Introduction
Pain is the most burdensome symptom of hidradenitis suppurativa (HS), accounting for greater impairment in HS-related quality of life (QoL) than even disease severity. Although it has received relatively less attention, pruritus is also common in HS and contributes to disability and poor health-related QoL among those living with HS. This chapter summarizes what is currently known about the epidemiology, clinical characteristics, and pathophysiology of HS-related pain and itch and synthesizes these data to suggest rational approaches to clinical management of these symptoms.
Pain in Hidradenitis Suppurativa
Epidemiology of Pain in Hidradenitis Suppurativa
Prevalence and Severity of Pain in Hidradenitis Suppurativa
Nearly all individuals living with HS experience lesion-associated pain during the disease course, and a large proportion report pain that occurs on a weekly basis (77% to 91%). In a cross-sectional study of 294 patients, the mean number of days with pain in the past month was 9.51 ± 9.67. A prospective international study of 1299 HS patients found that the majority of patients (61.4%) rate their pain severity as ≥ 5 on a numeric rating scale (NRS) of 0 to 10 and that 4.5% rate their pain as the “worst possible” or 10/10. When evaluating pain severity, other studies’ findings have been comparable, with average weekly NRS pain scores ranging from 3.6 to 5.0. Compared to other painful dermatologic conditions such as blistering diseases, leg ulcers, atopic dermatitis, lichen sclerosis, and skin tumors, those with HS had more frequent and severe pain. Individuals with HS are at increased risk for chronic opioid use as well as other substance misuse when compared to non-HS controls. Poorly controlled pain may contribute to this phenomenon, increasing the importance of recognizing and managing pain in patients with HS.
Risk Factors
Risk factors for HS pain include severe HS disease activity and a higher number of anatomic regions involved. Psychiatric comorbidities such as anxiety and depression further compound pain perception. Physical factors such as friction and tight-fitting clothing have also been reported to exacerbate HS pain.
Impact of Pain on Quality of Life
HS is highly debilitating, with worse patient-reported QoL than almost any other skin disease. Pain has been identified as the major contributor to reduced QoL and disability in HS, even more so than HS severity. Painful HS lesions directly impact physical function and daily activities. Individuals living with HS commonly report that HS lesional pain impacts mobility, participation in exercise and sports, work productivity, and sexual health. HS pain is also associated with insomnia and poor sleep quality. HS pain impacts psychological well-being, resulting in increased risks for depression, anxiety, and suicide among those living with HS. The HS pain experience and the uncertainty of its course contribute to a sense of powerlessness and worsened emotional function. Further, HS patients report feeling isolated and invalidated when others fail to understand and empathize with the severity of their pain.
Clinical Characteristics of Hidradenitis Suppurativa Pain
The quality of HS pain may vary between patients and may also vary over time within the same individual. The timing of HS pain may be acute with associated flare or worsening inflammatory activity, or chronic in nature. HS pain has been described by patients as nociceptive and neuropathic in character. Nociceptive pain results from a noxious stimulus that has potential for tissue damage, and is often described as “aching,” “gnawing,” or “throbbing.” 8 In HS, nociceptive pain may result from direct inflammatory tissue damage (see Pathophysiology of Pain in HS). Neuropathic pain arises from damage to the somatosensory nervous system. Neuropathic pain is classically described as “burning,” “stinging,” or “stabbing,” and these descriptors are common among HS patients experiencing pain. In a study of 92 HS patients, 31.5% of patients reported symptoms suspicious for neuropathic pain, 41.3% had no neuropathic symptoms, and 27.2% had unclear results. Those with neuropathic pain were more likely to experience moderate to severe pain and to report psychiatric comorbidities.
Pathophysiology of Pain in Hidradenitis Suppurativa
Overview of Pain Response
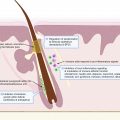
The physiologic response to pain resulting from tissue injury is an important evolutionary strategy in withdrawal from and avoidance of harmful stimuli. This response, which is not specific to HS, involves four major components: (1) transduction, (2) transmission, (3) modulation or transformation, and (4) perception. Transduction is the process by which primary afferent neurons or nociceptors convert noxious stimuli including chemical, mechanical, heat, and cold into nociceptive electrical signals. Pain, in addition to itch and temperature, are transmitted mainly by unmyelinated c-fibers and thinly myelinated Aδ fibers. If this electrical signal reaches the threshold for activation potential, transmission occurs, sending a nociceptive signal from peripheral nerve fibers to the central nervous system (CNS). Modulation or transformation modifies these signals at the level of the CNS. Perception is the final stage of the nociceptive process that integrates cognitive and affective responses, resulting in the subjective pain experience ( Fig. 19.1 ) . The sections that follow incorporate current knowledge of HS pathophysiology with clinical descriptions of HS pain to suggest mechanisms which may contribute to the pain response in HS.
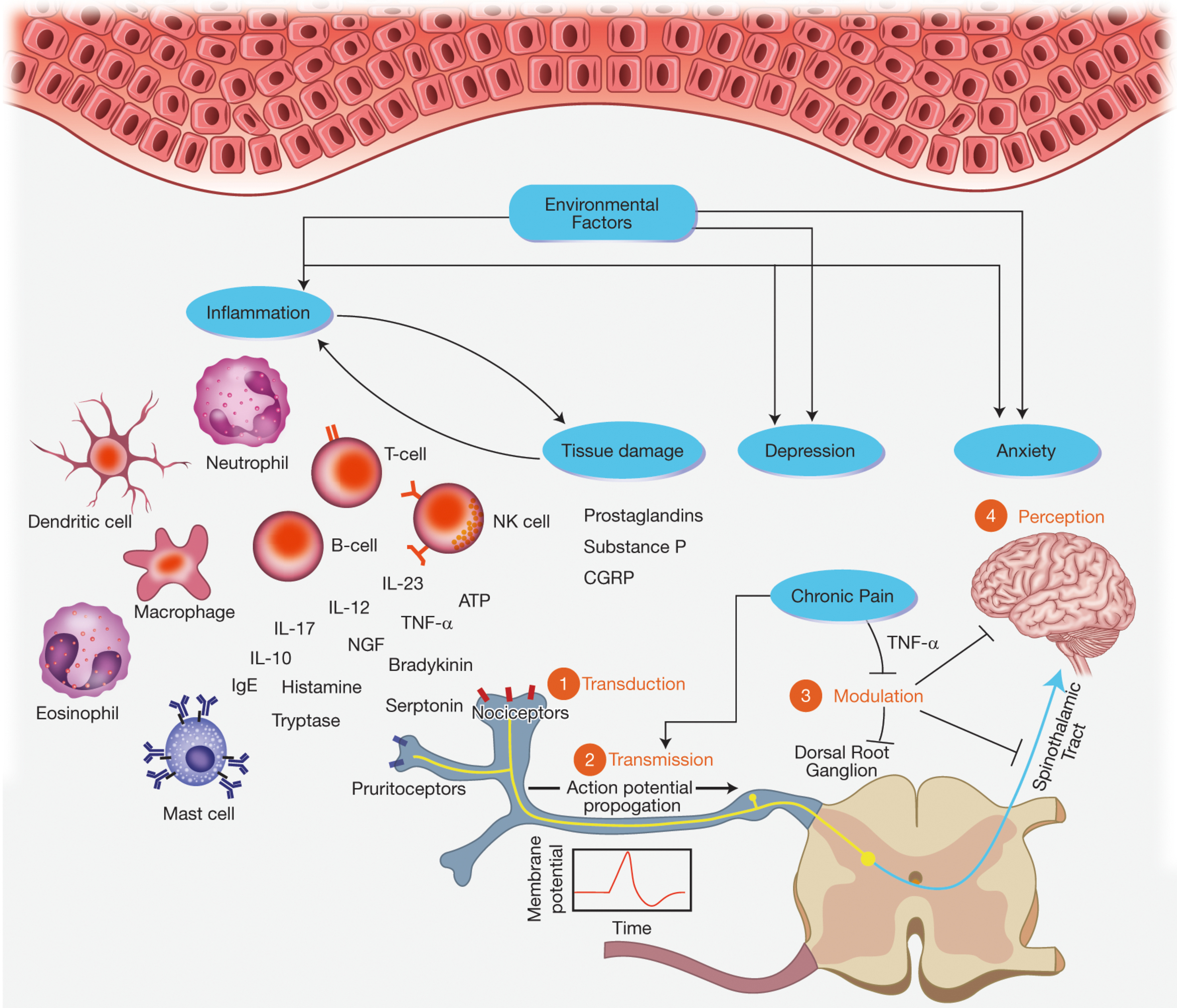
Nociceptors, Signal Transduction, and Transmission in Hidradenitis Suppurativa
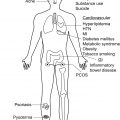
Inflammatory tissue damage likely plays a substantial role in generating the nociceptive signals that are the first step in the HS pain response pathway. HS is associated with a mixed inflammatory infiltrate including neutrophils, T-cells, B-cells, plasma cells, NK cells, mast cells, macrophages, and dendritic cells. These cells produce a variety of cytokines, chemokines, and growth factors which serve as stimuli to nociceptors. The role of inflammatory cells in causing HS pain is supported by the finding that antiinflammatory therapies can produce an analgesic effect in HS. Lifestyle factors and comorbid conditions, such as obesity, nicotine use, and metabolic syndrome, likely contribute to the stress response and enhance recruitment of inflammatory cells. Additionally, inflammatory cells recruitment results in damage to peripheral tissue, inducing keratinocytes to release pro-inflammatory factors such as prostaglandins, substance P, and calcitonin gene-related peptide (CGRP). These mediators subsequently bind to nociceptors, resulting in a series of electrical impulses at the afferent nerve terminal.
Of particular note, tumour necrosis factor-α (TNF-α) is a pro-inflammatory cytokine that has been found in lesional skin and serum of individuals with HS. Pharmacologic neutralization of TNF-α was shown to block nociceptive activity in the thalamus, somatosensory cortex, and limbic system within the first 24 hours and prior to any anti-inflammatory effect in peripheral tissues, suggesting that this cytokine may play a role in nociception, both peripherally and centrally.
Neuropathic Pain in Hidradenitis Suppurativa
Neuropathic pain arises from damage to the somatosensory nervous system, typically to peripheral fibers (Aβ, Aδ, and C-fibers) or central neurons. Studies of patient-reported pain descriptions suggest that neuropathic pain likely also plays a major role in pain in HS (see Clinical Characteristics of Pain). The precise mechanism for neuropathic pain in HS has not been elucidated. However, one explanation is that the inflammatory infiltrate, including mast cells, may promote neurogenic inflammation through release of mediators including histamine, tryptase, interleukin (IL)-31, and Nerve growth factor (NGF) which activate nociceptors on pain-sending neurons. Eosinophils and neutrophils may have similar, but less pronounced interactions with C-fibers. Additional studies are needed to fully understand the mechanism by which neuropathic pain occurs in HS.
Pain Perception in Hidradenitis Suppurativa
Pain experiences vary greatly between individuals. Differences in pain perception may in part be due to emotional distress, depression, and even socioeconomic predictors of health. In HS, these differences have been attributed to multiple causes including alterations in descending modulatory pathways as well as an exaggerated stress response. Modulatory pathways represent a complex system of feedback loops in the CNS that mediate top-down regulation of nociceptive processing. Altered descending modulatory pathways resulting in maintenance or amplification of pain signaling is implicated in various chronic pain states. Given that pain perception in HS does not necessarily correlate with disease severity, aberrant modulatory pathways may represent an important factor in HS pain.
Additionally, an overactive stress response in HS may result in a generalized inflammatory state that promotes pro-inflammatory cytokines including TNF-α, IL-1, and IL-6. Depression and anxiety are associated with higher serum levels of pro-inflammatory cytokines which may also contribute to a state of generalized inflammation. This theory is supported by the observations of worsening of HS disease in the setting of increased psychological stress as well as improvement in pain and psychological symptoms following treatment with adalimumab.
Pain Management in Hidradenitis Suppurativa
The pathophysiology of pain perception in HS is complex and closely linked with mental health. The most effective management strategies, therefore, likely require multimodal therapies that address nociceptive and neuropathic pain as well as the associated psychological distress. In other chronic pain syndromes, a multidisciplinary approach has been shown to yield superior pain outcomes. We therefore recommend early inclusion of non-pharmacologic treatments for HS pain such as physical therapy, appropriate wound care, and mental/behavioral health.
Overview of Evidence-Based Guidelines
Although multiple major international HS treatment guidelines have been published in recent years, few offer detailed guidance about management of HS pain. Some common themes and recommendations from these guidelines are highlighted in Box 19.1 .
|
|
|
|
|
|
|
Clinical Evaluation of Hidradenitis Suppurativa Pain
The best validated tools for evaluating pain severity across a wide spectrum of diseases include the Visual Analog Scale (VAS) and the Numeric Rating Scale (NRS). These are simple and practical for routine clinical use, making them a first choice for regular assessment of HS pain severity given the absence of HS-specific data currently. Pain was recently identified by the Hidradenitis SuppuraTiva cORe outcome set International Collaboration (HISTORIC) as one of six domains in the core outcome set for HS clinical trials. Additional effort is ongoing to determine the best measurement instrument and frequency evaluation for HS pain measurement in clinical trials.
In addition to evaluating pain severity, assessing the quality of HS pain may help distinguish nociceptive and neuropathic pain and help direct selection of pharmacologic analgesia. The McGill Pain Questionnaire and painDETECT survey have been used to help characterize HS pain, although neither has been specifically validated for use in HS. A patient interview including questions about the description of pain may be used to evaluate the pain character. Nociceptive pain is likely when the pain is localized to HS lesions and the patient describes pain as “aching,” “gnawing,” “pressure,” “squeezing,” or “throbbing.” Nociceptive pain is suggested by distal radiation, sensory deficits, use of temperature descriptors, and exaggerated hypersensitivity to classically non-painful stimuli. Common descriptors of nociceptive pain include “lancinating,” “shooting,” “electricity,” “tingling,” and “itching.” Clinically distinguishing nociceptive and neuropathic pain may have therapeutic implications, as many management guidelines for chronic non-cancer pain suggest different treatments for nociceptive and neuropathic pain.
Hidradenitis Suppurativa Pain Treatment Algorithms
One set of pain management algorithms has been published to date and has been reproduced in Figs. 19.2 and 19.3 . Although these algorithms provide a practical guide for healthcare providers in managing HS pain, improved knowledge of HS pain pathophysiology and effective therapies for HS pain management are critically needed. The strongest data for pain control in HS derive from clinical trials of HS disease-modifying therapies. First and foremost, HS pain control must begin with an appropriate medical management of the underlying disease.

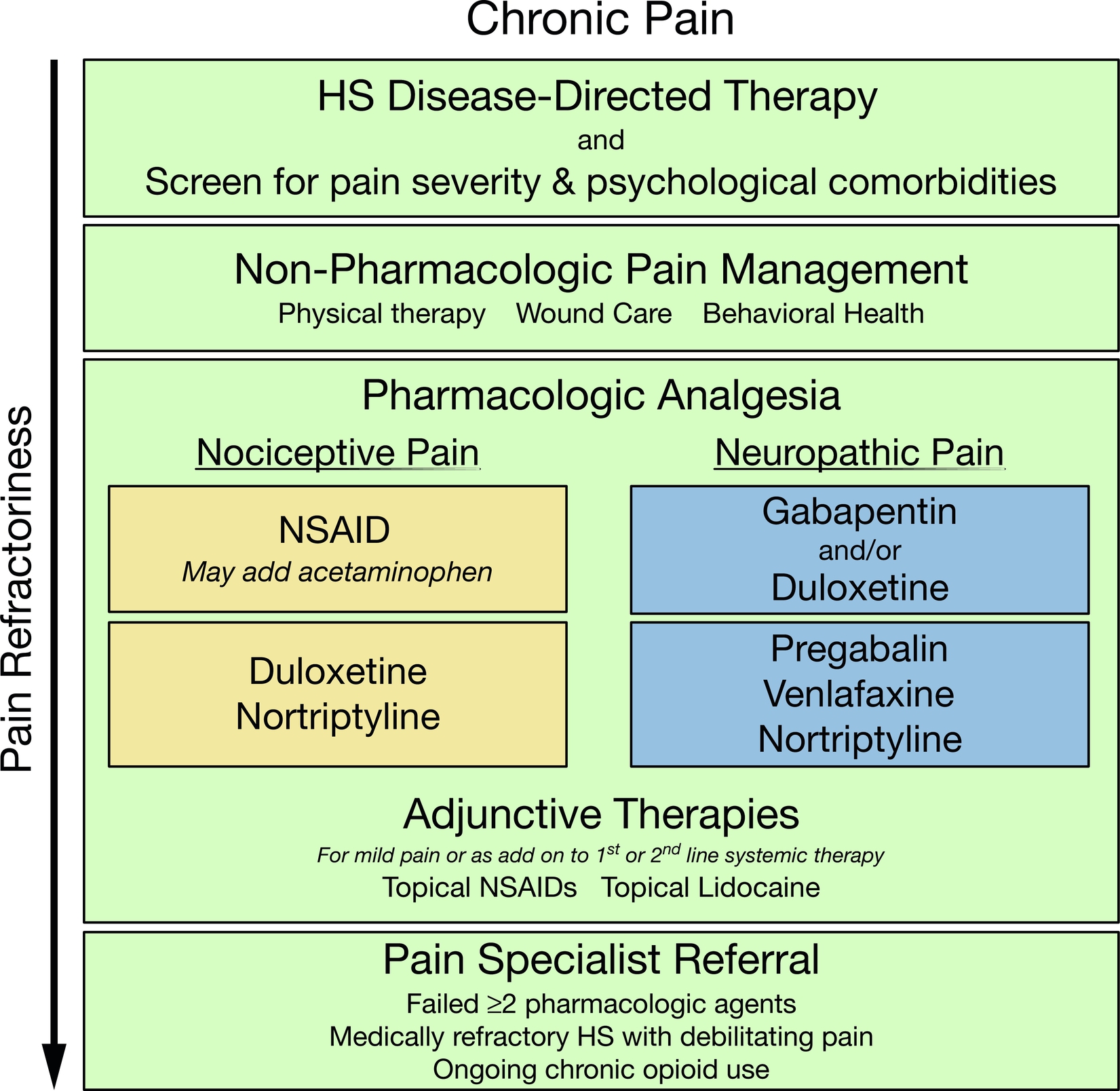
Pharmacologic Analgesia
Several reviews of the management of HS pain and chronic dermatologic pain provide comprehensive overviews of pharmacologic analgesia treatment options. Few disease-specific data in HS exist, and therefore pharmacologic therapies are often selected based on pain chronicity (acute vs. chronic) and character (nociceptive vs. neuropathic). Table 19.1 describes commonly employed analgesics, dosing guidelines, and risk profiles. Surgical management of HS including wide local excision and healing via secondary intent may result in increased acute pain symptoms. We recommend creating a pain management plan in advance for patients who are planning to undergo surgical management to ensure that pain is appropriately controlled in the pre- and post-operative periods.
| Medication | Dose | Selected Risks |
|---|---|---|
| Topical and Intralesional Therapies | ||
| Intralesional triamcinolone |
|
|
| Topical diclofenac |
|
|
| Lidocaine topical |
|
|
| Menthol topical |
|
|
| Non-Steroidal Antiinflammatory Drugs and Acetaminophen | ||
| Diclofenac sodium, delayed-release |
|
|
| Ibuprofen |
|
|
| Naproxen |
|
|
| Celecoxib |
|
|
| Acetaminophen |
|
|
| Calcium Channel Alpha-2 Ligands | ||
| Gabapentin |
|
|
| Pregabalin |
|
|
| Serotonin-Norepinephrine Reuptake Inhibitors | ||
| Duloxetine |
|
|
| Venlafaxine |
|
|
| Tricyclic Antidepressants | ||
| Desipramine and Nortriptyline |
|
|
| Amitriptyline |
|
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree