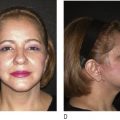
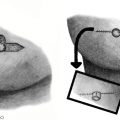
Orbital hypertelorism may be seen in isolation or more commonly within the context of other congenital craniofacial dyscrasias such as craniosynostosis, midface hypoplasia, exorbitism, atypical orofacial clefting, and malocclusion. Careful consideration of the preoperative cranial, orbital, and midface phenotypes, as well as the time of intervention, is imperative to choosing the optimal surgical approach to orbital repositioning. Virtual surgical planning serves as a powerful tool to increase surgical accuracy, decrease skeletal relapse, and mitigate historical complication profiles.
Key points
- •
Thoughtful analysis of the preoperative cranial, orbital, and midface phenotypes is imperative to choosing the optimal surgical approach to the management of hypertelorism.
- •
Careful consideration of the age at intervention can serve to decrease the operative risk profile, mitigate the rate of associated complications such as operative relapse, and reduce the need for subsequent operative interventions.
- •
Preoperative virtual surgical planning can be a powerful tool serving to increase operative accuracy, decrease operative intervals, and reduce postoperative risk of relapse in the surgical management of hypertelorism.
Background
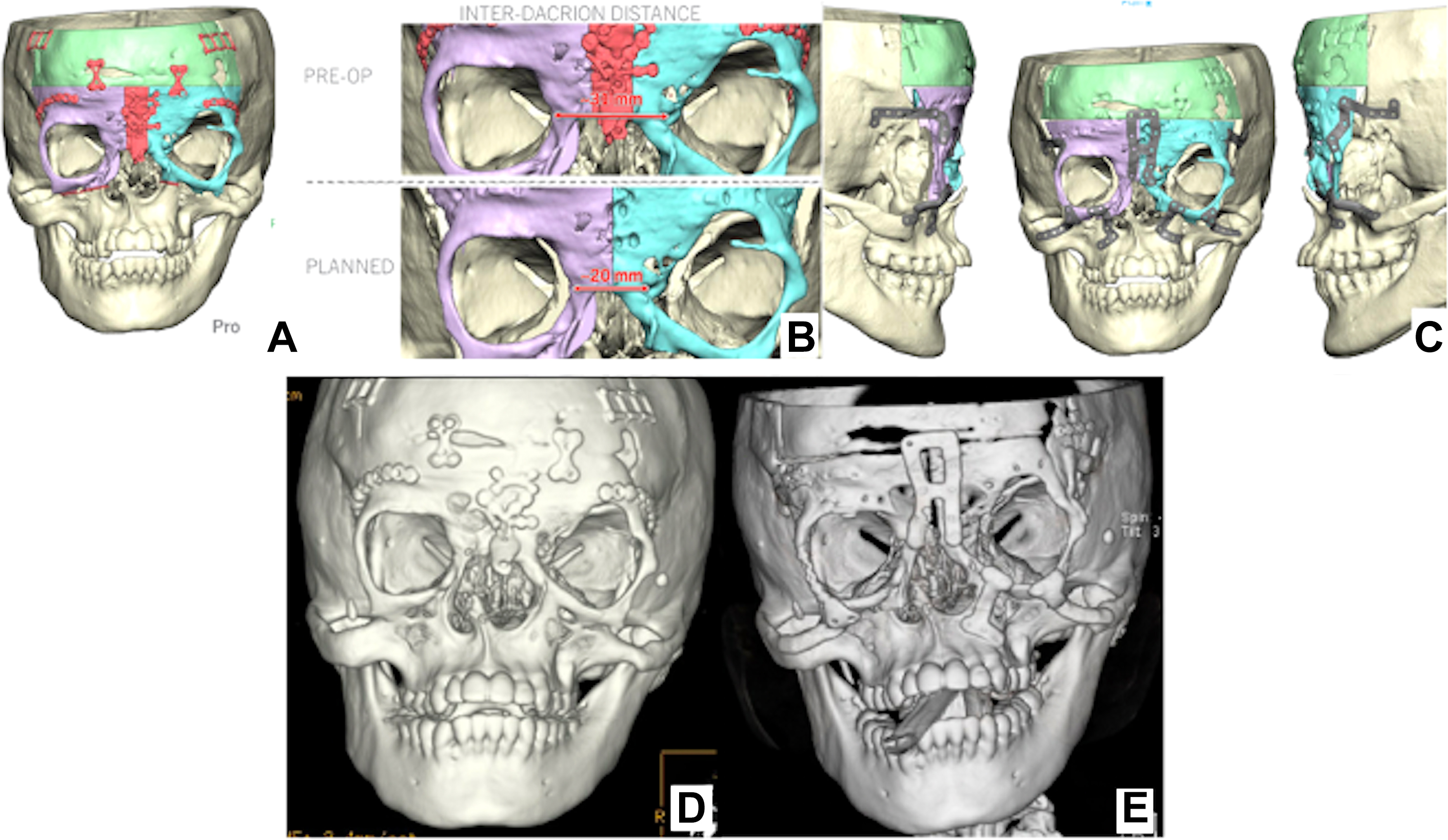
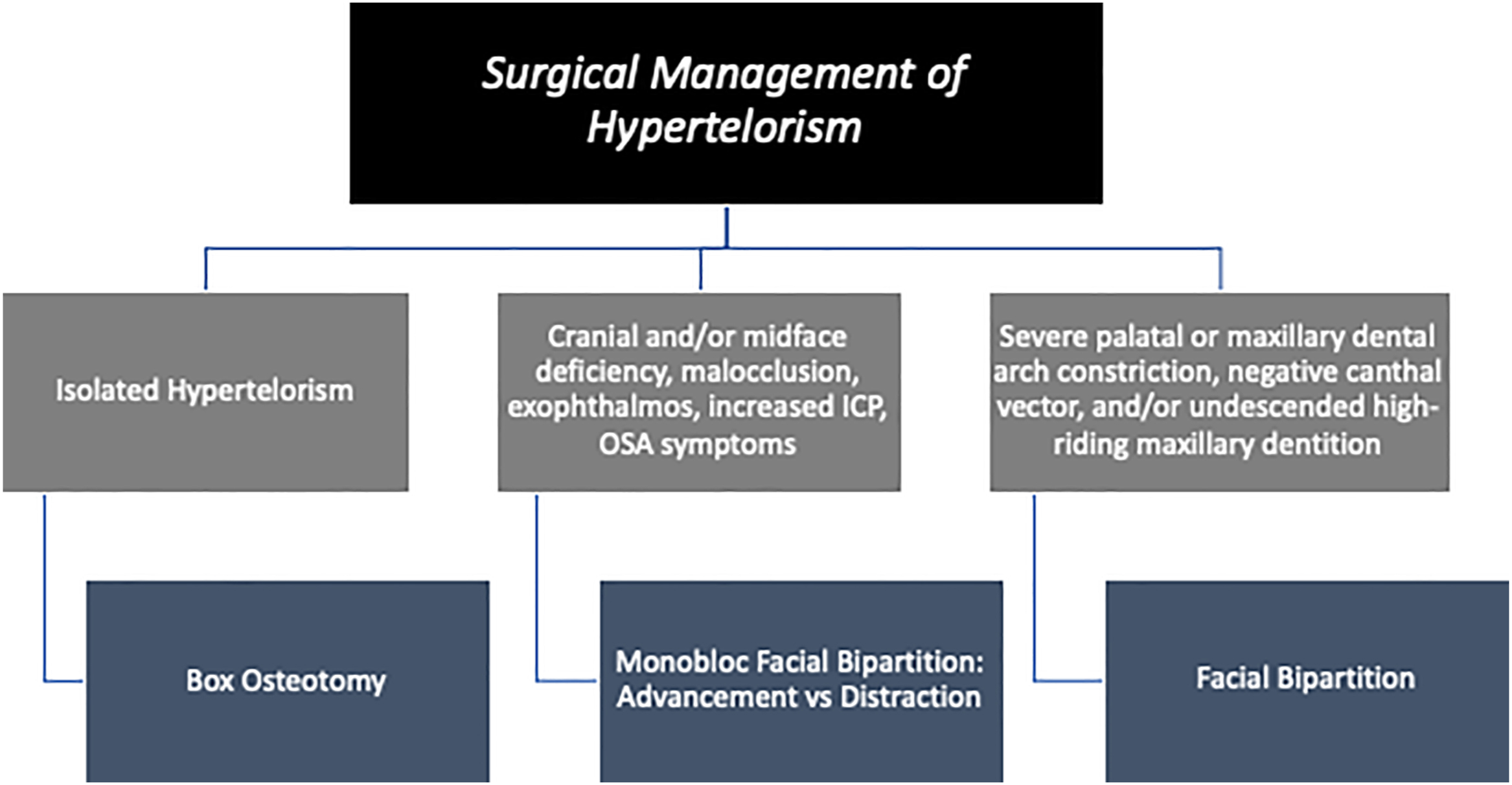
Orbital hypertelorism refers to the horizontal displacement of the orbital cones and their contents resulting in an increased interorbital distance. Interorbital distance is defined as the measurement between the anterior lacrimal crests or the interdacryon distance. The severity of hypertelorism can be classified by categorizing this distance into mild (grade I 30–34 mm), moderate (grade II 34–40 mm), and severe (grade III >40 mm). These measurements can be compared to normative values for adult female individuals (25 mm) and adult male individuals (28 mm). Orbital hypertelorism was first described by Greig in 1924 as a congenital craniofacial deformity. Surgical correction via both intracranial and extracranial approaches was subsequently described by Tessier in 1974. As malposition of the orbits is commonly seen in patients with craniofacial syndromes, correction of orbital hypertelorism is frequently considered within the context of other craniofacial dyscrasias such as midface retrusion, increased intracranial pressure, exophthalmos, exorbitism, malocclusion, or airway obstruction ( Table 1 ). Consequently, the most appropriate surgical treatment paradigm depends greatly on the constellation of clinical symptoms in conjunction with the presenting cranial and midface anatomy of the individual patient. Relevant procedures to address malpositioned orbits include (1) facial bipartition, (2) monobloc with facial bipartition, and (3) box osteotomy ( Fig. 1 ). As is common across the treatment paradigm of patients with complex craniofacial deformities, surgical outcomes are measured by both esthetic improvement in facial harmony as well as functional improvements in occlusion, ocular protection, relief of intracranial pressure and respiratory function.
| Diagnoses Associated with Hypertelorism | |
|---|---|
| Syndromes | Bohring–Opitz syndrome, Greig cephalopolysyndactyly, Noonan syndrome, 22q11 deletion syndrome, CHARGE, Loeys–Dietz syndrome, Wolf–Hirshorn syndrome, trisomy 18, Joubert syndrome, and Hurler syndrome |
| Midline dyscrasias | Frontonasal dysplasia, atypical orofacial clefting |
| Fibroblast growth factor receptor (FGFR) mutations | Crouzon syndrome, Apert syndrome, Pfeiffer syndrome, Antley–Bixler syndrome, Beare–Stevenson Cutis Gyrata syndrome, Jackson–Weiss syndrome, and Saethre-Chotzen-like syndrome |
Special anatomic considerations
The superior orbit is formed by frontal bone creating the superior orbital rim. Dissection of the area must consider the supraorbital notch and release of the supraorbital nerve if needed. The medial orbit consists of the anterior lacrimal crest of the maxillary bone, posterior lacrimal crest of the lacrimal bone, and the frontal bone. The medial canthus and the lacrimal apparatus must be carefully considered. Marking the medial canthus with methylene blue using a hypodermic needle prior to dissection can help to identify and avoid damage to the structure subsequently during the procedure. The inferior rim is formed from the zygomatic bone laterally and the maxillary bone medially. The lateral orbit consists of the zygomatic process of the frontal bone and the frontal process of the zygomatic bone. The lateral canthal tendon is released during dissection to complete the lateral orbital osteotomy. A canthoplasty to resuspend the lateral canthus is performed prior to closure of the bicoronal incision when appropriate.
Preoperative planning
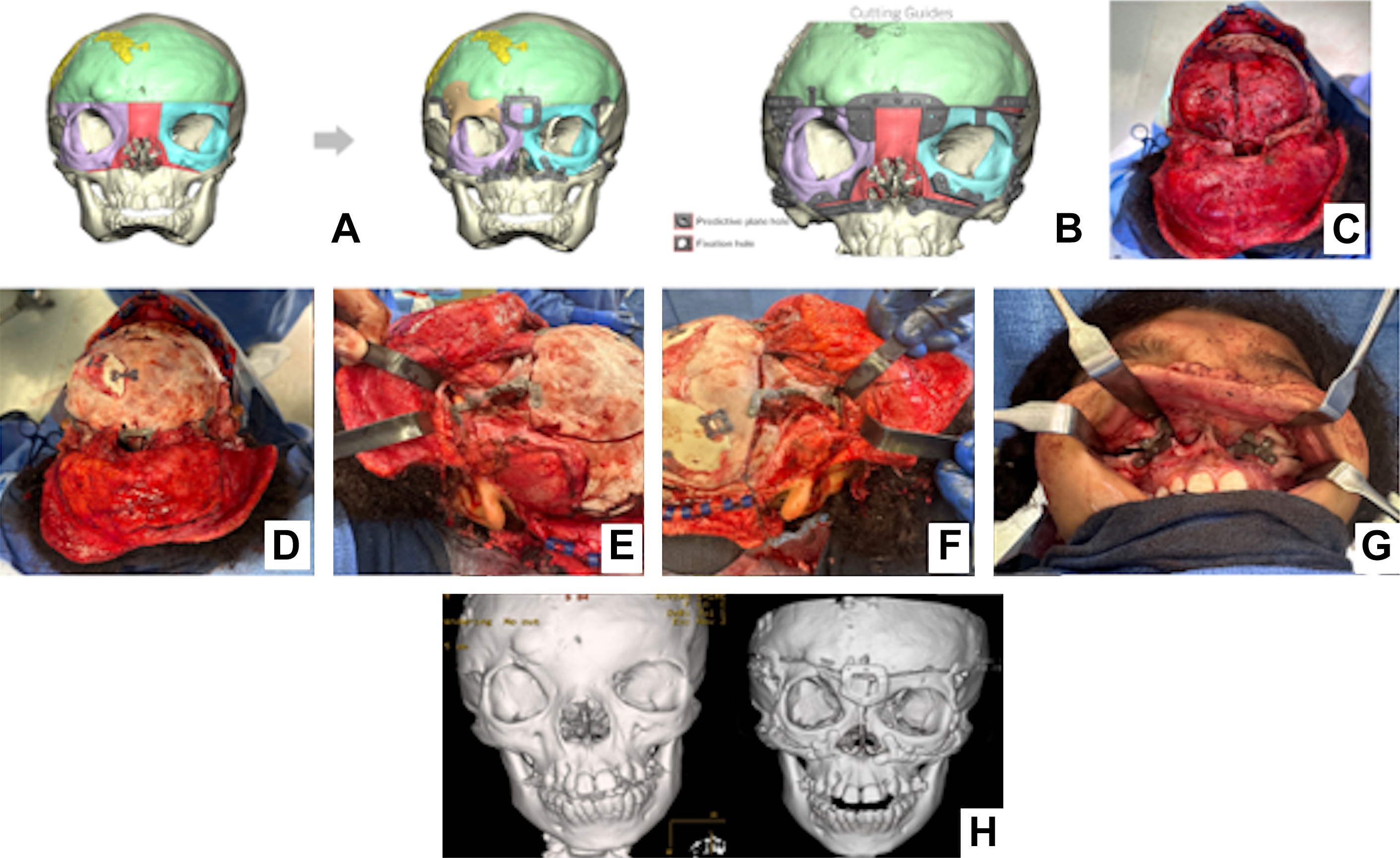
Preoperative workup includes a multidisciplinary preoperative assessment with the involvement of the neurosurgical team. Computed tomography of the head and maxillofacial skeleton is reviewed by all teams to assess associated cranial defects or congenital brain abnormalities. Virtual surgical planning may serve as a powerful tool to preoperatively determine the location of the osteotomies and the planned 3 dimensional (3D) translation of the orbital position prior to surgery. The ability to make exacting measurements to correct the interorbital distance decreases the margin of intraoperative error. A goal of 17 to 19 mm for the postoperative interorbital distance is commonly used to account for a possible bony and soft tissue relapse. Additionally, virtual surgical planning (VSP) allows the surgeon to compare the feasibility of advancement versus distraction depending on necessary movements and confidently establish occlusion when feasible with custom occlusal splints. The fabrication of custom cutting guides, a 3D skull model, and custom fixation hardware may further increase the efficiency and precision of the procedure lending to a more optimal outcome.
Discussion
Box Osteotomy
Isolated hypertelorism can be addressed with a unilateral or bilateral box osteotomy depending on preoperative orbital positioning. A box osteotomy allows for the movement of the 4 walls of each orbit as a single unit. Orbital translation can be multidirectional.
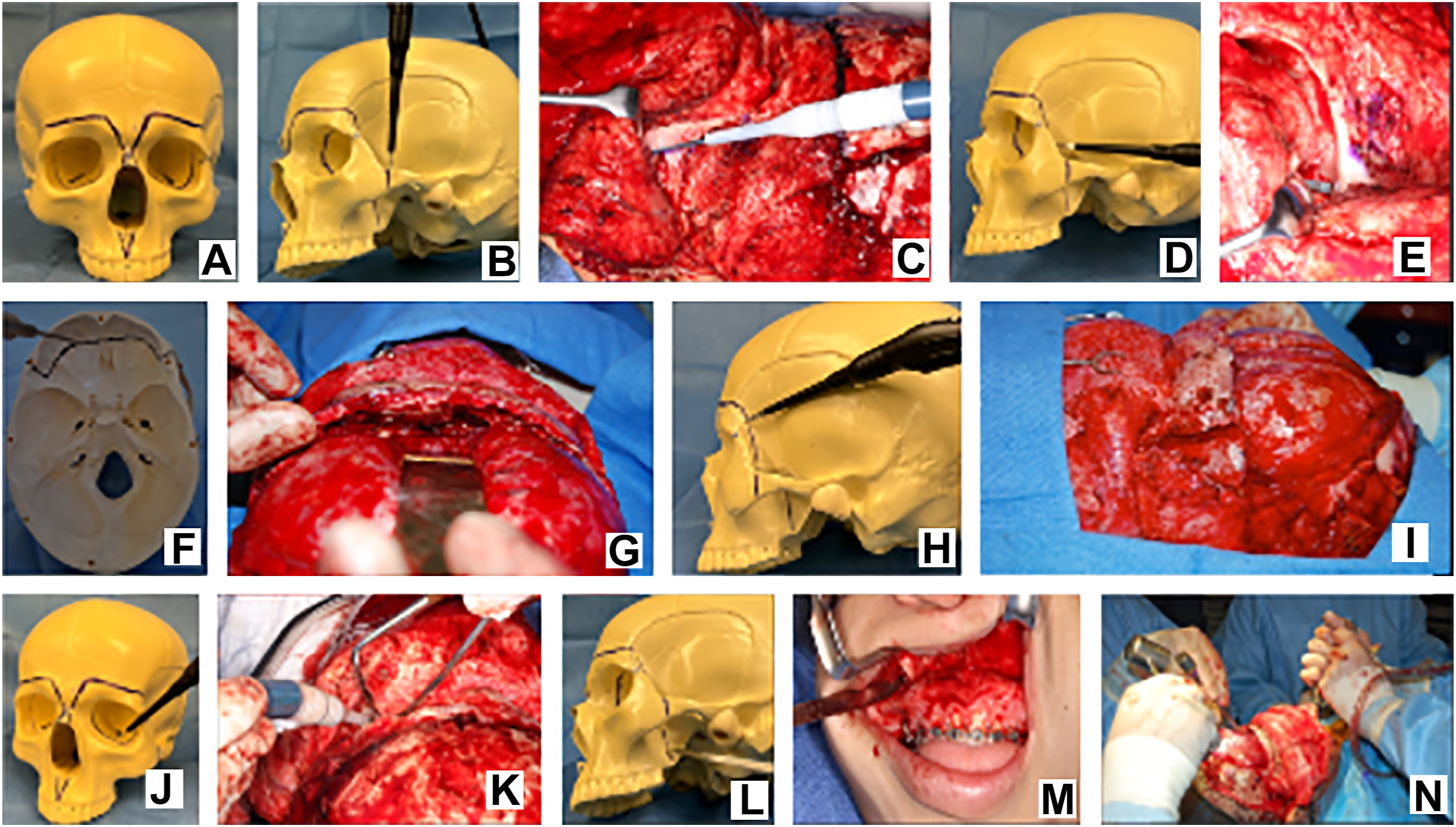
Intraoperatively, the patient is positioned in the supine position. A horseshoe headrest can be used to optimize exposure for the bicoronal incision. Eye protection during the procedure is vital, and therefore, bilateral suture tarsorrhaphies are performed. A coronal incision can be designed in a zigzag or curved manner to account for directionality of hair growth. This incision can be extended preauricularly as needed for exposure. Dissection is completed in a subperiosteal plane anteriorly to the supraorbital rim. Care is taken to identify the superior orbital neurovascular bundle and to release it from the superior orbital foramen with an osteotome if needed. This dissection is continued until the frontonasal junction is fully exposed. The lateral dissection is performed deep to the superficial layer of the deep temporal fascia within the superficial temporal fat pad. This allows access to the zygomatic arch and body while protecting the frontal branch of the facial nerve. The temporalis muscle is then elevated to allow access to the lateral orbit.
The craniotomy is then designed at the level of the height of contour of the calvarium; in coordination of neurosurgery, the frontal bone is removed. The anterior cranial base is then exposed through careful dissection to allow protection of the brain during completion of the osteotomies. The necessary osteotomies are then completed using an ultrasonic bone scalpel to minimize thermal injury and damage to soft tissues. The zygomatic arch vertical osteotomy is addressed first while leaving enough bone to allow for the fixation of plates to the lateral inferior orbital wall. The lateral orbital wall osteotomy is then completed starting from the lateral aspect of the inferior orbital fissure and extending superiorly through the temporal bone of the lateral orbital wall. Completion of the orbital roof osteotomy is performed via the anterior cranial fossa by connecting the lateral osteotomy and extending medially along the orbital roof while remaining anterior to the cribiform plate. An intraoral incision is made to access the maxilla for the inferior osteotomy. The osteotomy is designed inferior to the infraorbital nerve in order to preserve the nerve. An intracranial approach can be used to complete the medial osteotomy through the lateral nasal wall to connect to the inferior medial orbital osteotomy. Care should be taken that these osteotomies are not made too anterior. In order to assure that the movement of the skeletal box also adequately repositions the globe, the operator should strive to position the osteotomy behind the equator of the globe. The interval of planned midline bone resection is designed to allow for medialization of the orbital boxes and minding preservation of the medial canthus thereby avoiding telecanthus. Bony interferences, including the widened ethmoid sinuses, are removed as needed to allow for medial translation and reduction of the orbit. Once the orbital unit is mobile, the bony unit is then adjusted to its planned postoperative position and bony fixation is performed ( Figs. 2 and 3 ).
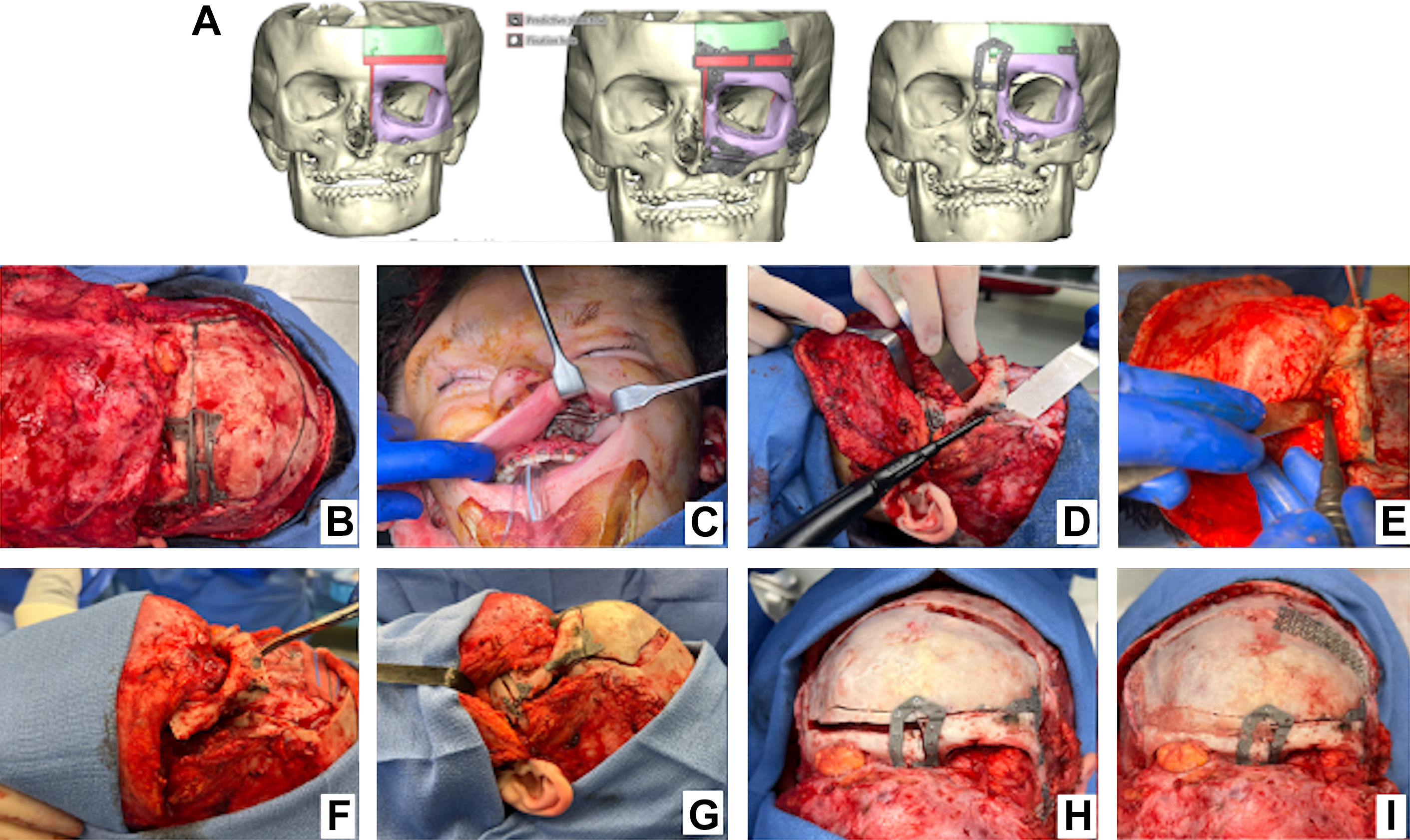
It should be noted that the actualized measurement of orbital box reduction does not translate in a 1:1 relationship to the overlying soft tissue envelope. After skeletal repositioning, excess midline paranasal soft tissue may give the continued appearance of hypertelorism. In cases of minimal soft tissue excess, a Vaseline gauze bolster dressing may be employed to narrow and reshape the soft tissue serving to mitigate soft tissue relapse. As the midline bone resection described earlier includes the nasal dorsum, the surgeon may consider the placement of a cantilevered bone graft in the midline to both restore nasal dorsal profile and serve to support and recontour the overlying soft tissue envelope. In patients in whom significant excess soft tissue is noted in the midline, direct excision may be indicated though the resulting vertical scar pattern is often not cosmetically ideal.
Monobloc with Facial Bipartition
Patients for whom monobloc with facial bipartition is indicated include patients with complex craniofacial differences who would benefit from concomitant correction of the midface, upper jaw, orbits, and the anterior cranium. This impactful intervention can simultaneously address increased intracranial pressure, exorbitism, exophthalmos, hypertelorism, midface retrusion, upper airway obstruction, and malocclusion. A monobloc movement can be performed as an advancement or as a distraction depending on the degree of correction indicated and the timing of intervention. Distraction is favored in the adolescent correction to mitigate the risks of anterior cranial dead space, infection, and skeletal relapse. Slight skeletal overcorrection is often favored to account for a degree of relapse over time depending on elasticity of the soft tissue. Rates of relapse have been anecdotally decreased in the era of VSP due to the improved operative accuracy and increased tensile strength of custom fixation hardware.
The surgical approach for the monobloc is as described earlier for the box osteotomy. The approach to the zygomatic arch and the orbital roof is the same as in the box osteotomy. However, the inferior osteotomy in the maxilla is not performed, and instead, the orbital floor osteotomy is performed starting medially from the inferior orbital fissure and extending medially in a curve mirroring the curve of the inferior orbital rim. Along the medial orbital wall, the orbital floor osteotomy is then connected to the orbital roof osteotomy. This is performed starting superiorly and taking care to remain positioned posterior to the medial canthus and lacrimal system.
The pterygomaxillary separation is then performed using a curved osteotome through a separate intraoral upper vestibular incision or by identifying the pterygomaxillary junction posterior to the zygomatic arch from above. After completion of all osteotomies, Rowe maxillary disimpaction forceps are used to apply gentle downward pressure while a double-guarded curved osteotome is used to perform the osteotomy along the perpendicular plate of the ethmoid to the posterior palate. Care is taken to release areas of soft tissue with Smith spreaders or laminar spreaders until segment is freely mobile. In patients with a high-arched palate, an intraoral palatal splint can be used to stabilize the maxillary arch and prevent injury during the use of the disimpaction forceps.
The predesigned osteotomy for the facial bipartition is then marked in a “V” shape in the nasofrontal region. Considerations when making this osteotomy include preserving the medial canthus and possible cranialization of the frontal sinus depending on the location of the nasofrontal osteotomy in relation to the nasofrontal ducts. An intraoral incision is then made to allow for split of the palate and the maxilla in the midline. This allows for rotation of the segments toward the midline and correction of both the dental arch and the interorbital distance and canthal vector.
Placement of fixation plates or the distraction devices for the monobloc distraction is then performed. The lateral canthus is then resuspended to the lateral orbital wall prior to closure. Alternatively, the lateral canthus can be resuspended at the time of distraction device removal if distraction is performed ( Figs. 4 and 5 ).
Facial Bipartition
Indications for facial bipartition includes patients with hypertelorism in addition to (1) disruption or severe constriction of the dental arch as in the case of midline facial clefts, (2) significant negative canthal vector, and/or (3) undescended/high-riding maxillary dentition. A facial bipartition allows for repositioning of the orbits in conjunction with adjustment of the upper dental arch and maxilla. In patients without a need to address increased intracranial pressure or frontal bandeau positioning, a monobloc is not indicated, and instead, a facial bipartition can be performed in isolation. Osteotomies for a facial bipartition mirror that of the monobloc with osteotomies along the zygomatic arch, the lateral orbital wall, orbital floor, orbital roof, and medial orbital wall and pterygomaxillary junction. The “V”-shaped nasofrontal osteotomy is marked, and the same considerations with regard to the medial canthus, the frontonasal ducts, the lacrimal system, and the palatal midline osteotomy are made as with the monobloc with facial bipartition.
Safety considerations
Preoperatively, the operating surgeon should be reminded of the importance of airway consideration during the skeletal manipulation of the orbits and midface. If occlusion is not being addressed with a facial bipartition or a monobloc with facial bipartition, an oral rae endotracheal tube may be utilized. This tube may be wired transdentally or around the mandible for additional security if needed. A submental airway utilizing an armored tube may be indicated if occlusion is to be addressed and maxillomandibular fixation is necessary.
Intraoperatively, separation of the intracranial contents from the nasal cavity following skeletal repositioning is imperative specifically to reduce the risk of ascending meningeal exposure to the sinonasal microbiome. Most commonly, pericranial flaps based anteriorly on the trochlear supply are elevated to provide vascularized tissue to fill this dead space and create a soft tissue barrier. In the multiply reoperated patient, a temporally based pericranial or even galeal flap may be utilized with similar efficacy.
Postoperative considerations
Postoperatively, the patient is admitted to the intensive care unit for close monitoring in the setting of blood loss, airway management, and pain management. The patient typically remains intubated for 24 to 48 hours postoperatively. Eye protection in the setting of postoperative soft tissue swelling is an important consideration. Efforts should be made to keep eyes well lubricated with ophthalmic ointment. Tarsorrhaphy sutures may be left in place postoperatively to protect the eyes until extubation; however, close monitoring is needed as the presence of tarsorrhaphy sutures can contribute to an increased ocular pressure.
Patients are kept on prophylactic antibiotics for up to 7 days postoperatively to prevent postoperative infections.
If distraction devices are placed, a latency period of 5 days allows for mucosalization of the nasal cavity prior to distraction. Distraction is then performed at 0.5 mm 2 times per day until the skeletal unit reaches a position of slight overcorrection.
Timing of surgery
The ideal age for the correction of hypertelorism is not clearly defined as there are a variety of contributing factors including severity of associated issues such as exorbitism, intracranial pressure, and airway compromise. By the age of 8 years, the cranium and upper midface have reached roughly 85% of their total growth potential, and therefore, waiting for this age can minimize the risk of growth disturbance, skeletal relapse, and the need for repeat procedures. In the case of the box osteotomy, the inferior osteotomy through the maxilla can damage unerupted teeth if performed too early. Therefore, in the setting of isolated hypertelorism, waiting until the child is older can prevent injury to teeth. Due to complications related to relapse, growth restriction, and infection in some clinical situations, staging the management of the affected skeleton may address the various craniofacial deformities while minimizing risk. In the case of syndromic craniosynostosis where the operative plan needs to address intracranial pressure, midface retrusion, exorbitism, and hypertelorism, a staged approach may be safer. The fronto-orbital advancement can be performed when the patient is aged 1 year, and the midface and orbits can be addressed when the patient is aged 8 to 10 years or older, depending on the severity of airway and ocular symptoms. A staged approach to skeletal repositioning presents less risk than a single stage with a monobloc.
Complications
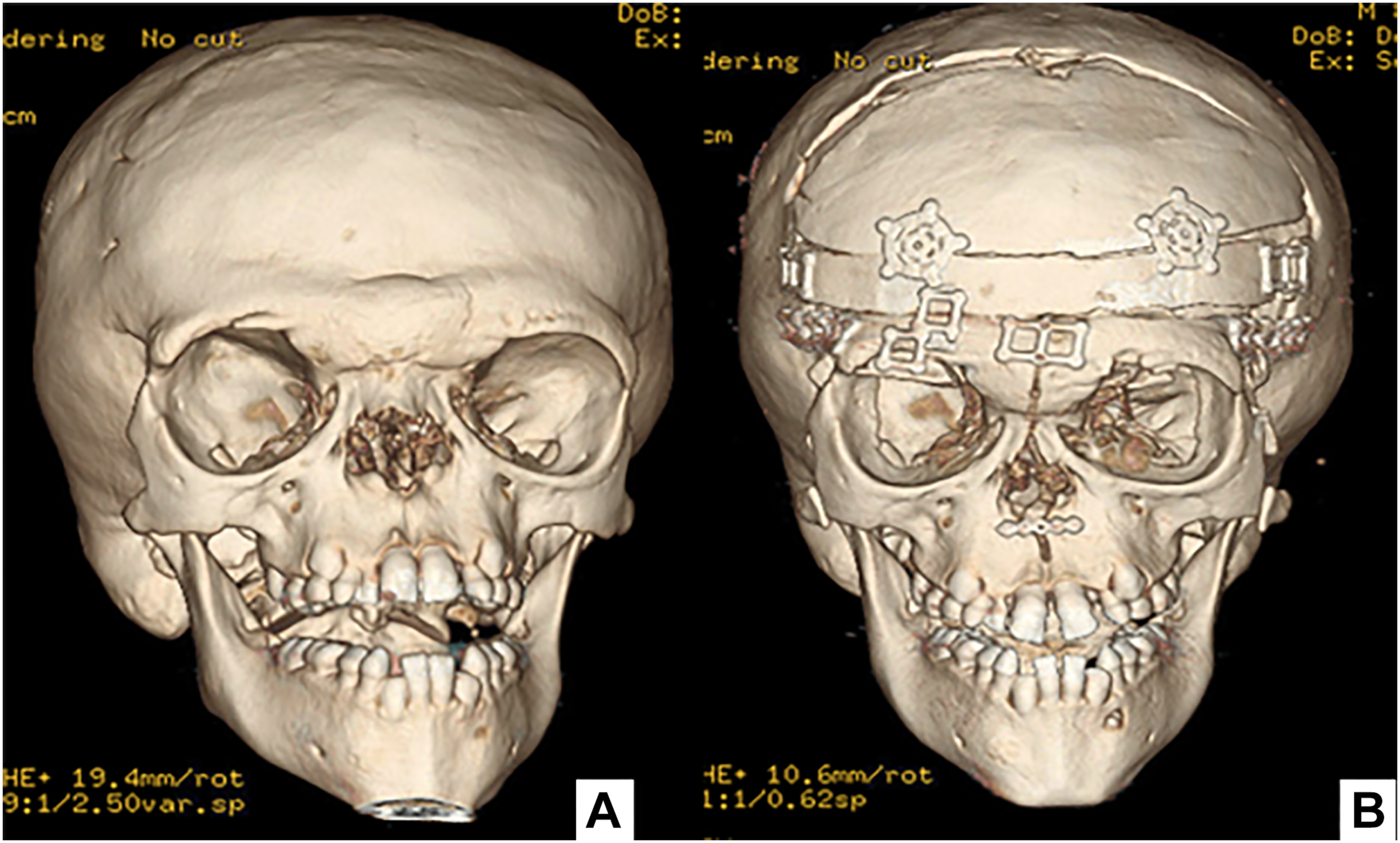
Generalized complications with any intracranial craniofacial procedure include bleeding from the bony osteotomies or from coagulopathy related to intraoperative blood transfusion. Cerebral spinal fluid (CSF) leak is another consideration with reports of up to 20% of patients developing rhinorrhea. Additionally, a fistula between the nasal cavity and the dura can be a source of ascending infection. A CSF leak should be treated with decompression through the placement of a lumbar drain and antibiotics. The risk of mortality and infection was historically increased with large monobloc movements; however, this risk has more recently been mitigated with the use of distraction for larger movements hence decreasing the amount of dead space postoperatively. , Additional complications involving the eye include enophthalmos, exophthalmos, blindness, and injury to the lacrimal ducts. Relapse is a consideration at both the soft tissue and skeletal levels. Historically, an estimated 35% of patients have bony relapse and 27% experience soft tissue relapse, and therefore, a slight overcorrection can be indicated. , No clear correlation among the type of procedure performed, age of surgery, and severity of presentation has been made to predict risk of relapse; however, in general, younger patients are at an increased risk of relapse and disruption to future growth potential ( Fig. 6 ). The use of VSP in preoperative planning is anecdotally serving to decrease relapse potential.