6 Mohs Micrographic Surgery’s Role in the Treatment of Skin Cancer Mohs micrographic surgery (MMS) is a specialized skin cancer excision technique that combines surgery with pathology. It is most commonly performed under local anesthesia in an outpatient setting and offers the advantages of higher cure rates for cutaneous tumors ( Advantages of Mohs micrographic surgery: • Tissue sparing. • Superior cure rates. • 100% margin examination. • Continuity of care: surgeon and pathologist. • Outpatient. • Local anesthesia. • Closure after histologic confirmation of clear margins. • Cost-effective. Skin cancer is the most common cancer. Of the nonmelanoma skin cancers (NMSCs), 95% are basal cell carcinomas (BCCs) and squamous cell carcinomas (SCCs). MMS has an evidence-based, 5-year cure rate of 99% for BCC9,10 and 94% for cutaneous SCC ( The mounting evidence of the benefits of MMS provides patients with quality, evidence-based care (see list above). However, MMS is one treatment modality in the large armamentarium of a cutaneous surgical oncologist. It is important to understand the advantages and disadvantages of multiple techniques including standard excision, MMS, radiotherapy, chemotherapy, and oral hedgehog pathway inhibitors. The objective of this chapter is to familiarize one with the MMS technique, illustrate histologic examination of the surgical margins in MMS, and discuss variations as well as tactics to allow integration in a multidisciplinary setting. Mohs surgery training can be obtained through an Accreditation Council for Graduate Medical Education (ACGME) approved fellowship,12 which has been renamed “micrographic surgery and dermatologic oncology” in September 2014. These fellowship-trained Mohs surgeons undergo 1 to 2 years of additional training after a 4-year dermatology residency. The fellowship involves gaining proficiency in cutaneous oncology, tumor resection with orientation on maps, tissue preparation, dermatopathology and histologic interpretation on frozen sections, and wound reconstruction. Dermatology residency, as a prerequisite, establishes a substantial clinical, surgical, and pathologic foundation for fellows to achieve expertise in clinicopathologic correlation. The dermatologic surgeon must also develop a mastery of frozen section histology interpretation, cutaneous and soft tissue anatomy, and advanced facial reconstruction. Central to surgical treatment of cutaneous neoplasms is the concept of surgical margins. It is crucial for the surgeon to be familiar with histologic grossing techniques and their implications. The final histologic reading of a surgical margin is only as informative as the method in which tumor tissue is processed and what is actually examined. Developing a false sense of security regarding negative margins may result in unexpected tumor recurrence. Strong communication between the surgeon and the pathologist is critical given there may be subjectivity and inconsistencies in the histologic interpretation of surgical margins. A clinical margin is defined as the clinically apparent edge of the tumor, while a surgical margin refers to the specific measurement of the tissue surrounding the clinically evident tumor.13 The term “close to the margin” illustrates the existing ambiguity. In a survey of pathologists, the definition of “close” surgical margins varied from tumor involvement of within a few cells to 0.1 to 5 mm adjacent to the tumor edge. In these cases of “close” surgical margins, SCCs were re-excised, while BCCs were not.14 Surgeons need to be aware of pathologists’ interpretation of margins because it will affect the need for subsequent surgical therapy or adjunctive therapy. Furthermore, regardless of the surgical technique, without clear understanding of how the tissue is processed, the final reading of clear margins is limited by the processing technique. A thorough discussion regarding laboratory techniques is discussed in Chapter 11. The focus here will be on the comparison between the most commonly used histological grossing method, vertical (perpendicular/bread loafing), and horizontal (Mohs) sectioning. The routinely utilized technique to process excision specimens involves transverse, vertical sections often compared to slices in a loaf of bread (“bread loafing”). This technique allows great visualization of both the tumor and the margins of each examined section. Its limitation is only a small percentage of the true surgical margin is viewed with this sectioning technique ( It is important to note that many laboratories are willing to cut more sections at narrower intervals to gain more precise important diagnostic information on specimens when indicated. Good communication is key and may require discussion with the dermatopathologist prior to grossing of the specimen. Of note, the actual grossing of the specimen may not actually be done by the dermatopathologist but may be done by a technician or resident.15 With the Mohs processing technique, 100% of the deep and peripheral margins are evaluated ( Tumors may have contiguous but asymmetric growth patterns and margins may be difficult to predict. The surgical margin is determined by the clinical margin, and with asymmetrical and infiltrative growth patterns, some tumors may extend histologically beyond the clinically evident margins. With standard vertical sectioning technique, areas of asymmetric infiltrative tumor growth may be missed ( Another advantage of MMS is the same physician serves as the pathologist and surgeon. The physician is in control of the specimen and is directly involved in sectioning and inking, and may assist in the grossing of the specimen. The Mohs surgeon is able to appreciate the histology during the procedure, which can be overlooked with the sampling errors in biopsies. For example, a biopsy of a tumor may reveal nodular BCC, but with the Mohs processing, it is common to discover that the tumor is actually an infiltrative BCC with further histologic extension than originally predicted. Mohs surgeons can correlate the clinical appearance of the tumor and note any discordance between the diagnosis of the biopsy sample and the tumor seen under frozen sections. Certain staging criteria like perineural invasion can also be appreciated during MMS, and adjunctive treatment can be recommended. Currently, approximately 25% of skin cancers are treated with MMS.19 MMS is ideally suited for skin cancers at high risk of recurrence, those located in regions requiring tissue conservation, and/or when complete margin control is necessary. Many NMSCs have aggressive local growth patterns and can invade vital structures such as the eye, nose, lips, and ears, leading to significant destruction and impairment. With time and neglect, metastasis may develop in more aggressive tumors. Fortunately, many of the NMSC are low risk and can be treated with superficial ablative techniques (e.g., electrodesiccation and curettage) with comparable cure rates and cost-effectiveness (see Section 6.4.3 Cost-Effective Care). Cutaneous tumors that can be treated with MMS are listed in Fig. 6.4 Mohs layer excision with narrow margin and subsequent flattening of skin edges. The tissue is horizontally sectioned so the skin edge (epidermis) is in the same plane as the deep margin, thus ensuring complete lateral and deep margin assessment. As 100% of the margin is examined, there is no concern that tumor may be missed with serial sectioning. Reprinted with permission from CCF. The definition of “high-risk” skin cancer is dependent on multiple factors: clinical and histologic characteristics of the tumor, the anatomic site, and history of prior treatments (see text box). MMS has the ability to track unpredictable superficial or deep projections of tumor with the meticulous and complete histologic examination of the surgical margins. Additionally, tumors that involve embryonic fusion planes, periorbital, nerves, and bone can be followed with MMS. In these situations, the adeptness of this technique is most evident as deep and irregular tumor extensions are responsible for the higher recurrence rates with other techniques. With recurrent tumors, it is especially difficult to achieve clear margins with recommended margins, given that the subclinical extension is often prominent and clinically it is difficult to delineate the tumor from scar. MMS has a superior cute rate for recurrent tumors ( Table 6.2 Cutaneous tumors treated with Mohs surgery and reported cure rates
6.1 Introduction
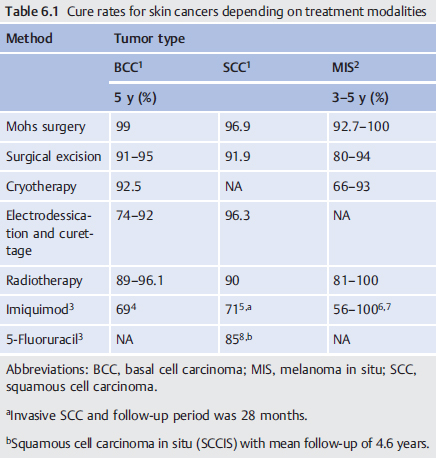
 Table 6.1) along with maximal tissue preservation. Conceptualized by general surgeon, Dr. Frederic E. Mohs, the method is based on the theory that a tumor arises from a single focus and has contiguous spread. By utilizing fresh frozen tissue processing and horizontal sectioning techniques, the Mohs surgeon excises the tumor and examines 100% of the microscopic tissue margin. The complete surgical margin examination in MMS provides the highest, evidence-based cure rate for cutaneous malignancies, while conserving the maximum amount of normal tissue (tissue-sparing). From a functional, aesthetic, and reconstructive perspective, tissue-sparing is especially important for skin cancers near the orbit, nose, lips, ears, anogenital region, and digits (
Table 6.1) along with maximal tissue preservation. Conceptualized by general surgeon, Dr. Frederic E. Mohs, the method is based on the theory that a tumor arises from a single focus and has contiguous spread. By utilizing fresh frozen tissue processing and horizontal sectioning techniques, the Mohs surgeon excises the tumor and examines 100% of the microscopic tissue margin. The complete surgical margin examination in MMS provides the highest, evidence-based cure rate for cutaneous malignancies, while conserving the maximum amount of normal tissue (tissue-sparing). From a functional, aesthetic, and reconstructive perspective, tissue-sparing is especially important for skin cancers near the orbit, nose, lips, ears, anogenital region, and digits ( Fig. 6.1a, b). The proven higher cure rate provided by MMS is especially valuable for recurrent tumors and those with more aggressive histologic features and subclinical extension (see list below).
Fig. 6.1a, b). The proven higher cure rate provided by MMS is especially valuable for recurrent tumors and those with more aggressive histologic features and subclinical extension (see list below).
 Table 6.1).1,11 MMS is also the preferred treatment method for skin cancers that are clinically poorly defined, recurrent, and/or have a higher risk of recurrence because of higher-risk histologic features, or specific anatomic location (
Table 6.1).1,11 MMS is also the preferred treatment method for skin cancers that are clinically poorly defined, recurrent, and/or have a higher risk of recurrence because of higher-risk histologic features, or specific anatomic location ( Fig. 6.2a,b).
Fig. 6.2a,b).
6.2 Training
6.3 Surgical Margins: Tissue Processing Techniques
6.3.1 Vertical (Bread Loafing) Sectioning
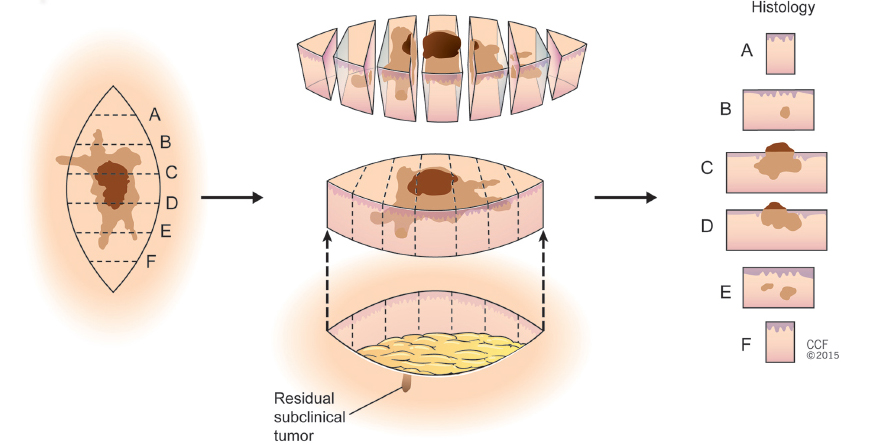
 Fig. 6.3). The specimen is typically placed in formalin, divided into 2 to 3 mm in width sections, placed in numbered cassettes and embedded in paraffin (formalin-fixed, paraffin-embedded [FFPE]). Within each section, a sample of tissue is sliced with the knife perpendicular to the epidermis and cut down to the base. Step sectioning cuts tissue at specified intervals, which can allow a faster view of different levels but can result in skip areas, while serial sectioning completely processes the specimen with unbroken sequence. With vertical sections, it is estimated that approximately 1% of the true surgical margin is evaluated. In theory, serial vertical sectioning could be done in a fashion that 100% of the margin is evaluated. This would be highly impractical, requiring sectioning and examination of hundreds of slices, and is not normally done in practice.15 For example, 1 cm of tumor would produce 2,000 5-μm sections. One study demonstrated that vertical sectioning is about 44% sensitive for distinguishing residual facial BCC at the surgical margin excised with a 2-mm margins.16
Fig. 6.3). The specimen is typically placed in formalin, divided into 2 to 3 mm in width sections, placed in numbered cassettes and embedded in paraffin (formalin-fixed, paraffin-embedded [FFPE]). Within each section, a sample of tissue is sliced with the knife perpendicular to the epidermis and cut down to the base. Step sectioning cuts tissue at specified intervals, which can allow a faster view of different levels but can result in skip areas, while serial sectioning completely processes the specimen with unbroken sequence. With vertical sections, it is estimated that approximately 1% of the true surgical margin is evaluated. In theory, serial vertical sectioning could be done in a fashion that 100% of the margin is evaluated. This would be highly impractical, requiring sectioning and examination of hundreds of slices, and is not normally done in practice.15 For example, 1 cm of tumor would produce 2,000 5-μm sections. One study demonstrated that vertical sectioning is about 44% sensitive for distinguishing residual facial BCC at the surgical margin excised with a 2-mm margins.16
6.3.2 Horizontal (Mohs) Sectioning
 Fig. 6.4). Tissue is excised with a surgical bevel of approximately 45 degrees. After the layer is removed and inked, the epidermal edge is pressed down to be in the same plane as the deep margin. The tissue is embedded in mounting medium (optimum cutting temperature [OCT] compound) and frozen. This frozen tissue block is sectioned in a horizontal fashion so that the knife is parallel to the deep margin of the subcutaneous tissue and the newly pressed epidermis. This bottom-up approach represents the true deep and peripheral surgical margin. Therefore, the specimen represents the entire deep and peripheral margin in one plane. Some critics argue that no tissue processing technique including Mohs consistently examines 100% of the margin due to incomplete sections, fragmented tissue, or orientation problems.15 However, with Mohs sectioning, at least 95 to 99% of the margin can be evaluated, which is far more precise than the 1% margin examined in the vertical sectioning.17,18 This complete margin examination is the basis for the maximal normal tissue preservation and higher cure rate for challenging tumors.
Fig. 6.4). Tissue is excised with a surgical bevel of approximately 45 degrees. After the layer is removed and inked, the epidermal edge is pressed down to be in the same plane as the deep margin. The tissue is embedded in mounting medium (optimum cutting temperature [OCT] compound) and frozen. This frozen tissue block is sectioned in a horizontal fashion so that the knife is parallel to the deep margin of the subcutaneous tissue and the newly pressed epidermis. This bottom-up approach represents the true deep and peripheral surgical margin. Therefore, the specimen represents the entire deep and peripheral margin in one plane. Some critics argue that no tissue processing technique including Mohs consistently examines 100% of the margin due to incomplete sections, fragmented tissue, or orientation problems.15 However, with Mohs sectioning, at least 95 to 99% of the margin can be evaluated, which is far more precise than the 1% margin examined in the vertical sectioning.17,18 This complete margin examination is the basis for the maximal normal tissue preservation and higher cure rate for challenging tumors.
 Fig. 6.3).
Fig. 6.3).
6.4 Indications and Contraindications for Mohs Micrographic Surgery
 Table 6.2. The indications for MMS are not absolute, and any treatment decision needs to be made in collaboration with the patient’s preferences, performance status, and understanding of other available treatment options and cure rates (
Table 6.2. The indications for MMS are not absolute, and any treatment decision needs to be made in collaboration with the patient’s preferences, performance status, and understanding of other available treatment options and cure rates ( Table 6.1).
Table 6.1).
 Table 6.2). The dilemma to clear the tumor with larger surgical margin resulting in a larger surgical defect and possible less aesthetic outcome and reduced functionality versus a more conservative margin with the possibility of positive margins and another surgery is a constant challenge for those treating facial tumors.37
Table 6.2). The dilemma to clear the tumor with larger surgical margin resulting in a larger surgical defect and possible less aesthetic outcome and reduced functionality versus a more conservative margin with the possibility of positive margins and another surgery is a constant challenge for those treating facial tumors.37
Cutaneous tumors treated with Mohs surgery and reported cure rates | ||
Cutaneous tumors | Cure rates | |
| 5 y | 10 y |
Basal cell carcinoma • Primary • Recurrent | • 99%9 • 93%10 | • 95.6%20 • 96.1%20 |
Squamous cell carcinoma • Primary • Recurrent • Erythroplasia of Queyrat • Keratoacanthoma | • 92–99%11 • 90%1 • 90%21 • 97.5%22 (2 y) | |
Melanoma in situ, including lentigo maligna | ||
Atypical fibroxanthoma | 93–100%26 | |
Dermatofibrosarcoma protuberans | ||
Merkel cell carcinoma | 84%30 | |
Microcystic adnexal carcinoma | ||
Sebaceous carcinoma | 89%34 | |
Extramammary Paget’s disease (EMPD) | 92%35 | |
Leiomyosarcoma (superficial) | 87%36 | |
Malignant fibrous histiocytoma | – | |
Angiosarcoma | – | |
Melanoma | – | |
Apocrine carcinoma | – | |
Lymphoepithelioma-like carcinoma | – | |
Eccrine porocarcinoma | – | |
Indications for Mohs Surgery for Basal Cell Carcinoma and Squamous Cell Carcinoma38
Tumor Characteristics
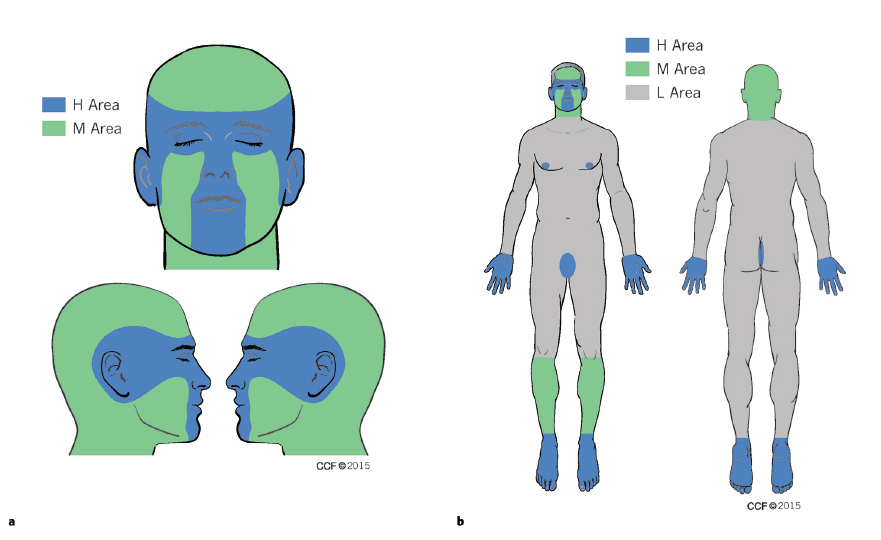
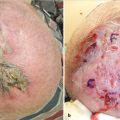
• High-risk* anatomic location (Area H): “Mask areas” of face (central face, eyelids, eyebrows, nose, lips, chin, ear, and periauricular skin/sulci, temple), genitalia (perineal and perianal), hands, feet, nail units, ankles, and nipples/areola ( Fig. 6.2a, b).
Fig. 6.2a, b).
• Large size: Area L ≥ 20 mm, Area M ≥ 10 mm, Area H ≥ 6mm ( Fig. 6.2a, b).
Fig. 6.2a, b).
• Aggressive histologic subtype:
 BCC: morpheaform (sclerosing), micronodular, or infiltrating.
BCC: morpheaform (sclerosing), micronodular, or infiltrating.
 SCC: poorly or undifferentiated, spindle cell, acantholytic, sclerosing, metatypical, small cell, clear cell, desmoplastic, KA type (central face), lymphoepithelioma-like, and sarcomatoid.
SCC: poorly or undifferentiated, spindle cell, acantholytic, sclerosing, metatypical, small cell, clear cell, desmoplastic, KA type (central face), lymphoepithelioma-like, and sarcomatoid.
• Recurrent.
• Positive margin on excision.
• Poorly defined clinical border.
• Perineural/vascular* invasion.
• Breslow depth ≥ 2 mm.*
• Rapid growth.*
• Neurologic symptoms.*
Patient Characteristics
• Prior exposure to ionizing radiation.*
• Chronic inflammatory process (scar).*
• Immunocompromised: human immunodeficiency virus (HIV), organ transplant, hematologic malignancy, or pharmacologic immunosuppression.
• Predisposing genetic syndromes: xeroderma pigmentosa, basal cell nevus syndrome.
• Patient known to have high-risk tumors without other health risk factors.
*Squamous cell carcinoma (SCC) specific.
According to the National Comprehensive Cancer Network (NCCN), a multidisciplinary expert panel, MMS is the preferred surgical technique for high-risk BCCs and SCCs as it provides superior intraoperative surgical margin analysis.39 This signifies the importance of complete tissue margin analysis for high-risk tumors. If MMS is not available, a viable alternative would be excision with recommended margins and complete circumferential peripheral and deep-margin (CPDM) assessment using intraoperative frozen sections. A meta-analysis of MMS demonstrated a 5-year disease-free survival rate of 99% for BCC and 97% for SCC.1,9 A prospective, randomized controlled trial with 10-year follow-up revealed significantly lower recurrence rates for recurrent and primary facial BCC treated with MMS compared to standard excision.20
In addition to BCCs and SCCs, MMS has been utilized effectively for recurrent benign tumors that tend to recur, such as desmoplastic trichoepithelioma, infantile digital fibroma, and granular cell tumor, as well as other rare cutaneous malignancies26,27,28,29,30,31,32,33,34,35,36 ( Table 6.2). In these cases, the complete margin examination MMS provides enhanced tumor clearance.
Table 6.2). In these cases, the complete margin examination MMS provides enhanced tumor clearance.
6.4.1 Melanoma Including Lentigo Maligna
The role of MMS with only frozen sections for the treatment of cutaneous melanoma is controversial because the margin taken is often narrower than published guidelines as well as the challenge in detecting atypical melanocytes in frozen sections (freeze artifact).40 The use of special immunohistochemical (IHC) stains, such as human melanoma black 45 (HMB-45), Mel-5, Melan-A (MART-1), and S100, can increase histologic sensitivity during MMS for melanocytic lesions ( Table 6.3).41 Many dermatologic surgeons still advocate for the examination of FFPE tissue margins to confirm that the final MMS margin is negative.42 Some Mohs surgeons with extensive experience with management of melanoma with MMS suggest that outcomes are equivalent to standard margins.43,44 MMS for melanoma may be advantageous in certain clinical scenarios such as locally recurrent melanoma, large diameter tumors, ill-defined or amelanotic melanoma, and melanoma near critical anatomic structures such as the genitals, digits, eyelids, nose, and ears.
Table 6.3).41 Many dermatologic surgeons still advocate for the examination of FFPE tissue margins to confirm that the final MMS margin is negative.42 Some Mohs surgeons with extensive experience with management of melanoma with MMS suggest that outcomes are equivalent to standard margins.43,44 MMS for melanoma may be advantageous in certain clinical scenarios such as locally recurrent melanoma, large diameter tumors, ill-defined or amelanotic melanoma, and melanoma near critical anatomic structures such as the genitals, digits, eyelids, nose, and ears.
The lentigo maligna subtype of melanoma in situ is considered an exception and MMS as a treatment is becoming increasingly accepted. Lentigo maligna (melanoma in situ) is often a clinically ill-defined tumor typically found on the head and neck with a high local recurrence rate with standard wide local excision, due to the unpredictable histologic tumor extension beyond the clinically predicted margins. Several studies have found the recommended 5-mm margin for melanoma in situ inadequate for complete removal of lentigo maligna.40,45 Given these challenges, conventional MMS is one possibility for treatment of lentigo maligna because it offers the possibility of complete margin examination and higher cure rate along with tissue sparing in critical areas. A challenge is that frozen tissue processing may result in morphologic artifacts where keratinocytes can resemble melanocytes and the true margin can be difficult to interpret. Rapid IHC stains41 are helpful, but it still may be challenging to reliably detect single cell change and nests without significant training and experience. One review demonstrated that nearly 25% of lesions initially diagnosed as melanoma in situ were found to have a dermal component upon re-excision.23 Therefore it is recommended that the debulked clinical tumor be sent for permanent sections to evaluate for dermal invasion.
Due to these challenges, there are several modifications to MMS for treatment of lentigo maligna and melanoma. Some techniques include wide excision Mohs surgery, “slow” Mohs staged excision using FFPE sections, and geometric staged excision (GSE; see the “Modifications of Mohs Micrographic Surgery” section),41,42,45 which aim to achieve complete margin control with FFPE pathology.46,47
Table 6.3 Commonly used rapid immunohistochemical stains for Mohs micrographic surgery
Tumor | Immunohistochemical stains |
Melanoma | HMB-45, Melan-A (MART-1), S100 |
Squamous cell carcinoma | Cytokeratin (AE1/AE3) |
Extramammary Paget’s disease | Cytokeratin 7 |
Dermatofibrosarcoma protuberans | CD34 |
Abbreviation: HMB-45, human melanoma black 45.
6.4.2 Appropriate Use Criteria
In 2012, evidence-based appropriate use criteria for Mohs micrographic surgery (Mohs AUC) was developed using the RAND Corp protocol with direction of the NCCN guidelines.38,48 Two hundred and seventy clinical scenarios based on tumor location ( Fig. 6.2a, b), size, recurrence history, histology, and patient health status were presented to an expert panel to be graded as appropriate, inappropriate, or uncertain based on available evidence. The goal of the Mohs AUC was to provide guidance in the rational use of MMS.38,49 The Mohs AUC tables can be downloaded at the American Academy of Dermatology World Wide Web site (http://www.aad.org/education/appropriate-use-criteria/mohs-surgery-auc). There is also a smartphone app (Mohs AUC) that can be downloaded and referenced for point-of-care decisions (http://www.aad.org/education/appropriate-use-criteria/mohs-auc-app).
Fig. 6.2a, b), size, recurrence history, histology, and patient health status were presented to an expert panel to be graded as appropriate, inappropriate, or uncertain based on available evidence. The goal of the Mohs AUC was to provide guidance in the rational use of MMS.38,49 The Mohs AUC tables can be downloaded at the American Academy of Dermatology World Wide Web site (http://www.aad.org/education/appropriate-use-criteria/mohs-surgery-auc). There is also a smartphone app (Mohs AUC) that can be downloaded and referenced for point-of-care decisions (http://www.aad.org/education/appropriate-use-criteria/mohs-auc-app).
6.4.3 Cost-Effective Care
With the escalating cost of health care, it is important to be mindful of meaningful use of therapies while providing evidence-based quality care to patients. The studies on the costeffectiveness of MMS compared to standard excision with local anesthesia are variable depending on the study location and methods.50,51 With the use of the Mohs AUC in addition to the aforementioned advantages of the procedure, MMS is a costeffective treatment for skin cancer.9,10 When standard excision with FFPE sections and immediate repair is compared with MMS, the cost differential is comparable or slightly less expensive with the former (standard excision with immediate repair). However, when factoring the physical location of where the surgery is performed (office-based vs. ambulatory center vs. operating room), type of anesthesia used (local vs. general), processing method for the specimen (frozen vs. permanent), type of reconstruction (immediate vs. delayed), the size of the defect (tissue-sparing vs. wide local excision), the need for additional surgery for recurrence, and the intangible aspects (stress associated with waiting for the results of permanent sections, returning for a second surgery), MMS is an economical skin cancer treatment.19,50,51,52,53
6.4.4 Contraindications
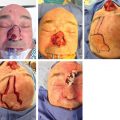
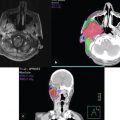
There are no absolute contraindications to MMS. There are limitations and challenges to the technique such as discontiguous tumors, certain histologic features, and patient characteristics. As the basis of MMS is grounded on a contiguous tumor, discontiguous tumors can result in false-negative margins and inadequate margin control. However, this is also a concern with other methods of evaluating surgical margins. If a tumor cannot reliably be identified histologically, especially on frozen sections, this can also be a relative contraindication. Patients who are unwilling or unable to undergo a time-intensive surgery may do better with a nonsurgical approach such as radiotherapy or standard excision. Nevertheless, for some patients with complex medical conditions and comorbidities, MMS done under local anesthesia may be preferable over surgery performed under general anesthesia.54,55,56 MMS may not offer a decreased morbidity or mortality in cases of metastases or tumors deeply invading structures such as the orbit, parotid, or bone. MMS can assist in peripheral margin clearance in conjunction with surgical colleagues performing the more extensive deep tumor resection possibly under general anesthesia. These cases are rare and MMS can be utilized to decrease tumor burden and prevent possible invasion of vital structures57 or assess peripheral margins (see below, Case 2).
6.5 Mohs Micrographic Surgery Technique
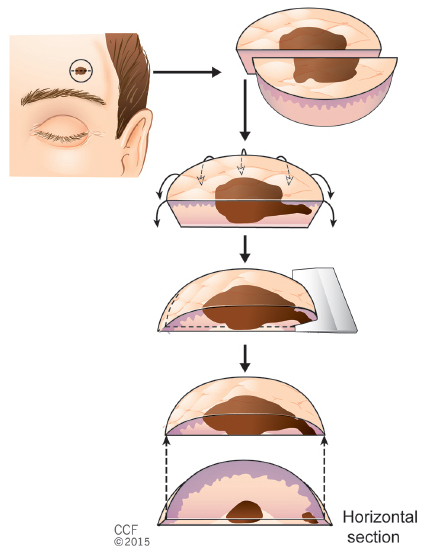
During MMS, the Mohs surgeon has complete control of the excised tissue, including laboratory processing and interpretation of the tissue sections, as well as precise mapping. MMS involves four major components: tumor extirpation, histopathologic examination, precise mapping, and wound management ( Fig. 6.5 and
Fig. 6.5 and  Fig. 6.6). The tumor extirpation component consists of removing a disc of tissue, often referred to as “layer” or “stage,” with a beveled surgical margin (
Fig. 6.6). The tumor extirpation component consists of removing a disc of tissue, often referred to as “layer” or “stage,” with a beveled surgical margin ( Fig. 6.6c). This ensures that the entire 360 degrees of tumor margin is circumferentially removed and examined. A beveled plane of excision allows the tissue specimen sides to be flattened completely into a single plane, allowing complete examination of the peripheral and deep margin of the excised tissue (
Fig. 6.6c). This ensures that the entire 360 degrees of tumor margin is circumferentially removed and examined. A beveled plane of excision allows the tissue specimen sides to be flattened completely into a single plane, allowing complete examination of the peripheral and deep margin of the excised tissue ( Fig. 6.4 and
Fig. 6.4 and  Fig. 6.6d). The histopathologic component involves cutting horizontal sections and mounting them onto glass slides, followed by staining and microscopic examination for residual tumor (
Fig. 6.6d). The histopathologic component involves cutting horizontal sections and mounting them onto glass slides, followed by staining and microscopic examination for residual tumor ( Fig. 6.6f). Precise mapping involves marking any residual tumor on the “map” of the excision (
Fig. 6.6f). Precise mapping involves marking any residual tumor on the “map” of the excision ( Fig. 6.6g). This three-step process is then repeated until there is no residual tumor identified microscopically (
Fig. 6.6g). This three-step process is then repeated until there is no residual tumor identified microscopically ( Fig. 6.5). In the fourth step, wound management, the tumor-free defect is surgically closed (
Fig. 6.5). In the fourth step, wound management, the tumor-free defect is surgically closed ( Fig. 6.6k) or allowed to heal by second intention. The specific way each step is achieved may vary between individual surgeons.
Fig. 6.6k) or allowed to heal by second intention. The specific way each step is achieved may vary between individual surgeons.
6.5.1 Preoperative Assessment
After a given tumor is deemed appropriate for MMS, a consultation including a history and focused skin examination should be performed, which may be done the same day as the surgery. Indications for preoperative and postoperative antibiotic prophylaxis should be evaluated.58 The management of any implanted devices including pacemakers, defibrillators, and deep brain stimulators should be considered and affect choice of hemostatic device. Medication and supplement history is noted. The use of anticoagulants should be managed accordingly. Typically, patients are asked to continue anticoagulants especially if used to prevent a life-threatening event as the risk/benefit ratio favors anticoagulation.59,60 However, in collaborative cases, it is important to discuss with other physicians involved. Patient should be thoroughly questioned regarding allergies including local anesthetics, antibiotics, iodine, latex, and tape products. A social history regarding smoking, alcohol use, and home assistance with wound care is necessary. Chronic alcohol dependence or abuse can result in increased intraoperative bleeding, and smoking can result in poor wound healing due to decreased tissue oxygenation. Gaining an appreciation of the patient’s understanding of the procedure will be important to prevent patient dissatisfaction.