Abstract
Treatment of facial pain must distinguish between classic trigeminal neuralgia, due to chronic, intracranial, vascular compression and posttraumatic facial pain due to direct injury to one or more branches of the trigeminal nerve. This chapter describes an approach to treating posttraumatic facial pain that is based upon the principles of peripheral nerve surgery. A neuroma is resected and the proximal end must be implanted in an area away from direct trauma. No randomized controlled studies exist. The evidence for the approach listed for each branch of the trigeminal nerve is Level IV from small cohort series of patients. Approaches to the cervical plexus and occipital nerves are included to complete the approach to pain in the entire craniofacial region.
Keywords
facial pain, neuroma, posttraumatic trigeminal neuralgia, migraine
Background
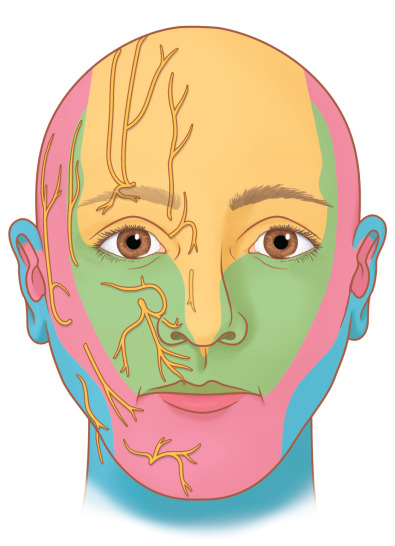
In order for a person to perceive facial pain, there must be a peripheral nerve pathway for neural impulses to travel from the site of injury into the central nervous system. The classic facial pain, “trigeminal neuralgia” or “tic doloureux” is not what this chapter is about, as that perceived pain originates within the central nervous system. Facial injury most often involves branches of the trigeminal nerve (TN) to the forehead (frontal branch, V 1 ), midface (maxillary branch, V 2 ), and mandible (mandibular branch, V 3 ) ( Fig. 3.15.1 ).
There is no published epidemiologic study about the incidence or prevalence of posttraumatic facial pain. The most recently reported description of 21,000 craniofacial injuries in almost 10,000 patients did not include facial pain. There have been observational studies related to the frontal bone (V 1 ) where 5 (13%) of 43 patients had chronic frontotemporal “headache pain.” These patients each had a fracture of the anterior table of the frontal sinus. In another observational study, related to zygoma fractures (V 2 ), 76% of 25 patients, at a mean of 6.3 months post-injury, were found to have abnormal cutaneous pressure thresholds in the distribution of the infraorbital nerve on the side with the fracture. The difference between the 13% prevalence of facial pain, and the 76% prevalence of sensory abnormalities is due to the difference in the question asked and the method of investigation in the study of craniomaxillofacial trauma.
While the TN is a cranial nerve, once its branches leave the cranium, they become covered with Schwann cells, and therefore behave like a peripheral nerve in that they can regenerate. If they can regenerate, then they can form a painful neuroma. This insight provides the basis of treatment of painful neuromas of the trigeminal nerve. While injuries and painful sequelae of injured trigeminal nerves have been described classically, and those descriptions remain valid, what remains unresolved is the surgical approaches to treat injuries to V 1 ,V 2 , and V 3 . An approach to the treatment of posttraumatic facial pain has been described in which the TN is considered as a peripheral nerve.
As with every peripheral nerve injury, if the nerve provides sensibility to an area of critical importance, like the lip, then reconstruction is the appropriate surgical choice. If the nerve does not provide critical sensibility to an area, like the forehead, then interruption of nerve function, with appropriate treatment to the proximal end of the nerve, is the surgical choice. The “appropriate treatment” for the proximal end of the injured TN will be discussed in detail later in this chapter ( Box 3.15.1 ).
- •
If a nerve provides critical sensibility to an area, like the lip, then reconstruction is the appropriate surgical choice.
- •
If a nerve does not provide critical sensibility to an area, like the forehead, then resection (interruption of function) of that nerve is the appropriate surgical choice.
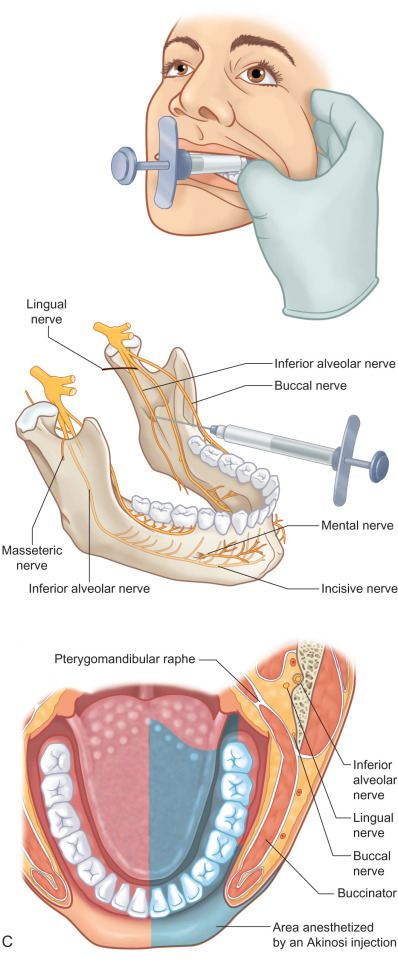
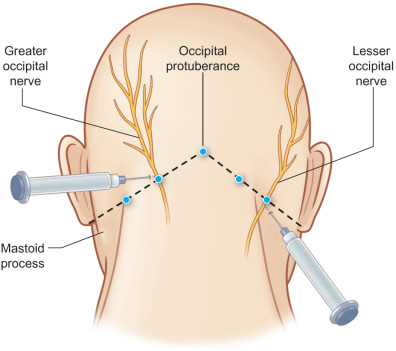
For the purposes of this chapter on posttraumatic facial pain, the “face” will be defined as including skin that is innervated by branches of the cervical plexus, as these can be injured and the pain perceived as facial in origin ( Fig. 3.15.1 ). Also for the purposes of this chapter, “headaches,” including frontal, temporal, nasal, and occipital, will be included to provide a basis for approaching posttraumatic pain in a surgical manner distinct from the approach to the classic migraine, which is not posttraumatic in origin. Additionally, posttraumatic occipital injuries may give pain that radiates to the frontal region, and needs to be distinguished for diagnosis and appropriate treatment by doing nerve blocks ( Figs. 3.15.2 and 3.15.3 ) or Botox® (onabotulinumtoxinA) injections.
Aesthetic surgery of the face and neck can be considered as trauma, and, as such, this chapter will include facial pain that results secondary to cosmetic surgery, and the surgical approaches to correct this origin of posttraumatic facial pain.
Finally, this chapter will not include discussion of intrinsic temporomandibular joint (TMJ) disorders, although a peripheral nerve approach to denervation of the TMJ has been described ; nor will this chapter discuss other rare pain disorders, such as Eagle’s syndrome (orofacial pain related to the stylohyoid ligament), or persistent burning mouth syndrome.
Differential Diagnosis of Posttraumatic Facial Pain
Posttraumatic facial pain must first be proven to be due to an injury to a peripheral nerve that is part of the craniofacial skeleton. This implies that there has to be a history of trauma to the head and neck region. A history of facial or cranial surgery is a history of trauma.
History
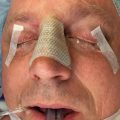
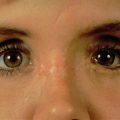
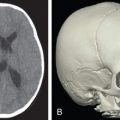
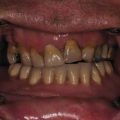
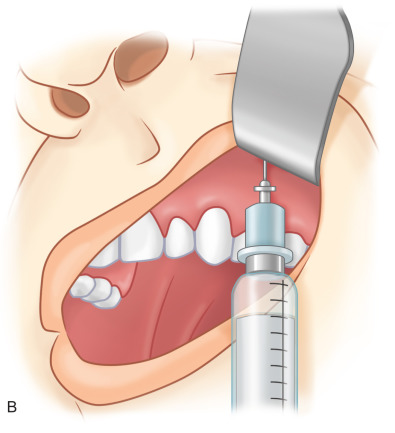
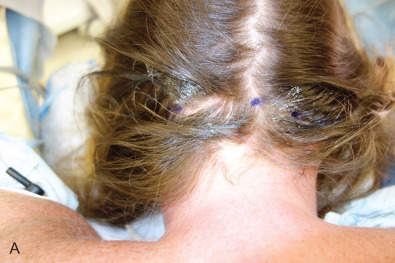
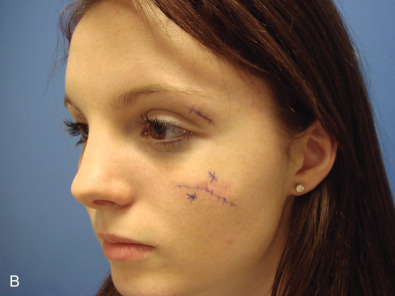
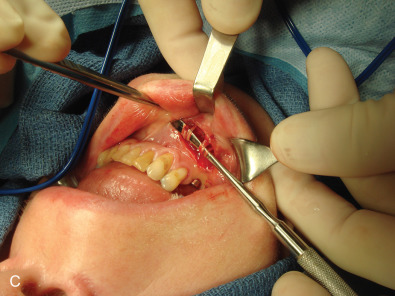
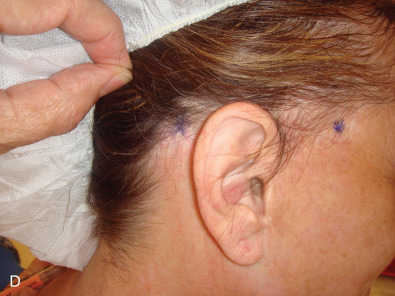
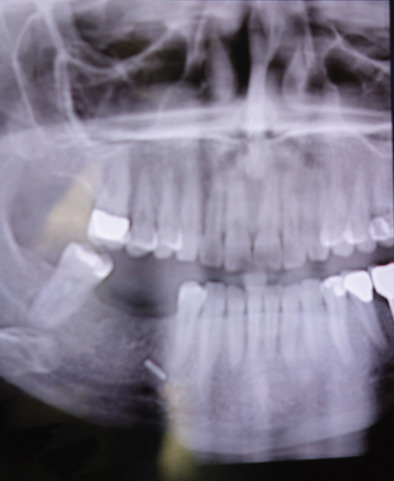
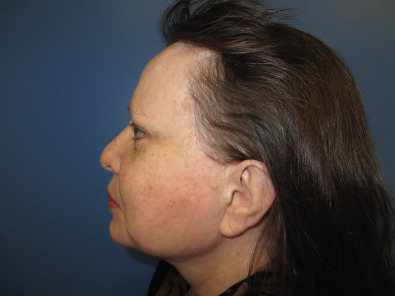
In obtaining the history from the patient, the most commonly overlooked source of posttraumatic facial pain is previous neurosurgery procedures , such as those for Chiari malformation ( Fig. 3.15.4A ), that require incisions at the base of the occiput, or those for brain tumor, such as acoustic neuroma, which are in the postauricular, mastoid area. Other less common causes related to neurosurgery are cerebrospinal shunt placements, burr holes, and the anterior site of tong placements. The next most frequently missed history point is a previous motor vehicle accident with a whiplash injury, which can cause pain related to the anterior scalene becoming adherent to the cervical and brachial plexus, or with an airbag inflation. Be sure to ask about childhood injuries to the craniofacial skeleton, such as a baseball ( Fig. 3.15.4B ) hitting the face, a fall from a bike, or other sports accidents, which must include heading a ball in soccer and concussion. Especially if the pain is related to V 2 or V 3 , inquire about dental surgery, particularly dental implants ( Fig. 3.15.4C ). Finally, when asking about previous facial surgery, ask directly about cosmetic surgery such as facelift ( Fig. 3.15.4D ), cheek implants, or jaw surgery, such as chin implant or orthognathic surgery.
Nerve Blocks
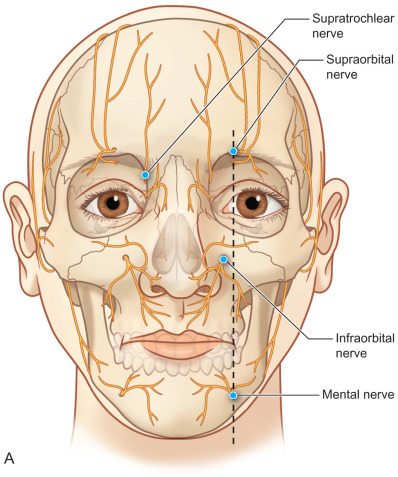
Compression of the trigeminal ganglion or its nerve roots intracranially will cause facial pain that will not be relieved by a nerve block of the peripheral TN branches. It is now accepted that traditional trigeminal neuralgia is a vascular compression of the Gasserian ganglia roots by a branch of the inferior cerebellar artery, which is best treated by an intracranial vascular decompression, as originally described by Jannetta, in 1967. Therefore, a nerve block, as depicted in Fig. 3.15.2 , will document that pain is from an injury to a peripheral branch of the TN, and not from intracranial vascular compression if (1) the expected skin territory goes numb after the local anesthetic is injected, and if (2) the pain is relieved. This holds true for nerve blocks to the cervical plexus ( Fig. 3.15.2 , and the occipital nerves ( Fig. 3.15.3 ).
Imaging
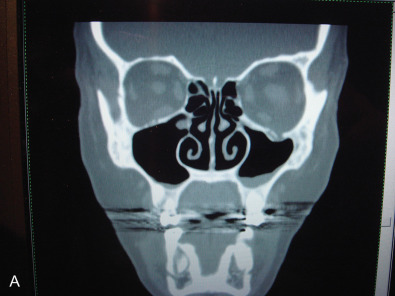
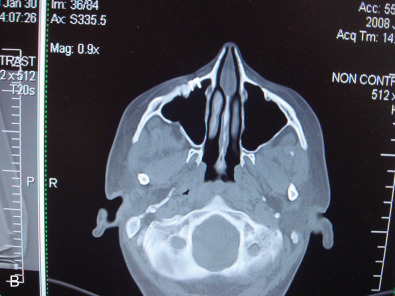
While the computed tomography (CT) scans usually cannot demonstrate nerves directly, the injury to a bone adjacent to the known anatomical location of a nerve supports the diagnosis of posttraumatic facial pain due to that nerve. This is demonstrated in Fig. 3.15.5A where the left orbital floor is lower than it is on the right, and the infraorbital canal is deformed on the left. In Fig. 3.15.5B , the right infraorbital nerve is injured at the location of the fixation plate at the infraorbital rim. Another example is given in Fig. 3.15.6 , where the right inferior alveolar nerve was injured twice; the first time with a screw designed to hold a bone graft prior to dental implant placement, and then a second time, when a metal clip was placed on the mental nerve at the mental foramen. The application of 3T MRI to the cranial nerves has brought a new dimension to being able to evaluate posttraumatic facial pain and is the best way to document vascular compression of the trigeminal ganglia and its roots.
Neurosensory Documentation of Trigeminal Nerve Function
An injured sensory nerve will develop a pattern of function that relates to the degree of nerve compression and a pattern that can identify nerve regeneration. Normative data for the one-point static- and moving-touch, and two-point static- and moving-touch was reported in 2007 using the Pressure-Specified Sensory Device™. The use of this device (previously marketed by Sensory Management Services, Inc., Baltimore, MD, now marketed as AcroVal™ Neurosensory and Motor Testing Systems by Axogen, Inc., Alachua, FL) has been reviewed extensively recently. By measuring the cutaneous pressure threshold of appropriate target skin areas, a patient’s subjective complaints can be documented, as related to complete loss of function, compression, or nerve regeneration for trigeminal nerve branches, as illustrated in Figs. 3.15.7–3.15.9 .
Psychological Evaluation
While it is appreciated that living with chronic facial pain can cause depression, frustration, anxiety, household problems, loss of work, and narcotic addiction, it is not so commonly appreciated that either the injury itself of the interactions with the medical profession can cause posttraumatic stress disorder (PTSD). At the time of the initial encounter with the patient, it is appropriate, indeed mandatory that medication either be prescribed for anxiety, depression, PTSD, or that referral to a psychiatrist be suggested so these medications can be prescribed. Most patients usually will have tried or already be on a neuropathic pain medication and a narcotic. It is critical that caring for this patient be done in conjunction with a pain management physician.
Surgical Treatment of A Painful Neuroma ( Box 3.15.2 )
Every divided peripheral nerve will sprout proximally and attempt to reconnect with its distal branches, guided by the gradient of new nerve growth factor produced by the distal Schwann cells. Nerve regeneration will proceed along the remaining distal basement membrane that survives after Wallerian degeneration. With the exception of the olfactory and optic nerves, which are extensions of the central nervous system, all other cranial nerves, will regenerate. Of course this applies to the cervical plexus (C 2 , C 3 , C 4 , and C 5 ) and occipital nerves, which are the dorsal cutaneous branches of the occipital nerves C 1 , C 2 , and C 3 .
- •
Treatment of a painful neuroma requires neuroma resection, as the neuroma is the source of the painful neural impulses.
- •
To prevent recurrent neuroma formation, the proximal end of the nerve must be placed into a “quiet” place, away from tension, traction, and touch.
- •
While muscle targets are available for cervical plexus and occipital nerves, muscle targets are not available for trigeminal nerve branches.
- •
Unique operative solutions are required for each trigeminal nerve branch that has a painful neuroma
When the nerve is first divided, there will be numbness in the distal skin target territory. Then, if the regenerating nerves connect to their distal targets, there will be the perception of buzzing and tingling as a normal consequence of regeneration. These are paresthesias. These perceptions may be perceived as uncomfortable or even painful, and then are termed dysesthesia. If during the course of regeneration the sprouts become entrapped in the healing process of the injury, a neuroma will form, which is just multiple small axon sprouts becoming embedded in scar. Not all neuromas are painful, in which case there is an area of numbness and no pain. If a localized area of pain exists at the site of an injury, with a distal decrease in sensibility, then a neuroma has formed.
While more than 200 papers have been published about techniques to prevent neuroma formation, the only approach that has proven successful over a long period of time, and for peripheral nerves of the upper and lower extremity, is to place the proximal end of the nerve into a muscle sufficiently large and with little excursion.
If a skin target is of critical sensibility, such as the lip, then the approach to the painful neuroma of the inferior alveolar nerve is to interrupt the pain pathway proximal to the injured nerve, and reconstruct the nerve, relieving pain and attempting to restore function.
For each of the specific nerve environments to be discussed in the rest of this chapter, this approach will be presented individually, but, in general, branches of the cervical plexus will be implanted into the sternocleidomastoid muscle, the occipital nerves will be implanted into the neck muscles deep to the trapezius (splenius capitus, and semispinalis capitus). There are no sufficiently large muscles on the facial skeleton into which a nerve can be implanted. In the hand, when no muscles sufficiently large are available, a digital nerve can be implanted into a medullary cavity of bone. There are no medullary bone cavities sufficient to do this in the facial skeleton. The approach to the TN branches is unique, and the author’s current approach will be presented in the sections to follow.
Frontal Branch of the Trigeminal Nerve (V 1 )
Supraorbital, Supratrochlear and Infratrochlear Nerves
The anatomy of these nerves is well described classically.
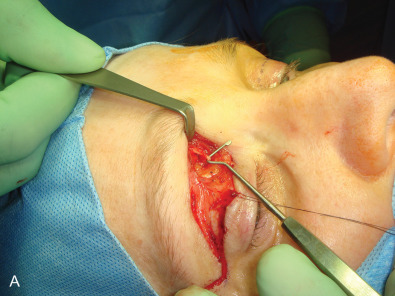
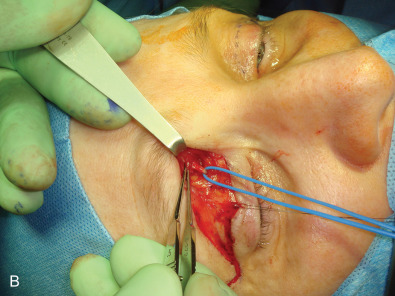
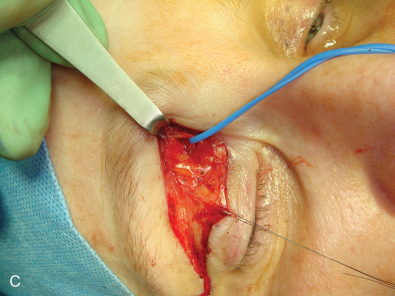
With a blunt injury to the forehead, these nerves may be contused, subsequently swell, and then become adherent to the surrounding corrugator muscles and superior orbital ridge/trochlea. This creates a chronic nerve compression , which will differ in its symptoms and approach from that for a painful neuroma. With compression, the forehead skin will have decreased, but not absent, sensation; the sensation present will still be normal, not dysesthetic, and there will be a mild Tinel sign or tenderness over the nerves at the superior orbital ridge. A nerve block will relieve the pain as a diagnostic test, Botox injection into the corrugator muscles may give sufficient relief for several months at a time, and neuropathic pain medication may be of value. The symptoms may be identical to the frontal headaches of migraine, but differ in the history of trauma. Frontal migraine patients do not have a history of trauma. With a diagnosis of compression, the surgical approach should be a neurolysis, attempting to preserve function while relieving pain ( Fig. 3.15.10 and Box 3.15.3 ). The patient should be counselled that the decompressed nerve may result, for about 3 months, in dysesthesias as neural regeneration occurs, although most commonly relief is experienced quickly. The patient should be counselled that if the symptoms persist 6 months or more after neurolysis, then it is appropriate to resect the injured nerves. This neurolysis differs from that typically described for the frontal migraine patient in that an upper eyelid incision, instead of an “endoscopic browlift” approach, is used, thereby permitting a neurolysis into the orbit including a division of the band across the supraorbital notch.
- •
The surgical treatment for a chronic nerve compression is neurolysis.
- •
If pain persists post-neurolysis, then nerve resection may be appropriate.
With a sharp or open injury to the forehead, these nerves will be directly damaged. They may or may not have been repaired primarily. The diagnosis of this form of chronic pain is a neuroma of the supraorbital and/or supratrochlear nerve. A resection of these neuromas and treatment by end-to-end coaptation by means of a neural conduit has been described. A series of six patients was reported. Preoperative mean pain (VAS) was 9.2 and postoperative mean pain was 1.5 ( p = .03). Mean follow-up was 14 months. No surgical complications were reported.
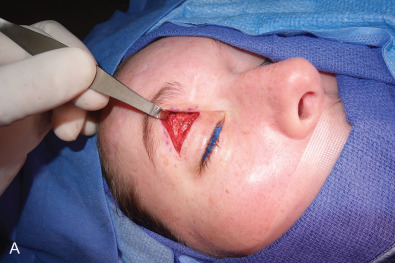
With or without a repair, if forehead and anterior scalp pain persist despite scar massage, neuropathic pain medication, and steroid injection, then a surgical approach to the painful neuroma is appropriate. This is illustrated in Fig. 3.15.11 . After protecting the globe, and injecting the local anesthetic, an upper eyelid incision is made, taken deep to the orbicularis muscle, and then the dissection is directed along the orbital septum to the superior orbital rim. The branches of the supraorbital nerve are identified within the orbit, blocked with 1% lidocaine with epinephrine, dissected about 1.5–2 cm into the orbit, cauterized with a bipolar coagulator set at low level, and then sharply divided with a microscissors, allowing the proximal end to retract into the orbit and above the orbital fat. The supratrochlear nerve is treated in a similar manner, taking care not to injure the superior oblique muscle as it courses around the trochlea. My personal experience with the first 5 patients having this approach has been reported. The mean preoperative pain (VAS) was 9 and the mean postoperative pain level was 0.6 (p <.001). The mean follow-up was 19.6 months, range 12–34 months. There were no surgical complications ( Table 3.15.1 ). This experience has been confirmed since then with two more patients.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree