Skin is the most voluminous organ of the body. It assumes several important physiological functions and represents also a “social interface” between an individual and other members of society. This is the main reason its age-dependent modifications are in the forefront of dermatological research and of the “anti-aging” cosmetic industry. Here we concentrate on some aspects only of skin aging, as far as the cellular and extracellular matrix components of skin are concerned. Most well studied mechanisms of skin aging can be situated at the postgenetic level, both epigenetic and post-translational mechanisms being involved. Some of these mechanisms will be reviewed as well as the capacity of fucose- and rhamnose-rich oligo- and polysaccharides (FROP and RROP) to counteract several of the mechanisms involved in skin aging.
Introduction
It is now largely accepted that aging is not “coded” in the genome although modifications of the coordination of gene functions are certainly involved. The hereditary genetic influences, put to about 25% a few decades ago, are now considered to represent no more than about 3%. Evolution apparently did not care much about aging, perhaps to some extent indirectly since the Paleolithic because of the “grandmother” effect. Most well studied, reproducible mechanisms can be situated at the epigenetic and postsynthetic (post-translational) level. Sirtuins, claimed to produce when stimulated an extension of lifespan, act clearly at the epigenetic level. Those mechanisms shown to be involved in skin aging are also driven by epigenetic and post-translational mechanisms. Some of these processes will be described.
Mechanisms of skin aging
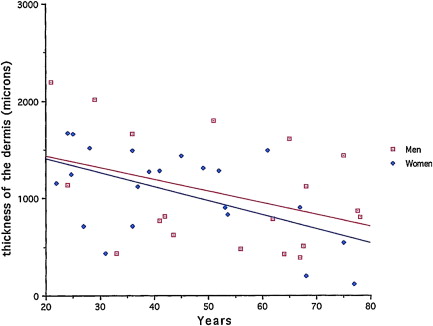
The most conspicuous process is certainly the progressive loss of skin tissue. Measured by image-analytical techniques on skin biopsies taken at sun-protected sites ( Fig. 1 ), skin loss amounts on the average to about 7% per decade with however large individual variations. This loss of skin tissue, which underlies most of the easily noticed morphological modifications of the skin, can be attributed to several factors such as loss of cells and loss of extracellular matrix (ECM). Cell loss concerns both the epidermal and dermal layers. Loss of ECM is evident when histological skin sections from young and old individuals are compared. Loss of ECM is the result of cell loss, decreased biosynthetic capacity of remaining cells and to a progressive increase of matrix degrading enzymes.
Loss of Cells
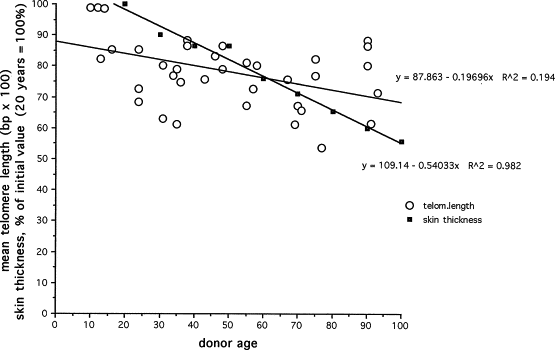
Loss of cells is currently attributed to two distinct processes: slow-down of cell division because of telomere loss and exit of cells from the mitotic pool mediated by some antioncogenes through a “switch mechanism” enabling cells to quit the mitotic pool entering the senescent phenotype, escaping thus from malignancy. This teleological presentation of the observed facts corresponds probably to a stress-mediated process of phenotypic switch. This process might well play an important role in the loss of mitotic cells as shown by our experiments, reproduced on Fig. 2 . It is clear that the rate of loss of telomeres is slower than the loss of skin tissue. The difference, of about 3.5 fold in the two slopes shown on Fig. 2 , is probably an indication of the importance of other mechanisms, such as the antioncogene mediated switch to the postmitotic phenotype. More recently, a third mechanism was described by McClintock et al. These authors showed that progerin accumulates in skin during aging. Progerin is a dominant negative form of lamin A, a nuclear membrane protein which is produced in cells of young individuals affected by the Hutchinson-Gilford syndrome (progeria) and who die young (12–15 years) with cardiovascular symptoms. Ninety percent of these cases carry the LMNA G608G (CGC > CCT) mutation within exon 11 of LMNA. This mutation activates a splice donor site resulting in the production of truncated lamin A designated progerin. The progressive accumulation of this molecular marker of cellular aging was achieved by the use of a specific antibody and immunolabeling of progerin in skin biopsies. Further studies will undoubtedly reveal the importance of this new biomarker of skin aging. Among the clinical symptoms in children affected by this rare disease is, among others the absence of subcutaneous adipose tissue which contributes to the senile appearence of the head and face of these children.
Increased Degradation of Skin-ECM
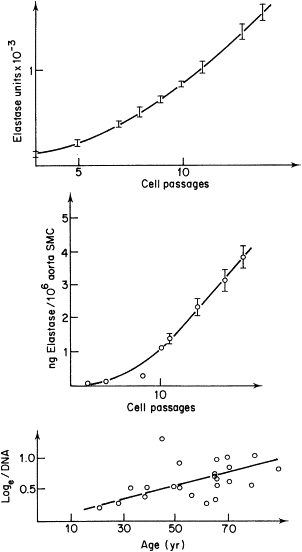
Among the early findings we made during our studies on aging of connective tissues, the most conspicuous was the progressive upregulation of elastase-type endopeptidase activity. This was first demonstrated on human aorta extracts devoid of atherosclerotic lesions. As shown on Fig. 3 , the elastase-type activity of aorta extracts increased exponentially with donor age. Similar findings were reported on mouse skin extracts, an exponential increase of elastase-type activity with age, further potentialised by UV-radiation. To our surprise, similar upregulation of elastase-type endopeptidase activity was seen when the determinations were carried out on successive passages of arterial smooth muscle cells or on human skin fibroblasts ( Fig. 3 ). Although the mechanism of this intrinsic cellular phenomenon is not yet elucidated, epigenetic modifications increasing the expression of MMP-2 and MMP-9 coding genes are the most plausible. There was a strong increase of elastase-type activity of vascular smooth muscle cell cultures in presence of atherogenic lipoproteins, LDL and VLDL. Lipid deposition in the skin, depending at least partially on the quality and quantity of dietary intake, may therefore represent an important factor for the regulation of skin proteolytic activity.
Mechanisms of skin aging
The most conspicuous process is certainly the progressive loss of skin tissue. Measured by image-analytical techniques on skin biopsies taken at sun-protected sites ( Fig. 1 ), skin loss amounts on the average to about 7% per decade with however large individual variations. This loss of skin tissue, which underlies most of the easily noticed morphological modifications of the skin, can be attributed to several factors such as loss of cells and loss of extracellular matrix (ECM). Cell loss concerns both the epidermal and dermal layers. Loss of ECM is evident when histological skin sections from young and old individuals are compared. Loss of ECM is the result of cell loss, decreased biosynthetic capacity of remaining cells and to a progressive increase of matrix degrading enzymes.
Loss of Cells
Loss of cells is currently attributed to two distinct processes: slow-down of cell division because of telomere loss and exit of cells from the mitotic pool mediated by some antioncogenes through a “switch mechanism” enabling cells to quit the mitotic pool entering the senescent phenotype, escaping thus from malignancy. This teleological presentation of the observed facts corresponds probably to a stress-mediated process of phenotypic switch. This process might well play an important role in the loss of mitotic cells as shown by our experiments, reproduced on Fig. 2 . It is clear that the rate of loss of telomeres is slower than the loss of skin tissue. The difference, of about 3.5 fold in the two slopes shown on Fig. 2 , is probably an indication of the importance of other mechanisms, such as the antioncogene mediated switch to the postmitotic phenotype. More recently, a third mechanism was described by McClintock et al. These authors showed that progerin accumulates in skin during aging. Progerin is a dominant negative form of lamin A, a nuclear membrane protein which is produced in cells of young individuals affected by the Hutchinson-Gilford syndrome (progeria) and who die young (12–15 years) with cardiovascular symptoms. Ninety percent of these cases carry the LMNA G608G (CGC > CCT) mutation within exon 11 of LMNA. This mutation activates a splice donor site resulting in the production of truncated lamin A designated progerin. The progressive accumulation of this molecular marker of cellular aging was achieved by the use of a specific antibody and immunolabeling of progerin in skin biopsies. Further studies will undoubtedly reveal the importance of this new biomarker of skin aging. Among the clinical symptoms in children affected by this rare disease is, among others the absence of subcutaneous adipose tissue which contributes to the senile appearence of the head and face of these children.
Increased Degradation of Skin-ECM
Among the early findings we made during our studies on aging of connective tissues, the most conspicuous was the progressive upregulation of elastase-type endopeptidase activity. This was first demonstrated on human aorta extracts devoid of atherosclerotic lesions. As shown on Fig. 3 , the elastase-type activity of aorta extracts increased exponentially with donor age. Similar findings were reported on mouse skin extracts, an exponential increase of elastase-type activity with age, further potentialised by UV-radiation. To our surprise, similar upregulation of elastase-type endopeptidase activity was seen when the determinations were carried out on successive passages of arterial smooth muscle cells or on human skin fibroblasts ( Fig. 3 ). Although the mechanism of this intrinsic cellular phenomenon is not yet elucidated, epigenetic modifications increasing the expression of MMP-2 and MMP-9 coding genes are the most plausible. There was a strong increase of elastase-type activity of vascular smooth muscle cell cultures in presence of atherogenic lipoproteins, LDL and VLDL. Lipid deposition in the skin, depending at least partially on the quality and quantity of dietary intake, may therefore represent an important factor for the regulation of skin proteolytic activity.