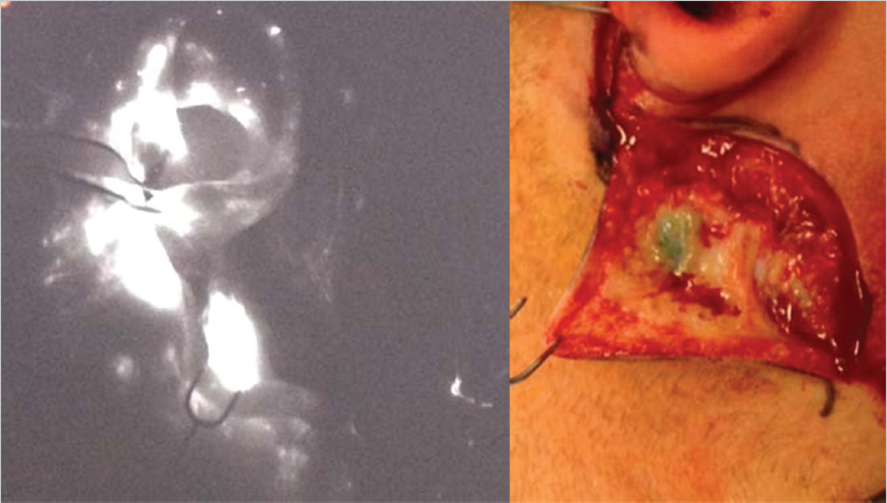
 Fig. 10.2). Further research is warranted to reliably establish the efficacy and utility of ICG for SLNB.
Fig. 10.2). Further research is warranted to reliably establish the efficacy and utility of ICG for SLNB.
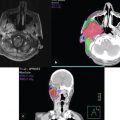
10.4 Performance of Lymphoscintigram
10.4.1 Timing of the Lymphoscintigram
10.4.2 Aberrant or Interval Drainage Patterns
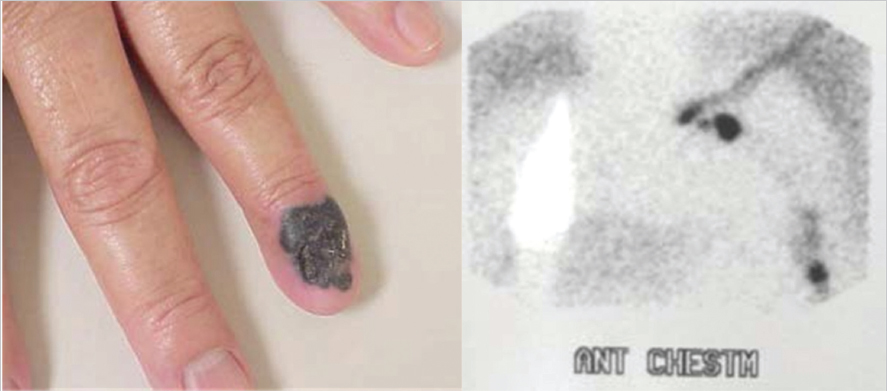
 Fig. 10.3).
Fig. 10.3).
10.5 Performance of the Sentinel Node Biopsy
10.5.1 Identification of the True Sentinel Node
10.5.2 Difficulties in Sentinel Node Identification
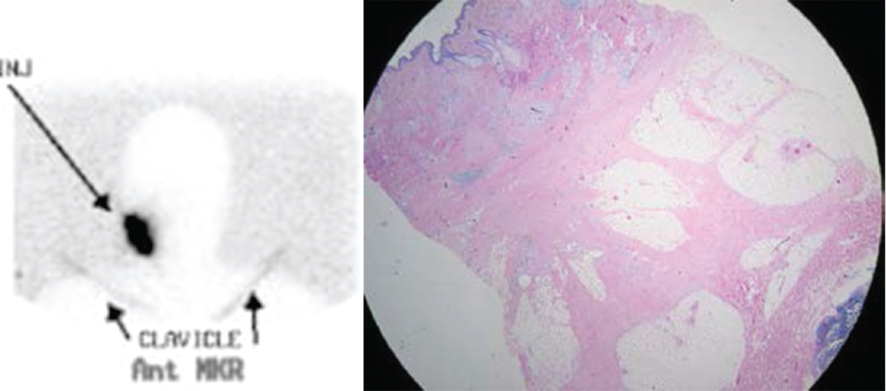
 Fig. 10.4). In this scenario, it may be helpful to excise the primary lesion (and the principal source of the obscuring radioactivity) before looking for the sentinel node as mentioned earlier. Another solution to the problem is to combine the use of vital dye with the radioactive tracer such that the sentinel lymph node can be visually confirmed without the distraction of heightened background radioactivity.
Fig. 10.4). In this scenario, it may be helpful to excise the primary lesion (and the principal source of the obscuring radioactivity) before looking for the sentinel node as mentioned earlier. Another solution to the problem is to combine the use of vital dye with the radioactive tracer such that the sentinel lymph node can be visually confirmed without the distraction of heightened background radioactivity.
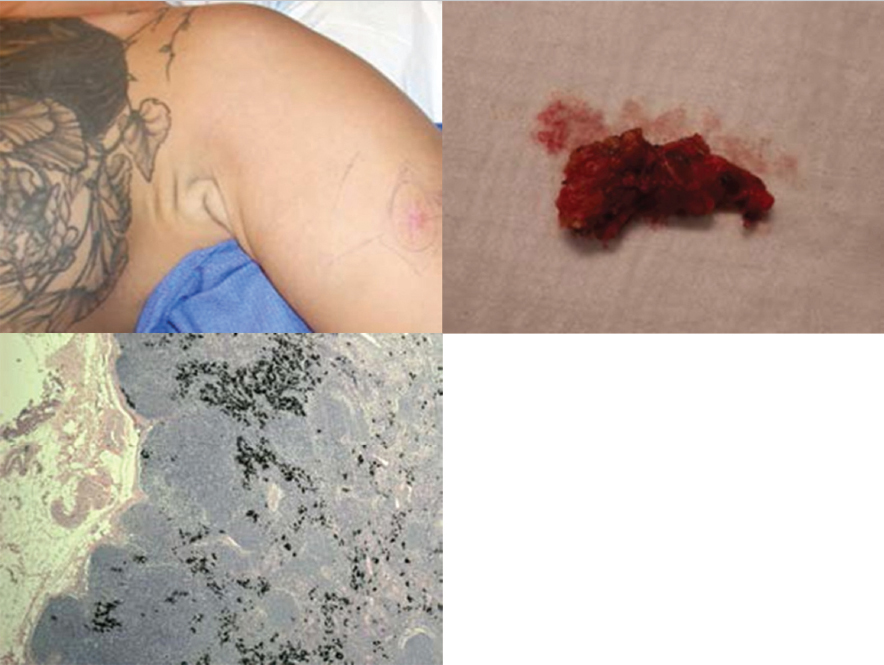
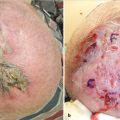
 Fig. 10.5). The presence of tattoos in the catchment area of the lymph node may lead to confusion with melanoma cells in the sentinel lymph nodes (
Fig. 10.5). The presence of tattoos in the catchment area of the lymph node may lead to confusion with melanoma cells in the sentinel lymph nodes ( Fig. 10.6). The presence of concurrent melanomas of differing prognostic significance, in the same anatomic region, may mandate the sequential lymphoscintigraphic assessment to accurately delineate the lymph nodal basins involved (
Fig. 10.6). The presence of concurrent melanomas of differing prognostic significance, in the same anatomic region, may mandate the sequential lymphoscintigraphic assessment to accurately delineate the lymph nodal basins involved ( Fig. 10.7).
Fig. 10.7).
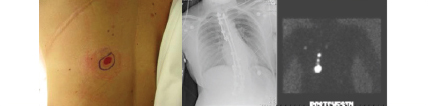
 Fig. 10.8).
Fig. 10.8).
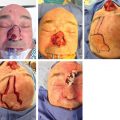
10.5.3 Special Considerations in the Head and Neck Region
10.5.4 Frozen Sections in the Identification of Sentinel Nodes
10.5.5 Complications of Sentinel Node Biopsy
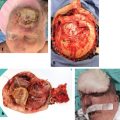
10.6 Operative Lymphadenectomy
10.6.1 General Considerations
10.7 Neck Dissection
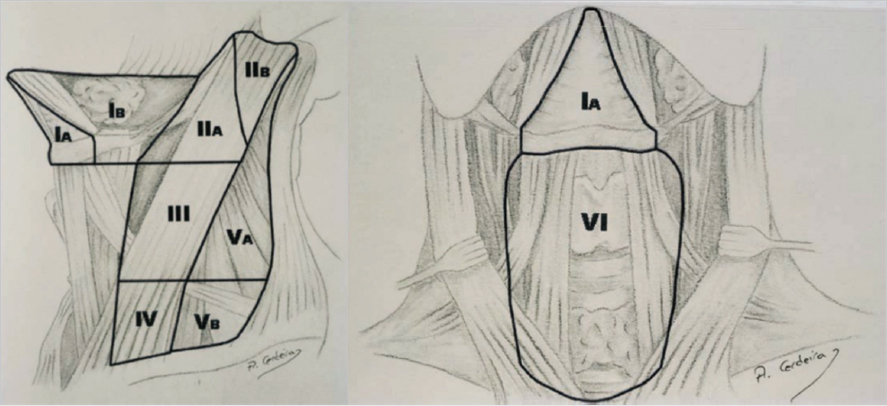
 Fig. 10.9). A complete (radical) neck dissection has become standard of care in patients with melanomas of the head and neck who have a positive SLNB or those patients with clinically positive lymph nodes.
Fig. 10.9). A complete (radical) neck dissection has become standard of care in patients with melanomas of the head and neck who have a positive SLNB or those patients with clinically positive lymph nodes.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree